Abstract
The cancer stem cell (CSC) hypothesis has gained significant recognition in describing tumorigenesis. Identification of the factors critical to development of breast cancer stem cells (BCSCs) may provide insight into the improvement of effective therapies against breast cancer. In this study, we aim to investigate the biological function of SLC34A2 in affecting the stem cell-like phenotypes in BCSCs and its underlying mechanisms. We demonstrated that CD147+ cells from breast cancer tissue samples and cell lines possessed BCSC-like features, including the ability of self-renewal in vitro, differentiation, and tumorigenic potential in vivo. Flow cytometry analysis showed the presence of a variable fraction of CD147+ cells in 9 of 10 tumor samples. Significantly, SLC34A2 expression in CD147+ BCSCs was enhanced compared with that in differentiated adherent progeny of CD147+ BCSCs and adherently cultured cell line cells. In breast cancer patient cohorts, SLC34A2 expression was found increased in 9 of 10 tumor samples. By using lentiviral-based approach, si-SLC34A2-transduced CD147+ BCSCs showed decreased ability of sphere formation, cell viability in vitro, and tumorigenicity in vivo, which suggested the essential role of SLC34A2 in CD147+ BCSCs. Furthermore, PI3K/AKT pathway and SOX2 were found necessary to maintain the stemness of CD147+ BCSCs by using LY294002 or lentiviral-si-SOX2. Finally, we indicated that SLC34A2 could regulate SOX2 to maintain the stem cell–like features in CD147+ BCSCs through PI3K/AKT pathway. Therefore, our report identifies a novel role of SLC34A2 in BCSCs’ state regulation and establishes a rationale for targeting the SLC34A2/PI3K/AKT/SOX2 signaling pathway for breast cancer therapy.
Introduction
Breast cancer is the most common malignancy in women. Currently, 115 million patients have been diagnosed with breast cancer, resulting in 410,000 deaths annually worldwide. 1 Although current anti-tumor therapies have greatly improved the 5-year survival rate of breast cancer patients, recurrence and long-distance metastasis of the primary breast cancer are often incurable and are the leading causes of mortality in cancer patients. 2 Thus, it underscores the urgent need for refined investigation on molecular mechanisms that provide novel targets for advancing anti-breast-cancer therapy.
Many solid tumors, including breast cancer, exhibit a functional hierarchy of cells of which only a small subpopulation of stem-like cells, cancer stem cells (CSCs), or tumor-initiating cells (TICs), give rise to the differentiated cells that make the bulk of the tumor. 3 This is also popularly known as the CSCs model, which proposes that CSCs are the unique source of all tumor cells at earlier stages of tumorigenesis and are responsible for tumor propagation and relapse at advanced stages. 4 Meanwhile, there are a number of reports showing that CSCs are more resistant to conventional cancer therapies, therefore, placing these cells at the root of tumor recurrence and metastases. 5 Current breast cancer therapies frequently fail to eradicate tumors because they either fail to effectively target CSCs or CSCs are resistant to the chemotherapeutic agents. In essence, breast cancer treatment is hindered by the lack of understanding main regulators and mechanisms that are critical to the stem cell–like state of breast cancer stem cells (BCSCs).
SLC34A2 is a member of the solute carrier gene family, which encodes the type II Na/Pi co-transporter, NaPi2b. 6 NaPi2b, a multi-transmembrane sodium-dependent phosphate transporter, which is responsible for transcellular inorganic phosphate absorption, maintenance of phosphate homeostasis, and tumorigenesis. 7 The expression level of SLC34A2 in breast cancer patient cohort is an issue of controversy,8,9 although in ovarian cancer 10 and non-small cell lung cancer 11 an enhanced expression of SLC34A2 has been found. SLC34A2 was found necessary for BCSC-like cells to induce chemotherapeutic resistance to doxorubicin. 9 However, it is unclear whether SLC34A2 plays an important role in affecting the stem cell–like phenotypes in BCSCs.
On the other hand, there are some controversies about the choice of cell-surface markers to sort BCSCs. Although CD44+ CD24−, EpCAM, and so on are supposed to be the markers to select BCSCs, in an attempt to associate CSC content and prognosis, the presence of CD44+ CD24− cells in clinical samples of breast cancer is not associated with aggressive tumor behavior and poor clinical outcome, suggesting that it could not be the surface-marker to sort BCSCs. 12 CD147 was reported to contribute to therapy resistance, angiogenesis, inflammatory signaling, cytoskeletal remodeling, and migration/invasion in different types of cancers.13–15 However, whether CD147 is the cell-surface marker that can be used to identify BCSCs is difficult to pinpoint as yet.
Here, we aim to investigate the biological function of SLC34A2 in affecting the stem cell–like phenotypes of BCSCs and its underlying mechanisms. We demonstrated that CD147+ cells from breast cancer tissue samples and cell lines possessed BCSC-like features, including the ability of self-renewal in vitro, differentiation, and tumorigenic potential in vivo. Flow cytometry analysis showed the presence of a variable fraction of CD147+ cells in 9 of 10 tumor samples. Significantly, SLC34A2 expression in CD147+ BCSCs was enhanced compared with that in differentiated adherent progeny of CD147+ BCSCs and adherently cultured cell line cells. In breast cancer patient cohorts, SLC34A2 expression was found increased in 9 of 10 tumor samples. By using lentiviral-based approach, si-SLC34A2-transduced CD147+ BCSCs showed decreased ability of sphere formation, cell viability in vitro, and tumorigenicity in vivo, which suggested the essential role of SLC34A2 in CD147+ BCSCs. Furthermore, PI3K/AKT pathway and SOX2 were found necessary to maintain the stemness of CD147+ BCSCs by using LY294002 or lentiviral-si-SOX2. Finally, we indicated that SLC34A2 could regulate SOX2 to maintain the stem cell–like features in CD147+ BCSCs through PI3K/AKT pathway.
Methods and materials
Sample collection
In all, 10 patients with primary breast cancer, who did not show other tumor occurrences, in Department of Vascular and Endocrine Surgery, Department of Orthopaedic, and Institute of Clinical Laboratory Medicine, Xijing Hospital, Fourth Military Medical University, were enrolled into this study from January 2009 to June 2015. A consecutive series of informed consent for the core-needle biopsy and experimental use of tumor samples was obtained from all patients, following a protocol approved by the Ethics Committee of Xijing Hospital, Fourth Military Medical University. These patients were not treated with chemotherapy before surgery. All patients were diagnosed with primary breast cancer and did not show other tumor occurrences. All tissue samples were flash-frozen in liquid nitrogen immediately after collection and stored at −80°C until use.
Sphere formation and propagation
Single-cell suspensions were obtained from breast cancer primary tissue samples and breast cancer cell lines (MCF7 and T47D, respectively). Breast cancer primary tissue samples were shipped to laboratory in cold RPMI-1640 medium with penicillin/streptomycin within 1 h of removal from patients. Surgical specimens were washed three times with cold phosphate-buffered saline (PBS) supplemented with high doses of penicillin/streptomycin, chopped with a sterile blade, and incubated in 1 mg/mL collagenase II (Sigma–Aldrich, USA) for 30 min at 37°C. After incubation, the suspensions were repeatedly triturated, and passed through 70 µm cell strainers (BD Falcon, USA). Recovered cells were cultured at clonal density in serum-free medium containing N2 (100×) supplementary (Gibco-Invitrogen, USA), 0.4% bovine serum albumin (BSA; Gibco-Invitrogen, USA), 100 U/mL penicillin, 1 mg/mL streptomycin in Dulbecco’s modified Eagle medium (DMEM)–F12 medium (Gibco-Invitrogen) and supplemented with 20 mg/mL epidermal growth factor (EGF), 20 mg/mL insulin-like growth factor (IGF) and 20 mg/mL basic fibroblast growth factor (bFGF). Non-treated flasks were used to reduce cell adherence and support growth as undifferentiated tumor spheres.
For breast cancer cell lines (MCF7 and T47D), after enzymatic digestion, all the cells were washed with PBS several times to remove the serum. Recovered cells were cultured at clonal density in serum-free medium as above. Single cells were cultured in serum-free DMEM/F12 medium with supplements as discussed above.
The medium of all the spheres was replaced with fresh growth factors twice a week until cells started to form floating aggregates. Cultures were expanded by enzymatic digestion of spheres with TrypLE Express (Gibco-Invitrogen, USA), followed by re-plating of both single cells in complete fresh medium. All cells were cultured at 37°C in a 5% CO2 humidified incubator.
Cell growth assay
Adherent cell lines (MCF7 and T47D) and sphere cells were digested to single cells and seeded into 96-well plates at a density of 2 × 103 cells/well in 200 µL of medium containing DMEM/F12/10% fetal bovine serum (FBS) on day 0, and cell growth was measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay daily until day 7. Adherent breast cancer cell line cells served as controls. At the indicated time points, 20 µL MTT (Sigma-Aldrich, USA) solution (5 mg/mL) was added and cells were further incubated at 37°C for 4 h. After the removal of culture medium, 150 µL dimethyl sulfoxide (DMSO) (Sigma–Aldrich) was added in each well. Absorbance (A) was measured spectrophotometrically in a microplate reader (Bio-Rad, USA) at a wavelength of 490 nm.
Xenotransplantation
Mice were maintained at the Animal Core Facility at Xijing Hospital, Fourth Military Medical University under specific pathogen-free (SPF) condition. All studies on mice were conducted in accordance with the National Institutes of Health “Guide for the Care and Use of Laboratory Animals” and followed a protocol approved by the Ethics Committee of Xijing Hospital, Fourth Military Medical University. A concentration of 104 sphere cells was injected subcutaneously into the left flank of nude mice. Each group contained 3–4 mice. In vivo serial transplantation assay, a total of 104 CD147+ cells or CD147− cells, purified from MCF7 and T47D, were injected subcutaneously into nude mice. Derived tumor xenografts were dissociated to single-cell suspension and then serially re-injected in mice (104 cells), generating secondary and then tertiary tumors. The tumor volume was calculated by the formula V (mm3) = 0.52 × length (mm) × width2 (mm2). About 3–4 weeks after injection, the animals were sacrificed and flank tumors were removed.
Flow cytometry analysis and fluorescence activated cell sorting
Differentiation of stem cell progeny
To obtain differentiated cells, spheres were cultured in DMEM/F12/10% FBS medium instead of serum-free medium in tissue culture–treated flasks, to allow cell attachment and differentiation.16–18 The loss of stem cell markers was evaluated by flow cytometry as indicated above.
Quantitative real-time reverse transcription polymerase chain reaction analysis
Total RNA was isolated by using Trizol reagent (Invitrogen, USA) according to the manufacturer’s protocol. Total RNA in 10 primary breast cancer samples, along with their respective adjacent breast tissues, was extracted using Trizol reagent (Invitrogen) according to the manufacturer’s protocol at low temperature. Complementary DNA (cDNA) was synthesized with reverse transcriptase using PrimeScript RT reagent Kit Perfect Real Time (TaKaRa, China). For genes expression analysis, quantitative real-time polymerase chain reaction (PCR) analysis was done using the iCycler iQ5 real-time PCR Detection system (Bio-Rad) with SYBR Green Reagents (Bio-Rad). β-actin was amplified as an internal control. Comparative gene expression analysis was done using the 2−ΔΔCt method with normalization to the level of the internal control, that is, β-actin. Primer sequences of SLC34A2 are described in Table S1, supplementary information.
In vitro migration and invasion assays
In vitro cell migration assays were performed using millicell chambers (Millipore, USA). After being treated with LY294002, BCSCs treated with LY294002 and the same number of BCSCs treated without LY294002 were re-suspended in serum-free medium, and 200 µl of the cell suspension (7 × 104 cells) was added to the upper chamber. The cells in the chamber were then cultivated in 5% CO2 at 37°C for 24 h. Then, the cells in the upper chamber were removed, and the attached cells in the lower section were stained with 0.1% crystal violet. The invasion assay was the same as the migration assay except that matrigel (Clontech, USA) was used in the transwell chambers (Millipore), and the cell suspension for the upper chambers had a concentration of 2.5 × 105 cells. The migration and invasion rates were quantified by counting the migration cells in six random fields under a light microscope.
Lentiviral transduction
Dissociated MCF7 and T47D CD147+ spheres were spin-infected with 1 mL of NC-si-RNA (Shanghai GenePharma Co. Ltd, China) or
Western blot
The different treated cells were washed with ice-cold PBS and then lysed using protein lysate with radioimmunoprecipitation assay (RIPA) buffer (Pierce, USA). After centrifugation at 12,000 rpm for 10 min at 4°C, the protein concentration was measured by bicinchoninic acid (BCA) protein assay kit (Pierce). Then, all proteins were resolved on a 10% sodium dodecyl sulfate (SDS) denatured polyacrylamide gel and were then transferred onto a polyvinylidene difluoride (PVDF) membrane (Millipore). Membranes were incubated with blocking buffer for 60 min at room temperature and were then incubated with a specific primary antibody against SLC34A2 (catalog No.: 66445; Cell Signal Technology, USA), phospho-AKT (Ser473)(catalog No.: 4070; Cell Signal Technology), PI3K (catalog No.: 4249; Cell Signal Technology), β-catenin (catalog No.: 8480; Cell Signal Technology), Notch-1 (catalog No.: 3608; Cell Signal Technology), AKT (catalog No.: 4685; Cell Signal Technology), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (catalog No.: 5174; Cell Signal Technology) with Blotto overnight at 4°C. The membranes were washed and incubated with a horseradish peroxidase (HRP)-conjugated secondary antibody. Blots were then exposed to secondary antibodies and visualized by an enhanced chemiluminescence (ECL) blotting analysis system (GE Healthcare Life Sciences, China).
Statistical analysis
All the assay results represent the arithmetic mean ± SD standard error of triplicate determinations of at least three independent experiments done under the same conditions. The significant difference of the experimental results was calculated using one-way analysis of variance (ANOVA) and an unpaired Student’s t-test with SPSS13.0 software. Significances are as follows: ***, p < 0.001; **, p < 0.01; *, p < 0.05.
Results
CD147+ spheres from breast cancer primary tissue samples and cell lines display stem cell-like features
To characterize the surface phenotype of BCSC-like cells in breast cancer primary tissue samples and cell lines (MCF7 and T47D), we tested whether CD147 could be the surface marker for sorting BCSC-like cells by using flow cytometry analysis. We found the presence of a variable fraction of CD147+ cell populations in 9 out of 10 tumor samples, varying from 0.1% to a maximum of 10.3% (Figure 1(a), Table S2, Supplementary Information). The rates of CD147+ cells in MCF7 and T47D cell lines are 2.5% and 4.1%, respectively (Figure 1(a)). Because the definition of CSCs relies mainly on stem cell–like properties, we further fractionated CD147+ and CD147− cells by FACS to corroborate the capacity of CD147+ spheres for self-renewal, differentiation in vitro, and tumorigenic ability in vivo. We found that CD147− cells remained viable in non-serum culture medium, but only CD147+ cells were able to form compact self-renewing spheres (Figure 1(b)). Additionally, we found that the percentage of CD147+ cells decreased gradually when CD147+ spheres were cultured in a serum-containing medium (Figure 1(c)). To corroborate the in vivo tumorigenic ability, nude mice 4- to 5-weeks old were subcutaneously injected with 104 cells (CD147+ and CD147− cells, respectively). We found that tumorigenic potential in CD147+ group increased compared with that in CD147− group (Figure 1(d)). Then, to test the long-term tumorigenic potential of CD147+ cells, we performed serial transplantation assays of cells isolated from MCF7 and T47D tumor xenografts originally derived from CD147+ and CD147− cells injection in mice. Cells derived from CD147+ tumors were able to generate tumors in primary, secondary, and tertiary transplantation, whereas cells from CD147− tumors lost tumorigenic potential during serial transplantations (Figure 1(d)). Thus, all these data demonstrated that CD147+ spheres from breast cancer primary tissue samples and cell lines possessed BCSC-like features, including the ability of self-renewal, differentiating in vitro, and tumorigenic potential in vivo.

CD147+ spheres from breast cancer primary tissue samples and cell lines display stem cell–like features. (a) Flow cytometer analysis of CD147 expression in fresh breast cancer primary tissue samples and cell lines (MCF7 and T47D). (b) Phase-contrast images of CD147+ sphere cells and CD147− cells. Scale bar, 100 µm. (c) The percentage of sphere cells, carrying CD147 antigen with and without serum in the culture medium after 3 or 6 days, respectively, were detected by flow cytometer. (d) In vivo serial transplantation assay. A total of 104 CD147+ cells and CD147−cells, purified from MCF7 and T47D, were injected subcutaneously into nude mice. Derived tumor xenografts were dissociated to single-cell suspension and then serially re-injected in mice (104 cells), generating secondary and then tertiary tumors. Tumor growth curves of primary and tertiary tumors are shown.
CD147+ BCSCs express high level of SLC34A2
To obtain expression profile of SLC34A2 in CD147+ BCSCs, we compared SLC34A2 expression in CD147+ BCSCs with that in differentiated adherent progeny of CD147+ BCSCs and adherently cultured cell line cells. We found that the expression of SLC34A2 in CD147+ BCSCs (sph) was enhanced at both mRNA and protein level, compared with that in differentiated adherent progeny of CD147+ BCSCs (dif) and adherently cultured cell line cells (adh) (Figure 2(a) and (b)). These results suggested that SLC34A2 might play an essential role in maintaining the stem cell–like features in CD147+ BCSCs. In patient cohort, SLC34A2 expression was found increased in 9 of 10 tumor samples, which suggested a potential role of SLC34A2 in breast cancer prognosis (Table S2, Supplementary Information). SLC34A2 expression was, however, not significantly associated with age, nor advanced disease stage (Table S2, Supplementary Information).

CD147+ spheres express high level of SLC34A2 and SLC34A2 promotes the ability of sphere formation and cell viability. (a) Relative expression of SLC34A2 in CD147+ BCSCs, their differentiated adherent progeny, and adherent cell line cells was examined by qPCR. Columns, mean of three individual experiments; SD, **p < 0.01. (b) Relative expression of SLC34A2 in CD147+ BCSCs (sph), their differentiated adherent progeny (dif), and adherent cell line cells (adh) was examined by western-blot. (c) qPCR was used for selection of si-SLC34A2 stable expressed cell strains. Columns, mean of three individual experiments. (d) Phase-contrast images of CD147+ BCSCs with lentiviral-si-SLC34A2 knockdown (si-SLC34A2), lentiviral-si-NC knockdown (si-NC), and CD147+ BCSCs without treatment (Non-T). Scale bar, 100 µm. (e) The viability of CD147+ BCSCs and CD147+ BCSCs transduced with si-SLC34A2 or empty vector (si-NC) within 7 days was measured by MTT. Columns, mean of three individual experiments; SD, **p < 0.01.
SLC34A2 contributes to maintaining the stemness in CD147+ BCSCs
To further determine the biological function of SLC34A2 in CD147+ BCSCs, a lentiviral-based approach was used to knockdown SLC34A2 in CD147+ BCSCs with si-RNAs. The knockdown efficiency was confirmed by Quantitative real-time reverse transcription polymerase chain reaction (qPCR; Figure 2(c)). si-SLC34A2-transduced CD147+ BCSCs showed decreased ability of sphere formation in serum-free cultivation compared with non-treated CD147+ BCSCs (Non-T) or CD147+ BCSCs transduced with si-NC (Figure 2(d)). Meanwhile, we found that the cell viability in si-SLC34A2-transduced CD147+ BCSCs (si-SLC34A2-BCSCs) decreased compared with that in non-treated CD147+ BCSCs (BCSCs) and CD147+ BCSCs transduced with si-NC (si-NC-BCSCs) (Figure 2(e)). Then, we implanted different group cells, including si-SLC34A2-BCSCs, si-NC-BCSCs, and BCSCs, into the flank of nude mice, and the tumorigenicity of si-SLC34A2-BCSCs was significantly reduced in comparison with other groups (Figure 3(a)). Furthermore, the expression of stem cell–associated genes, including OCT4, SOX2, and Bmi-1, reduced in si-SLC34A2-BCSCs compared with those in si-NC-BCSCs (Figure 3(b)). However, the expression of Bmi-1 was almost unchanged at protein level (Figure 4(a)). Additionally, the expression of key proteins in Notch signaling pathway in si-SLC34A2-BCSCs was almost unchanged (Figure 3(b)). The expression of key proteins in Wnt/β-catenin, PI3K/AKT, and STAT3 signaling pathway in si-SLC34A2-BCSCs reduced dramatically compared with that in si-NC-BCSCs (Figure 3(b)). Nevertheless, the expression of β-catenin and p-STAT3 was almost unchanged at protein level, which was determined using western-blot (Figure 4(a)). Thus, these results implied that SLC34A2 contributed to maintaining stem cell–like feature in CD147+ BCSCs by regulating stem cell–associated genes via PI3K/AKT cell signaling pathway.

High level of SLC34A2 promotes tumorigenicity and the expression of stem cell–associated genes in CD147+ BCSCs. (a) The potential of tumor initiation of CD147+ BCSCs and CD147+ BCSCs transduced with si-SLC34A2 (si-SLC34A2-BCSCs) or empty vector (si-NC-BCSCs) were measured by subcutaneous injection. Representative tumor growth curves of xenografts derived from different cell subpopulations were determined. Columns, mean of three individual experiments; SD, **p < 0.01. (b) qPCR analysis was performed to determine the stem cell–associated gene expression in CD147+ BCSCs transduced with si-SLC34A2 (si-SLC34A2-BCSCs) or empty vector (si-NC-BCSCs). Columns, mean of three individual experiments; SD,**p < 0.01; SD,*p < 0.05.
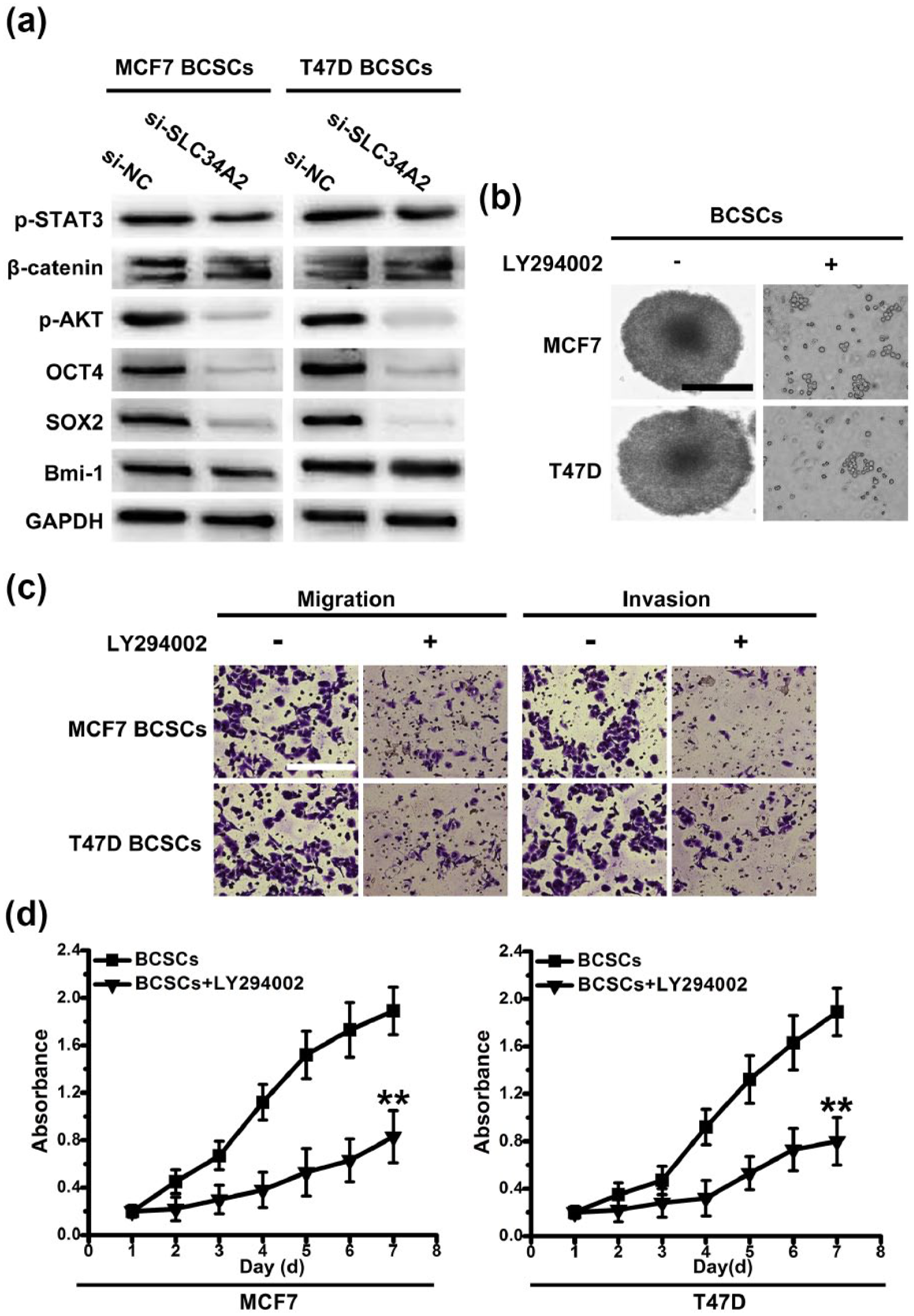
PI3K/AKT cell signaling pathway contributes to the stemness maintenance in CD147+ BCSCs. (a) Western blot analysis was performed on stem cell–associated genes expression and key proteins of stem cell–associated signaling pathways expression. (b) With or without the presence of LY294002, phase-contrast images of CD147+ BCSCs. Scale bar, 100 µm. (c) Transwell assays without matrigel were used to detect the migration activity of CD147+ BCSCs with or without the presence of LY294002. Transwell assays with matrigel were performed to detect the invasion activity of CD147+ BCSCs with or without the presence of LY294002. Scale bar, 50 µm. (d) The viability of CD147+ BCSCs and CD147+ BCSCs with or without the presence of LY294002 within 7 days was measured by MTT.
SLC34A2 regulates SOX2 through PI3K/AKT cell signal pathways to maintain the stemness of CD147+ BCSCs
To gain a deeper understanding of the molecular basis for CD147+ BCSCs state maintenance, we furthered our study in relative cell signaling pathways and stem cell–associated genes. As it shown in Figure 4(b), in the presence of LY294002 (the inhibitor of PI3K/AKT signaling pathway), the ability of sphere formation of CD147+ BCSCs decreased. Similarly, the potential of migration and invasion also reduced with the presence of LY294002 (Figure 4(c)). We found that cell viability of CD147+ BCSCs decreased in the presence of LY294002 (Figure 4(d)). These data implied that PI3K/AKT signaling pathway might have an important role in maintaining the stem cell–like phenotypes, including sphere formation, the ability of self-renewal, and migration and invasion, in CD147+ BCSCs.
Next, we determined the change of OCT4 and SOX2 expression in the presence of LY294002. We found that the expression of SOX2 reduced, while the expression of OCT4 remained unchanged (Figure 5(a)). It implied that SOX2 could be the downstream target of PI3K/AKT and could affect the state of CD147+ BCSCs. To determine whether SOX2 could be exploited by SLC34A2 to maintain the stemness of CD147+ BCSCs, we knocked SOX2 down in CD147+ BCSCs with lentiviral-based si-RNAs. Stable transduction with a lentiviral vector containing si-SOX2 in CD147+ BCSCs was done. We found that CD147+ BCSCs transduced with si-SOX2 showed lower ability of sphere formation. Moreover, tumorigenic potential was also significantly reduced upon knockdown of SOX2, which suggested that tumorigenic potential of CD147+ BCSCs was dependent on SOX2 (Figure 5(d)). These data implied that SLC34A2 could regulate SOX2 to maintain the stemness of CD147+ BCSCs through PI3K/AKT pathway.

SOX2 is necessary for maintaining the stemness of CD147+ BCSCs. (a) Relative expression of SOX2 and OCT4 in CD147+ BCSCs with or without the presence of LY294002 were examined by qPCR. Columns, mean of three individual experiments; SD, **p < 0.01. (b) qPCR was used for selection of si-SOX2 stable expressed cell strains. Columns, mean of three individual experiments. (c) Phase-contrast images of CD147+ BCSCs transduced with lentiviral-si-SOX2 knockdown (si-SOX2) and lentiviral-si-NC knockdown (si-NC). Scale bar, 100 µm. (d) The potential of tumor initiation of CD147+ BCSCs and CD147+ BCSCs transduced with si-SOX2 or empty vector (si-NC) by subcutaneous injection, and representative tumor growth curves of xenografts derived from different cell subpopulations. Columns, mean of three individual experiments; SD, **p < 0.01.
To test the hypothesis, we characterized the relationship among SLC34A2, PI3K/AKT pathway, and SOX2. We found that the expression of SLC34A2, p-AKT, and SOX2 in CD147+ BCSCs reduced upon knockdown of SLC34A2 with lentiviral-si-SLC34A2, which suggested that both PI3K/AKT pathway and SOX2 were downstream targets of SLC34A2 (Figure 6). With the presence of LY294002, the expression of p-AKT and SOX2 in CD147+ BCSCs was downregulated, while the expression of PI3K and AKT remained unchanged (Figure 6). Additionally, the expression of SOX2 in CD147+ BCSCs reduced upon knockdown of SOX2, whereas the expression of SLC34A2, p-AKT, PI3K, and AKT remained unchanged, which implied that SOX2 was a downstream target of PI3K/AKT pathway (Figure 6). In the presence of LY294002, CD147+ BCSCs transduced with si-SLC34A2 showed reduced expression of SLC34A2, p-AKT, and SOX2, whereas PI3K and AKT expression remained unchanged (Figure 6). Taken together, these results suggested that SLC34A2 regulated SOX2 to maintain the stem cell–like phenotypes in CD147+ BCSCs through PI3K/AKT pathway.

SLC34A2 regulates Sox2 through PI3K/AKT cell signaling pathways to maintain the stemness of CD147+ BCSCs. With or without the presence of LY294002, western-blot analysis was performed to determine the expression of SLC34A2, PI3K, AKT, p-AKT, and SOX2 in CD147+ BCSCs and CD147+ BCSCs transduced with si-SLC34A2 or si-SOX2. GAPDH was used as a normalization control.
Discussion
It has been approximately 150 years since the idea of CSCs was first proposed. During this time, advances in stem cell biology have redefined the CSC hypothesis. This hypothesis now centers on the concept that a small fraction of tumor cells retain a self-renewing capacity that drives the tumorigenic potential. The identification of distinct phenotypic and molecular signature associated with tumorigenicity of BCSCs would be instrumental for developing therapeutic targeting strategies in breast cancer. Our work sheds new light on the nature of the BCSCs state and the role of SLC34A2 in maintaining stem cell–like features. We provide further insight into the regulatory mechanisms that govern the stem cell niche, and its effects on SOX2 expression in BCSCs in this study.
We first confirmed the presence of CSC-like cells by identifying CD147 in breast cancer primary tissue samples and breast cancer cell lines (MCF7 and T47D). CD147 (extra-cellular matrix metalloproteinase inducer (EMMPRIN); basigin) is a variably glycosylated transmembrane protein that is involved in both physiological and pathological cellular processes.6,14,15 CD147 is associated with an invasive phenotype in various types of cancers, including breast cancer.19,20 CD147 were reported to contribute to therapy resistance, angiogenesis, inflammatory signaling, cytoskeletal remodeling and migration/invasion.14,21 Meanwhile, Yang et al., 22 suggested that CD147 could facilitate the malignant proliferation of breast cancer cell by regulating cell cycle distribution and might be a molecular target for breast cancer gene therapy. However, no studies had revealed whether CD147 could be the surface marker to sort BCSCs. Here, in contrast to the CD147− cells, CD147+ spheres possessed CSC phenotypes, including high ability of sphere formation in vitro, high tumorigenic potential in vivo, differentiation in serum, and overexpression of OCT4, SOX2, and Bmi-1 pluripotency factors. We found that the percentage of CD147+ cells decreased gradually when CD147+ spheres were cultured in a serum-containing medium, which revealed that CD147+ cells generated large and differentiated cells closely resembling the main cellular population of the original tumor under appropriate conditions. Additionally, CD147+ cells possessed the long-term tumorigenic potential, which was determined by using serial transplantation assays of cells isolated from tumor xenografts originally derived from CD147+ cells injection in mice, indicating the existence of a precise hierarchical model for the formation of lung cancer tissue, based on the generation of a vast cell progeny by a small number of self-renewing undifferentiated cells. It is likely that CD147+ cells comprise two populations of cells with similar phenotype, but different potential: a tumorigenic population of stem-like cells able to self-renew and a non-tumorigenic population of progenitor/precursor cells.
Additionally, we found the presence of a variable fraction of CD147+ cell populations in 9 out of 10 tumor samples. Thus, we concluded that CD147 could be a marker to sort BCSCs both in breast cancer cell lines and freshly resected breast cancer specimens. Moreover, CD147 was reported to be upregulated in breast cancer and has been associated with tumor progression, 23 but little is known about its regulatory mechanisms, which needs to be focused on in further studies.
Importantly, we found that when comparing SLC34A2 expression in CD147+ BCSCs with that in differentiated adherent progeny of CD147+ BCSCs and adherently cultured cells, the expression of SLC34A2 in CD147+ BCSCs was enhanced, suggesting that SLC34A2 might play an essential role in maintaining stem cell–like feature in CD147+ BCSCs. In the patient cohort, SLC34A2 expression was found increased in 9 of 10 tumor samples. SLC34A2 expression was, however, not significantly associated with age, or advanced disease stage. The prognosis value of SLC34A2 in breast cancer needs to be reproduced across a large number of samples in further studies. Due to the short follow-up period after the fresh clinical specimens were collected, the association between SLC34A2 expression and disease-free survival or overall survival could not be calculated.
Furthermore, by using lentiviral-based approach, si-SLC34A2-transduced CD147+ BCSCs showed decreased ability of sphere formation in serum-free cultivation, reduced cell viability in vitro, and declined tumorigenicity in vivo. Additionally, the expression of p-AKT (the key protein of PI3K/AKT cell signaling pathway) and pluripotency factor SOX2 and OCT4 were found downregulated in si-SLC34A2-transduced CD147+ BCSCs. These results suggested that SLC34A2 contributed to maintaining stemness of CD147+ BCSCs by regulating stem cell–associated genes via PI3K/AKT cell signaling pathway.
Next, we determined the role of PI3K/AKT pathway in maintaining the stemness of CD147+ BCSCs by using LY294002. The ability of sphere formation of CD147+ BCSCs, the potential of migration and invasion, and the cell viability of CD147+ BCSCs were all decreased in the presence of LY294002. We found that the expression of SOX2 reduced in the presence of LY294002, while the expression of OCT4 remained unchanged. It implied that SOX2 could be the downstream target of PI3K/AKT that affected the state of CD147+ BCSCs. Transcriptional regulatory networks consist of functional interactions between regulatory genes and a much larger set of downstream target genes. In the context of cancer, SOX2 overexpression has been demonstrated in ovarian, lung, liver, and breast carcinomas and is a predictor of increased malignancy and poor prognostic outcome.24,25 However, it is unknown whether SOX2 plays a role in regulating BCSCs. To further characterize the underlying mechanisms by which SLC34A2 regulated the stemness of CD147+ BCSCs, we used lentiviral-si-SOX2 to test the hypothesis above. When SOX2 was knocked down, CD147+ BCSCs transduced with si-SOX2 showed lower ability of sphere formation in vitro and tumorigenic potential in vivo. Finally, we characterized the relationship among SLC34A2, PI3K/AKT pathway, and SOX2 by western-blot. Taken together, these results indicated that LC34A2 could regulate SOX2 to maintain the stem cell–like features in CD147+ BCSCs through PI3K/AKT pathway. However, how these factors react with each other, directly or indirectly, needs to be focused on in further studies.
In conclusion, we demonstrated that CD147+ cells from breast cancer tissue samples and cell lines possessed BCSC-like features, including the ability of self-renewal in vitro, differentiation, and tumorigenic potential in vivo. Flow cytometry analysis showed the presence of a variable fraction of CD147+ cells in 9 of 10 tumor samples. Significantly, SLC34A2 expression in CD147+ BCSCs enhanced compared with that in differentiated adherent progeny of CD147+ BCSCs and adherently cultured cell line cells. In breast cancer patient cohorts, SLC34A2 expression was found increased in 9 of 10 tumor samples. By using lentiviral-based approach, si-SLC34A2-transduced CD147+ BCSCs showed decreased ability of sphere formation, cell viability in vitro, and tumorigenicity in vivo, which suggested the essential role of SLC34A2 in CD147+ BCSCs. Furthermore, PI3K/AKT pathway and SOX2 were found necessary to maintain the stemness of CD147+ BCSCs by using LY294002 or lentiviral-si-SOX2. Finally, we indicated that SLC34A2 could regulate SOX2 to maintain the stem cell–like features in CD147+ BCSCs through PI3K/AKT pathway. Therefore, our report identifies a novel role for SLC34A2 in BCSCs state regulation and establishes a rationale for targeting the SLC34A2/PI3K/AKT/SOX2 signaling pathway for breast cancer therapy.
Footnotes
Acknowledgements
Yonggang Lv, Ting Wang, Jing Fan, and Zhenzhen Zhang contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Fund (81272899, 81172510, 81470120, 81502546 and 81672593); Shaanxi Fund 2013K12-03-03, 2014JM4087 and Xi’an Found SF1323(3); and Discipline Booster Plan of Xi Jing Hospital XJZT12Z07. The funding agencies had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
