Abstract
Long non-coding RNAs have been proved to be closely associated with different cancers. This study was designed to elucidate the function and mechanisms of colon cancer–associated transcript-1 in the progression of human laryngeal squamous cell cancer. Expressions of colon cancer–associated transcript-1, microRNA-218, and zinc finger protein, X-linked messenger RNA were measured using quantitative real-time polymerase chain reaction, and the expression level of zinc finger protein, X-linked protein was detected using western blot. Proliferation and invasion of laryngeal squamous cell cancer cell lines were detected by Cell Counting Kit-8 assay and Transwell invasion assay, respectively. Luciferase assay was used to confirm whether microRNA-218 is a target of colon cancer–associated transcript-1 and whether microRNA-218 directly binds to 3′-untranslated region of zinc finger protein, X-linked messenger RNA. Effect of colon cancer–associated transcript-1 on tumor growth was observed through xenograft mice models in vivo. The results showed that expressions of colon cancer–associated transcript-1 and zinc finger protein, X-linked were significantly higher while microRNA-218 expression was significantly lower in the laryngeal squamous cell cancer tissues than those in the adjacent normal tissues. MicroRNA-218 overexpression or zinc finger protein, X-linked silencing significantly suppressed proliferation and invasion of laryngeal squamous cell cancer cells. Moreover, knockdown of colon cancer–associated transcript-1 significantly inhibited proliferation and invasion of laryngeal squamous cell cancer cells, which were reversed by microRNA-218 downregulation or zinc finger protein, X-linked upregulation. Finally, colon cancer–associated transcript-1 silencing inhibited xenograft tumor growth of laryngeal squamous cell cancer in vivo. In conclusion, colon cancer–associated transcript-1 knockdown inhibits proliferation and invasion of laryngeal squamous cell cancer cells through enhancing zinc finger protein, X-linked by sponging microRNA-218, elucidating a novel colon cancer–associated transcript-1–microRNA-218–zinc finger protein, X-linked regulatory axis in laryngeal squamous cell cancer and providing a promising therapeutic target for laryngeal squamous cell cancer patients.
Keywords
Introduction
Laryngeal squamous cell cancer (LSCC) is one of the most commonly diagnosed malignancies in the head and neck region, 1 with a rapid increase in incidence and mortality all over the world. 2 Despite considerable improvements in the laryngeal carcinoma treatment, the survival rates remained unfavorable during the past three decades. 3 It was reported that the overall 1- and 2-year survival rates for LSCC patients without treatment are only 56.4% and 26.5%, respectively. 4 Frequent metastasis and recurrence are the feature of LSCC and the main factors resulting in treatment failure. 5 Therefore, it is required to develop effective therapeutic methods against LSCC and to understand the underlying mechanisms of laryngeal carcinogenesis.
MicroRNAs (miRNAs), a class of small ncRNAs, could promote messenger RNA (mRNA) degradation or suppress mRNA translation by interacting with the 3′-untranslated region (3′-UTR) of target mRNAs. 5 Increasing evidence has demonstrated that miRNAs are aberrantly expressed and function as oncogenes or tumor suppressor genes involved in the cell proliferation, apoptosis, development, and differentiation in almost all cancers.6,7 Among these cancer-associated miRNAs, microRNA-218 (miR-218) is served as an anti-cancer gene and generally downregulated in different types of cancers, such as breast cancer, 8 colorectal cancer, 9 and gastric cancer. 10 Growing documents have demonstrated that abnormal expression of miR-218 is closely associated with the proliferation, apoptosis, metastasis, invasion, and resistance in cancers. 11 For instance, miR-218 expression level was decreased in prostate cancer cells and miR-218 overexpression obstructed interleukin (IL)-6-induced cell proliferation and invasion of prostate cancer cells via directly targeting LGR4 and modulating the phosphatidylinositol-3 kinase (PI3K)/Akt and Wnt/β-catenin signaling pathways. 12 MiR-218 suppressed the proliferation, migration, and invasion of LoVo colon cancer cells by targeting the PI3K/Akt/mTOR signaling pathway and matrix metalloproteinase (MMP)-9. 13 However, the function and mechanism of miR-218 in LSCC remain unclear.
Long noncoding RNAs (lncRNAs) are a group of non-coding RNA molecules with length longer than 200 nucleotides, which are involved in normal development, differentiation, as well as the tumorigenesis process.14,15 LncRNAs exert their physiological and pathological functions through their interactions with genomic DNA, miRNAs, mRNAs, and proteins. 16 Several lncRNAs have been identified to act as miRNA sponges involved in LSCC.17,18 For example, lncRNA H19 was upregulated in LSCC and promoted LSCC migration, invasion, and proliferation through releasing DNA methyltransferase-1 (DNMT1) by sponging miR-148a-3p. 17 However, this particular function of lncRNAs involved in LSCC progression is largely elusive.
Colon cancer–associated transcript-1 (CCAT1), also known as cancer-associated region long noncoding RNA-5 (CARLo-5) or CCAT1-S, is a newly discovered lncRNA with 2628 base pairs in length, which is located on chromosome 8q24.21 and in the vicinity of c-MYC, a well-known transcription factor.19,20 CCAT1 has been demonstrated to be upregulated and act as an oncogenic lncRNA in different types of cancer, including colon cancer, 21 breast cancer, 22 and hepatocellular carcinoma (HCC). 23 Moreover, CCAT1 has been found to be upregulated and promote the proliferation, migration, and invasion of LSCC cells. 24 However, the concrete mechanism by which CCAT1 exerts its role in LSCC cells remains obscure.
In this study, the biological function of lncRNA CCAT1 in LSCC cells and its possible molecular mechanisms were explored.
Materials and methods
Tissue samples and cell lines
Fresh LSCC tissues and adjacent non-cancer tissues were gained from 43 histopathologically confirmed LSCC patients receiving surgical removal in Huaihe Hospital of Henan University. Patients enrolled in this study had no treatment history. Harvested tissues were quickly frozen in liquid nitrogen and preserved at −80°C for subsequent experiments. Informed written consents from all participants and approval of Huaihe Hospital Research Ethic Committees were obtained prior to the use of these clinical samples.
Two human LSCC cell lines, AMC-HN-8 and TU212, were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). Both cell lines were maintained in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, Carlsbad, CA, USA) with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin at 37°C/5% CO2.
Quantitative real-time polymerase chain reaction
TRIzol reagent (Invitrogen, Carlsbad, CA, USA) was used to extract the total RNA from LSCC cell lines or tissues. RevertAid Reverse Transcriptase (Thermo Fisher Scientific, Waltham, MA, USA) was used for synthesis of complementary DNA (cDNA) of mRNA and long non-coding RNA (lncRNA) by Oligo (dT18) RT primer, as well as cDNA of miR-128 by Stem-loop RT primer. mRNA and lncRNA expression was detected by a SYBR Premix Ex Taq™ kit (Takara Bio, Otsu, Japan), while miRNA levels were quantified using TaqMan miRNA assays (Applied Biosystems, Foster City, CA, USA). qRT-PCR reactions were conducted on the 7500 Real Time PCR System (Applied Biosystems). β-actin and U6 were used as endogenous controls.
Western blot analysis
Total protein was isolated from cells or tissues using radioimmunoprecipitation assay (RIPA) reagents (Thermo Scientific, Rockford, IL, USA). Protein samples were separated by 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and then electrotransferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Bedford, MA, USA). The membranes were blocked with skim milk and incubated with primary antibodies against zinc finger protein, X-linked (ZFX; Sigma-Aldrich, St. Louis, MO, USA), PCNA (Santa Cruz Biotechnology, Santa Cruz, CA, USA), MMP-2 (Santa Cruz Biotechnology), MMP-7 (Santa Cruz Biotechnology), and β-actin (Cell Signaling Technology, Danvers, MA, USA) overnight at 4°C, followed by treatment with secondary antibody conjugated to horseradish peroxidase for 2 h at room temperature. The protein bands were visualized and quantified by the ImageQuant software (Molecular Dynamics, Sunnyvale, CA, USA).
Cell transfection
MiR-218 mimics (miR-218), miR-218 inhibitor (anti-miR-218), and small interfering RNA against ZFX (si-ZFX) and CCAT1 (si-CCAT1) were purchased from GenePharma (Shanghai, China). The pcDNA-CCAT1 and pcDNA-ZFX expression plasmids were obtained from GeneChem (Shanghai, China). Their respective controls (miR-control, si-control, and pcDNA-control) were also designed. Sh-CCAT1 and sh-control were synthesized by System Biosciences (Mountain View, CA, USA). All transfections were performed by Lipofectamine 2000 reagent (Invitrogen) according to the manufacturer’s protocol.
Cell proliferation assay
Cell proliferation was determined by Cell Counting Kit-8 (CCK-8; Beyotime, Jiangsu, China). AMC-HN-8 and TU212 cells (5000 per well) were seeded in the 96-well plates and maintained for 48 h. Then, 10 µL CCK-8 solutions were added to each well for co-culture for another 1 h. A Bio-Rad Microplate Reader (Bio-Rad, Hercules, CA, USA) was used to assess the cell viability through absorbance detection at the 450 nm.
Cell invasion assay
To measure cell invasion, Transwell invasion chamber (Bectin Dickinson, Bedford, MA, USA) was used. Cells (2 × 105) cultured in serum-free media were placed into the top compartment coated with 10 µg/mL Matrigel. Culture medium containing 10% FBS as a chemoattractant was added into the lower chamber. After 24 h, cells on the upper side of the membrane were removed with a cotton swab. Cells migrating to the lower surface of the membrane were stained with crystal violet and counted under a microscope (Olympus, Tokyo, Japan).
Luciferase reporter assays
To verify the interaction between miR-218 and CCAT1, the pMIR-report luciferase vectors containing the binding sequences of wild-type (WT) or mutant CCAT1 fragments on miR-218, named pMIR-CCAT1-WT or pMIR-CCAT1-MUT, were constructed and co-transfected with miR-control or miR-218 into AMC-HN-8 cells. To confirm ZFX was a direct target of miR-218, the WT or mutant 3′-UTR (3′-UTR-WT or 3′-UTR-MUT) of ZFX was amplified and inserted into the luciferase reporter vectors. Then, AMC-HN-8 cells were co-transfected with miR-control or miR-218 or (miR-218 + pcDNA-control) or (miR-218 + pcDNA-CCAT1) and ZFX-3′-UTR-WT or ZFX-3′-UTR-MUT reporter vectors. The luciferase activity was measured using the dual-Luciferase reporter assay system (Promega, Madison, WI, USA) 48 h after transfection.
In vivo study
Male athymic BALB/c mice (6-week-old) were purchased from Shanghai Experimental Animal Center (Shanghai, China). All mice were maintained in aseptic environment with a steady temperature and humidity. For xenograft analysis, total 3 × 106 AMC-HN-8 cells stably transfected sh-CCAT1 or sh-control were subcutaneously injected into the flanks of these nude mice (
Statistical analysis
Student’s
Results
CCAT1 and ZFX expressions are upregulated, and miR-218 expression is downregulated in LSCC tissues
The expression levels of CCAT1, miR-218, and ZFX mRNA and ZFX protein level were measured using 45 pairs of LSCC tissues and adjacent non-cancerous tissues. The results showed that CCAT1 expression in LSCC tissues was significantly higher than normal counterparts (Figure 1(a)). As shown in Figure 1(b), miR-218 was significantly decreased in LSCC tissues compared to normal controls. ZFX mRNA (Figure 1(c)) and protein (Figure 1(d)) were significantly upregulated in LSCC tissues compared with adjacent non-neoplastic tissues. Moreover, CCAT1 expression is negatively related to miR-218 expression (Figure 1(e)) and positively correlated with ZFX level (Figure 1(f)) in LSCC tissues. These results suggested that the dysregulated expression of CCAT1, miR-218, and ZFX may be associated with the tumorigenesis of LSCC.

Expression levels of CCAT1, miR-218, and ZFX in LSCC tissues. (a, b, and c) qRT-PCR analysis of CCAT1, miR-218, and ZFX expression in LSCC tumor tissues (
MiR-218 overexpression inhibits proliferation and invasion of LSCC cell lines
In order to investigate the role of miR-218 in LSCC progression, AMC-HN-8 and TU212 cells were transfected with miR-control or miR-218. CCK-8 assay revealed that miR-218 overexpression resulted in a significant decrease in cell viability in AMC-HN-8 (Figure 2(a)) and TU212 cells (Figure 2(c)) compared with the control groups. Moreover, the expression of proliferation-related protein PCNA was obviously suppressed by transfection of miR-218 into AMC-HN-8 (Figure 2(b)) and TU212 cells (Figure 2(d)). Transwell invasion assay was also performed in AMC-HN-8 and TU212 cells transfected with miR-218 mimics. Compared with the control groups, ectopic expression of miR-218 significantly hindered cell invasion in AMC-HN-8 (Figure 2(e)) and TU212 cells (Figure 2(g)). Additionally, overexpression of miR-218 dramatically downregulated the expression of invasion-related proteins MMP-2 and MMP-7 in AMC-HN-8 (Figure 2(f)) and TU212 cells (Figure 2(h)). Taken together, these results suggested that upregulation of miR-218 expression led to an inhibition of proliferation and invasion in LSCC cell lines.
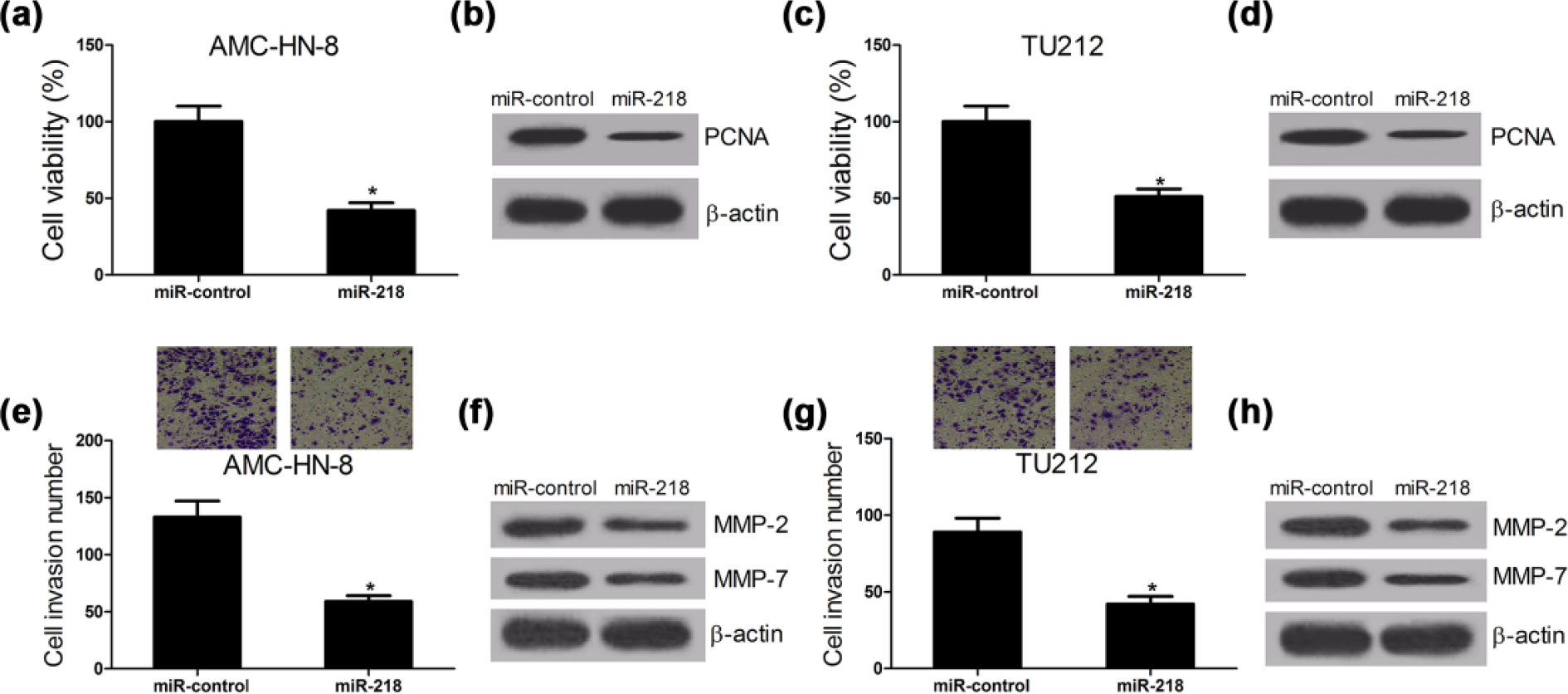
MiR-218 overexpression hinders proliferation and invasion of LSCC cell lines. AMC-HN-8 and TU212 cells were transfected with miR-control or miR-218. (a and c) CCK-8 assay was performed to detect the cell viability 48 h after transfection in AMC-HN-8 and TU212 cells. (b and d) Western blot was conducted to measure the proliferation-related protein PCNA in AMC-HN-8 and TU212 cells. (e and g) Cell invasion was determined by Transwell assay 24 h post transfection in AMC-HN-8 and TU212 cells. (f and h) The level of invasion-related proteins MPP-2 and MMP-7 in AMC-HN-8 and TU212 cells was detected by western blot (*
ZFX knockdown suppresses proliferation and invasion of LSCC cell lines
Based on the elevated expression of ZFX in LSCC tissues, si-ZFX was transfected into AMC-HN-8 and TU212 cells to downregulate ZFX expression. CCK-8 assay was performed to explore the effect of ZFX on the proliferation of LSCC cells. The results demonstrated that the cell viability of AMC-HN-8 and TU212 cells was significantly inhibited by ZFX silencing compared with the control groups (Figure 3(a)). Similarly, ZFX downregulation decreased the expression of PCNA in AMC-HN-8 and TU212 cells (Figure 3(b)). Transwell assay showed that si-ZFX-transfected AMC-HN-8 and TU212 cells had a significantly lower invasion ability than si-control-transfected cells (Figure 3(c)). Consistently, ZFX depletion resulted in a prominent repression in expression MMP-2 and MMP-7 in AMC-HN-8 and TU212 cells (Figure 3(d)). Collectively, these results revealed that ZFX knockdown hindered proliferation and invasion in LSCC cell lines.
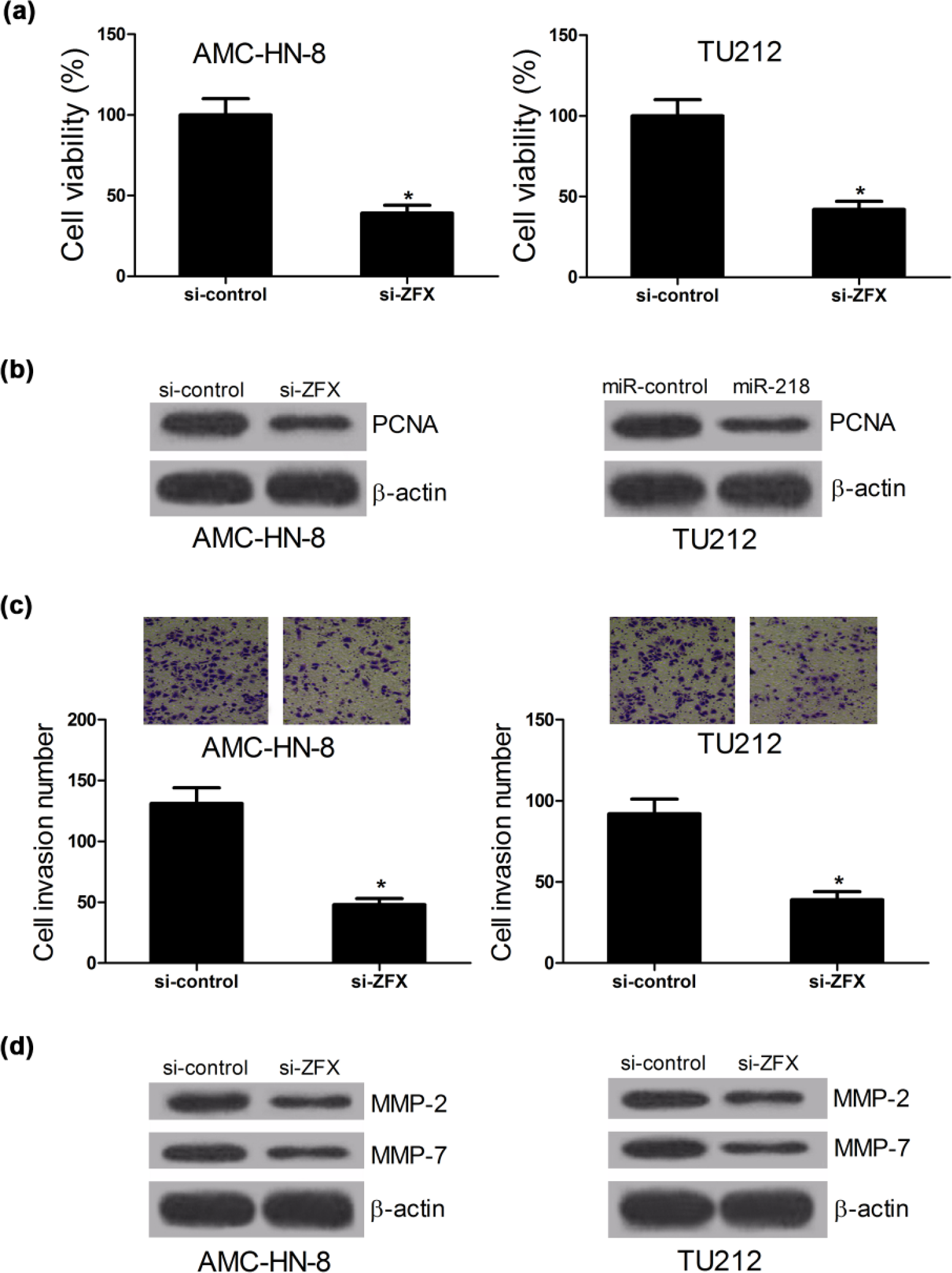
ZFX knockdown obstructs proliferation and invasion of LSCC cell lines. AMC-HN-8 and TU212 cells were transfected with si-control or si-ZFX. (a) CCK-8 assay of cell viability 24 h after transfection in AMC-HN-8 and TU212 cells. (b) Western blot assay of PCNA expression in AMC-HN-8 and TU212 cells. (c) Transwell assay of cell invasion ability 24 h post transfection in AMC-HN-8 and TU212 cells. (d) Western blot assay of MPP-2 and MMP-7 in AMC-HN-8 and TU212 cells (*
MiR-218 inhibition reverses the effects of CCAT1 knockdown on proliferation and invasion in LSCC cell lines
LncRNAs have been illuminated to contain complementary binding sites of miRNAs and suppress miRNAs expression and activity. 25 Online database starBase v2.0 was used to predict potential lncRNA–miRNA interactions. The results presented that CCAT1 contains one conserved binding site of miR-218 (Figure 4(a)). Then, dual-luciferase reporter assay was performed to verify the mutual effect between miR-218 and CCAT1 in AMC-HN-8 cells. Results demonstrated that miR-218 overexpression was able to significantly suppress the luciferase activity of the WT reporter (CCAT1-WT), whereas mutant reporter gene (CCAT1-MUT) showed no obvious difference in luciferase expression compared with miR-control in AMC-HN-8 cells (Figure 4(b)). To clarify the direct interaction between CCAT1 and miR-218, qRT-PCR was performed to detect the miR-218 expression in si-CCAT1 or pcDNA-CCAT1 transfected AMC-HN-8 cells. As expected, CCAT1 knockdown significantly improved miR-218 expression, while enforced expression of CCAT1 significantly lowered expression of miR-218 (Figure 4(c)). All these data imply that CCAT1 may act as a regulator of miR-218. To further confirm the functional effect of CCAT1 in the progression of LSCC cells, AMC-HN-8 cells were transfected with either si-CCAT1 or in combination with anti-miR-218. CCK-8 assay and western blot revealed that CCAT1 knockdown triggered a significant decrease in cell viability (Figure 4(d)) and a notable suppression in PCNA expression (Figure 4(e)) of AMC-HN-8; however, miR-218 inhibition upon anti-miR-218 reversed these effects. Transwell invasion assay demonstrated that CCAT1 knockdown mediated by si-CCAT1 significantly repressed the invasion ability of AMC-HN-8 cells; however, downregulation of miR-218 abolished this inhibitory effect of CCAT1 depletion (Figure 4(f)). Likewise, anti-miR-218 attenuated the inhibition of MPP-2 and MPP-7 levels caused by CCAT1 knockdown (Figure 4(g)). All these data suggested that CCAT1 promoted proliferation and invasion of LSCC cells through suppressing miR-218 expression.

MiR-218 downregulation reversed the inhibitory effects of CCAT1 knockdown on proliferation and invasion in LSCC cell lines. (a) Putative miR-218 binding sequence of CCAT1 RNA is shown. (b) The relative luciferase activity was detected in AMC-HN-8 cells co-transfected with CCAT1-WT or CCAT1-MUT and miR-218 mimic or miR-control. (c) qRT-PCR was performed to measure the miR-218 expression in AMC-HN-8 transfected with pcDNA-control or pcDNA-CCAT1. AMC-HN-8 cells were transfected with either si-CCAT1 or combined with anti-miR-218. (d) CCK-8 assay was performed to determine cell viability. (e) The level of PCNA was detected by western blot. (f) Transwell assay was conducted to test cell invasion ability. (g) Western blot was performed to measure MPP-2 and MMP-7 expression (*
CCAT1 promotes proliferation and invasion of LSCC cells through releasing ZFX by inhibiting miR-218 in LSCC cell lines
According to the online bioinformatic software TargetScan, it is found that there existed a conserved binding sequence of miR-218 on 3′-UTR of ZFX (Figure 5(a)). Dual-luciferase reporter assay implied that overexpression of miR-218 significantly refrained the luciferase activity of ZFX-3′-UTR-WT reporter vector in AMC-HN-8 cells, while transfection of pcDNA-CCAT1 generated a reversion of luciferase activity (Figure 5(b)). However, there were no apparent changes in luciferase activity of ZFX-3′-UTR-MUT reporter vector when AMC-HN-8 cells were transfected with miR-218 or combined with pcDNA-CCAT1. Further western blot analysis indicated that compared with the controls, ZFX expression was significantly downregulated with either CCAT1 knockdown or miR-218 overexpression, while CCAT1 overexpression induced a significant increase in ZFX level. Moreover, co-transfection of CCAT1 abrogated miR-218-mediated inhibition in ZFX expression (Figure 5(c)). In summary, these data suggested that CCAT1 regulated ZFX expression via modulating miR-218. To further confirm the regulatory role of CCAT1-miR-218-ZFX axis in cell proliferation and invasion, rescue experiments were performed in AMC-HN-8 cells. As presented in Figure 5(d), CCAT1 knockdown led to a significant inhibition in cell viability, while this effect was abated by simultaneous CCAT1 knockdown and ZFX overexpression. Moreover, ZFX overexpression overturned the decrease in PCNA level induced by CCAT1 knockdown (Figure 5(e)). Transwell invasion assay confirmed that ZFX overexpression reversed the inhibitory effect of CCAT1 downregulation on AMC-HN-8 cell invasion capacity (Figure 5(f)). Moreover, pcDNA-ZFX could recover MPP-2 and MPP-7 expression inhibited by CCAT1 depletion (Figure 5(g)). All these data suggested that CCAT1 promoted proliferation and invasion of LSCC cells through serving as a competing endogenous RNA (ceRNA) for miR-218, thereby changing expression of its target ZFX.

CCAT1 enhances proliferation and invasion of LSCC cells through increasing ZFX expression by sponging miR-218 in LSCC cells. (a) Binding sequence of wild-type (WT) 3′-UTR of ZFX on miR-218 are presented. (b) Luciferase activity analysis in AMC-HN-8 cells co-transfected with WT or Mut ZFX-3′-UTR and miR-218 mimic or miR-218 mimic + pcDNA-CCAT1. (c) Western blot was performed to detect the ZFX expression in AMC-HN-8 cells transfected with pcDNA-CCAT1, si-CCAT1, and miR-218 or co-transfected with miR-218 and pcDNA-CCAT1. AMC-HN-8 cells were transfected with si-CCAT1 or in combination with pcDNA-ZFX. (d) CCK-8 assay of cell viability. (e) Western blot of PCNA expression. (f) Transwell assay of cell invasion. (g) Western blot analysis of MPP-2 and MMP-7 expression (*
CCAT1 knockdown suppresses tumorigenesis of LSCC cells in vivo
To explore the oncogenic role of CCAT1 in LSCC in vivo, a xenograft mice model was constructed. AMC-HN-8 cells with sh-control or sh-CCAT1 transfection were injected subcutaneously into nude mice. The tumor growth was significantly inhibited by CCAT1 silencing compared to the negative control group, evidenced by significantly reduced tumor volume (Figure 6(a)) and weight (Figure 6(b)). Furthermore, a significant increase in miR-218 expression (Figure 6(c)) and a dramatic decrease (Figure 6(d)) in ZFX expression were observed in sh-CCAT1-treated LSCC xenografts compared with the control group. Together, CCAT1 depletion impeded LSCC tumor growth in vivo.

CCAT1 silencing suppresses LSCC tumor growth in vivo. Mice were inoculated with AMC-HN-8 cells transfected with sh-control or sh-CCAT1. (a) Tumor growth curves were depicted. (b) Tumor weight was measured. (c and d) Expressions of miR-218 and ZFX in resected tumors (*
Discussion
In this study, the function of lncRNA CCAT1 in LSCC cells was explored. CCAT1 was significantly upregulated in LSCC tissues compared with the adjacent non-cancer tissues, indicating that CCAT1 may act as an oncogene in LSCC. Previous documents revealed that CCAT1 promoted cell proliferation and invasion in colon cancer and HCC.21,26 Similarly, this study found that CCAT1 silencing exerted inhibitory effects on the proliferation and invasion of LSCC cells. A previous study reported that CCAT1 was overexpressed in LSCC tissues and knockdown of CCAT1 promoted the proliferation and invasion of Hep-2 cell, 22 which was consistent with the findings in this study. These findings suggested that CCAT1 could act as an oncogene and stimulate the progression of LSCC, providing a promising diagnostic biomarkers and therapeutic target for LSCC.
It has been illuminated that lncRNAs could act as ceRNAs to involve in post-transcriptional regulation by interaction with miRNA.27,28 For instance, LncRNA urothelial carcinoma-associated 1 (UCA1) could promote the growth and metastasis of HCC cell lines through the activation of extracellular signal–regulated protein kinase (ERK) signaling pathway by sponging miR-216b and releasing miR-216b target fibroblast growth factor receptor 1 (FGFR1). 29 LncRNA metastasis-associated lung adenocarcinoma transcription 1 (MALAT1) could act as a sponge of miR-124 to control the cyclin-dependent kinase 4 (CDK4) expression in breast cancer. 30 In this study, CCAT1 functioned as a sponge of miRNA, participating in post-transcriptional regulation of target mRNA in the development of LSCC. Considering that CCAT1 RNA contains one conserved seed sequence of miR-218 (Figure 4(a)) and that the expression levels of CCAT1 and miR-218 were inverse in LSCC tissues (Figure 1(a) and (b)), CCAT1 might act as an miRNA sponge to de-repress miR-218 expression. The interaction of miR-218 and CCAT1 was confirmed by dual-luciferase reporter analysis in this study. Moreover, CCAT1 silencing could significantly boost miR-218 expression, and CCAT1 upregulation could significantly lower miR-218 expression. All these results indicated that CCAT1 could act as an endogenous sponge, blocking miR-218 expression. Previous studies also indicated that CCAT1 could function as miRNA sponges in different cancers, such as miR-155 in Acute Myeloid Leukemia and let-7 in HCC.23,31
ZFX is a member of the zinc finger protein super family, which was originally identified as a critical regulator of self-renewal in both embryonic and hematopoietic stem cells. 32 Emerging evidence suggested that ZFX was a novel oncoprotein frequently overexpressed in human malignancies, including hepatocellular, renal, and breast carcinoma.33–35 In addition, ZFX has been found to be involved in the proliferation and metastasis of human gallbladder cancer and non-small-cell lung cancer.36,37 ZFX was upregulated in LSCC tissues, and ZFX silencing inhibited cell proliferation and migration and induced apoptosis of LSCC cells.38,39 However, how ZFX is regulated in LSCC remains unclear. In this study, ZFX was verified to be a target of miR-218 by dual-luciferase reporter analysis. CCAT1 overexpression abrogated the suppression of luciferase activity of the ZFX 3′-UTR induced by miR-218 as well as abolished the inhibitory effect of miR-218 on ZFX expression. These data suggested that CCAT1 could lead to the de-repression of ZFX by sponging miR-218. Furthermore, CCAT1 knockdown significantly inhibited proliferation and invasion ability of LSCC cell lines, which were reversed by miR-218 inhibition or ZFX overexpression. Taken together, CCAT1 could promote proliferation and invasion of LSCC cells through regulating ZFX mediated by miR-218. Therefore, these findings provide a new CCAT1-miR-218-ZFX clue for understanding the pathogenesis of LSCC. In acute myeloid leukemia, CCAT1 repressed monocytic differentiation and promoted cell growth of HL-60 by sequestering miR-155 and releasing c-Myc. 31 CCAT1 was also implied to promote the proliferation and migration of HCC cells through mediating HMGA2 and c-Myc by sponging let-7. 23 All these studies suggested that CCAT1 could function as an miRNA sponge to release the miRNA targets in cancers.
In summary, CCAT1 contributed to the occurrence and development of LSCC. Moreover, a novel CCAT1-miR-218-ZFX regulatory axis was demonstrated in the proliferation and invasion of LSCC cells in vitro and in vivo. This study gave further evidence of lncRNA as a ceRNA to enhance target gene of miRNA via directly suppressing miRNA, thus facilitating the progression of LSCC.
Footnotes
Acknowledgements
Y.Z. and H.H. have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was obtained from Huaihe Hospital of Henan University
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
