Abstract
Somatostatin receptors are overexpressed in neuroendocrine tumors, whose endogenous ligands are somatostatin. DOTA-TATE is an analogue of somatostatin, which shows high binding affinity to somatostatin receptors. We aim to evaluate the 68Ga/177Lu-labeling DOTA-TATE kit in neuroendocrine tumor model for molecular imaging and to try human–positron emission tomography/computed tomography imaging of 68Ga-DOTA-TATE in neuroendocrine tumor patients. DOTA-TATE kits were formulated and radiolabeled with 68Ga/177Lu for 68Ga/177Lu-DOTA-TATE (M-DOTA-TATE). In vitro and in vivo stability of 177Lu-DOTA-TATE were performed. Nude mice bearing human tumors were injected with 68Ga-DOTA-TATE or 177Lu-DOTA-TATE for micro–positron emission tomography and micro-single-photon emission computed tomography/computed tomography imaging separately, and clinical positron emission tomography/computed tomography images of 68Ga-DOTA-TATE were obtained at 1 h post-intravenous injection from patients with neuroendocrine tumors. Micro–positron emission tomography and micro-single-photon emission computed tomography/computed tomography imaging of 68Ga-DOTA-TATE and 177Lu-DOTA-TATE both showed clear tumor uptake which could be blocked by excess DOTA-TATE. In addition, 68Ga-DOTA-TATE–positron emission tomography/computed tomography imaging in neuroendocrine tumor patients could show primary and metastatic lesions. 68Ga-DOTA-TATE and 177Lu-DOTA-TATE could accumulate in tumors in animal models, paving the way for better clinical peptide receptor radionuclide therapy for neuroendocrine tumor patients in Asian population.
Introduction
Neuroendocrine tumors (NETs) are a group of heterogeneous tumors originated from the diffuse neuroendocrine system, and their occurrence has been increasing in recent years. Over 70% NETs express somatostatin receptors (SSTRs), which is one of the biomarkers for NETs.1–5
Somatostatin (SST) is a hormone secreted in the pancreas and pituitary gland, which binds SSTRs—a class of G-protein-coupled receptors. Five distinct subtypes of SSTRs (SSTR1-5) have been characterized, with SSTR2 as the most frequently expressed type. SST can inhibit cell growth, and it is widely distributed in the central nervous system and the endocrine tissues outside the brain. 5 Since the natural SST has a short half-life (about 2 min) in vivo, and needs complex process to prepare in laboratory, it has limited use. This creates the need to develop clinically useful somatostatin analogues (SSAs).6,7
The synthetic peptide DOTA-TATE is a clinically useful SSA developed recently, which is structurally stable and has better SSTR-targeting affinity and selectivity especially for SSTR2—which is the most often expressed SSTRs in NETs; the binding affinity of DOTA-TATE for SSTR2 is 0.38 nM (half maximal inhibitory concentration (IC50)).8,9 Moreover, because DOTA-TATE has a longer biological half-life (about 2 h) compared with natural SST, as well as the clinical safety property of the peptide, its radioactive analogue, 68Ga-DOTA-TATE, has been approved to detect NETs for clinical application in patients by Food and Drug Administration (FDA) in 2016. 10
Peptide receptor radionuclide therapy (PRRT) is a form of molecular targeted therapy which is performed using a small peptide (a SSA similar to octreotide) that is coupled with a radionuclide emitting beta radiation.11,12 The first radionuclide used in PRRT was 90Y; however, its high-energy electron-emitting property is problematic for treating some small tumors. 13 111In is another radionuclide used in PRRT, but it has high abundance of photon emission. 14 The radioactive property of 177Lu makes it suitable both for therapeutic and diagnostic purposes. Its emission of γ photons with energies of 113 keV (6.4%) and 208 keV (11%) provides the opportunity to carry out in vivo tumor imaging. 15 It also emits of β− particles, a half-life of 6.7 days, with maximum energies of 497 keV (78.6%), 384 keV (9.1%), and 176 keV (12.2%) which contributes to the in vivo targeted therapy of NETs. Mostly, its β− particles have shorter penetration range in tissues than those of 90Y and 111In.
68Ga-DOTA-TATE has been approved by FDA as an excellent positron emission tomography (PET) tracer for SSTRs overexpressed tumors; in addition, 177Lu-DOTA-TATE holds promise in NET therapy. Here, we report the kit formulation, quality control, and molecular imaging of 68Ga/177Lu-DOTA-TATE (M-DOTA-TATE) for both therapeutic and diagnostic purposes of NETs. The preparation of the radioactive material takes the kit formulation approach, which has used extensively recently, such as prostate-specific membrane antigen (PSMA) kits for prostate cancer diagnosis. 16 The new agent was evaluated biologically for in vitro and in vivo stability, in vivo biodistribution in mice, micro-PET/micro-single-photon emission computed tomography (SPECT)/computed tomography (CT) imaging in mice bearing tumors, and, especially, clinical PET/CT imaging in patients with NETs.
Materials and methods
Materials
68Ga was eluted from a 740 MBq 68Ge/68Ga generator from Isotope Technologies Garching (Munich, Germany). 177Lu was purchased from PerkinElmer (Boston, MA, USA). DOTA-TATE was purchased from ABX (Heinrich, Germany). Acetonitrile was purchased from Sigma-Aldrich (St. Louis, MO, USA); other reagents, including sodium acetate (NaOAc), sodium chloride, potassium chloride, disodium hydrogen phosphate dodecahydrate, and sodium dihydrogen phosphate, were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China); all chemicals and solvents used in our work were of analytical reagent (AR) grade.
The Agilent Technologies 1200 series high-performance liquid chromatography (HPLC) was purchased from Agilent Technologies (Forest Lake, CA, USA). The 68Ga/177Lu compound was analyzed by YMC-Pack ODS-AQ AA12S05-2546WT column (Shimadzu Corp., Kyoto, Japan) coupled with the HPLC system. The HPLC system was equipped with both ultraviolet (UV) absorption detector (280 nm) and B-Fc-1000 HPLC radioactivity detector obtained from Bioscan (Washington, DC, USA). All the solvents used in HPLC were of HPLC grade and purchased from local manufacturers, and they were degassed and filtered before use. The radio-thin-layer chromatography (TLC) system together with the TLC–silica gel (SG) strips was purchased from Gelman Sciences (Washington, DC, USA).
Cell culture and animals
The HT-29 tumor cells were obtained from American Type Culture Collection (New York, NY, USA), the human pulmonary giant cell carcinoma cell line (PG tumor cells) was provided by the Peking University Cancer Hospital (Beijing, China), and they were cultured at 37°C with 5% CO2 in RPMI-1640 medium containing 10% (v/v) fetal bovine serum, which were obtained from Invitrogen (Carlsbad, CA, USA). Animal experiments were performed in BALB/c nude mice (16–18 g) which were reared in the laboratory animal facility of our center under standard care. All the animal experiments were approved by the Institutional Animal Care and Use Committee of Peking University Cancer Hospital & Institute.
Kit formulation
DOTA-TATE was dissolved in water to form the pre-kit solution (1.0 mg/mL). Each glass vial was added with 20 µg of DOTA-TATE prepared above and 65 µL of 1.0 M NaOAc (pH = 4.0), and all vials described above were freeze-dried by freeze-dryer at −70°C for 16 h. The kits were kept at −20°C and were stable (>99%) for at least 1 year, as confirmed by MALDI-TOF. The kits were used for 68Ga/177Lu labeling.
Radiosynthesis of 68Ga-DOTA-TATE
68GaCl3 solution (1.0 mL, 370 MBq), which was eluted from the 68Ge/68Ga generator, was added into a single kit vial as described above with a plastic syringe, and the mixture was heated at 95°C for 15 min (Scheme 1). 68Ga-DOTA-TATE solution was prepared and quality control was performed. The reaction progress and final product were monitored by a combination of radio-HPLC and radio-TLC as described earlier. For radio-HPLC, 0.1% trifluoroacetic acid (TFA) in water as solvent A and 0.1% TFA in acetonitrile as solvent B were used (gradient program: 0–8 min, 20%–65% solvent B, and flow rate: 1.0 mL/min). For radio-TLC, the strips were developed using saline solution containing 4.0 mM of ethylenediaminetetraacetic acid (EDTA).
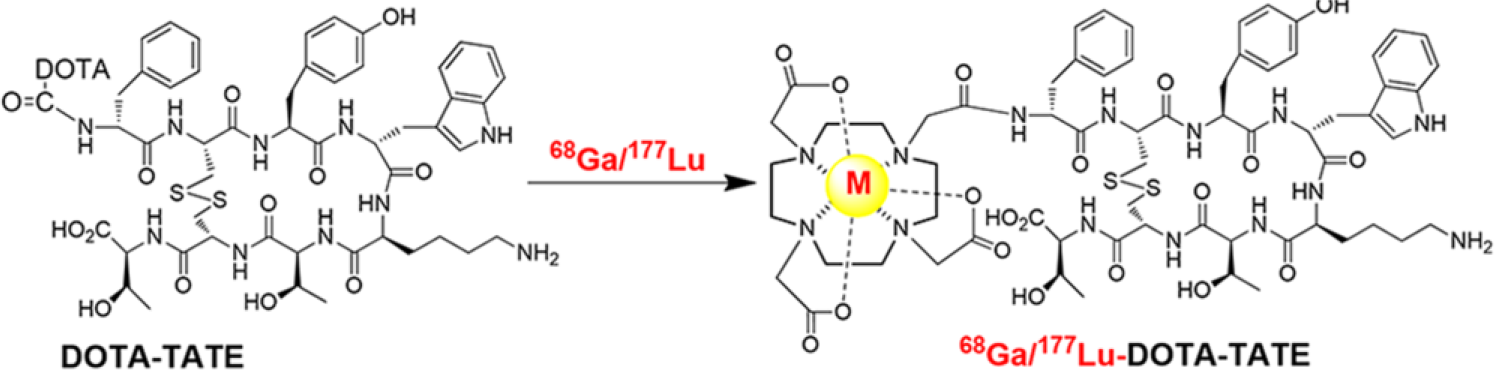
The radiosynthesis of M-DOTA-TATE.
Radiosynthesis of 177Lu-DOTA-TATE
500 µL of 177LuCl3 (74 MBq) solution was added into a single kit vial to dissolve the materials (Scheme 1). The mixture was heated at 95°C for 15 min. 177Lu-DOTA-TATE solution was prepared and quality control was performed. The product was monitored by radio-HPLC and radio-TLC procedures, just as 68Ga-DOTA-TATE, as described above.
In vitro and in vivo stability of 177Lu-DOTA-TATE
The in vitro stability of 177Lu-DOTA-TATE was evaluated in phosphate-buffered saline (PBS; pH = 7.4), or NaOAc (pH = 5.5), for a total of 240 h post-preparation. A solution of 177Lu-DOTA-TATE (3.0 µL, 14.8 MBq) was added into 300 µL of PBS (1.0 M) or 300 µL of NaOAc (1.0 M). Each reaction mixture was then incubated at room temperature (25°C), and the chemical composition was determined by radio-HPLC at different time points during the 240 h incubation, with three repeats for each time point. For in vivo stability in normal BALB/c mice, 177Lu-DOTA-TATE (3.75 µL, 18.5 MBq) in 200 µL solution of saline was injected intravenously into the tail vein of each mouse; they were transferred back to the cage with proper food and water. At the time of 5, 20, 40, 60, 120, and 240 h post-injection, the urine of each mouse was collected by a hot compress on its abdomen. The urine was centrifuged at 3000 r/min for 2 min, and a specific amount of supernatant was collected for evaluation by radio-HPLC. Each experiment point was repeated three times.
Biodistribution of 177Lu-DOTA-TATE
The BALB/c mice were divided into three groups, with three mice in each group. 177Lu-DOTA-TATE (3.75 µL, 18.5 MBq) in 200 µL solution of saline was injected intravenously via the tail vein. At 1, 4, and 24 h post-injection, the mice were sacrificed, and the tissues and organs of interest were collected, including blood, heart, liver, spleen, kidneys, lung, stomach, small and large intestines, thigh muscle, bone, and brain. All tissues and organs were weighed, and the radioactivity in each sample was measured using a γ counter, with an energy window 10% above 208 keV photo peak and background radiation correction. The results were presented as percentage of injected dose (ID) per gram of tissue or organ (% ID/g).
Micro-PET imaging in nude mice
The nude mice tumor model was made by injecting 2 × 106 viable human PG tumor cells subcutaneously under the right axilla of each nude mouse. When the tumor had reached about 1.0 cm in diameter, 68Ga-DOTA-TATE (3.0 µL, 11.1 MBq) in 200 µL solution of saline was injected intravenously via the tail vein in each mouse. Each animal was placed prone on the bed of a PET system (SuperArgus, Sedecal, Spain). The PET images were acquired for different time periods at 1 and 2 h post-injection of 68Ga-DOTA-TATE under the anesthesia of 4%–5% isoflurane for induction and 1%–2% for maintenance, both balanced by oxygen. After micro-PET imaging, the nude mice were sacrificed by cervical dislocation; the organs of interest were dissected for ex vivo PET imaging, including tumor, heart, liver, lung, spleen, kidneys, and intestines. Each test was repeated three times.
Micro-SPECT/CT imaging in nude mice
The nude mice tumor model was prepared by injecting human colon HT-29 tumor cells in a similar way as described above. 177Lu-DOTA-TATE (3.75 µL, 18.5 MBq) in 200 µL solution of saline was injected intravenously via the tail vein of each mouse. The whole-body static SPECT/CT images were taken at 1 and 4 h post-injection under anesthesia, with three repeats for each time point. For blocking study, 177Lu-DOTA-TATE (18.5 MBq) and DOTA-TATE (1.0 mg) in 200 µL solution of saline were co-injected into nude mice bearing HT-29 tumor. Each animal was placed prone on the bed of a SPECT system (NanoSPECT/CT; Mediso, Budapest, Hungary) equipped with high-resolution parallel-hole collimators used to acquire whole-body SPECT/CT scans. The whole-body static SPECT/CT images were taken at 1 and 4 h post-injection under anesthesia.
Clinical applications
Patients, who were suspected with SSTR positive tumors, were recruited from Peking University Cancer Hospital & Institute, from June 2013 to August 2014, excluding those of pregnant or lactating women or those with severe liver or kidney dysfunction. The histopathological tumor grade of NET patients was determined using Ki-67 index according to the World Health Organization. 17 Overall, 10 patients were enrolled, with detailed clinical history. The patients’ informed consents were obtained before they participated in the study. This study was approved by the Ethics Committee of Peking University Cancer Hospital & Institute (Permission No. 2014011313). PET/CT images were obtained at 60 min post-intravenous injection of 1.85–3.7 MBq/kg of 68Ga-DOTA-TATE, using the Philips Gemini TF 16 PET/CT tomograph (Eindhoven, Netherlands). The 68Ga-DOTA-TATE radiotracer solution was filtered before injection with a 0.22 µm Millex-LG filter obtained from EMD Millipore (Darmstadt, Germany). PET/CT imaging scanned from vertex to the mid-thigh. The CT acquisition and reconstruction parameters were 120 keV, 100 mAs, 0.5 s tube rotation, and 3 mm thickness, and ordered subset expectation maximization (OSEM) was used for PET reconstruction, with background radiation corrected. At last the Fusion Viewer software (extended brilliance workspace nuclear medicine, EBW NM 2.0, Philips, Netherlands) was used to fuse the PET image with the CT image for each patient.
Statistical analysis
Statistical analysis was conducted using GraphPad Prism 6 software (GraphPad Software, San Diego, CA, USA). Data in the article are expressed as mean ± standard deviation, and one-way analysis of variance (ANOVA) was applied to determine the mean values. p < 0.05 was considered to be statistically significant.
PET/CT images were evaluated by two experienced nuclear medicine physicians blinded to the initial reports, and they could reach an agreement through discussion when inconformities arise. After region of interest (ROI) was delineated, maximum standardized uptake value (SUVmax) was calculated using the software provided by the instrument manufacturer.
Results
Radiosynthesis of M-DOTA-TATE
Both 68Ga-DOTA-TATE and 177Lu-DOTA-TATE have shown high radiochemical yields (Table 1 and Supplementary Figure 1–4 available online). The chemical and radiochemical yields reached >99% as measured by radio-HPLC and radio-TLC, as reported. 18 Other quality controls of M-DOTA-TATE such as organic residues, endotoxins, and sterility are presented in Table 1. Before the radiolabeling experiment, the chemical identities within the kits were confirmed by MALDI-TOF with the molecular ion at 1435.8 for DOTA-TATE (Supplementary Figure 5 available online).
Quality control of M-DOTA-TATE for injection.

QC: quality control; HPLC: high-performance liquid chromatography; TLC: thin-layer chromatography.
In vitro and in vivo stability of 177Lu-DOTA-TATE
177Lu-DOTA-TATE showed in vitro stability in PBS and NaOAc solution, with >96% radiochemical purity within 240 h incubation at room temperature (Figure 1). It also showed good in vivo stability (Figure 1). The radiochemical purity collected in urine showed >90% within 240 h post-injection.

In vitro and in vivo stability of 177Lu-DOTA-TATE.
Biodistribution of 177Lu-DOTA-TATE
The radioactivity in tissues and organs of normal mice decreased with time post-injection (Table 2). The radioactivity level in blood dropped fairly quickly with time, with rather low levels at 4 h post-injection. No significant retention was observed in liver, suggesting that 177Lu-DOTA-TATE was fairly water-soluble. Significant uptake of 177Lu-DOTA-TATE was observed in kidneys, most likely due to excretion through the renal system, consistent with previous work. 19
Biodistribution of 177Lu-DOTA-TATE in percentage of injected dose per gram of organs at 1, 4, and 24 h post-injection in mice. Values are mean ± standard deviation (n = 3/group).

Micro-PET imaging in nude mice
Micro-PET imaging showed that high uptake of 68Ga-DOTA-TATE was observed in tumor, liver, and kidneys in nude mice bearing PG tumor cells (Figure 2). The tumor was clearly visible after administration of 68Ga-DOTA-TATE at 1 h and was decreased rapidly at 2 h post-injection. The clear high uptake of the radiotracer in liver and bladder might indicate the metabolic excretion of 68Ga-DOTA-TATE in nude mice. Tumor uptake was further confirmed by ex vivo micro-PET imaging at 1 h post-injection; after the nude mice were dissected, important organs were taken out for an ex vivo micro-PET imaging (Figure 3).

Micro-PET imaging of 68Ga-DOTA-TATE in nude mice bearing PG tumors at (a) 1 h and (b) 2 h after intravenous injection. The white arrows indicate the tumors.

Ex vivo micro-PET imaging of 68Ga-DOTA-TATE after dissecting organs in nude mice bearing PG tumors.
Micro-SPECT/CT imaging in nude mice
Micro-SPECT/CT imaging of 177Lu-DOTA-TATE in nude mice bearing HT-29 colorectal tumors have shown that high uptake in the tumor was visible at 1 h post-injection (Figure 4). Tumor uptake increased with time and was clearly visualized by 4 h post-injection. No purification of radiotracer synthesized using the kit formulation was needed for micro-SPECT/CT.

Micro-SPECT/CT imaging of 177Lu-DOTA-TATE in nude mice bearing HT-29 tumors at (a) 1 h and (b) 4 h and blocking at (c) 1 h and (d) 4 h after intravenous injection. The white arrows indicate the tumors.
After blocking the SSTRs using DOTA-TATE, the tumor uptake was barely visible in micro-SPECT/CT images at 1 and 4 h post-injection, indicating that the radiotracer binds with SSTRs in the tumor. Relatively high uptake was observed in kidneys, indicating that the radiotracer was cleared mainly through the renal route.
Clinical PET/CT imaging of 68Ga-DOTA-TATE
Representative PET/CT imaging of 68Ga-DOTA-TATE was obtained from a 38-year-old man with multiple liver metastasis and unknown primary lesion as shown in Figure 5. The histopathological tumor grade of the patient was G2 with a Ki-67 index of 3%. PET/CT imaging of 68Ga-DOTA-TATE showed that the mean SUVmax in the liver ranged from 4.3 to 14.3 (Figure 5(a) and (b)), and the SUVmax in left lower rectal wall reached 8.6 (Figure 5(c)); the latter high uptake indicated the location of high SSTRs expression, which was the location of primary NET.
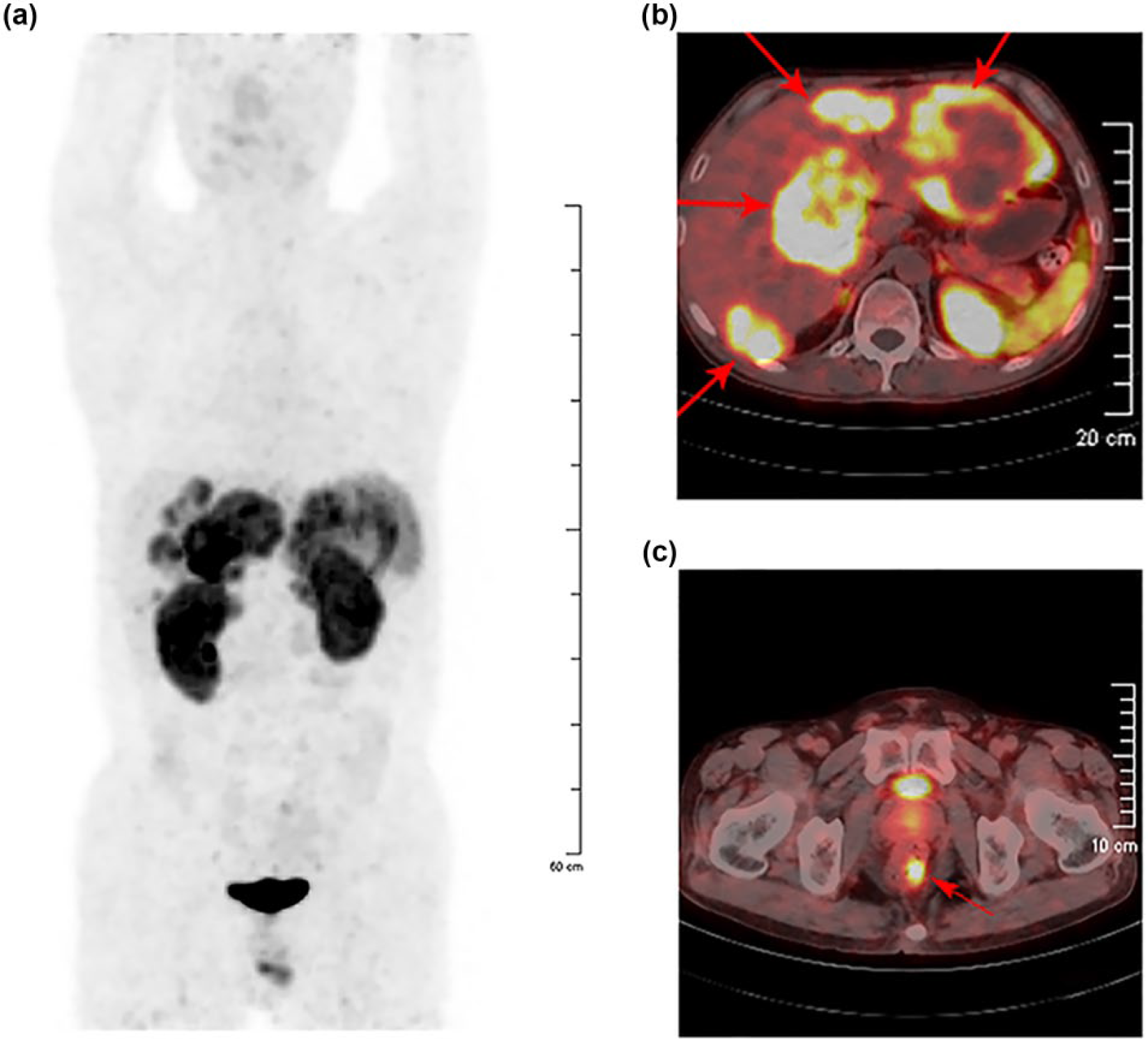
(a) PET imaging of NET patient after injection of 68Ga-DOTA-TATE. PET/CT images show (b) liver lesions and (c) rectum lesion. Red arrows show the positions of lesions.
Discussion
In recent decades, the morbidity and mortality rates of NETs have increased markedly; however, the lack of specific symptoms often results in the misdiagnosis of the disease, which presents a big challenge for early and accurate diagnosis.20,21 In recent years, the diagnosis of NETs includes blood test (CgA), pathologic diagnosis, molecular imaging, and others. The tumor marker of chromogranin A (CgA) in human blood can be affected by several other factors such as SSAs and proton pump inhibitors (PPIs); thus, blood test can cause misdiagnosis of NETs. 22 Though pathologic diagnosis can determine the staging and grading of the tumors, it cannot achieve whole-body diagnosis and locate the position of primary and metastatic lesions. Molecular imaging, with the property of non-invasiveness, rapid-clearance, and thorough diagnosis, has been a common diagnostic method of NETs in clinical application. 21
SSAs labeled with radionuclides for molecular imaging and PRRT have become a significant theranostic method targeting SSTR overexpressed NETs; besides, other SSTR positive tumors such as lung cancer,23–25 melanoma, 26 breast cancer, 27 and lymphoma 28 can also be visualized with the use of nuclear medicine techniques, so we choose the nude mice bearing HT-29 and PG tumor cells as NET model in the research.
DOTA-TATE, the most commonly used SSA in clinic, presents improved pharmacokinetics properties with fast clearance from non-tumor organs and high accumulation in targeted tumor when radiolabeled with radionuclide.29,30 In this research, we prepared the DOTA-TATE kits for 68Ga/177Lu labeling, which were easily prepared, preserved stable for a long time, and offered an effective way to achieve high radiochemical yields of M-DOTA-TATE.
For the micro-PET and micro-SPECT/CT imaging, tumor uptake of M-DOTA-TATE increased with time, and can be blocked by co-injection of excess DOTA-TATE, which is in accordance with previous study.31,32 Even though the old nude mice bears small HT-29 tumor, good tumor targeting visualization of the 177Lu-DOTA-TATE probe can be found as shown in Figure 4. For the human PET/CT imaging, first, no side effects were observed in the study post-injection of 68Ga-DOTA-TATE; moreover, the probe could detect primary (rectal cancer showed in Figure 5(c)) and metastatic lesions (liver cancer showed in Figure 5(a) and (b)), which showed high targeting specificity using 68Ga labeled SSA DOTA-TATE for diagnosis of NETs.
In summary, we synthesized 68Ga-DOTA-TATE and 177Lu-DOTA-TATE in high radiochemical yields from freeze-dried kit formulation. Both micro-PET imaging of 68Ga-DOTA-TATE and micro-SPECT/CT imaging of 177Lu-DOTA-TATE have shown clear uptake from tumor, which can be blocked by excess DOTA-TATE, supporting our hypothesis that 68Ga-DOTA-TATE and 177Lu-DOTA-TATE may have similar biodistribution within an organism, to serve for both therapeutic and diagnostic purposes of NETs. Clinical PET/CT imaging of 68Ga-DOTA-TATE in NET patients have shown that the radiotracer can detect primary and metastatic lesions.
Footnotes
Acknowledgements
F.L., H.Z., N.L., and Z.Y. designed the research; F.L., H.Z., X.H., and Q.X. conducted the experiments; F.L., H.Z., J.Y., and T.L. performed data analysis; F.L. and H.Z. wrote and revised the manuscript. F.L. and H.Z. contributed equally to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All the animal experiments were approved by the Institutional Animal Care and Use Committee of Peking University Cancer Hospital & Institute. All procedures performed in studies involving human patients were in accordance with the Ethics Committee of Peking University Cancer Hospital & Institute; the patients’ informed consents were obtained before they participated in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Major State Research Development Program of China (2016YFC0100400), National Natural Science Foundation of China (No. 81371592, No. 81401467, No. 81501519, and No. 81571705), Beijing Natural Science Foundation (No. 7154188 and No. 7162041), and Beijing Municipal Commission of Health and Family Planning (215 backbone program). Scientific Research Foundation for Returned Scholars of Beijing Municipal Human Resources and Social Security Bureau (2016) and 2017 Beijing Nova Program also supported this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
