Abstract
Circular RNAs are a special class of endogenous RNAs characterized by jointing 3′ and 5′ ends together via exon or intron circularization. Recent studies found that circular RNAs are involved in the development of some human diseases. However, little is known about their roles in human gastric cancer. In this study, we chose hsa_circ_0001895 as a targeted circRNA to investigate its clinical significances in gastric cancer patients. Hsa_circ_0001895 expression levels in five gastric cancer cell lines and 257 specimens of tissues were measured by real-time quantitative reverse transcription polymerase chain reaction. Then, the potential relationship between hsa_circ_0001895 expression levels and patients’ clinicopathological factors was investigated. A receiver operating characteristic curve was constructed for evaluating the diagnostic value of hsa_circ_0001895. Hsa_circ_0001895 expression levels in five detected gastric cancer cell lines (AGS, BGC-823, HGC-27, MGC-803, and SGC-7901) were all significantly downregulated than those in normal gastric epithelial GES-1 cells. Besides, compared with healthy control tissues, it was downregulated not only in 69.8% (67/96) gastric cancer tissues but also in gastric precancerous lesions. Moreover, hsa_circ_0001895 expression levels were significantly correlated with cell differentiation, Borrmann type, and tissue carcino-embryonic antigen expression. Our results suggested that hsa_circ_0001895 may play crucial roles during gastric cancerogenesis and is a potential biomarker for clinical prognosis prediction.
Introduction
Gastric cancer is one of the most common malignant tumors in human digestive system. It remains the second leading cause of cancer-related deaths worldwide. 1 Although there are several clinical treatment options to gastric cancer, patients’ prognosis is still poor especially at tumor advanced stage. The 5-year overall survival rate of gastric cancer patients is still <25%. 2 This is mainly due to the less understanding of cancer heterogeneity and absence of desirable biomarkers for early detection.3,4 Thus, identifying molecular characterizations and searching new targets for diagnosis and treatment of gastric cancer should be a priority of current research.
Circular RNAs (circRNAs) are a special class of endogenous RNAs characterized by jointing 3′ and 5′ends together via exon or intron circularization. 5 Compared to linear RNAs, circRNAs are more stable and conserved than linear RNAs. 6 Majority of circRNAs are resistant to exonucleases and often exhibit tissue and developmental stage–specific expression.6,7 Recent studies have found that circRNAs can participate in the RNR–RNA regulation network or RNA–protein complex formation and thus are involved in the development of some human diseases.8,9 However, still little is known about their roles in human gastric cancer.
Hsa_circ_0001895 (Alias, hsa_circ_000760; http://circrna.org/) is one of circRNAs with 348 nt in spliced sequence length. Its gene is located at chr9:134305476-134308181, and its associated gene symbol is PRRC2B (proline-rich coiled-coil 2B). In this study, we chose hsa_circ_0001895 as a targeted circRNA in gastric cancer because it is one of circRNAs that may be associated with gastric cancer according to the bioinformatics analysis in CircBase database.
Based on the above information, we first verified hsa_circ_0001895 expression levels in gastric cancer cell lines and gastric cancer tissues. Then, its expression levels in tissue with various stages of gastric tumorigenesis were explored, and the potential relationship between hsa_circ_0001895 expression levels and patients’ clinicopathological factors was investigated. Moreover, a receiver operating characteristic (ROC) curve was constructed for evaluating its diagnostic value. Our data indicate that hsa_circ_0001895 plays crucial roles during gastric cancerogenesis and may be a potential biomarker for screening and predicting prognosis of gastric cancer.
Material and methods
Specimens and clinical information collection
Evaluated by clinical gastroenterologists, a total of 257 specimens were collected from the Affiliated Hospital of Ningbo University School of Medicine, China, from November 2014 to February 2016. The paired 96 gastric cancer tissues and their adjacent non-tumorous tissues 5 cm from the edge of tumor were collected from surgical patients. Another 35 healthy gastric mucosa and 30 gastric dysplasia (GD) tissues were obtained from biopsy specimens. All specimens were immediately immersed into RNA fixer (Bioteke, Beijing, China) and stored at −80°C until use.
All specimens in this study were histopathologically confirmed. Tumors were staged according to the tumor–node–metastasis (TNM) staging system (7th ed.). Histological grade was assessed following the National Comprehensive Cancer Network (NCCN) clinical practice guideline of oncology (V.1.2012). No patient received treatment before upper gastrointestinal endoscopy or surgical excision. This study was approved by the Human Research Ethics Committee of Ningbo University. Written informed consents were obtained from all participants.
Total RNA extraction and quantitative reverse transcription polymerase chain reaction detection
Total RNA was extracted from tissue and cell samples by TRIzol reagent (Ambion, Carlsbad, CA, USA), following the manufacturer’s instruction. Then, complementary DNA (cDNA) was synthesized by random primers and the GoScript Reverse Transcription (RT) System (Promega, Madison, WI, USA). The conditions of RNA reverse transcription were as follows: incubation at 42°C for 1 h, 70°C for 10 min, and 4°C for forever. No template reaction was used as the control at the same time.
Real-time quantitative reverse transcription polymerase chain reaction (qRT-PCR) was achieved by using GoTaq qPCR Master Mix (Promega) on an Mx3005P real-time PCR System (Stratagene, LaJolla, CA, USA). Divergent primers of hsa_circ_0001895 were designed and synthesized by Sangon Biotech (Shanghai) Co., Ltd. The sequences of hsa_circ_0001895 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) primers were as follows: 5′-GCAAACAAGCAGGATCAGCAA-3′ and 5′-CCCATCCTTGCCCTTGGTAA-3′ for hsa_circ_0001895; 5′-ACCCACTCCTCCACCTTTGAC-3′ (sense) and 5′-TGTTGCTGTAGCCAAATTCGTT-3′ (antisense) for GAPDH. The conditions of thermal cycling were as follows: 95°C at 10 min for a hot-start, then 45 cycles at 94°C for 15 s, 55°C for 30 s, and 72°C for 30 s. The qRT-PCR products of hsa_circ_0001895 were confirmed by sequencing; and its expression levels were calculated by the ΔCt method with GAPDH as the control. 10 Larger ΔCt values indicate lower expression. All data in this study were expressed as the mean ± standard deviation (SD) of three independent experiments.
Cell culture
Normal human gastric mucosa epithelial cell line (GES-1) and five gastric cancer cell lines (AGS, BGC-823, HGC-27, MGC-803, and SGC-7901) were purchased from Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, China). All cells were cultured in RPMI-1640 Medium (Life Technologies, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (Life Technologies) at 37°C in a humidified atmosphere with 5% CO2.
Statistical analysis
Statistical data were analyzed by Statistical Product and Service Solutions (SPSS) 20.0 software (SPSS, Chicago, IL, USA). GraphPad Prism 5.0 (GraphPad Software, LaJolla, CA, USA) software was used to draw graphs. SigmaPlot12.3 (Systat Software, San Jose, CA, USA) was used to construct ROC curve. Student’s t-test, one way analysis of variance (ANOVA) test, and rank-sum test were flexible and were used as appropriate. The value of p < 0.05 was regarded as statistically significant.
Results
Hsa_circ_0001895 expression in gastric cell lines and tissues
To verify the bioinformatics analysis results of hsa_circ_0001895, we first used qRT-PCR method to detect hsa_circ_0001895 expression levels in gastric cancer cell lines and tissues. Our results showed that hsa_circ_0001895 expression levels in five gastric cancer cell lines (AGS, BGC-823, HGC-27, MGC-803, and SGC-7901) were all significantly downregulated than those in normal gastric epithelial GES-1 cells (Figure 1). Similar to the situations in cell lines, its expression levels were downregulated in 69.8% (67/96) gastric cancer tissues compared with the paired adjacent normal tissues (p < 0.001; Figure 2).
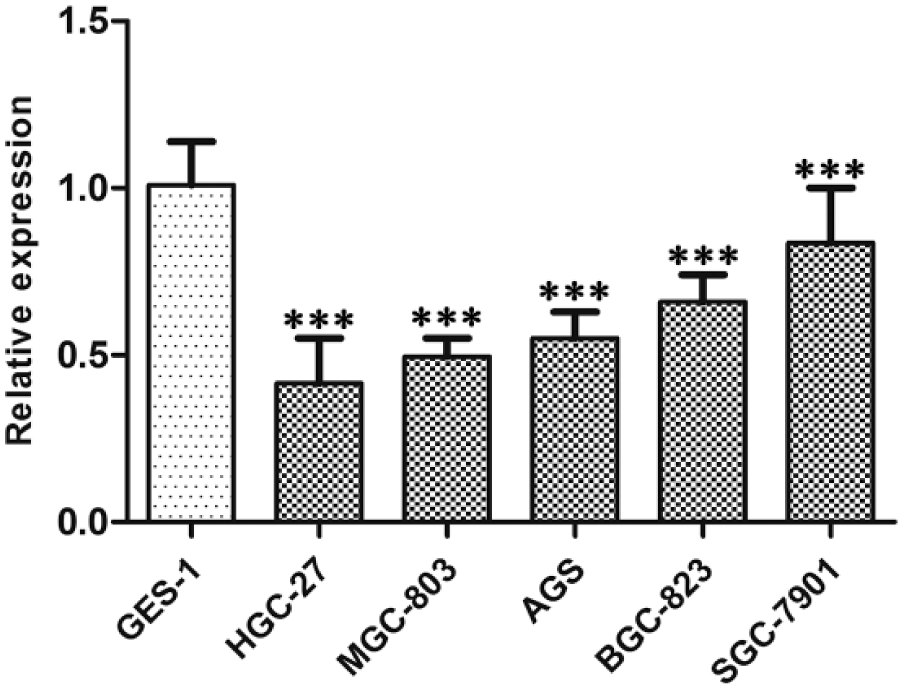
Relative expression of hsa_circ_0001895 in gastric cancer cell lines. Hsa_circ_0001895 expression levels in five gastric cancer cell lines (AGS, BGC-823, HGC-27, MGC-803, and SGC-7901) and normal gastric epithelial cell line (GES-1) were determined by qRT-PCR. Data are expressed as mean ± SD of three independent experiments. Asterisks indicate values that are significantly different when compared with that in GES-1. ***p < 0.001.

Hsa_circ_0001895 expression in gastric cancer tissues and adjacent normal tissues. Hsa_circ_0001895 expression in gastric cancer tissues compared with adjacent normal tissues (n = 96, ***p < 0.001). Data are expressed as the mean ± SD of three independent experiments.
Then, we explored hsa_circ_0001895 expression levels in the tissues from various stages of gastric carcinogenesis. As shown in Figure 3, compared with healthy control group, hsa_circ_0001895 expression level was significantly decreased in GD and gastric cancer tissues.
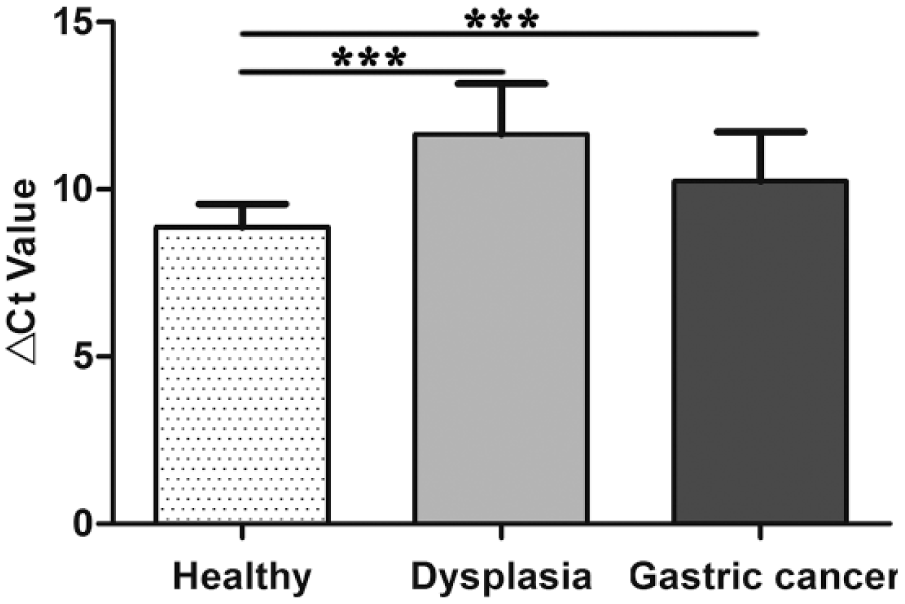
Hsa_circ_0001895 expression in gastric carcinogenesis. Compared with healthy control, hsa_circ_0001895 expression level was downregulated not only in gastric cancer tissues but also in gastric dysplasia. Hsa_circ_0001895 expression levels in healthy gastric mucosa (n = 35), dysplasia (n = 30), and gastric cancer tissues (n = 96) were determined by qRT-PCR. Data are mean ± SD of three independent experiments. Asterisks represent significant statistical difference (***p < 0.001).
Relationship between hsa_circ_0001895 levels and clinicopathological factors
An analysis was performed to assess the relationship between hsa_circ_0001895 expression level and clinicopathological factors of gastric cancer patients. As shown in Table 1, hsa_circ_0001895 level was related to cell differentiation (p = 0.042), Borrmann type (p = 0.047), and tissue carcino-embryonic antigen (CEA) expression (p < 0.001), but not associated with other important clinicopathological features, such as tumor diameter, invasion, or TNM stage.
Relationship of hsa_circ_0001895 expression levels (ΔCt) in cancer tissues with clinicopathological factors of GC patients.

GC: gastric cancer; SD: standard deviation; CEA: carcino-embryonic antigen; CA19-9: carbohydrate antigen 19-9.
ROC curve of hsa_circ_0001895
To further estimate the diagnostic value of hsa_circ_0001895 in gastric cancer, a ROC curve was constructed for differentiating gastric cancer tissues from healthy controls. The area under the ROC curve (AUC) was up to 0.792 (95% confidence interval (CI) = 0.717–0.867; p < 0.001; Figure 4). When the optimal cutoff value was set at 9.53, the sensitivity and specificity of hsa_circ_0001895 were 67.8% and 85.7%, respectively.

The ROC curve for differentiating gastric cancer tissues from healthy controls. ROC curve was constructed for evaluating the diagnostic value of hsa_circ_0001895. The area under the ROC curve (AUC) was up to 0.792 (95% CI = 0.717-0.867; p < 0.001). The optimal cutoff value was 9.53. The sensitivity and specificity were 67.8% and 85.7%, respectively.
Discussion
CircRNAs are a special class of endogenous RNAs and widely exist in human cells. Recent studies have demonstrated that circRNAs play a crucial role in various biological processes thus are involved in the development of some human diseases. 11 Several associations between altered circRNAs and human diseases have been reported.7,12–15 Wang et al. 12 found that heart-related circRNA (HRCR) can function as an endogenous miR-223 sponge to inhibit cardiac hypertrophy and heart failure. Cui et al. 13 reported that hsa_circRNA_103636 could be a biomarker for the diagnosis and treatment of major depressive disorder (MDD). In the field of colorectal cancer, the dysregulated hsa_circ_001569 and hsa_circ_001988 were correlated with cell differentiation, proliferation, and invasion.14,15 All these findings strongly suggest an important role for circRNAs in pathophysiology and the great potential in clinical application.
Hsa_circ_0001895 is one of circRNAs associated with gastric cancer according to our bioinformatics analysis. In this study, we first chose gastric cancer cell lines to verify the actual expression level of hsa_circ_0001895 in gastric cancer. The results showed that hsa_circ_0001895 in five gastric cancer cell lines were significantly downregulated than those in normal gastric epithelial GES-1 cells (Figure 1). Our data are completely consistent with the bioinformatics analysis results. Then, we explored hsa_circ_0001895 expression levels in the tissues from various stages of gastric carcinogenesis. As shown in Figure 2, it was also downregulated in 69.8% (67/96) gastric cancer tissues compared with the paired adjacent normal tissues (p < 0.001; Figure 2). Moreover, hsa_circ_0001895 expression level was also decreased in GD tissues (Figure 3). Our results showed that low expression of hsa_circ_0001895 in tissues is closely related to gastric cancerogenesis.
Tumor markers and their combined detection are effective methods for screening of gastric cancer. CEA, carbohydrate antigen 19-9 (CA19-9), and carbohydrate antigen 72-4 (CA72-4) are the most commonly used biomarkers for gastric cancer screening in clinic. However, the sensitivity of CEA, CA19-9, and CA72-4 in the diagnosis of gastric cancer was only 20.1%–27.6% individually and increased to 48.2% when they were considered in combination. 16 In this study, a ROC curve was constructed for differentiating gastric cancer tissues from controls. The results showed that the AUC was up to 0.792 (Figure 4). The sensitivity and specificity of hsa_circ_0001895 were up to 67.8% and 85.7%, respectively. These are better than the commonly used biomarkers of gastric cancer.
Current researches indicated that some clinicopathologic features such as cell differentiation, Borrmann type, and tissue CEA expression appear as independent prognostic factors affecting gastric cancer–free survival and overall survival.17–19 Shorter postoperative survival patients frequently have poor pathological condition. Ryu et al. 19 reported histologic differentiation was a prognostic indicator in gastric cancer patients with curative resection. Patients with the poorly differentiated type had more prominent serosal invasion, much more lymph node involvement, and more advanced stage than patients with the well-differentiated type. 19 Another study conducted by Li et al. 20 found that Borrmann type is associated with lymph node metastasis and is a simple and valuable predictor for survival in advanced gastric cancer patients. Moreover, Wang et al. 18 reported that tissue CEA in gastric cancer is significantly correlated with cancer invasion, metastasis, and TNM staging, and patients with negative tissue CEA staining had a better prognosis. The 5-year survival rates of gastric cancer patients were 67.6%, 53.9%, and 40.1% for negatively, moderately, and intensely positively stained tissues. 18 In our study, we performed an analysis to assess the relationship between hsa_circ_0001895 expression level and clinicopathological factors of gastric cancer patients. We found that hsa_circ_0001895 expression level was associated with cell differentiation, Borrmann type, and tissue CEA expression. Our data indicated that hsa_circ_0001895 is a potential biological molecule for clinical prognosis prediction.
In conclusion, our research showed that hsa_circ_0001895 was significantly decreased in gastric cancer and its precancerous lesion. Its expression was closely associated with cell differentiation, Borrmann type, and tissue CEA expression. Our results suggested that hsa_circ_0001895 may play crucial roles during gastric cancerogenesis and is a potential biological molecule for clinical prognosis prediction.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Human Research Ethics Committee of Ningbo University School of Medicine.
Funding
This study was supported by grants from the Social Development Research Project of Ningbo (No. 2016C51015), the Medical Research Project of the Affiliated Hospital of Ningbo University School of Medicine (No. XYY16022), the Foundation of Zhejiang Key Laboratory of Pathophysiology (No. 201601), the Medical and Health Research Project of Zhejiang Province (No. 2017KY598), Applied Research Project on Nonprofit Technology of Zhejiang Province (No. 2016C33177), and the K. C. Wong Magna Fund in Ningbo University.
