Abstract
Wnt1-inducible signaling pathway protein-1 is a cysteine-rich protein that belongs to the CCN family, which has been implicated in mediating the occurrence and progression through distinct molecular mechanisms in several tumor types. However, the association of Wnt1-inducible signaling pathway protein-1 with gastric cancer and the related molecular mechanisms remain to be elucidated. Therefore, this study aimed to clarify the biological role of Wnt1-inducible signaling pathway protein-1 in the proliferation, migration, and invasion in gastric cancer cells and further investigated the associated molecular mechanism on these biological functions. We first detected the expression level of Wnt1-inducible signaling pathway protein-1 in gastric cancer, and the reverse transcription polymerase chain reaction have shown that Wnt1-inducible signaling pathway protein-1 expression levels were upregulated in gastric cancer tissues. The expression of Wnt1-inducible signaling pathway protein-1 in gastric cancer cell lines was also detected by quantitative real-time polymerase chain reaction and Western blotting. Furthermore, two gastric cancer cell lines with high expression of Wnt1-inducible signaling pathway protein-1 were selected to explore the biological function of Wnt1-inducible signaling pathway protein-1 in gastric cancer. Function assays indicated that knockdown of Wnt1-inducible signaling pathway protein-1 suppressed cell proliferation, migration, and invasion in BGC-823 and AGS gastric cancer cells. Further investigation of mechanisms suggested that cyclinD1 was identified as one of Wnt1-inducible signaling pathway protein-1 related genes to accelerate proliferation in gastric cancer cells. In addition, one pathway of Wnt1-inducible signaling pathway protein-1 induced migration and invasion was mainly through the enhancement of epithelial-to-mesenchymal transition progression. Taken together, our findings presented the first evidence that Wnt1-inducible signaling pathway protein-1 was upregulated in gastric cancer and acted as an oncogene by promoting proliferation, migration, and invasion in gastric cancer cells.
Background
Gastric cancer (GC) is one of the most common type of cancer and the third leading cause of cancer-related mortality. 1 Despite the improvement of surgical techniques and adjuvant treatment, the 5-year survival rate of patients with GC remains as a low level. Since the early stages of GC are normally asymptomatic, a majority of GC patients are often diagnosed in advanced stages and the available treatments are mostly inefficient. 2 The early diagnosis of GC is essential for effective medical treatment, which may contribute to the decrease in the high morbidity and mortality rates of GC. Therefore, potential molecular biomarkers that may act as novel therapeutic targets for patients with early stages of GC are crucial for the effective clinical diagnosis and treatment.
WISP-1 (Wnt1-inducible signaling pathway protein-1) has already been identified as a member of the CCN family and contains four cysteine-rich modular domains, which include insulin-like growth factor binding domain, thrombospondin domain, von Willebrand factor type C module domain, and C-terminal cysteine knot-like domain. 3 The members of CCN family have been reported to be involved in part of complex biological processes such as cell proliferation, differentiation, adhesion, migration, and invasion. 4 Moreover, this family was also found to have regulatory roles in angiogenesis and tumorigenesis. 5 WISP-1 was first demonstrated as an oncogene and acted as a downstream gene of Wnt signaling to induce colon cancer occurrence and progression; and the molecular mechanism was through the specific combination of Wnt-1 and β-catenin with WISP-1 promoter, which in turn activated the initiation of WISP-1 transcription.6,7 The relationships between WISP-1 protein expression and breast cancer, endometrial endometrioid adenocarcinoma, and fibroblasts were gradually reported in recent studies.8,9 In addition, more complex molecular mechanisms of WISP-1 in mediating cancer occurrence and progression by activating the signaling pathways, such as mitogen-activated protein kinase (MAPK), nuclear factor-kappa B (NF-κB), and activator protein-1 (AP-1), were also found in different tumor types.10,11 With the help of the further extensive investigation, WISP-1 was no longer only recognized as an oncogene that promoted malignant biological processes, and it was also demonstrated to function as a tumor suppressor that inhibited the progression of tumorigenesis in various tumor types. 12 However, it seemed that the diverse roles of WISP-1 demonstrated by previous studies were tumor-type-dependent. Moreover, previous associated researches of some CCN family members, such as CYR611 (CCN1) and WISP-2 (CCN5), were already studied in GC, whereas the biological role of WISP-1 in GC was so far still unknown.13,14 Therefore, this study investigated the association of WISP-1 protein expression with the essential biological functions, such as cell proliferation, migration, and invasion, to determine the potential role of WISP-1 in the development and progression of GC.
Materials and methods
Gastric tissue specimens
Frozen GC tissues were obtained from the BioBank of Peking University Cancer Hospital. All human samples were obtained through written informed consent from patients, and the ethics committee of Peking University Cancer Hospital approved these tissues for research use.
Cell lines and cell culture
All the GC cell lines were provided by the Peking University Cancer Hospital & Institute. SGC-7901, BGC-803 and AGS cell lines were cultured in Dulbecco’s modified Eagle medium (DMEM; MAC GENE, China, http://macgene.com) supplemented with 10% fetal bovine serum (FBS; HyClone, USA) and BGC-823 cell line was cultured in DMEM supplemented with 5% FBS. All the cell lines were maintained at 37°C, 5% CO2.
RNA interference
The small hairpin RNA (shRNA) was used for the knockdown of endogenous WISP-1 in BGC-823 and AGS cells. The target sequences were as follows: sh-WISP-1-1: 5′-GCTTCTGTAACCTGAGCTGTA-3′ and sh-WISP-1-2: 5′-CCACTCGGATCTCCAATGTTA-3′. GV248 was used as sh-control. Cells with depleted endogenous WISP-1 expression were selected by being cultured in puromycin at the final selection concentration of 2 µg/mL.
RNA extraction and reverse transcription polymerase chain reaction
Total RNA was extracted from tissue samples and cell lines using the TRIzol reagent (15596-026; Ambion, USA). Reverse transcription polymerase chain reaction (RT-PCR; AT301, Transgene, China) was used to determine the messenger RNA (mRNA) level of WISP-1 in the gastric tissues and GC cell lines. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a control. PCR was performed in PCR reaction mix (A012-100, GenStar, China) with initial heating at 94°C for 5 min, followed by 30 cycles at 94°C for 30 s, 62°C for 30 s, 72°C for 30 s, and finally 72°C for 10 min. The primers for WISP-1 and GAPDH were as follows—WISP-1: CATCAGCACACGCTCCTATC (forward), GCTCAGGTTACAGAAGCAGG (reverse); GAPDH: GCATCCTGGGCTACACT (forward), CACCACCCTGTTGCTGT (reverse).
Quantitative real-time PCR
Quantitative real-time PCR was performed with the ABI PRISM 7500 Sequence Detection System using the SYBR Green method (AQ141, Transgene, China). The mRNA levels of all detected genes were normalized against GAPDH. Specific primers used in PCR amplification were as follows—WISP-1: CATCAGCACACGCTCCTATC (forward), GCTCAGGTTACAGAAGCAGG (reverse); Slug: CTACAGCGAACTGGACACACA (forward), GCCCCAAAGATGAGGAGTATC (reverse); Snail: TCCAGAGTTTACCTTCCAGCA (forward), CTTTCCCACTGTCCTCATCTG (reverse); Twist: GTCCGCAGTCTTACGAGGAG (forward), GTCTGAATCTTGCTCAGCTTGTC (reverse); Vimentin: GGACCAGCTAACCAACGACA (forward), AAGGTCAAGACGTGCCAGAG (reverse); β-catenin: ACGGAGGAAGGTCTGAGGAG (forward), AGCCGCTTTTCTGTCTGGTT (reverse); cyclinD1: GATGCCAACCTCCTCAACGA (forward), GGAAGCGGTCCAGGTAGTTC (reverse); proliferating cell nuclear antigen (PCNA): CTGACAAATGCTTGCTGACCT (forward), CTTCTTGAGGATGGAGCCCTG (reverse); and GAPDH: GCATCCTGGGCTACACT (forward), CACCACCCTGTTGCTGT (reverse).
Western blotting
Cells were lysed completely in lysis buffer (50 mM Hepes pH 7.5, 150 mM NaCl, 2 mM ethylenediaminetetraacetic acid (EDTA), 2 mM ethylene glycol-bis(β-aminoethyl ether)-N,N,N’,N’-tetraacetic acid (EGTA), 1% Triton X-100, 50 mM sodium fluoride (NaF), 5 mM Sodiun pyrophosphate, 50 mM sodium β-glycerophosphate, 1 mM DTT (dithiothreitol), 1 mM PMSF (phenylmethylsulfonyl fluoride), 10 µg/mL Leupeptin, and 10 µg/mL Aprotinin) at 4°C. The protein content was determined using a BCA Protein Assay Kit (Thermo Fisher Scientific, USA). Total proteins were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, USA). The blotted membranes were incubated with primary antibodies and then with secondary antibodies. Antibodies against GAPDH (sc-166545) and WISP-1(sc-25441) were purchased from Santa Cruz Biotechnology (USA).
Cell proliferation assay
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was performed to determine cell proliferative activity. BGC-823 and AGS cells were seeded onto 96-well plates at 1 × 103 per well and detected at 24, 48, 72, and 96 h. At the detected time points, MTT solution was added to react for 4 h. The formazan crystals were dissolved with 100 µL dimethyl sulfoxide per well. The absorbance was read using a microplate reader (Bio-Rad Laboratories, USA).
Colony formation
For colony formation assay, 500 of BGC-823 and AGS cells were seeded in triplicate in 60-mm dishes. At the detected time, the cells were washed with phosphate-buffered saline (PBS) for three times, fixed with 4% paraformaldehyde for 20 min before being stained with 0.1% crystal violet for 20 min. Finally, PBS was used to wash the dishes for three times. Photographs were taken and the cell clones were counted.
Scratch wounding migration assay
BGC-823 and AGS cells were grown in six-well culture plates. Cell layers were scraped with a sterile pipette tip. Photographs were taken at indicated time points. The migration of the cells was quantified by measurement of the distance between each wound edge.
Transwell chamber migration and invasion assay
Transwell chamber migration assay was performed using a transwell chamber with 8 µm filter inserts (BD Biosciences, USA). For transwell chamber invasion assay, transwell inserts with 8 µm filter were pre-coated with 50 µg of Matrigel (Becton Dickinson, USA). A volume of 5 × 104 cells were added to the upper chamber containing serum-free DMEM medium. The lower chamber was filled with 600 µL of DMEM medium containing FBS. Following the appropriate incubation period (migration: 24 h for AGS and BGC-823; invasion: 24 h for AGS and 48 h for BGC-823), the upper chamber was cleaned thoroughly, and the migrated or invasive cells were fixed with 4% paraformaldehyde for 20 min, stained with 0.1% crystal violet for 20 min. Photographs were captured, and the cell number was counted in at least three random fields.
Statistical analysis
Statistical analysis was performed using the SPSS 21.0 software. All data were represented as mean value ± standard deviation (SD). A two-tailed p < 0.05 was considered to be statistically significant.
Results
Expression of WISP-1 in GC tissues and GC cell lines
The expression levels of WISP-1 in normal and tumor tissues were diverse in different tumor types.6,12 WISP-1 was always upregulated in most of the cancer types, but some studies also reported that WISP-1 was expressed lower in tumor tissue from certain types of cancer. Therefore, it was necessary to identify the expression profile of WISP-1 in GC. We first assessed the expression level of WISP-1 mRNA in GC tissues by RT-PCR. WISP-1 mRNA was upregulated in GC tissues compared with its adjacent noncancerous tissues (Figure 1(a)). The mRNA and protein expression levels of WISP-1 were also examined in four GC cell lines by RT-PCR, real-time PCR, and Western blotting. The results were revealed in Figure 1(b) and Figure 1(c), WISP-1 mRNA was expressed in all GC cell lines and especially highly expressed in two GC cell types—BGC-823 and AGS. The protein expression level was correlated well with the mRNA expression level.
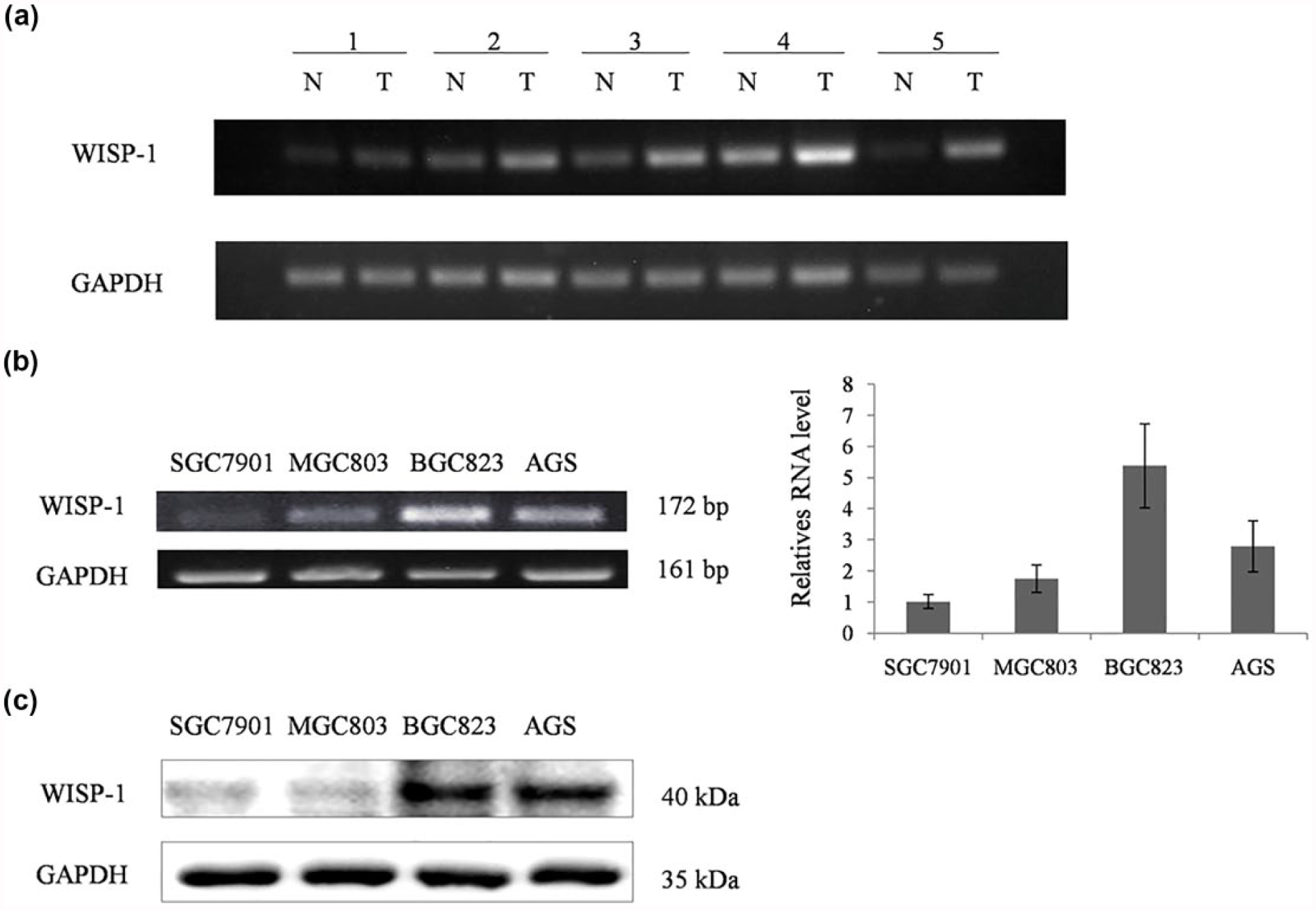
The expression of WISP-1 in GC tissues and GC cells. (a) The mRNA expression of WISP-1 in adjacent noncancerous tissues and GC samples. (b) The mRNA expression of WISP-1 in different GC cell lines by RT-PCR and real-time PCR. (c) The protein expression of WISP-1 in different GC cell lines by Western blotting.
Loss of WISP-1 reduced GC cell proliferation, migration and invasion
Aberrant cell proliferation was always a typical characteristic of the cancer occurrence. Therefore, in order to investigate the effect of WISP-1 on GC cell proliferation, we utilized a loss of function approach in two WISP-1 high expression GC cell types as mentioned above. The WISP-1 expression of two selected GC cell lines which were infected with a lentivirus expressing anti-WISP-1 small hairpin RNA (shRNA) was confirmed by RT-PCR, real-time PCR and Western blotting (Figure 2(a) and (b)). The effect of WISP-1 on proliferation of the two GC cell lines was examined through MTT assay. As shown in Figure 2(c), knockdown of WISP-1 significantly reduced the proliferation of BGC-823 and AGS cells as compared with their respective sh-control cells (p < 0.01). Moreover, to corroborate the above analysis, we performed colony formation assay and the result was consistent with the MTT assay (Figure 2(d)). Similar results were also found in breast cancer and acute lymphoblastic leukemia. 15 Previous research has confirmed that the expression of WISP-1 was related to the expression of cyclinD1, PCNA, and some other regulated factors that were involved in cell proliferation.9,16 However, our results have shown that decreased WISP-1 expression only significantly downregulated the expression of cyclinD1 but not the expression of PCNA in BGC-823 and AGS GC cells, which probably contributed to the reduced GC cell proliferation (Figure 2(e)).

Loss of WISP-1 reduced GC cell proliferation. (a) The mRNA expression of WISP-1 was significantly reduced in BGC-823 and AGS GC cells transfected with sh-WISP-1 and -2, detected by RT-PCR and real-time PCR. Sh-control served as the negative control. (b) The protein expression of WISP-1 was significantly decreased in BGC-823 and AGS GC cells transfected with sh-WISP-1-1 and sh-WISP-1-2, detected by Western blotting. (c and d) The MTT proliferation assay and colony formation assay revealed a growth inhibitory effect with the knockdown of WISP-1. (e) The mRNA expression of cyclinD1 and PCNA in WISP-1 knockdown BGC-823 and AGS GC cells detected by real-time PCR.
WISP-1 has been reported to be associated with the progression of various cancer cell migration, invasion, and metastasis, but its roles in regulating this series of biological progression were disparate. Recent researches revealed that WISP-1 inhibited non–small cell lung cancer cell migration and invasion, whereas it accelerated the cell invasion and metastasis of colorectal carcinoma and breast cancer.12,16,17 Therefore, to explore the role of WISP-1 in GC progression, we investigated the relationship of WISP-1 with migration and invasion, which were essential characteristics in mediating tumorigenesis. As expected, in scratch wounding assay, knockdown of WISP-1 significantly inhibited GC cell migration (Figure 3(a)), and this result was corresponding to the result which was demonstrated by the transwell chamber migration assay (Figure 3(b)). Furthermore, the effect of WISP-1 on GC cell invasion was also determined. As shown in Figure 3(c), transwell chamber invasion assay demonstrated that knockdown of WISP-1 in GC cells abolished the invasive ability of BGC-823 and AGS in comparison with the sh-control cells (p < 0.01).

WISP-1 promoted both GC cell migration and invasion. (a) Knockdown of WISP-1 reduced wound closure in scratch wounding migration assay in BGC-823 and AGS GC cells. Photographs were taken at 24 and 48 h, 12 and 24 h in BGC-823 and AGS GC cells, respectively. (b) Knockdown of WISP-1 decreased migration through transwell chamber migration assay. (c) WISP-1 knockdown inhibited the GC cell invasion ability through transwell chamber invasion assay.
WISP-1 accelerated GC cell migration and invasion through inducing epithelial-to-mesenchymal transition progression
The mechanism of WISP-1 on regulating cell migration, invasion, and metastasis was diverse according to the previous research reports in different cancer. On the basis of the above indicated results, we further utilized the WISP-1 knockdown GC cell model to test the potential molecular mechanism which might be mediated by WISP-1. The epithelial-to-mesenchymal transition (EMT) is one of the major complex processes, which always occurs in different kinds of cancer that promotes the progression of invasion and metastasis.18,19 Therefore, we detected the EMT-related molecules in WISP-1 knockdown GC cell models. As shown in Figure 4, the mRNA expression of Slug, Snail, Twist, Vimentin, and β-catenin was all significantly decreased following WISP-1 knockdown in BGC-823 GC cells compared with the sh-control cells. In addition, the same result was verified by AGS GC cells. In summary, these results suggested that one potential pathway of WISP-1 promoting GC cell migration and invasion was through upregulating EMT-related molecules.

WISP-1 accelerated GC cell migration and invasion by inducing EMT progression. The expression of EMT markers Slug, Snail, Twist, Vimentin, and β-catenin in WISP-1 knockdown and sh-control BGC-823 and AGS GC cells were analyzed by real-time PCR.
Discussion
GC is one of the most common types of cancer with poor clinical outcome prediction. In recent years, new approved drugs have been shown to reduce the progression of GC; however, the extensive application of such drugs is limited due to the little effect on halting or reversing the pathological changes in GC. Therefore, further understanding of the molecular mechanisms involved in GC development and progression is required for the emergence of novel therapeutic strategies. In previous studies, WISP-1 has already been identified as a member of the connective tissue growth factor and was a target of the Wnt/β-catenin pathway. 7 However, recent researches also revealed that the roles of WISP-1 in cancer occurrence and progression were diverse in different kinds of cancer. WISP-1 was recognized as an oncogene due to the fact that it could not only promote cancer cell growth through the activation of protein kinase B (Akt/PKB) but also attenuate p53-mediated apoptosis. Furthermore, WISP-1 was indicated to induce morphological transformation and to accelerate both cancer cell invasion and metastasis, resulting in more advanced tumor stage. 20 On the contrary, WISP-1 was also found to negatively regulate the progress of cell motility and invasion by the inhibition of Rac function through integrins in lung cancer.16,20 Although WISP-1 has been increasingly investigated in different types of cancer, because of the complicated functions and mechanisms of WISP-1 on mediating the occurrence and progression in different types of cancer, the accurate biological effect of WISP-1 on GC was still completely unrevealed in current research.
This study was the first report to explore the biological functions and potential mechanisms of WISP-1 in GC progression. We identified the expression patterns of WISP-1 in both the adjacent noncancerous tissues and GC tissues from GC patients. The results have shown that WISP-1 was upregulated in GC tissues compared with their adjacent noncancerous tissues, suggesting that WISP-1 acted as an oncogene in GC (Figure 1(a)). This result was supported by the previous studies on endometrial endometrioid adenocarcinoma, breast cancer, and some other tumor types.8,16 The associations of WISP-1 with cancer cell biological processes were also revealed in recent studies, but the exact biological role and the related molecular mechanism were disparate. Oral squamous cell carcinoma (OSCC) was one tumor type that clearly presented the molecular mechanisms which were mediated by WISP-1 in recent years. Chuang et al. 10 indicated that WISP-1 was involved in promoting OSCC cell migration by upregulating the expression of intercellular adhesion molecule 1 (ICAM1) through the activation of αvβ3 integrin receptor and signaling transduction pathways of apoptosis signal regulating kinase-1 (ASK-1), c-Jun N-terminal kinase/p38 (JNK/p38), and AP-1. Chuang et al. 21 supplemented that WISP-1 could also accelerate the OSCC development by inducing the expression of vascular endothelial growth factor A (VEGF-A). Moreover, WISP-1 was also demonstrated to enhance the migration progression in osteosarcoma, whereas the mechanism involved in this process was mainly via increasing the expression of matrix metalloproteinase 2 (MMP2) and MMP9 by the activation of the αvβ3 integrin receptor and extracellular–signal regulated protein kinase (ERK) or NF-κB signaling pathways—an alternative mechanism differed from the one revealed in OSCC. In our study, we found that WISP-1 promoted cell proliferation by altering the expression of cell growth–regulated protein factors such as cyclinD1 (Figure 2(c)–(e)). Similar results were found in the previous studies in melanoma, liver cancer, breast cancer, and acute lymphoblastic leukemia,15,22,23 whereas the molecular mechanisms that mediated this biological process were not the same. Chiang et al. 16 revealed that WISP-1 accelerated breast cancer cell proliferation mainly through skewing cells toward S phase by the downregulating P21 and P27 expression, whereas no significant alteration was found in the expression of cyclinD1 and cyclinE. However, WISP-1 was also proved to be mainly associated with the expression of cyclinD1 both in lung cancer and colorectal carcinoma, which was in agreement to our study in GC.9,17 Migration and invasion are essential characteristics of malignant tumor, which also indicate the malignant degree of tumor. Majority of the GC deaths were caused by local invasion and distant metastasis of tumor cells. Our result also identified that WISP-1 exactly enhanced GC cell migration and invasion by upregulating the expression of EMT molecules such as Slug, Snail, Twist, Vimentin, and β-catenin, which was not in line with the above mentioned molecular mechanisms involved in other tumor types (Figures 3 and 4). EMT is an important biological process that enables epithelial cells to break through the basal membrane and achieves increased potential for cell motility and invasion. This process is always accompanied by the aberrant expression of some characteristic genes and occurs in a large part of tumor types. Surprisingly, β-catenin, one of the important molecules during EMT progression, was also decreased in WISP-1 knockdown GC cells. Similar result was also found in the study performed by Chiang et al., 16 demonstrating the protein expression profile of WISP-1 in breast cancer. As revealed previously, WISP-1 was a downstream target gene regulated by Wnt-1 and β-catenin at the initiation of transcription level.7,24 Our result implied that WISP-1 probably downregulated β-catenin at transcription level and formed a negative feedback loop with β-catenin to enhance the occurrence and progression of migration and invasion through direct or indirect pathway in GC, nevertheless further study will be done to support this evidence.
In summary, we identified that WISP-1 was upregulated in GC tissues, and more importantly, we discovered multiple novel functional impacts of WISP-1 in GC cells, such as accelerating GC cell proliferation, migration, and invasion. Moreover, the molecular basis of WISP-1 on triggering these aggressive biological behaviors, migration, and invasion, was probably via promoting EMT progression. Our results first underscored the importance of fully elucidating the role of WISP-1 in GC progression and provided more clinical evidence for potential biomarker or therapeutic target for GC therapy.
Footnotes
Acknowledgements
S.J., T.Q., and M.F. contributed equally to this work and shared the first authorship.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Young Talents of Science and Technology Support Project of Colleges and Universities of Inner Mongolia Autonomous Region (NJYT-12-B21, 2012), the Great Project of the Affiliated Hospital of Inner Mongolia Medical University (NYFY ZD 2012014), the National Natural Science Foundation of China (81260363), Beijing Municipal Administration of Hospitals’ Youth Programme (QML20151003), the Project supported by National Science and Technology Ministry (2014BAI09B02), and Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201701).
Informed consent
Informed consent was obtained from all individual participants included in the study.
