Abstract
Melanoma is a devastating disease with few therapeutic options in the advanced stage and with the urgent need of reliable biomarkers for early detection. In this context, circulating microRNAs are raising great interest as diagnostic biomarkers. We analyzed the expression profiles of 21 selected microRNAs in plasma samples from melanoma patients and healthy donors to identify potential diagnostic biomarkers. Data analysis was performed using global mean normalization and NormFinder algorithm. Linear regression followed by receiver operating characteristic analyses was carried out to evaluate whether selected plasma miRNAs were able to discriminate between cases and controls. We found five microRNAs that were differently expressed among cases and controls after Bonferroni correction for multiple testing. Specifically, miR-15b-5p, miR-149-3p, and miR-150-5p were up-regulated in plasma of melanoma patients compared with healthy controls, while miR-193a-3p and miR-524-5p were down-regulated. Receiver operating characteristic analyses of these selected microRNAs provided area under the receiver operating characteristic curve values ranging from 0.80 to 0.95. Diagnostic value of microRNAs is improved when considering the combination of miR-149-3p, miR-150-5p, and miR-193a-3p. The triple classifier had a high capacity to discriminate between melanoma patients and healthy controls, making it suitable to be used in early melanoma diagnosis.
Introduction
MicroRNAs (miRNAs) are small, non-coding, single-stranded RNAs produced by eukaryotic cells that play a key role in regulating the expression of hundreds of target genes.1,2 MiRNAs are involved in numerous processes including proliferation, differentiation, and apoptosis, and their altered expression and function have been linked to the development and progression of various types of cancers.3–9 In particular, miRNAs may act as oncomirs or tumor suppressor molecules since their expression in tumor cells can be up- or down-regulated, compared with normal tissues. 10 MiRNAs can be released into circulation as a consequence of tumor cell death or, in some cases, actively secreted by viable tumor cells. Moreover, miRNAs might also derive from blood and/or endothelial cells. 11 MiRNAs display several properties that make them potentially valuable biomarkers including stability in body fluids, resistance to endogenous RNase activity, prolonged life at room temperature, and resistance to multiple freeze–thaw cycles. 12 A growing body of evidence shows a different pattern of expression of circulating miRNAs between cancer patients and healthy donors11,13–19; however, only a limited number of studies have been focused on melanoma.
Melanoma is the fourth and sixth most common malignancy in men and women, respectively. Disease incidence is increasing by 3% every year, and melanoma accounts for 75% of skin cancer deaths. The 5-year survival rate critically depends on disease stage, and it has been estimated to be around 95% for stage I and 15%–20% for stage IV. 20 Although several circulating diagnostic and prognostic biomarkers have been investigated in melanoma patients, the only one routinely used in clinical setting is lactate dehydrogenase (LDH). 21 Serum LDH has a significant prognostic value only in the advanced disease (stage IV). 21 At present, no circulating marker is available to help clinicians in detecting melanoma in early stage when survival is at the highest rate.
Many recent papers reported circulating miRNAs as being dysregulated in melanoma cancer patients with respect to healthy subjects (Carpi et al., 2016). 22 Nevertheless, further studies are urgently needed to better define a clearer role of specific miRNAs in melanoma diagnosis and prognosis.
In this pilot study, the expression profiles of 21 miRNAs selected from the literature for their role in melanoma progression were analyzed in plasma samples collected from both melanoma patients, at different disease stages, and healthy controls. The data obtained allowed the identification of some plasma miRNAs with high sensitivity and specificity as potential diagnostic biomarkers in melanoma patients worthy of being validated in a large prospective clinical trial.
Materials and methods
Study participants
Control blood samples from 32 healthy volunteers with similar age and sex distribution were obtained from the Blood Donor Centre of the University Hospital of Pisa. Healthy subjects who had taken systemic drugs in the week prior to enrollment and those affected by other medical conditions were excluded by sample collection.
Patient recruitment was carried out at the Department of Oncology of the University Hospital of Pisa (Pisa, Italy). Ten American Joint Committee on Cancer (AJCC) stage I–II, 10 stage III, and 10 stage IV (https://cancerstaging.org/) subsequent patients were included this study. Blood of 2.5 mL was drawn from each patient, collected in BD Vacutest Kima tubes (VACUTEST KIMA S.r.l., Arzergrande, Padua, Italy) containing ethylenediaminetetraacetic acid (EDTA), and centrifuged at 4°C for 10 min at 1900g for 1 h. Plasma supernatant was then aliquoted into 1.5 mL DNA LoBind tubes (Eppendorf AG, Amburgo, Germany) and stored at −80°C until analysis.
The timing of blood collection in patients was (1) within a month following the surgical removal of the lesion and the availability of the pathology report for patients with stage I–II melanoma, (2) within a month following the surgical removal of the sentinel lymph node and the availability of the pathology report for patients with stage III melanoma, and (3) within a month following histologically proven melanoma metastasis for patients with stage IV disease.
The study protocol was approved by the local ethics committee and conducted in accordance to the principles of the Declaration of Helsinki. Informed consent was obtained from all individual participants included in the study.
Selection criteria for miRNAs panel
For the purpose of this analysis, 21 miRNAs were selected from the literature according to at least two of the following properties: dysregulation in the presence of BRAF mutations, negative correlation with metastatic melanoma patient survival, and potential involvement in drug resistance phenotype (please refer to supplementary material). Among all miRNAs selected, 11 have already been individuated as circulating in plasma melanoma patients and the others had only been identified as dysregulated in melanoma cells or tissue samples but they were not yet detected in plasma.
MiRNA extraction and real-time polymerase chain reaction analyses
All 21 selected miRNAs were isolated from plasma samples (200 µL) using the miRNeasy Serum/Plasma Kit (Qiagen, Valencia, CA, USA) following the manufacturer’s instructions and eluted with 14 µL RNase-free water. Sample concentration was measured by Qubit® microRNA HS Assay (Thermo Fisher Scientific, MA, USA). Reverse transcription was carried out using miRCURY LNA™ Universal RT microRNA PCR System (Exiqon, Vedbaek, Denmark) according to the manufacturer’s instructions. Complementary DNA (cDNA) synthesis was conducted by incubation at 42°C for 60 min and 95°C for 5 min. Prior to real-time quantitative polymerase chain reaction (RT-qPCR) analysis, cDNA was diluted 1:40. RT-qPCRs were run on a 7900HT thermocycler (Applied Biosystems, Waltham, MA, USA) using the miRCURY LNA Universal RT microRNA PCR assays with the thermal-cycling parameters recommended by Exiqon. Raw Cq values were calculated, as recommended by Exiqon, using the RQ manager software version 1.2.1 (Applied Biosystem Instruments, Waltham, MA, USA) with manual settings for threshold and baseline.
Data analysis and statistics
Data analysis was carried out by GenEx software (MultiD Analyses). Since there is no validated reference miRNA for miRNA analysis in serum/plasma, we used two different methods for data normalization: global mean normalization (GMN) and NormFinder model. The first method was based on the normalization of each single miRNA expression value over the mean expression of all analyzed miRNAs (i.e. difference between single Cq and global mean Cq values), assuming that the latter was constant when the same amount of total RNA from patients and healthy controls was analyzed. Such a strategy has been reported to reduce technical variations providing a more accurate assessment of biological changes than using endogenous small nuclear RNAs such as U6. 23 The second method data were normalized according to miRNA expression stability using the algorithm NormFinder.
Statistical analysis was carried out using the Wilcoxon test (non-parametric) followed by Bonferroni correction for multiple comparisons. Linear regression followed by receiver operating characteristic (ROC) analyses was carried out to evaluate whether selected miRNAs were able to discriminate between cases and controls. To this aim, the optimal cut-off value for sensitivity and specificity was determined based on the highest Youden’s Index in ROC curve analysis. ROC curves were drawn using GraphPad Prism 6, and the area under the ROC curve (AUC) was calculated to evaluate the specificity and sensitivity of selected miRNAs.
Results
Characteristics of study population
From January 2015 to February 2016, blood samples were collected from 30 consecutive melanoma patients in different disease stages. Patients’ characteristics, including sex, age, and other clinical information, are reported in Table 1. The median age of the melanoma patient and healthy volunteers was 63 and 57 years, respectively.
Clinical characteristics of study subjects.

LDH: lactate dehydrogenase.
Expression levels of miRNAs in patients and healthy controls
Data normalization by GMN method provided 13 out of the 21 miRNAs significantly dysregulated (p < 0.05, analysis of variance (ANOVA); Table 2). The NormFinder algorithm identified eight out of 21 miRNAs as normalizers, and the subsequent comparison provided seven significantly dysregulated miRNAs (p < 0.05; Table 2). Only five miRNAs resulted significantly dysregulated (p < 0.01) with both methods of data analysis after the Bonferroni correction for multiple tests (Figure 1). Specifically, circulating miR-15b-5p, miR-149-3p, and miR-150-5p were significantly up-regulated, and miR-193a-3p and miR-524-5p were significantly down-regulated in plasma of melanoma patients compared with healthy controls. No significant difference in miRNA expression was observed among different disease stages. These miRNAs were selected for a further statistical analysis to evaluate their diagnostic power.
MiRNAs found differentially expressed in melanoma patients compared with healthy subjects.
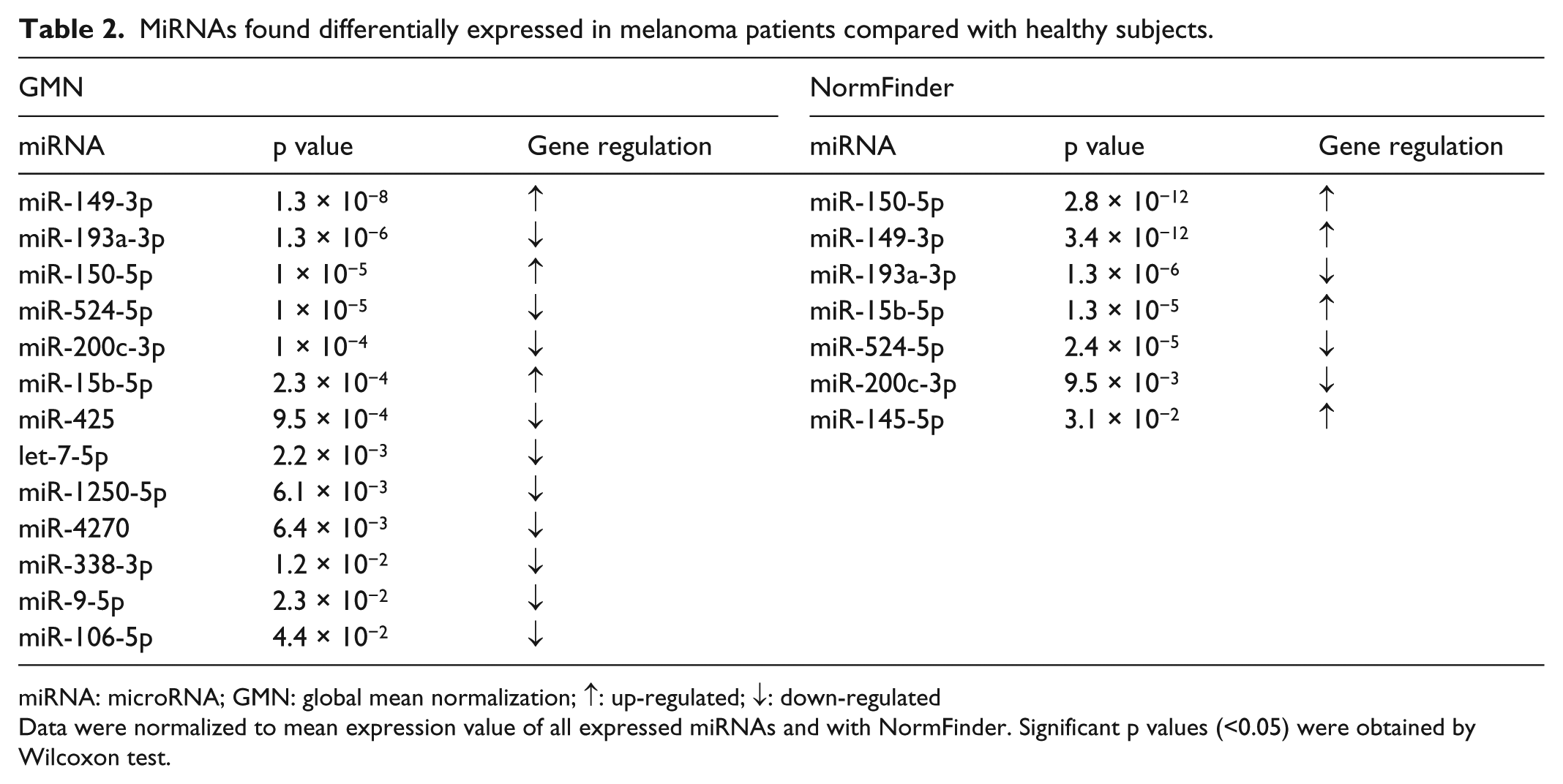
miRNA: microRNA; GMN: global mean normalization; ↑: up-regulated; ↓: down-regulated
Data were normalized to mean expression value of all expressed miRNAs and with NormFinder. Significant p values (<0.05) were obtained by Wilcoxon test.

Expression levels of the five selected miRNAs in plasma samples from melanoma patients (MP) and healthy subjects (C). p value is reported in Table 2.
As opposed to the others, miR-200c-3p was not dysregulated in plasma melanoma patients compared with healthy donors, but its expression was significantly down-regulated in different stages of the disease. Particularly, while the expression of miR-200c-3p in plasma of stage I–II melanoma patients was not different from healthy donors, its expression in plasma of stage III–IV melanoma patients was significantly down-regulated compared with that of stage I–II melanoma patients (p = 0.001) and of healthy donors (p = 0.00004) (Figure 2). Diagnostic power of miR-200c-3p was not considerate since its value did not differ in plasma of melanoma patients compared with healthy donors.

Expression levels of miR-200c-3p in plasma samples from melanoma patients at different stages and healthy subjects (C).
No significant miRNA dysregulation due to gender, age, or any clinical-pathological factors was observed (data not shown).
Diagnostic value of plasma miR-150-5p, miR-149-3p, miR-193a-3p, miR-15b-5p, and miR-524-5p in melanoma patients
The AUC for each of these five miRNAs ranged from 0.80 to 0.95 (Table 3). Although high AUC values were attributed to each miRNA, miR-150-5p, miR-149-3p, and miR-193a-3p present the best performance in terms of diagnostic power. Noteworthy, diagnostic power was further improved when considering miR-150-5p, miR-193a-3p, and miR-149-3p signature. The triple classifier has increased AUC, sensitivity, and specificity (Table 3) with an accuracy of 91%.
The AUC for single miRNAs and for triple classifier with corresponding sensitivity and sensibility.

AUC: area under the ROC curve; miRNA: microRNA; CI: confidence interval; ROC: receiver operating characteristic.
Triple classifier: miR-149-3p + miR-150-5p + miR-193a-3p.
Level of significance: p < 0.0001.
Discussion
The incidence of melanoma is increasing all over the world. This disease is curable when detected in early stages, and it might be deadly when diagnosed in advanced stages. Although lately new and very effective therapies came in clinical use, mortality rate of stage IV melanoma patients is still high. 20 Since melanoma must be treated in early stages to maximize the chances of patient survival, the ability to identify early signs of melanoma progression would be very valuable in clinical setting. To date, no reliable biomarker has been identified as sensitive or specific enough to be beneficial for early detection of melanoma (all stages). Reliable circulating biomarker(s) to detect melanoma with distant or regional spread prior to clinical evidence of metastasis could improve treatment and outcome for melanoma patients. Stability, sensitivity, and specificity together with resistance to degradation are the main characteristics to look for a potential circulating biomarker.
The potential diagnostic role of plasma miRNAs in melanoma has been investigated in a few studies23,24 with different results due mainly to differences in analytical methods that lead to the identification of different miRNA panels with diagnostic value. A major source of difference in analytic methods is the normalization in RT-qPCR. Up to now, there are no validated normalizers for the analysis of circulating miRNA expression levels. Therefore, we used different methods for data normalization (GMN and NormFinder) not involving the use of a single molecule as a control, given the great biological variability in expression of circulating miRNAs. Indeed, we did not use miR-16, as in many studies conducted by others, since this miRNA is particularly susceptible to hemolysis. 25
In this pilot study, we identified five miRNAs with promising diagnostic power in plasma of melanoma patients at different stages of disease. Three of them, that is, miR-149-3p, miR-150-5p, and miR-15b-5p, were significantly up-regulated, while two, that is, miR-193a-3p and miR-524-5p, were significantly down-regulated in plasma of patients compared with healthy controls. To the best of our knowledge, this study is the first to report on dysregulation of miR-149-3p, miR-524-5p, and miR-193a-3p in plasma samples of melanoma patients. Moreover, the expression of miR-200c-3p in plasma of stage I–II melanoma patients is not different from healthy donors, while its expression in plasma of stage III–IV melanoma patients was significantly down-regulated, compared with both stage I–II melanoma patients (p = 0.001) and healthy donors (p = 0.00004). Therefore, miR-200c-3p is not suitable as diagnostic biomarker, but instead, it could be studied as a prognostic biomarker.
While we found significant levels of circulating miR-149-3p from plasma samples of melanoma patients compared with healthy controls, Pfeffer et al. 26 found higher miR-149-5p levels in peripheral exosomes extracted from plasma samples of metastatic melanoma patients compared with healthy controls. This suggests that miR-149-3p and miR-149-5p species may have a similar functional role as oncomirs in human melanoma. Mature miRNA-3p and miRNA-5p are generated from the same pre-miRNA precursor, but more often, one of them is degraded, although co-existence of these two species has also been reported. 27 The reason of this apparent discrepancy is currently unclear. It has been hypothesized that the main (-3p) and the passenger (-5p) strands might influence the expression of different targets and, in some cases, they can regulate invasiveness of cancer cells. 28
Concerning miR-149-5p, several lines of evidence suggest that in some cancer types other than melanoma, it may function as a tumor suppressor. For example, miR-149-5p expression was decreased in gastric cancer cell lines and in clinical specimens compared with normal counterparts. 29 Furthermore, low miR-149-5p expression levels were correlated with lymph node or distant metastasis and advanced disease stages in colorectal 30 and breast cancers. 31 In this work, high levels of miR-149-3p have been detected in plasma of melanoma patients according to the work of Jin et al, which described miR-149-3p in melanoma cells and found that it induced a p53-dependent survival by increasing the expression of the anti-apoptotic Mcl-1 protein. The same authors described elevated expression of miR-149-3p in fresh human metastatic melanoma isolates. 32
In this study, plasma miR-150-5p was up-regulated in plasma of patients compared with healthy controls. MiR-150 was also found to be overexpressed in formalin-fixed paraffin-embedded melanoma metastases and in primary melanoma compared with nevi. 24 In line with the hypothesis of its oncogenic role, high levels of circulating miR-150 were also detected in melanoma patients with high risk of recurrence. 33 Preclinical evidence showed that tumor growth and metastasis were reduced in immunodeficient mice injected with miR-150−/− NK (natural killer) cells, compared with non-injected mice. 34 Noteworthy, miR-150 has been reported to increase tumor immunoresistance by post-transcriptionally down-regulating perforin-1 in mouse NK cells. 34 Although these findings suggest an oncogenic role for miR-150 in human melanoma, some evidence seems to go in the opposite direction. For instance, miR-150-5p up-regulation was found both in serum and tissues derived from melanoma patients with longer disease-free survival after resection of the metastatic lymph node disease.24,35 Recently published evidence on the ability of miR-150-5p to suppress glioma cell proliferation and migration by targeting membrane-type-1 matrix metalloproteinase 36 reinforces the tumor suppressor role of miR-150-5p. These apparent discrepancies may be due to the complex functional role of miR-150-5p in tumor biology. Indeed, this specific miRNA is expressed in the lymph nodes, spleen, and thymus, and it is highly up-regulated during lymphocyte maturation and down-regulated during the activation of mature B and T cells. 37 Thus, elevated circulating expression of miR-150 could also be the consequence of its role in the modulation of T-cell response. In line with these findings, in vivo studies show that miR-150 is released in the external environment after T cell activation, and this may cause an increase of its blood levels during immune system stimulation. 38 Further experiments aimed at clarifying the precise role of miR-150 in human melanoma immunity are warranted.
The third circulating miRNA found up-regulated in this study is miR-15b-5p, whose levels were significantly higher in plasma samples from melanoma patients than in healthy donors, with a trend for a more pronounced increase in advanced stage patients (data not shown). Our results are consistent with data of Fleming et al. showing that circulating miR-15b levels were increased compared with the levels found at diagnosis in melanoma patients whose disease recurred compared with disease-free patients. 39 Finally, evidence showing miR-15b-5p overexpression in melanoma tissues compared with melanocytic nevi suggests a potential oncogenic role for this specific miRNA. 40 In line with this notion, the same authors clearly demonstrated that up-regulation of miR-15b-5p is responsible for increasing cell proliferation and decreasing apoptosis in melanoma cell lines. 40
MiR-193a-3p is a member of the miR-193 family whose expression was linked to BRAF mutation status in melanoma tissues. 41 MiR-193a-3p was found to be significantly up-regulated in both tissue and blood samples of colorectal cancer patients, and there was an increasing trend of expression of circulating miR-193a-3p in advanced stages of colon cancer patients compared with healthy individuals. 42 Up-regulation of miR-193 was also found in the high-grade prostate tumors (Gleason score ≥8) 43 as well as in serum samples obtained from patients with esophageal squamous cell carcinoma. 44 In apparent contrast with these findings but in agreement with those of this study, in lung and oral squamous cell carcinoma,45,46 acute myelogenous leukemia, 47 breast cancer, 48 and Wilms’ tumor, 49 circulating miR-193a-3p levels were found to be reduced in plasma of patients compared with healthy controls. The report that methylation is responsible for miR-193a down-regulation46,47,50 reinforces the notion that miR-193a-3p might have a role as tumor suppressor in a variety of human cancers, including melanoma.
The functional role of miR-524-5p in cancer development and progression is largely unknown. A possible tumor suppressor role for miR-524-5p may be in line with findings of this study showing how circulating miR-524-5p levels were significantly lower in plasma of patients compared with healthy controls. Preclinical data demonstrating that miR-524-5p overexpression inhibited melanoma cell proliferation and migration in human melanoma cell lines 51 are likely to support this notion.
Although not significantly down-regulated in plasma of all melanoma patients compared with healthy controls, miR-200c-3p showed different levels of expression in plasma of patients with advanced melanoma stages compared with those detected in patients with early stages of the disease and healthy individuals. This suggests that miR-200c-3p can undergo a process of dynamic change at different tumor stages. In agreement with this hypothesis are findings by Xu et al. 52 who observed that miR-200c-3p was down-regulated in metastatic melanoma compared with benign melanocytic nevi. Noteworthy, several lines of evidence support the hypothesis that miR-200c may play a role in epithelial–mesenchymal transition by modulating expression of E-cadherin. 17 Therefore, up-regulation of circulating miR-200c-3p in patients with early melanoma stages may correlate with the presence of an epithelial phenotype in the majority of melanoma cells, whereas down-regulation of circulating miR-200c-3p in patients with melanoma in advanced stages may indicate the predominance of the mesenchymal cell phenotype with increased capacity of tissue invasion and distant metastasis. Such a biological behavior suggests that miR-200c-3p could be an interesting biomarker of prognosis in melanoma patients. Finally, miR-200c have been reported to be under-expressed in melanoma tissues collected from patients considered non-responders to BRAF-inhibitors compared with responders, 17 suggesting a possible involvement of miR-200c in drug resistance phenotype.
Compared with Stark et al., 11 we were able to identify the signature of three different miRNAs with comparable diagnostic sensitivity and specificity to the MELmiR-7. Moreover, although limited by the small sample size, our findings are to be considered reliable since we utilized two different normalization methods compared with the one chosen by Stark et al.
In conclusion, our pilot study identified five circulating miRNAs, of which three detected for the first time in plasma of melanoma patients. MiR-149-3p, miR-150-5p, and miR-193a-3p, either alone or as a signature, are suitable to be considered as potential diagnostic biomarkers in human melanoma and worthy to be validated in future prospective clinical trials.
Footnotes
Acknowledgements
S.F., B.P., A.R., and P.N. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Associazione Contro il Melanoma (ACM) Onlus (Italy). The funding source had no role in study design; collection, analysis, and interpretation of data; writing of the report; and the decision to submit the article for publication.
