Abstract
Receptor for advanced glycation end products and glyoxalase I metabolizing advanced glycation end product precursors may play important role in the pathogenesis and progression of cancer. Potential relation between soluble forms of receptor for advanced glycation end products (sRAGE), receptor for advanced glycation end products, glyoxalase I polymorphisms, and long-term outcome (median follow-up of 10.3 years) was studied in 116 patients with breast cancer. Gly82Ser and 2184 A/G RAGE polymorphisms were related to the mortality due to the breast cancer and -419 A/C glyoxalase I polymorphism was related to the overall mortality of the patients suggesting their role not only in the risk of breast cancer but also in the outcome of patients with breast cancer.
Keywords
Introduction
Receptor for advanced glycation end products (RAGE), member of the immunoglobulin superfamily, is normally involved in the regulation of tissue regeneration and resolution of inflammation; but under pathological condition, activation of RAGE by some of its many ligands (not only advanced glycation end products, but also, for example, HMGB1 or S100 proteins) may induce diminished apoptosis, enhanced autophagy, and cell necrosis and thus contribute to the malignant transformation, cancer progression, and metastasis.1–5 Soluble forms of RAGE (sRAGE) may bind natural RAGE ligands and counteract some of the RAGE-mediated effects. Activation of RAGE was demonstrated in different types of cancer (including breast cancer), 6 and tissue expression of RAGE and serum levels of soluble RAGE were suggested as putative biomarkers of cancer. Strategies aimed at interfering with RAGE signaling might be promising in terms of their anticancer potential. Glyoxalase I is an enzyme involved in the degradation of AGE precursors metabolizing glyoxal and methylglyoxal. 7
In our recent paper, 8 we demonstrated that the natural inhibitor of RAGE activation soluble RAGE (sRAGE) was decreased in patients with breast cancer compared to healthy controls, and its serum levels were influenced genetically (Gly82Ser and 2184 polymorphisms of the RAGE gene). Moreover, patients with better outcome (low grade and positive estrogen receptors) had higher sRAGE levels. Importantly, patients who died during relatively short follow-up (median 3.6 years) had lower presenting sRAGE compared to patients who were at the end of this follow-up in clinical remission.
We were also able to show that glyoxalase I Glu111Ala polymorphism was related to the absence of estrogen receptors and more advanced stages of breast cancer and suggested that it could be associated with poor outcome. 9
Based on these preliminary data, we hypothesized that sRAGE and polymorphisms of RAGE and glyoxalase I could also be predictors of long-term outcome in patients with breast cancer and looked at the long-term outcome of the patients from our original cohort.8,9
Subjects and methods
Study population
We originally examined sRAGE and RAGE polymorphisms in 116 unrelated Caucasian women regularly followed in one large tertiary center in the Department of Oncology of the General University Hospital in Prague. Detailed characteristics of our original cohort were described in our previous paper. 8
In short, all patients were examined after the breast surgery and after the end of the adjuvant chemotherapy and divided based on the expression of estrogen and the C-erb B2/Her2-neu receptors, clinical stage of the disease (based on tumor–node–metastasis (TNM) classification—stages I–IV), and histological grading. All patients suffering from type 2 diabetes and with serum creatinine exceeding 120 µmol/L were excluded. sRAGE and genetic polymorphisms of RAGE were also assessed in 92 unrelated healthy women (not shown here, see our previous paper). The patients were recruited to the study (serum samples were collected) between 21 October 2003 and 3 March 2005, and database was closed on 30 March 2015.
The study was performed in accordance with the principles of the Declaration of Helsinki and approved by the Institutional Ethical Committee. All patients have given their informed consent prior to entering the study (extra informed consent for biochemical and genetic analysis).
Mean age of breast cancer patients at entry to the study was 61.2 ± 11.9 years and mean sRAGE of breast cancer patients at presentation was 1568 ± 754 pg/mL, significantly lower than in controls: 1803 ± 634 pg/mL (p < 0.05). 8
Laboratory parameters
Assessment of RAGE, glyoxalase I polymorphisms, and sRAGE was also described in detail in our previous papers.8,9 In brief, we evaluated four RAGE gene polymorphisms (-429 T/C—rs1800625, -374 T/A—rs1800624, Gly82Ser—557G/A—rs2070600, and 2184 A/G—rs3134940) and 419 A/C (Ala111Glu—rs 2736654 glyoxalase I polymorphism) from DNA extracted from samples of peripheral blood after appropriate amplification using polymerase chain reaction (PCR) and subsequent restriction fragment analysis. sRAGE was measured with sandwich enzyme-linked immunosorbent assay (ELISA) using standard kits (Quantikine; R&D Systems, USA) according to the manufacturer’s protocol.
Statistical analysis
Statistical analysis was performed using SPSS v.16 statistics software. Descriptive results of biochemical parameters were expressed as mean ± standard deviations. Patient’s survival was evaluated using Kaplan–Meier analysis and univariate and multivariate Cox regression. Risk ratios (RR, relative risks of mortality) and appropriate confidence intervals (CIs) were calculated using the Cox proportional risk model. Kaplan–Meier analysis was used to construct survival curves.
Patients were stratified for clinical stages of the breast cancer at presentation. sRAGE was compared between different subgroups using generalized linear model (GLM) analysis with bootstrap extension. Comparison of subgroups was performed using Kruskal–Wallis and Mann–Whitney test. All results were considered as statistically significant at p < 0.05.10–12 Post hoc power for Kaplan–Meier survival results was computed based on log-rank test via the method recommended by Cantor. 13
Results
Data on long-term outcome were collected from 110 patients (6 patients from the original cohort lost follow-up) with a median follow-up of 125.9 months (10.3 years). At the end of follow-up, 62 patients were in clinical remission, 11 patients were living with progressive disease (either local recurrence or generalization), 24 died of breast cancer, and 13 died of other causes unrelated to breast cancer, mostly cardiovascular disease. Clinical characteristics of the subgroups of breast cancer patients based on their outcome are provided in Table 1.
Basic clinical parameters of patients with breast cancer in relation to their outcome.

At the end of follow-up, 62 patients were in remission, 11 were living with progressive disease, 24 out of 116 patients died of breast cancer, 13 died of other causes, and 6 patients lost follow-up.
Not assessed in all patients.
Patients who died of breast cancer had significantly more advanced diseases (stages II–IV) more frequently compared to patients who were in remission (p < 0.001), and patients who died of other causes unrelated to breast cancer were significantly older compared to patients in remission (p < 0.001).
Mean and median serum levels of sRAGE are shown in Table 2. Although median values of sRAGE were nominally lower in patients who died (both of breast cancer and other causes unrelated to breast cancer) compared to surviving patients, the difference did not reach statistical significance.
AGEs and sRAGE in different subgroups of patients based on their outcome.

AGE: advanced glycation end products; sRAGE: soluble forms of receptor for advanced glycation end products; BC: breast cancer; FU: follow-up
Distribution of alleles of -429 T/C, -374 T/A, G82S, 2184 A/G of RAGE, and 419A/C of glyoxalase 1 is shown in Table 3.
RAGE and GLO polymorphisms in different subgroups of patients based on their outcome.

Data are available for 116 patients (in case of RAGE – 374 T/A, RAGE G82S G/S, and RAGE 2184 A/G) and 113 patients (GLO1 419A/C).
In Kaplan–Meier analysis, there was no difference in overall survival between patients due to all polymorphisms which were studied. With respect to breast-cancer mortality, AG heterozygotes of 2184 A/G RAGE polymorphism had lower mortality than both AA and GG homozygotes (p = 0.014, post hoc power 1 − β = 0.776; Figure 1), and GS heterozygotes of G82S RAGE polymorphism had higher mortality than GG homozygotes (p = 0.042, post hoc power 1 − β = 0.592; Figure 2).
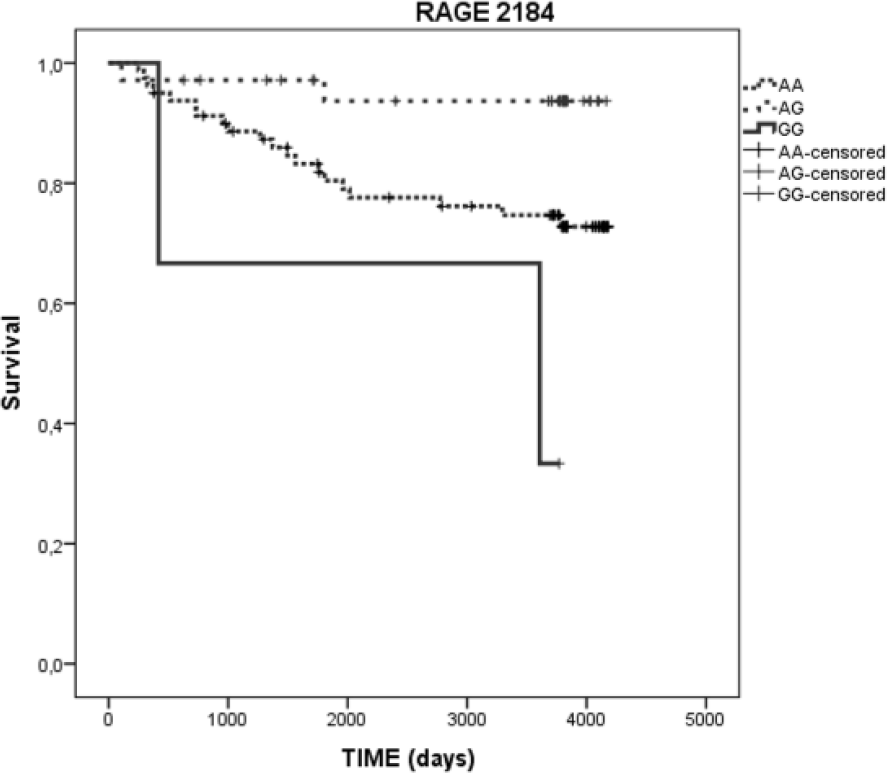
Kaplan–Meier curves demonstrating lower mortality due to breast cancer in patients with AG compared to AA (and GG) genotype of the RAGE 2184 polymorphism (p = 0.014, log-rank test).

Kaplan–Meier curves demonstrating overall mortality of patients with breast cancer with GG and GS genotypes of the RAGE G82S (557G/A) polymorphism (p = 0.042, log-rank test).
Cox regression analysis (Table 4) demonstrated higher overall mortality in patients with AC genotype compared to AA genotype of 419A/C GLO1 polymorphism (RR = 2.91, p = 0.037), and AG heterozygotes of RAGE 2184 A/G polymorphism show lower mortality due to breast cancer than AA homozygotes of this genotype (RR = 0.209, p = 0.035).
Overall mortality and mortality due to the breast cancer in patients with different RAGE and glyoxalase I polymorphisms.

RR: risk ratio; CI: confidence interval.
When the patients were stratified according to the clinical stage of breast cancer (0–IV), the presence of GG genotype of 2184 A/G RAGE polymorphism was conferred with significantly higher risk of mortality due to the breast cancer compared to AA genotype (RR = 28.520, p = 0.001), and on the opposite, AG heterozygotes had lower overall mortality compared to AA homozygotes (RR = 0.226, p = 0.046).
Discussion
Data on the relation between sRAGE serum levels and RAGE polymorphisms and the outcome of patients are controversial. RAGE polymorphisms (-429T/C and G82S) were demonstrated to be related to sRAGE levels 14 and outcome (-374T/A polymorphism) in terms of future cardiac events in non-diabetic patients with coronary artery disease 14 and in terms of macrovascular complications in type 2 diabetic patients. 15
In patients with end-stage renal disease treated with hemodialysis, RAGE gene polymorphisms (-429CC and 2184GG) were demonstrated to be related to sRAGE levels. 16 In these patients, sRAGE was negatively associated with glomerular filtration rate, acute phase reactants, but it was not related to the presence of diabetes mellitus, cardiovascular disease, nutritional status, and mortality. 16 With respect to these data, RAGE ligand S100A12 (EN-RAGE), but not sRAGE, was related to cardiovascular mortality.17,18 However, in another study, alternatively spliced variant of RAGE (endogenous secretory RAGE) was a predictor of cardiovascular mortality. 19
Increased expression of RAGE on breast cancer cells was repeatedly demonstrated 6 and may be partly estrogen dependent. 20 Activated RAGE stimulates cell proliferation due to increased expression of cell cycle protein cyclin D1 and prolongs survival of breast cancer cells by increased phosphorylation of prosurvival protein Akt and increased expression of antiapoptotic protein Bcl-2. 20 Also, RAGE ligands are overexpressed in breast cancer, namely, HMGB1. 21 HMGB1 protein levels in breast cancer tissue were shown to be correlated with cancer progression. 22 Of the RAGE ligands of another group, S100 proteins, S100P was demonstrated to be expressed on breast cancer cell lines, and its expression was associated with hormone independence, resistance to chemotherapy, and metastatic potential. 23 Another S100 protein, S100A7/psoriasin, was shown to be overexpressed in invasive estrogen receptor–negative breast cancers, 24 and its expression stimulated ductal hypeplasia, tumor growth, and metastasis. Invasion of breast cancer cell could also be stimulated by S100A8/A9 binding of RAGE which is associated with nuclear factor-κB (NF-κB)-mediated enhancement of epithelial–mesenchymal transition and formation of distant metastases. 25
In view of this increasing evidence of the involvement of AGE-RAGE axis in the pathogenesis of (not only 26 ) breast cancer, RAGE and its ligands could also be very attractive putative therapeutic targets using monoclonal antibodies against RAGE, 27 recombinant soluble RAGE, 28 or RAGE small interfering RNA 29 and also antibodies for other interventions directed against HMGB1 and S100 proteins. 30
sRAGE physiologically counteracts activation of RAGE on cancer cells and may serve as a natural antagonist of multiple RAGE ligands. In patients with metastatic breast cancer, lower sRAGE (and higher HMGB1) was shown to be a predictor of no response to neoadjuvant chemotherapy. 31
Clinical data relating sRAGE with the outcome of breast cancer are rare. In our previous paper, 8 we demonstrated that patients with breast cancer have lower levels of sRAGE compared to healthy controls and patients with higher RAGE had better outcome (lower grade and positive estrogen receptor). Based on the higher levels of sRAGE in patients with advanced breast cancer (stage III), we, however, speculated that, on one hand, decreased sRAGE levels in patients with breast cancer may contribute to the progression of the disease, but, on the other hand, progression may increase sRAGE levels, possibly as a compensatory mechanism to counteract further progression. Of course, these equivocal relations may confound any prediction of the patients’ outcome based on the sRAGE serum levels.
In our original study, 8 at the end of relatively short median follow-up of only 3.2 years, serum levels of sRAGE during the study were significantly lower in (only 9) patients who died of breast cancer compared to the patients in remission (1081 ± 514 vs 1628 ± 846 ng/L, p < 0.05). Serum levels of sRAGE were also negatively associated with advanced breast cancer, higher grade, and positivity of estrogen receptors and indicated that sRAGE could be a prognostic marker in breast cancer. In this study, we were unable to confirm the relation of sRAGE levels with long-term outcome of our patients with breast cancer. As the serum levels of sRAGE in our patients were influenced genetically (were related to Gly82Ser and 2184 A/G polymorphisms of the RAGE gene 8 ), we also looked at the relation between the outcome and RAGE gene polymorphisms.
In our cohort of patients with breast cancer with long-term follow-up, we were able to demonstrate putative prognostic value of glyoxalase 419 A/C polymorphism in terms of overall mortality and G82S RAGE and 2184 A/G RAGE polymorphism in terms of mortality due to breast cancer. At least to our knowledge, this is the first study showing the relation between RAGE and glyoxalase polymorphisms and long-term outcome in patients with breast cancer as other studies looked only at the relation between RAGE polymorphisms and susceptibility to the breast cancer.
After stratification for the presenting clinical stage of breast cancer GG, and also AA homozygotes of 2184 A/G polymorphism had compared to heterozygotes very high risk of mortality due to the breast cancer. This is in keeping with other data from our group, higher mortality of GG homozygotes of 2184 RAGE A/G polymorphism among patients with end-stage renal disease on hemodialysis 32 and higher grade of renal cancer. 33
In the study by Hashemi et al. 34 with small cohort, there was no difference between RAGE (-374T/A, -429T/C, and 63 bp Ins/del) polymorphisms among patients with breast cancer and healthy controls, but recent large study in Han Chinese 35 did find a significant difference in one out of four studied polymorphisms (rs184003) between breast cancer patients and controls suggesting that larger studies are necessary to demonstrate any putative relation between RAGE gene polymorphisms and susceptibility to breast cancer in Caucasian patients. Very recently, RAGE -374 T/A polymorphism was found to be associated with decreased risk of breast cancer in another Chinese study. 36 Recent meta-analysis revealed that G82S polymorphism is associated with increased risk of cancer (namely, lung cancer) and -374T/A polymorphism is associated with the decreased risk of cancer (namely, lung and breast cancer). 37 Another meta-analysis confirmed the association of RAGE – 374T/A polymorphism with decreased susceptibility to the breast cancer and demonstrated for the same polymorphism increased susceptibility to the lung cancer. 38 Definitely, polymorphisms related to the outcome may not be in the same time associated with the susceptibility to cancer, and in breast cancer, further studies are warranted.
In our previous study with the shorter follow-up of this cohort of patients with breast cancer, 9 we demonstrated that minor allele of glyoxalase I 419A/C (Glu111Ala) polymorphism was associated with the absence of the expression of the estrogen receptor on the cells of our patients with breast cancer and more advanced stages of breast cancer. In this study with long-term follow-up of the same cohort of patients, AC genotype compared to wild AA genotype of C419A GLO1 polymorphism was associated with higher overall mortality of affected patients with breast cancer.
Breast cancer proteomics revealed a positive correlation between glyoxalase I expression in breast cancer cells, and high tumor grade 39 and glyoxalase I inhibition induced apoptosis in MCF-7 breast cancer cells. 40 Glyoxalase I was found to be the most frequently amplified gene in different types of cancer including breast cancer. 41 Glyoxalase I 419A/C polymorphism was also associated with the biomarkers of the oxidative stress in cell lines of aggressive and invasive prostate cancer. 42 In a large Italian study, glyoxalase I 419A/C polymorphism was shown to be associated with the susceptibility to the breast cancer, 43 but in another study from Malaysia, 44 this polymorphism was not associated with the risk of breast cancer, but only with the absence of progesterone receptor.
We have to admit some limitations of our study, mainly the relatively small sample size. However, the main advantage of the study is long-term follow-up, a median of more than 10 years. The topic would surely deserve further investigation with a larger number of patients.
In conclusion, serum levels of sRAGE did not predict the outcome in our cohort of patients with breast cancer, but the association of G82S, 2184 A/G RAGE, and 419A/C GLO1 polymorphisms with mortality of our patients suggests that activation of RAGE and impaired degradation of AGEs may both play a role in the progression of breast cancer. Our data should be confirmed in other cohorts of patients with breast cancer with long-term follow-up.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by research projects MH CZ DRO VFN 64165 and PROGRES Q27.
