Abstract
Epithelial carcinomas occasionally have sarcomatous components that consist primarily of spindle and cuboidal cells, which often resemble osteoblasts. Sarcomatoid carcinomas consist of similar cells. Recent studies have characterized these phenomena as a manifestation of epithelial–mesenchymal transition in carcinoma cells, but the mesenchymal phenotypes that manifest in sarcomatous cells of epithelial carcinomas are not well understood. Here, we examined the expression profiles of four osteoblastic differentiation biomarkers in the sarcomatous components of multiple carcinoma types, including five renal clear cell, four breast invasive ductal, two esophageal, one maxillary squamous cell, three larynx, three lung, one liver, and one skin sarcomatoid carcinoma. Expression was analyzed by immunohistochemistry using antibodies against cell adhesion molecule 1, a member of the IgCAM superfamily, osterix transcription factor (Osterix), cluster of differentiation 151, a transmembrane 4 superfamily member, and alkaline phosphatase. Immunostaining intensity was rated in scale 0 (negative), 0.5 (weak), and 1 (strong) for each marker, and the four scale values were summed to calculate osteoblastic scores. In all, 10 cases had a osteoblastic score ≥3, and all of these 10 cases were cell adhesion molecule 1- and Osterix-positive. Eight and five of the nine samples with a osteoblastic score <3 were negative for cell adhesion molecule 1 (
Keywords
Introduction
Epithelial carcinomas occasionally have sarcomatous components that are thought to be an important factor for clinical prognosis in kidney, esophagus, pancreatic, and liver carcinomas.1–6 In addition, these organs are occasionally affected by sarcomatoid carcinomas, which are often referred to as spindle cell variants of epithelial carcinomas. Morphological assessment by hematoxylin and eosin (H&E) staining is the standard method for pathological diagnosis of these lesions and often reveals cuboidal or spindle cells. However, H&E staining is incapable of accurately determining mesenchymal characteristics in the sarcomatous component of epithelial and sarcomatoid carcinomas. To our knowledge, few systematic trials have attempted to classify the sarcomatous and mesenchymal phenotypes of these lesions.
The focal appearance of sarcomatous cells in epithelial carcinomas can be explained by epithelial–mesenchymal transition (EMT), which is used to describe the process of an epithelial cell undergoing multiple biochemical changes to become a mesenchymal cell (non-epithelial cell). 7 Mesenchymal features include enhanced invasiveness, increased migration, elevated resistance to apoptosis, and greater extracellular matrix production.7,8 There are three EMT subtypes. The first subtype is necessary for normal morphogenesis during critical phases of embryonic development, the second subtype is associated with tissue regeneration and fibroblast activation (fibrosis), and the third subtype is related to cancer mesenchymal changes. Mixed malignant tumors, such as sarcomatoid and epithelial carcinomas, often contain a sarcomatous component. 8 Most studies on EMT have used in vitro techniques in various carcinoma cell lines.9–13 These studies have revealed a diversity of phenotypes and an increase in malignant potential through the loss of epithelial characteristics. The loss of epithelial phenotype in several carcinomas has also been confirmed through in vivo studies of human epithelial carcinomas. Mesenchymal traits in epithelial carcinomas are associated with increased invasiveness and metastasis, which results in poor prognosis.8,14 EMT can occur through multiple mechanisms, even in epithelial carcinomas that originate from the same organ tissue, and the phenotype switch from epithelial to mesenchymal seems to be irreversible. 15
A representative mesenchymal cell is the osteoblast, which differentiates from stromal cells and is essential for bone development and homeostasis. Common osteoblast markers include alkaline phosphatase (ALP), 16 cluster of differentiation 151 (CD151),17,18 and Osterix.19–21 ALP, produced mostly in the liver and in bone, reflects a variety of conditions, such as rapid bone growth and bone disease. ALP is used in clinical settings to estimate bone metabolism and osteoblast maturation. CD151 is a member of the transmembrane 4 superfamily, also known as the tetraspanin family, and has been used experimentally as a diagnostic marker for osteosarcoma.17,18 Osterix is a member of the Sp1 transcription factor family and is upregulated with cell adhesion molecule 1 (CADM1) when calvarial stromal cells differentiate into osteoblasts. CADM1 is expressed in developing mouse bones and remodeling human bones. 22 CADM1 is an intercellular adhesion molecule that belongs to the immunoglobulin superfamily and is expressed in various cell types, including lung alveolar cells, biliary epithelial cells, pancreatic islet cells, ameloblasts, neurons, mast cells, and bone cells.23–25
In this study, we evaluated 11 epithelial carcinomas with a sarcomatous component and 8 cases of sarcomatoid carcinoma. The sarcomatous/sarcomatoid lesions were analyzed through an H&E-based morphological assessment and immunohistochemistry using antibodies against the previously mentioned osteoblastic and EMT markers. To verify the histological data, we prepared protein extracts from the pathological specimens and analyzed these samples by western blotting for CADM1.
Materials and methods
Patient characteristics
All patients examined in this study underwent a standard operation that included complete dissection. The patients were not treated with irradiation or chemotherapy before surgery. Cases were selected according to a computerized search for sarcomatoid carcinoma and/or carcinoma with sarcomatous components in the surgical pathology databases of Kindai University Hospital and Izumiotsu Municipal Hospital between January 2005 and December 2014. A total of 13 males and 6 females, with a mean age of 66.15 years (range: 35–84 years), were included in this study.
The sarcomatous component of epithelial carcinomas and sarcomatoid carcinomas were diagnosed independently by two pathologists (Y.T. and A.I.) according to World Health Organization (WHO) classifications (Supplementary references 1–7), and samples with lesions large enough for histological analysis were selected. There enrolled cases included 11 epithelial carcinomas, consisting of 4 renal clear cell carcinomas, 4 breast invasive ductal cell carcinomas, 2 esophageal and 1 maxillary squamous cell carcinomas, and 8 sarcomatoid carcinomas, consisting of 3 larynx, 1 lung, and 1 skin squamous cell carcinoma, 1 lung pleomorphic carcinoma, 1 lung adenocarcinoma, and 1 hepatocellular carcinoma. Patient characteristics are shown in Table 1. All experiments were approved by the Ethics Committees of Kindai University Hospital and Izumiotsu Municipal Hospital (#25-088).
Clinicopathological features and immunohistological analysis.
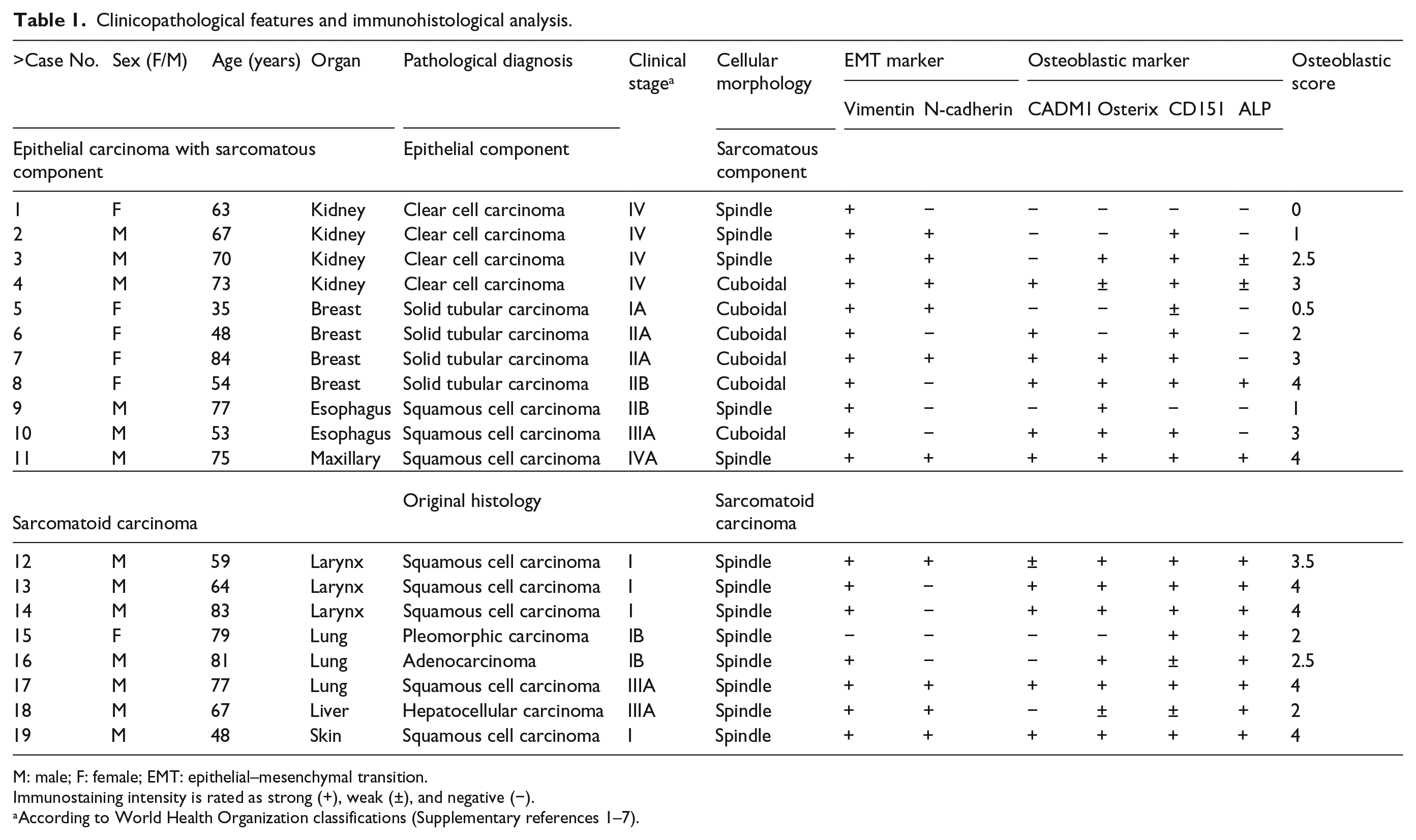
M: male; F: female; EMT: epithelial–mesenchymal transition.
Immunostaining intensity is rated as strong (+), weak (±), and negative (−).
According to World Health Organization classifications (Supplementary references 1–7).
Histological analysis
Carcinoma samples were fixed in 4% paraformaldehyde and 10% natural phosphate-buffered formalin and embedded in paraffin. H&E staining and immunohistochemistry for vimentin, N-cadherin, CADM1, CD151, Osterix, and ALP were performed as described previously. 22 Briefly, deparaffinized sections (thickness = 3 µm) were autoclaved in 0.01 M citrate buffer, blocked with bovine serum albumin (BSA), and incubated with a primary antibody. A list of antibodies used in this study can be found in Table 2. Secondary antibody staining was performed using a Histofine Simple Stain MAX-PO (R) kit (Nichirei Biosciences, Tokyo, Japan). The sections were incubated with ImmPACTTM AEC (Vector Laboratories, Burlingame, CA, USA) and counterstained with hematoxylin. All experiments were completed with adequate positive and negative controls, including omission of primary antibodies, and representative examples are shown in Supplementary Figure 1.
List of primary antibodies used.

CADM1: cell adhesion molecule 1; ALP: alkaline phosphatase.
The sarcomatous component was classified based on the appearance of osteoblastic immunohistochemical phenotypes, using a scale ranging from 0 to 4. The osteoblastic score was quantified based on the intensity of individual osteoblastic marker expression (CADM1, CD151, Osterix, and ALP) as negative (score
Western blot analysis
Paraffin-embedded tumor tissues were cut into 10-µm-thick sections. When the section contained both the sarcomatous and epithelial components, one of the components was removed by manual scratching. Protein was extracted from the section according to the method described by Rodriguez-Rigueiro et al.
26
with minor modifications.
27
Briefly, the section was deparaffinized by incubation in mineral oil at 95°C for 2 min, and the supernatant was removed by centrifugation at 11,320
Statistical analysis
Fisher’s exact test was used to analyze the association of osteoblastic score with osteoblastic marker expression and cellar morphologic changes. Statistical significance was denoted by
Results
The sarcomatous component consisted of cuboidal and spindle cells
The sarcomatous component of epithelial carcinomas and sarcomatoid carcinomas had either cuboidal or spindle cells as the major constituent. In the sarcomatous component of epithelial carcinomas (cases 1–11, Table 1), 6 out of 11 cases were mainly composed of cuboidal cells, whereas all sarcomatoid carcinomas (cases 12–19, Table 1) were composed primarily of spindle cells. Representative microphotographs of the cuboidal and spindle-type lesions are shown in Figure 1.

Histology of epithelial carcinoma sarcomatous components. Hematoxylin and eosin staining revealed that the sarcomatous component was composed of cuboidal cells or spindle cells. Representative histology for each cell type is shown. Spindle form, case 8; cuboidal form, case 11 (numbered as in Table 1). The boxed areas in the left photomicrographs are enlarged in the right column. (Original magnification: left, 100×; right, 400×; bar
Osteoblastic phenotypes in sarcomatous component
Immunohistochemistry was performed on the sarcomatous lesions. More than half of the sarcomatous components and sarcomatoid carcinomas were positive for vimentin (18/19) and N-cadherin (11/19), suggesting that EMT may be involved in these sarcomatous changes (Figure 2). Immunohistochemistry for four osteoblastic markers (CADM1, CD151, Osterix, and ALP) was rated on a scale (0, 0.5, and 1), and the osteoblastic score was calculated by summing the scale value for each marker. The osteoblastic score ranged from 0 (one case) to 4 (six cases), suggesting that sarcomatous epithelial–carcinoma cells express varying levels of osteoblastic phenotypes (Table 1). The correlation between cellular morphology (cuboidal form or spindle form) and osteoblastic score in the sarcomatous components (quantified as osteoblastic score ≥3) derived from epithelial carcinomas was investigated, but there was no statistically significant correlation (

Immunohistochemistry of epithelial carcinoma sarcomatous components. The sarcomatous components were immunohistochemically stained for cell adhesion molecule 1 (CADM1), Osterix, CD151, alkaline phosphatase (ALP), vimentin, and N-cadherin and counterstained with hematoxylin. Representative photomicrographs from two tumors are shown. Spindle form, case 8; cuboidal form, case 11 (numbered as in Table 1; original magnification: 400×).
Relation between osteoblastic score and cellular morphology.

Sample number is expressed in Arabic numerals.
Statistical significance was evaluated by Fisher’s exact test.
CADM1 reflects osteoblastic differentiation in the sarcomatous component
Each of the four osteoblastic markers was examined for its association with osteoblastic scores. CADM1 and Osterix were significantly associated with a high osteoblastic score (≥3) in the sarcomatous component (
Correlation between osteoblastic score and osteoblastic marker–stained grade.

Sample number is expressed in Arabic numerals.
Statistical significance was evaluated by Fisher’s exact test.
p<0.0001, **p

Western blot analysis of the sarcomatous and epithelial components for CADM1. Protein extracts were prepared from paraffin sections of the sarcomatous (S) and epithelial (E) components of cases 4, 8, 10m and 11 (numbered as in Table 1) and subjected to western blot analyses with an anti-CADM1 antibody (upper panel). The blots were reprobed with a β-actin antibody as the control (lower panel; P, positive control (HOS cell lysate); M, molecular size marker (precision plus protein standard marker; Bio-Rad, CA, USA); circled 1, 150 kDa; circled 2, 100 kDa).
Discussion
Sarcomatous components in epithelial carcinomas tend to be composed of cuboidal or spindle cells, which are assumed to develop through EMT.2,30 Vimentin and N-cadherin, well-known EMT markers, have been consistently detected in the sarcomatous components of epithelial carcinomas. 31 However, little is known about the major mesenchymal phenotypes of the sarcomatous component, including bone cells, chondrocytes, muscle cells, and adipocytes. Therefore, EMT-related mesenchymal characteristics are not efficient for differential diagnosis of sarcomatous components of epithelial carcinomas.
Cuboidal cells in the sarcomatous component often resemble osteoblasts in morphology.
22
Osteoblasts exhibit maturation stage-specific phenotypes over the course of development and differentiation from mesenchymal stem cells to mature osteocytes.
32
To assess whether cuboidal and spindle cells have osteoblast-like phenotypes, we analyzed immunohistochemical data for CADM1, Osterix, CD151, and ALP and expressed them as osteoblastic scores. The osteoblastic score is a parameter that we developed to quantify the degree of acquisition of osteoblastic phenotypes and the loss of epithelial characteristics in the sarcomatous component. The score varied considerably between samples. Since the four markers individually represent different or partly overlapping stages of osteoblastic differentiation/maturation (Supplementary Figure 2), the scores should reflect different stages of osteoblastic transformation of sarcomatous lesion cells. This means that individual lesions should consist of cells that are in lesion-specific stages of osteoblastic differentiation. Alternatively, the transformed cells could also aberrantly express osteoblastic markers. Most epithelial carcinoma sarcomatous components composed of cuboidal cells had an osteoblastic score ≥2 (83%; 5/6). In addition, the proportion of patients with an osteoblastic score ≥3 was higher when the sarcomatous component was composed of cuboidal cells (66%; 4/6) compared to those composed of spindle cells (20%; 1/5), although the difference was not statistically significant (
We examined which osteoblastic markers had the highest sensitivity for detecting osteoblastic phenotypes in the sarcomatous components and identified CADM1 as the best candidate because it was the marker most highly related to osteoblastic score in the lesions. CADM1 may play a role in the development of the sarcomatous component as an adhesion molecule. Osterix, the second candidate, is an Sp1-type transcription factor that plays a critical role in osteoblastic differentiation.
19
We previously reported that CADM1 and Osterix were co-upregulated during osteoblastic differentiation of mouse calvarial stromal cells.
22
Osterix may transcriptionally transactivate the
Overall, this study strongly suggests that osteoblastic phenotypic cells are present in the sarcomatous component of epithelial carcinoma and sarcomatoid carcinomas. Since these cells are considered to be “differentiated” rather than “undifferentiated,” sarcomatous components composed of these cells may be biologically more benign than components lacking any differentiation. In fact, osteosarcoma, a mesenchymal tumor arising from osteoblasts or their progenitors, has clinicopathological features consistent with this notion. Poorly differentiated osteosarcomas (i.e. conventional osteosarcomas) autonomously produce osteoid or bone and have a poor prognosis, while well-differentiated osteosarcomas have a benign prognosis.33,34 In contrast, EMT in epithelial carcinomas has been previously associated with poor clinical prognosis due to the associations among EMT, invasiveness, and metastasis.10,11 Further experiments are needed to examine whether EMT results in sarcomatous changes in epithelial carcinomas.
In conclusion, our data demonstrate that sarcomatous epithelial–carcinoma cells can exhibit varying osteoblastic phenotypes. These results could lead to new classifications of epithelial carcinoma sarcomatous components and sarcomatoid carcinomas, which should include parameters indicating the degree of osteoblastic phenotype manifestation. Future studies are needed to reveal associations between osteoblastic phenotypic manifestations with biological malignancy and clinical prognosis in sarcomatous epithelial carcinomas.
Footnotes
Acknowledgements
The authors would like to thank Dr Mikihiro Uenishi (Izumiotsu Municipal Hospital) for surgical material preparation. Y.T. and T.M. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Technology-Supported Program for the Strategic Research Foundation at Private Universities 2015-17 (to Akihiko Ito).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
