Abstract
Glioblastoma is the most common and most malignant primary brain tumor with a median survival of 15 months. Moschamine is an indole alkaloid that has a serotoninergic and cyclooxygenase inhibitory effect. In this study, we sought to determine whether moschamine could exert cytotoxic and cytostatic effects on glioma cells in vitro. Moschamine was tested for toxicity in zebrafish. We investigated the effect of moschamine on U251MG and T98G glioblastoma cell lines. Viability and proliferation of the cells were examined with trypan blue exclusion assay, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, and the xCELLigence system. Apoptosis (annexin–propidium iodide), cell cycle, and CD24/CD44/CD56/CD15 expression were tested with flow cytometry. Treatment with moschamine significantly reduced cell viability in both cell lines tested. Induction of cell death and cell cycle arrest was confirmed with flow cytometry in both cell lines. After treatment with moschamine, there was a dose-dependent decrease in CD24 and CD44 expression, whereas there was no change in CD56 and CD15 expression in T98G cell line. The zebrafish mortality on the fifth post-fertilization day was zero even for 1 mM of moschamine concentration. The treatment of glioblastoma cell lines with moschamine may represent a novel strategy for targeting glioblastoma.
Keywords
Introduction
Glioblastoma is the most common and most malignant primary brain tumor. Despite intensive clinical investigation and several novel therapeutic approaches, the patient’s median survival remains poor in the range of 15 months. 1 The standard of care involves surgical resection followed by radiotherapy with concurrent and adjuvant chemotherapy. 2 Many chemotherapeutic agents have been used against glioblastoma including, among others, temozolomide (TMZ). 3 However, the genetic heterogeneity and the diverse molecular pathology create difficulties to treat and virtually all tumors recur. Thus, new treatments are urgently needed to battle this devastating disease.
A significant number of drugs with anti-tumor activity have been isolated from plants. Moschamine (Figure 1) is an indole alkaloid isolated mainly from the seeds of Centaurea species.4–7 Since antiquity, many Centaurea species have been used in folk medicine, menstrual disorders, vaginal candidiasis, as laxative, tonic, diuretic, expectorant, and stimulant for liver and gallbladder function. 8 The molecular weight (MW) of moschamine is 352.3 Daltons and thus able to cross the intact blood–brain barrier (BBB). New drug development for glioma should focus on substances with MW less than 600 in order to pass the intact BBB. 9 Herewith, we investigated the anti-tumor effects of moschamine, which was isolated from the seeds of C. vlachorum as previously reported 10 in glioblastoma cell lines.

Structure of moschamine.
Materials and methods
Cell lines and treatment conditions
The human glioma cell line U251MG was obtained from Dr W.K. Alfred Yung (Department of Neuro-Oncology, MD Anderson Cancer Center, Houston, TX, USA), whereas the T98G, MRC5, and HFL1 cell lines were obtained from American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were cultured in Dulbecco’s Modified Eagle’s Medium (Gibco BRL, Life Technologies, Grand Island, NY, USA) supplemented with 10% fetal bovine serum, 100 µg/mL of streptomycin, and 100 Units/mL of penicillin (Gibco BRL) at 37°C in a humidified atmosphere regulated at 5% CO2. TMZ was purchased from Schering–Plough Ltd (Welwyn Garden City, UK). Moschamine was dissolved in dimethyl sulfoxide (DMSO) to obtain stock solutions. Cultures of malignant glioma cells were treated with increasing concentrations of moschamine.
Cell adhesion assay
We used the xCELLigence Real-Time Cell Analyzer (Roche Diagnostics, GmbH, Germany) for cell adhesion assay. The system can monitor cell number and adhesion by calculating the electrical impedance at the bottom of tissue culture E-plates. The E-plates contain wells relevant to that of 96-well plates. The analyzer automatically calculated the electrical impedance as a cell index (CI). As more cells attach to the bottom of E-plates, the impedance value increases and thereby increasing the CI number. Thus, CI represents the attachment and spreading of the cells. Normalized CI can be calculated as the quotient of CI at each point to CI at the point of treatment. Cell cultures were treated 24 h after dispersion of cells in the wells of E-plates and monitored for 72 h. Values of normalized CI are presented as the mean of two different measurements.
Viability assays
Cultures of human glioma cells were treated with DMSO (0.126 µM) alone and with moschamine at concentration of 50, 150, 250, 500, and 1000 µM. Cell viability was assessed by trypan blue exclusion assay 11 and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. 12 Each assay was performed at least three times and is represented as the mean of different experiments. Cell cultures were observed every day by light microscopy, and viability tests were performed at 24 and 72 h post-treatment with moschamine. Cell proliferation was also continuously monitored for every 30 min for 72 h using the xCELLigence system via calculation of the cell index.
Flow cytometric analysis of DNA cell cycle
Cells were treated with DMSO (0.126 µM) alone and with moschamine at concentration of 150, 200, 250, and 300 µM. Untreated cells were used as a negative control. All samples were run in triplicate in at least three independent experiments. Flow cytometric analysis for propidium iodide (PI) was performed at 72 h. For the DNA cell cycle, the cells were trypsinized, centrifuged, washed with phosphate-buffered saline (PBS), and incubated with PI-working solution (50 µg/mL PI, 20 mg/mL RNase A, and 0.1% Triton X-100) for 20 min at 37°C in the dark. Using a flow cytometer (FACScalibur, BD Biosciences, San Jose, California, USA) the PI fluorescence of 10,000 individual nuclei was calculated. The fractions of the cells in G0/G1, S, G2/M, and sub-G0/G1 phase were analyzed using CellQuest software program (BD Biosciences) and were determined for each histogram as the mean peak fluorescence intensity. Cisplatin was used as a positive control to induce cell cycle arrest in glioma cell lines.
Flow cytometric analysis of apoptosis
Apoptosis induction in control, DMSO-treated (0.126 µM), or treated with moschamine cells was assayed by the detection of membrane externalization of phosphatidylserine with the Annexin V–FITC/PI Apoptosis Detection Kit I (BD Biosciences Pharmingen, San Diego, CA, USA) following the manufacturer’s instructions. The analysis by flow cytometry of PI and annexin was performed 72 h post-treatment. Cells were trypsinized, centrifuged, washed with PBS, and incubated with PI (50 mg/mL) and ribonuclease A (20 mg/mL) for 20 min at 37°C. A total of 10,000 nuclei were measured in a FACSCalibur flow cytometer (BD Biosciences). Cisplatin was used as a positive control for induction of apoptosis.
CD24/CD44/CD15/CD56 analysis by flow cytometry
Cells were treated with DMSO (0.126 µM) alone and moschamine at concentration of 150, 200, 250, and 300 µM. Untreated cells were used as negative control. Cells were dissociated by trypsinization, washed twice with PBS, and in order to block Fc receptors they were incubated with 10% human serum for 20 min on ice. FITC Mouse Anti-Human CD24 (ML5), FITC Mouse Anti-Human CD44 (Leu-44), FITC Mouse Anti-Human CD56 (NCAM16.2), FITC Mouse Anti-Human CD15 (W6D3) antibodies (BD Biosciences Pharmingen) were added and incubated for 20 min on ice in the dark. The cells were washed twice with PBS, finally resuspended in PBS, and analyzed by a flow cytometer (FACScalibur, BD Biosciences). The analysis was performed using CellQuest software program (BD Biosciences) and the results were determined for each histogram as the geometric mean peak fluorescence intensity. Quantitative measurement of bound anti-CD24, anti-CD44, anti-CD15, and anti-CD56 antibodies was achieved using the flow cytometry based QIFIKIT® assay (DAKO, Glostrup, Denmark) according to the manufacturer’s instructions. A standard curve comparing the geometric mean of fluorescence to known fluorescein isothiocyanate (FITC) content of the Quantibrite beads was constructed using QuantiCALC software (BD Biosciences).
Zebrafish
Zebrafish (Danio rerio) were maintained according to standard animal care protocols and were used in our experiments. Embryos were obtained by random pairwise mating of zebrafish in a humidified incubator at 28.5°C. The embryos were maintained in embryo water at 28°C. The moschamine toxicity was evaluated for 96 h exposure during the period 24 h-post-fertilization (hpf) to 5 days-post-fertilization (dpf) as described previously. 13 Healthy embryos were loaded onto 24-well plates with two embryos/well and incubated at 28°C for 96 h in 0.5 mL of embryo water. Embryos were treated with moschamine (10, 50, 100, 200, 500, and 1000 µM) or blank. We used 10 embryos for each concentration and 10 for control.
Statistical analysis
Data were expressed as mean ± standard deviation (SD). The significance of differences between experimental conditions was determined using Mann–Whitney test. Differences were considered significant at p values less than 0.05. The half maximal inhibitory concentration (IC50) value of moschamine was calculated using the software of the xCELLigence RTCA DP system (Roche, Mannheim, Germany).
Results
Sensitivity of glioblastoma cells to moschamine and IC50 calculation
In order to investigate the sensitivity of U251MG and T98G cell lines to moschamine, we utilized xCELLigence system, 2 × 103 U251MG and T98G cells were seeded and were exposed to moschamine (50, 150, 250, 500, and 1000 µM) after 24 h. Both cell lines were sensitive to treatment with moschamine in a dose-dependent manner. Cell proliferation was analyzed in the xCELLigence system for 72 h. The IC50 value of reduced proliferation for moschamine was 59 × 10−5 M in U251MG cells and 73 × 10−5 M for the T98G cells during the 72-h treatment. Human fetal lung fibroblasts (HFL1 and MRC5) were incubated in the absence or presence of increasing concentrations of moschamine for 72 h, and cell proliferation was used as a marker for toxicity. Moschamine had no adverse effect on the non-cancer human fibroblast cell line MRC5 (IC50 = 343 × 10−5 M) and HFL1 (IC50 = 290 × 10−5 M). MTT assay was used to calculate cytotoxicity effects on U251MG and T98G cells after moschamine treatment. We found that moschamine induced cytotoxicity 72 h post-treatment (Figure 2). IC50 values of moschamine induced cytotoxicity based on MTT assay were similar for both U251MG (IC50 = 238 µM) and T98G (IC50 = 193 µM) cell lines. To examine the cytotoxic effects of moschamine, we also deployed the trypan blue exclusion test. We determined the cell survival rate, which was defined as the number of living cells after a 72-h exposure to escalating doses of moschamine compared with the number of live control cells. The number of both U251MG and T98G cells was significantly decreased even after treatment with 50 µM of moschamine (Figure 3(a)). Collectively, moschamine reduces cell proliferation and induces cytotoxicity in glioma cells in lower concentrations than in normal human fibroblasts.

Results of the MTT assay on U251MG and T98G cells after moschamine treatment.
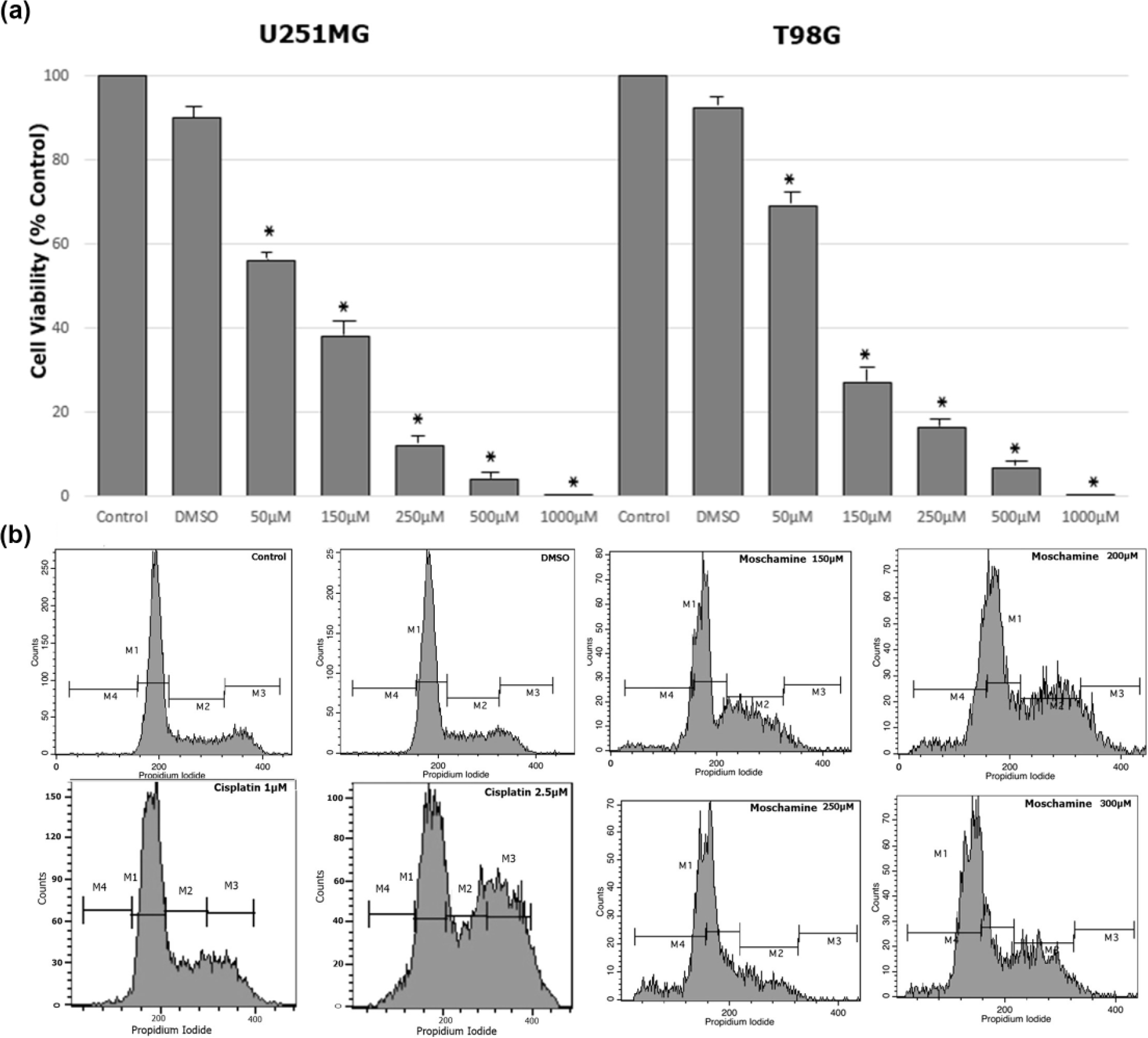
(a) Viability of glioma cells following moschamine treatment. Cell viability was assessed by the trypan blue exclusion test in U251MG and T98G glioma cells. A total of 104 glioma cells were seeded on a 24-well plate. After 24 h of seeding glioma, the cells were treated with escalating concentrations of moschamine (150, 200, 250, and 300 µM). Viability tests were performed at day 1 and 3. Values shown are the means and standard deviations from three independent experiments. Values are normalized to non-treated cells (*p < 0.05 vs control). (b) The effect of moschamine in cell cycle. U251MG cells (105) were seeded on 24-well plate and after 24 h were exposed to DMSO (0.126 µM), cisplatin (1 and 2.5 µM), and escalating concentrations of moschamine (150, 200, 250, and 300 µM) for another 72 h. At 72 h, cells were stained with propidium iodide and the DNA content was evaluated by flow cytometry. Cisplatin induced a dose-dependent cell cycle arrest. Moschamine concentration equal to or higher than 150 µM produced S-phase cell cycle arrest in a dose-dependent manner. An escalating increase in the sub-G0/G1 phase was observed.
Moschamine induced cell cycle arrest
To examine the cell cycle events underlying these observed growth inhibitory effects, we assessed the effect of moschamine on cell cycle progression of the U251MG and T98G cells. Both cell lines were exposed to DMSO alone and increasing concentrations of moschamine (150, 200, 250, and 300 µM) for 72 h (Figure 3(b)). In the absence of drug, DMSO, alone, had no effect on the cell cycle distribution of both cell lines. In U251MG and T98G cells, moschamine induced an increase in cell populations in sub-G0/G1 (M4) and S-phase arrest in a dose-dependent manner. At a concentration of 300 µM, there was an apoptotic peak in both cell lines (Table 1).
Modulation of cell cycle by moschamine. The analysis of cell cycle distribution in U251MG and T98G cells. Data are given as mean ± SD and are representative of three separate experiments.

SD: standard deviation; DMSO: dimethyl sulfoxide.
p < 0.05, **p < 0.01 versus control.
Comparison of cell cycle arrest induced by moschamine and TMZ
TMZ is the chemotherapy of choice to treat high-grade gliomas. Having this in mind, we compared the action of TMZ and moschamine and also their combination effect. U251 or T98G cells were treated with 100 µM or 200 µM TMZ, 100 µM moschamine, or 100 µM TMZ combined with 100 µM of moschamine (Figure 4). In T98G, TMZ-resistant cells, moschamine had a similar action with or without TMZ. On the contrary, we found that in U251 cells, moschamine induced a similar G2/M cell cycle arrest with TMZ for ~30% of the cell population. More importantly, the combination of 100 µM moschamine and 100 µM TMZ had a stronger effect than 200 µM TMZ, suggesting a possible synergistic effect.

The effect of TMZ, moschamine, and their combination on the cell cycle. U251MG and T98G cells (105) were seeded on a 24-well plate and after 24 h were exposed to TMZ (100 µM) alone, TMZ (200 µM), moschamine (100 µM) alone, and TMZ (100 µM) and moschamine (100 µM) for 72 h. At 72 h, the cells were stained with propidium iodide and the DNA content was evaluated by flow cytometry. In T98G, temozolomide-resistant cells, moschamine had a similar action with or without temozolomide. On the contrary, we found that in U251 cells, moschamine induced a similar G2/M cell cycle arrest with temozolomide for ~30% of the cell population. The combination of 100 µM moschamine and 100 µM temozolomide had a stronger effect than 200 µM temozolomide, suggesting a synergistic effect in U251MG cells.
Moschamine induced apoptosis and significant mitochondrial membrane depolarization
We then examined the ability of moschamine to promote an apoptotic effect in the U251MG and T98G cell lines. Apoptotic cells were determined by flow cytometry after a 72-h treatment using PI/annexin V staining. Our results have shown that moschamine significantly induced apoptosis in a dose-dependent manner starting with concentrations of 150 µM in both the cell lines. Apoptosis was more pronounced in T98G cell line (Figure 5). To study whether the apoptosis, induced by moschamine, is associated with decreased mitochondrial membrane potential, untreated U251MG and T98G cells or moschamine-treated cells (150 and 200 µM for 24 h) were stained with JC-1 and analyzed by flow cytometry (Figure 6). JC-1 fluorescence in FL-2 and FL-1 channels corresponds to mitochondria with a polarized Δψ (gated region R4), while JC-1 that fluoresces in the FL-1 channel and lacks fluorescence in the FL-2 channel corresponds to mitochondria with depolarized Δψ (gated region R5). Moschamine treatment led to a significant increase in the number of cells with lowered red fluorescence, 38.6% in 150 µM and 28% in 200 µM, compared to 16.7% in untreated control cells in U251MG cells. In T98G cells, moschamine also led to a significant increase in the number of cells with lowered red fluorescence, 28.7% in 150 µM and 32.6% in 200 µM, compared to 18.2% in untreated control cells (Figure 1 in Supplementary material). Thus, cell death induced by moschamine is associated with mitochondrial membrane depolarization, indicative of the intrinsic, mitochondrial-dependent apoptotic pathway.

Moschamine induces apoptosis in glioma cell lines. Proportion of apoptosis calculated by flow cytometric analysis of annexin/PI in U251MG and T98G cells. U251MG and T98G cells (105) were seeded on a 24-well plate and after 24 h were exposed to DMSO (0.126 µM) and escalating concentrations of moschamine (150, 200, 250, and 300 µM). Apoptosis was assessed after 72 h of treatment by annexin V/PI staining. Moschamine increased the percentage of annexin V–positive cells in a dose-depended manner in both the cell lines (*p < 0.05 vs control).

Mitochondrial membrane potential analysis with JC-1 in untreated and moschamine (MM)-treated U251MG Cells. Untreated U251MG cells or coumarol-treated cells (150–200 µM for 24 h) were stained with JC-1 and analyzed on a BD FACSCalibur. JC-1 fluorescence in FL-2 and FL-1 channels was analyzed using gated region R4 (cells with high FL-2 fluorescence, corresponding to high mitochondrial membrane potential) and region R5 (cells with decreased fluorescence in the FL-2 channel, corresponding to mitochondria with a depolarized Δψ). The effects of escalating concentrations of moschamine in U251MG cells (20×).
Moschamine suppressed CD24 and CD44 expression
We then analyzed the expression of markers for cancer stem cells, CD24, and CD44 by flow cytometry. CD24 is a heavily glycosylated cell surface protein that is elevated in various malignancies, and high expression of CD24 is associated with enhanced invasiveness and proliferation. The mean number of CD24 molecules on the surface of T98G cells was 73,000 molecules/cell and the mean number of CD44 on the surface of T98G cells was 191,000 molecules/cell. We found a dose-dependent decrease in the number of CD24 molecules/cell after moschamine treatment, which was significant at a concentration equal to or over 200 µM. For the CD44 expression, we found a dose-dependent decrease in the number of CD44 molecules/cell after moschamine treatment, which was significant even at a concentration of 150 µM (Figures 7 and 8).

Flow cytometry analysis of the expression of CD24 and CD44 in T98G cells. U251MG and T98G cells (105) were seeded on 24-well plate and after 24 h were exposed to DMSO (0.126 µM) and escalating concentrations of moschamine (150, 200, 250, and 300 µM) for another 72 h. Using anti-human CD24 and CD44 monoclonal antibodies, we measured, by flow cytometry, the number of CD24 and CD44 molecules on the surface of T98G cells (see section “Material and methods”). Box and whisker plots display median (horizontal black line) and quartiles with maximum and minimum values (*p < 0.05 vs control).

Histograms of CD24, CD44, CD15, and CD56 expression in a representative experiment after a 72-h treatment of T98G cells with escalating concentrations of moschamine.
Moschamine does not affect CD56 and CD15 expression
We then analyzed the expression of CD56 and CD15 by flow cytometry. The mean number of CD56 molecules on the surface of T98G cells was 1100 molecules/cell and the mean number of CD15 on the surface of T98G cells was 210 molecules/cell. After moschamine treatment, there was no significant change in the expression of CD56 and CD15 in T98G cells (Figure 8).
Zebrafish
To evaluate the possible toxicity of zebrafish embryos exposure to moschamine, we measured the mortality during observation period. The zebrafish mortality on the fifth post-fertilization day was zero even for 1 mM of moschamine concentration.
Discussion
This study showed for the first time that moschamine is a promising treatment for malignant glioma cell lines in vitro. Moschamine induced suppression of glioma growth, cell cycle arrest, and apoptosis in both U251MG and T98G cell lines. Moschamine also decreased CD24/CD44 expression in T98G cells. In zebrafish model, no mortality was observed even at a moschamine concentration of 1 mM. Since glioblastoma is difficult to treat tumors, moschamine seems to be a promising novel treatment.
Moschamine is a phenylpropenoic acid amide that can be isolated from plants. Csapi et al. studied the anti-proliferative effects of extracts prepared from the whole plant of C. arenaria. The preparative work also resulted in the isolation of moschamine which was found to produce anti-proliferative effects on a skin epidermoid carcinoma (A431) cell line but not in cervical epithelial adenocarcinoma (HeLa) or breast epithelial adenocarcinoma (MCF7) cell lines. 14 Another study has also proved that moschamine may be a promising cytotoxic agent for colon (Caco2) cancer cells. 5 Park showed that moschamine holds serotonergic and cyclooxygenase (COX) inhibitory activities by suppressing cyclic adenosine monophosphate (cAMP) formation, via binding to 5-hydroxytryptamine (5-HT1) receptors, and by inhibiting COX-1 and COX-2. The oral bioavailability of moschamine was also investigated in mice. 15 In this study, we tested moschamine in two non-cancer human fibroblast cell lines. Moschamine appeared to be significantly less toxic in normal cells than in cancer cells. This is of paramount importance, since a plant extract could be considered as a potential anti-cancer drug, if it can kill cancer cells without causing excessive damage to normal cells.
COX-2 protein is upregulated in glioma cells and COX-2 inhibitors such as NS-398, celecoxib, and meloxicam have been demonstrated to hold anti-proliferative activities. 16 Furthermore, these agents enhanced radiation-induced cell death, when glioma cell lines were incubated before radiation. 17 Since moschamine has COX inhibitory activities, this might be a possible mechanism of action for this compound. Further research to investigate whether moschamine holds additional radiosensitization properties is warranted.
Several natural phytochemicals have been demonstrated to block cell cycle progression and to inhibit the growth of cancer cells. A hallmark of cancer is escaping cell cycle arrest. 17 There are several checkpoints, such as those at G1/S and G2/M, in cell cycles, that hold the role of surveillance mechanisms. In this study, moschamine triggered S-phase cell cycle arrest and a dose-dependent increase in the sub-G0/G1 cell fraction. Further research is needed to elucidate the exact mechanism by which moschamine exerts this action. In a first attempt to elucidate the mechanism by which moschamine exerts this action, we found that moschamine treatment significantly decreased mitochondrial membrane potential in U251MG and T98G cells. Taking into account the cell death pathways that a cancer cell undergoes, 18 this result supports an intrinsic, mitochondrial-dependent apoptotic pathway.
TMZ is the first-line treatment for glioblastoma, since it prolongs survival and delays progression without affecting the quality of life. Even in the elderly, TMZ is comparable to radiotherapy regarding overall survival and progression-free survival. 3 In this study, two glioma cell lines were exposed to moschamine. In the U251MG cell line, there is no O 6 -methylguanine DNA methyltransferase (MGMT) protein expression and this cell line is TMZ sensitive, whereas in T98G cell line, there is MGMT expression and these cells are resistant to TMZ.19–21 We compared the action of TMZ and moschamine and found that in U251 TMZ-sensitive cells they have a synergistic effect. This result suggests that moschamine may also be used as a combination therapy with TMZ. In addition, moschamine induced cell cycle arrest and apoptosis on both TMZ-sensitive U251MG and TMZ-resistant T98G, whereas apoptosis was more pronounced in T98G cells. Thus, moschamine could be useful as a first-line treatment or an additional/combinatory treatment in TMZ-resistant tumors.
The NCAM, also known as CD56, is present in the intercellular junctions of neurons and glial cells and is expressed on several brain tumors, including gliomas. 22 Todaro et al. 23 studied several brain tumors and reported NCAM immunopositivity, which was inversely correlated with the histological grade of malignancy. In this study, we found that moschamine had no effect on the expression of CD56 in T98G cells. Knüpfer et al. 24 investigated the anti-tumor effect of valproic acid in glioma cells. Valproic acid markedly increased CD56 and decreased the CD44 expression in T98G cells, while it had a non-significant effect on cell proliferation. Based on this study and the fact that in our study we observed a cytotoxic effect without change in CD56 expression, we suggest that CD56 expression may not be implicated in glioma cell resistance to chemotherapy. CD15 is a marker of glioma stem cell together with CD133 and nestin. 25 In this study, we found low expression of CD15. This expression was not affected by moschamine. This finding is in accordance with the study of Reifenberger et al. 26 who found that anaplastic gliomas and glioblastomas consistently did not express CD15 contrary to low-grade gliomas. In addition, low CD15 expression, may represent a barrier to metastasis, 27 an intrinsic feature of many gliomas that is not altered by moschamine treatment.
CD24 is a heavily glycosylated surface protein holding an important role in cell–matrix and cell–cell interactions, thus having a role in metastasis and cell adhesion in cancer cells. 28 Deng et al. 29 reported overexpression of CD24 in higher grade gliomas, whereas patients with CD24 positive tumors showed poor prognostic outcome after surgery. CD44 is a transmembrane glycoprotein expressed in nearly every cell and is a receptor for hyaluronic acid. In cancer cells, CD44 confers a growth advantage. 30 In glioblastoma tissue, CD44 has been found unregulated, whereas its depletion blocks tumor growth and sensitizes tumor cells to cytotoxic drugs in vivo. 31 Both CD24 and CD44 are associated with cancer stem cells. Treatment of T98G cells with moschamine resulted in a decrease in CD24 and CD44 expression. This is an important preliminary result, since glioma cancer stem cells show resistance to chemotherapy 32 and have been shown to propagate glioblastoma after chemotherapy 33 through distinct mechanisms. 34 Collectively, taking into account that moschamine does not affect the already low CD15 in glioma cells, but downregulates CD24 and CD44, markers associated with cancer stem cells, indicate that its action may involve reduction of glioma cancer stem cells without affecting their metastatic ability.
In conclusion, glioblastoma constitutes an extremely heterogeneous tumor comprising both differentiated and stem cells. A multifaceted approach combining several treatment strategies might be eventually required for better results. The present study indicated that exposure of glioma cells to moschamine reduces cell viability significantly and induces cell cycle arrest at S phase and apoptosis. Thus, moschamine seems to be a promising compound against glioma, and there is a need for our preliminary observations to be validated in glioma xenograft models.
Footnotes
Acknowledgements
The authors thank Miss Eleni Dimoka for English language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Joseph and Esther Gani Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
