Abstract
The purpose of our study was to assess the effect of controlled-release chemotherapy on the growth and viability of peritoneal carcinomatosis treated by subperitoneal injection in a rabbit VX2 model. A model of peritoneal carcinomatosis was created by laparoscopic injection of VX2 tumor in the left and right broad ligaments of 12 White New Zealand rabbits. At day 12, each tumor was randomly treated with a peritumoral injection of 0.5 mL microspheres loaded with doxorubicin (DEM-DOX) or unloaded (DEM-BLAND). Seven days after treatment, tumor volume, tumor viability in histology, local tumor necrosis in contact with DEM, and doxorubicin concentration profile around the drug eluting microspheres (DEM) were measured. Tumor volume was significantly lower in the DEM-DOX group (3.6 ± 3.2 cm3) compared with the DEM-BLAND group (8.9 ± 5.4 cm3) (p = 0.0425). The percentage of viable tumor tissue was significantly lower in the DEM-DOX group (38% ± 17%) compared with the DEM-BLAND group (56% ± 20%) (p = 0.0202). Tissue necrosis was observed around all DEM-DOX up to a distance of 1.094 ± 0.852 mm and never observed around DEM-BLAND. Drug concentration was above the therapeutic level of 1.0 µM up to a distance of 1.4 mm from the DEM to the tumor. Laparoscopic subperitoneal injection of chemo-loaded particles is feasible and lowers tumor growth and viability in a rabbit model of peritoneal carcinomatosis after 1 week.
Introduction
Epithelial ovarian cancer is the sixth most common cancer in women. Up to 75% of the patients are diagnosed at an advanced stage (stage III-IV) with peritoneal involvement or distant metastasis and have poor long-term survival rates (10%–30%). 1
The standard treatment of advanced epithelial ovarian cancer is cytoreductive surgery (CRS) combined with intravenous chemotherapy that improves survival rates in advanced cases. The limit of this therapy is the recurrence of the peritoneal carcinomatosis (PC) after CRS in 60%–70% of patients and the systemic toxicity of the intravenous chemotherapy. 1
Direct intraperitoneal chemotherapy by pressured aerosol 2 or heated chemotherapy 3 has been proposed with the intent to expose the tumor to high concentrations of antitumor agents with less systemic toxicity. However, these procedures of intraperitoneal chemotherapy are difficult to set up and associated with local toxicities. 4
Several trials of local chemotherapy have been performed with drug-eluting devices to provide a sustained delivery of the drug. A phase I trial with paclitaxel-loaded microspheres (Paclimer®) administered intraperitoneally for the treatment of PC in women showed peritoneal lesions as extensive adhesions, fat necrosis, fibrous connective tissue, and foreign body giant cell reaction. 5 In small experimental models of PC from an ovarian carcinoma, direct intraperitoneal application of drug-eluting devices proved efficacy for the higher doses but was associated with general toxic effects.6–9
In large animals, direct intraperitoneal application of drug-eluting devices showed high local toxicity at the tested dose. 10
All these trials used the intraperitoneal route to deliver the drug-eluting devices, in other words a direct injection inside the abdominal cavity. One way to improve the treatment of PC could be to place the drug formulation directly in contact with the tumors, and to expose only the lesion while preserving the healthy tissue. Our aim was to bring the proof of concept that subperitoneal peritumoral injection of a drug-eluting system is efficient with an acceptable safety profile.
Doxorubicin was chosen as the anticancer agent because it is one of the most efficient chemotherapy drugs used in clinical trials via the intraperitoneal route.11,12 The drug-eluting system consisted of polymeric microspheres calibrated between 200 and 400 µm, currently approved for the treatment of hypervascular tumors such as liver cancer. 13 These microspheres proved in vivo a sustained delivery of high concentrations of drug for days to weeks.13–15
This study evaluated the efficacy and the safety of local subperitoneal injection of doxorubicin-loaded microspheres in a model of PC on the broad ligament in the female rabbit.
Materials and methods
Animal model
The experimental procedures were performed at the Center of Research in Interventional Radiology (Cr2i/APHP/National Institute of Agronomic Research INRA; Jouy-en-Josas, France). Ethical approval number 08-008 was obtained from the Institutional Animal Care and Use Committee of the Center and was conducted according to European Community rules of animal care (Directive EC 86/609).
Twelve White New Zealand adult female rabbits were selected for tumor implantation. Rabbit VX2 carcinoma, a squamous epithelioid carcinoma with rapid growth, was used to create peritoneal tumours. 16
Anesthesia
Anesthesia was induced by ketamine 35 mg/kg and 3 mg/kg Rompun (xylazine 2%), and maintained with isoflurane 3%/97% oxygen after endotracheal intubation with a 3-mm cannula with continuous monitoring of respiration and cardiac frequency. For analgesia, subcutaneous buprenorphine (0.03–0.05 mg/kg) was injected.
Laparoscopy
Laparoscopy was performed with Aesculap® Endoscopy Units and Accessories (B. Braun, Boulogne Billancourt, France).
The rabbit was placed in a supine position, the head down to ensure that stomach and intestines were cranially located. After shaving off the abdominal region, a 5-mm incision was made into the abdominal wall 1 cm below the navel to the white line. A 5-mm endoscopic disposable trocar was introduced through the abdominal wall (open laparoscopy) for CO2 insufflation and fiberscope insertion. Two other incisions were made on each side of the abdomen at 5 cm symmetrically to a point located on the white line, 10 cm below the first incision. In both incisions, disposable endoscopic trocars were inserted, one for the injection of the cells or treatment and the other one for the endoscopic forceps. The CO2 pressure was maintained up to 10 mmHg during processing. The broad ligaments were localized on each side of the two uteri.
VX2 implantation
The VX2 tumor was serially passed into a hind paw of White New Zealand rabbits. At 15 days, hind paw tumor was removed in sterile conditions and minced into small fragments that were further passed through a tissue strainer. The cellular suspension was washed with phosphate-buffered saline (PBS) with 1% calf serum. A volume of 0.1 mL cellular suspension corresponding to 25 × 106 cells was implanted by a subperitoneal puncture on the anterior part of the right and left broad ligaments using an 18G × 6″ spinal needle (Becton Dickinson, New Jersey, USA; Figure 1). After implantation, the trocar orifices were washed with saline solution to prevent tumor dissemination and sutured with Vicryl 2.0 (Ethicon, Cornelia, GA, USA). The tumor was grown for 11 to 13 days.
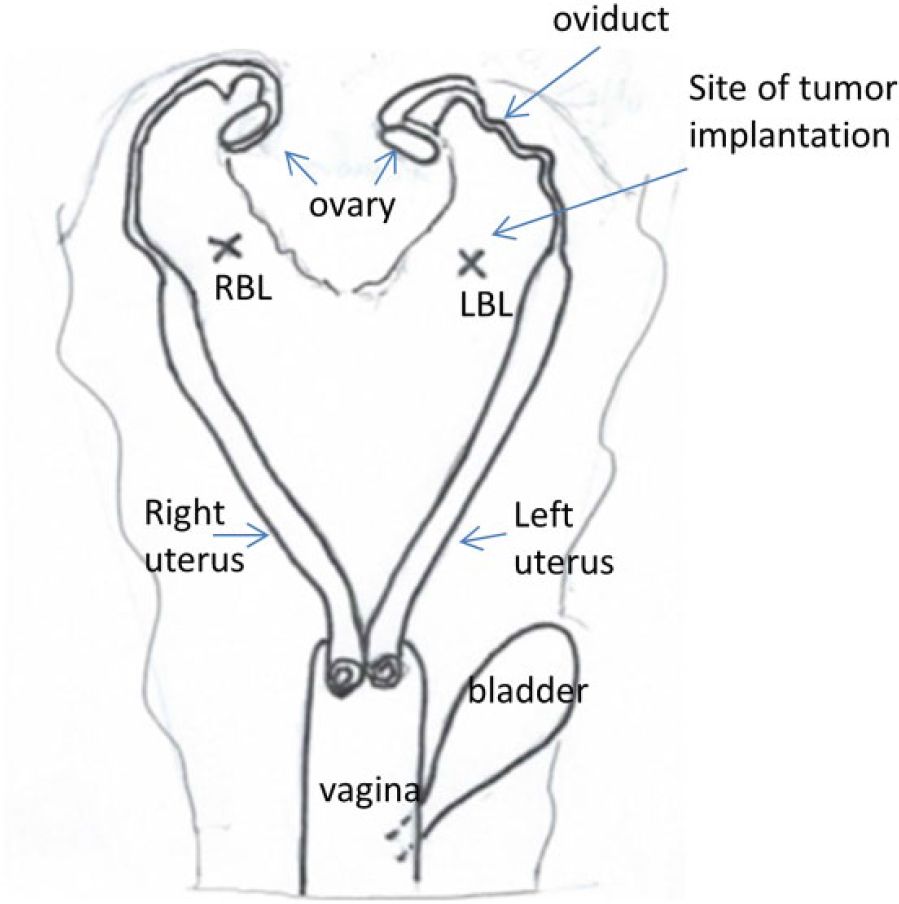
Schematic representation of genital tract in female rabbit with site of tumor implantation (cross) in left broad ligament (LBL) and right broad ligament (RBL).
Treatment
The treatment was performed by laparoscopy following the procedure described above. After the insertion of the trocars, the injection was made in a single point under the tumor site by insertion of the needle between tumor and connective tissue (Figure 2). Each animal received a subperitoneal injection of 0.5 mL microspheres (50–100 µm that measure 200–400 µM after reconstitution; HepaSphere; Biosphere Medical, Roissy-en-France, France) right under the tumor site, on each broad ligament. For each animal, one tumor was treated with microspheres loaded with doxorubicin (Adriblastine, 25 mg/mL, Teva, 25 mg/mL microsphere; DEM-DOX group) and the tumor on contralateral side was treated with 0.5 mL blank microspheres (DEM-BLAND group).

Laparoscopic view of broad ligament with VX2 tumor on the day of treatment. Dotted line represents the needle trajectory.
Animal’s weight, appearance, appetite, and behavior were monitored as an indication of general toxicity. After 7 days, euthanasia was performed by an intravenous injection of 180 mg/kg pentobarbital (Dolethal; Vétoquinol, Lure, France) after flash mask anesthesia with isoflurane 5%/oxygen 95%. Broad ligaments with the tumors, uteri, and ovaries were dissected for pathology examination. The length and the width of the tumors were measured with a caliper. The volume of the tumor was calculated using the formula: π / 6 (ab2), where a and b were the length and the width of the tumor. The orifices of the trocars, the intestines, the bladder, the liver, and the lungs were palpated for the presence of metastasis.
Pathology examination
The tissue specimens were fixed in 4% neutral buffered formalin for at least 48 h and embedded in paraffin. Tissue sections were cut from paraffin blocks and stained with hematoxylin–eosin–saffron (HES). Only the slides that were qualitatively appropriate were subsequently analyzed. Stained sections were then digitized with a slide scanner (NanoZoomer 2.0 HT; Hamamatsu, Hamamatsu City, Japan) at magnification ×20. The surface of viable tumor was assessed on each digitized section using the NDP view2 U12388-01 viewing software (Hamamatsu Photonics, Hamamatsu City, Japan) and expressed as the percentage of the total tumor surface.
Doxorubicin concentration in tissue
Doxorubicin concentration in the tissue around the microspheres was assessed using microspectrofluorimetry as previously described. 17
Two jointed tissue slides of 10 µm in thickness were cut from tumor samples embedded in paraffin. One was left unstained and kept into the dark for doxorubicin quantification, and the other was stained with HES to visualize the microspheres and to measure the tissue necrosis from the microsphere edges to the viable tumor. The unstained section was placed on the stage of a microspectrofluorimeter (V45 Dilor; Jobin-Yvon/Horiba, Lille, France) coupled to an argon ion laser at λ = 488 nm excitation. Fluorescence emission spectra were recorded linearly every 60 µm from the edge of the microspheres up to a distance of 3 mm. The contribution of doxorubicin to the fluorescence spectra recorded was determined and converted to a concentration of drug using standard collagen type I phantoms loaded with different concentrations of doxorubicin for calibration. 17 The lowest limit of quantification (LLOQ) of the drug was 100 nM.
The tissue concentration of doxorubicin was then plotted against the distance to the DEM-DOX to obtain a profile of tissue drug concentration around the DEM-DOX.
Statistical analysis (StatView Version 5.0; SAS Institute Inc., Cary, NC, USA) was performed using the non-parametric Mann–Whitney test. Results were considered significant for p < 0.05.
Results
Tumors
All animals developed tumors at the time of treatment measured about 1 cm in length (Figure 2). The treatment could be performed for all tumors, without technical issues (bleeding, leakage).
However, particles were found at pathology examination in 19 cases out of the 24 treated tumors. The five tumors without microspheres were considered as technical failure and excluded from the study. A total of 19 tumors were included for analysis. All along the treatment period, animals were doing fine, with no significant weight loss between the treatment date (4.14 ± 0.16 kg) and the sacrifice date (4.03 ± 0.23 kg, p < 0.1734) (Table 1).
Global data.

SD: standard deviation; DOX: doxorubicin; MW test: Mann–Whitney test.
Pathology examination
No metastases were found on the abdominal organs or on the lungs. Few tumoral nodules measuring 2–3 mm were observed into the peritoneum, in the vicinity of primary tumors. No peritoneal necrosis, adherences, or other lesions of inflammation were observed at macroscopic examination of abdominal cavity.
Tumor volume on the day of sacrifice was significantly lower in the group treated by subperitoneal injection of DEM-DOX (4.6 ± 4.6 cm3) compared with the group treated by subperitoneal injection of DEM-BLAND (8.9 ± 5.4 cm3) (p = 0.0425, Mann–Whitney; Table 1, Figure 3).

Tumor volume at sacrifice (D+7). (a) Tumor volume, represented as a boxplot with median, upper and lower quartiles, and error bars as 10th and 90th percentiles, is significantly lower for DEM-DOX versus DEM-BLAND treated group. (b) Picture of explanted treated tumors (area surrounded by the white dotted line). DEM-DOX tumor is smaller and has area less vascularized compared with the DEM-BLAND tumor.
The percentage of viable tumor tissue was significantly lower in the DEM-DOX group (37.6% ± 17%) compared with the DEM-BLAND group (55.5% ± 20.3%) (p < 0.0202; Figure 4, Table 1).

Percentage of the viable tissue, represented as a boxplot with median, upper and lower quartiles, and error bars as 10th and 90th percentiles, was significantly higher in DEM-BLAND than in DEM-DOX treated tumors.
Microscopically, there were no tissue modifications around the deposits of DEM-BLAND (Figures 5 and 6(a)), while DEM-DOX were systematically associated with coagulative necrosis of the surrounding tissue (Figures 5 and 6(b)). The thickness of the necrosis layer between DEM-DOX and viable tissue was 1094 ± 852 µm (Figure 5).

The thickness of the necrotic tissue between the beads and the viable tumor (µm) was represented as a boxplot with median, upper and lower quartiles, and error bars as 10th and 90th percentiles. Only DEM-DOX induced tissue necrosis which was measured at 1094 ± 852 µm.

Digitized images of hematoxylin–eosin–saffron (HES) stained tumors show the tumors and deposit of beads in contact with the tumor: (a) DEM-BLAND (arrow) and (b) DEM-DOX microspheres (arrow head). (a) DEM-BLAND were associated with viable tumor tissue (VT). (b) DEM-DOX were associated with tumor necrosis (TN). The red line indicates the limit between viable tissue and necrosis.
Doxorubicin concentration in tumor tissue
No doxorubicin was detected in the tissue around DEM-BLAND (Figure 6).
Doxorubicin tissue concentration around DEM-DOX decreased with the distance from the microspheres to the tissue. It was higher than the LLOQ (0.1 µM) up to a distance of 2.6 mm from the bead edge (Figure 7). Maximum concentration was measured in the first 100 µm around the beads where it reached 15 µM. Doxorubicin concentration decreased rapidly and reached 1.5 µM at a distance of 1 mm from the beads. Drug concentration was higher than the therapeutic level of 1.0 µM up to a distance of 1.4 mm from the microsphere edge (Figure 7).

Doxorubicin concentration profile in tumor tissue (n = 9) of DEM-DOX treated animals, measured from the bead edges to the tumor. Each point of the curve accounted for the mean of the total number of measurements for doxorubicin concentrations every 60 µm from the edge of the microspheres up to a distance of 3 mm. Doxorubicin was detected above the lowest limit of quantification level (dotted red line) up to 2.6 mm deep from the beads. Continuous red line represents the therapeutic level (IC50) of doxorubicin of 1 µM as detected in vitro in ovariocarcinoma and VX2 cell lines (Serova et al. 18 ; Pascale et al. 19 ). Doxorubicin level was higher than the cytotoxic level up to 1.4 mm from the beads.
There was no significant difference for the distance between the microspheres deposit and the tumor between DEM-BLAND (1101 ± 801 µm) and DEM-DOX (2392 ± 2549 µm) (p < 0.2416; Table 1).
Comment
Our objective was to prove that the peritumoral injection of doxorubicin-eluting microspheres lowers the growth of peritoneal tumor nodules with an acceptable safety profile in a preclinical model.
Animal model
To this aim, a model of PC on the broad ligament of the female rabbit was developed. The broad ligament is a wide fold of peritoneum connecting the reproductive organs to the walls of the pelvis. It is the first site of metastases in ovarian cancer in women since the ovarian surface epithelium is contiguous with the peritoneal epithelium. 20 The broad ligament therefore appears as an ideal site for tumor implantation to mimic peritoneal dissemination from an ovarian tumor. We further aimed to treat the tumors with the same procedure as used in clinical practice. The intraperitoneal chemotherapy in women is currently performed by laparoscopic injection. The setting up of the real treatment conditions in an animal model was not possible in rodents due to their small size. The rabbit was chosen because a tumor model is available and it allows the use of the same materials and procedures as in humans. 21 Tumor implantation by laparoscopy was feasible, and nodules measuring approximately 1 cm in length were found at all sites of injection after 2 weeks.
Tumors were treated by an injection of DEM representing 50% of the tumor volume. Paclimer and OncoGel® are paclitaxel formulations developed for intratumoral (IT) injection, and the dose of administration is 20%–50% of the tumor volume. 22 In this study, the drug delivery system was delivered around the tumor and not inside the tumor; therefore, a dose of 50% of the tumor volume was chosen.
General toxicity
The treatment of the tumors by subperitoneal injection of doxorubicin-eluting microspheres was well tolerated with no sign of general toxicity. All animals were doing fine until sacrifice, and did not show any sign of pain or significant weight loss. Previously, preclinical reports of doxorubicin-eluting beads in the treatment of PC described severe local toxicity and even death of the animals8,10 when the particles were injected directly into the abdominal cavity. In a clinical trial of paclitaxel-eluting beads in patients with PC, the intraperitoneal treatment was associated with major local tissue lesions. On the opposite, the same drug delivery system was very well tolerated when injected directly inside the tumours. 23 OncoGel, a formulation of paclitaxel in a biodegradable gel, showed no toxicity when injected by intratumoral route. 24 In this study, the drug-eluting microspheres were injected subperitoneally at the tumor border.
No peritoneal lesions, necrosis, or adherences were macroscopically observed at the time of sacrifice. Microscopic lesions of necrosis were observed around the loaded microspheres but were limited to a 1-mm distance from the bead deposit.
Altogether, these data suggest that the choice of the site of treatment injection is a crucial point for safety issue. The intratumoral and peritumoral injection may be better tolerated than the intraperitoneal route.
Antitumoral effects
The intra-/peritumoral administration may further enhance the efficacy of the treatment compared with the intraperitoneal route. After intraperitoneal injection of doxorubicin-eluting beads in a rodent PC model, tumor growth reduction was only 5%. 8 Increasing the dose induced 15% decrease of tumor growth but resulted in 10% mortality. 8 In this study, DEM-DOX treatment induced 65% tumor growth reduction when compared with controls. In line with our findings, Glage et al. 25 reported a 44% reduction of tumor growth after direct intratumoral injection of doxorubicin-eluting beads in a rat glioma model. Strong antitumoral effects were also observed after IT injection of other doxorubicin-eluting devices in mice. 26
Doxorubicin concentration in tumor tissue
Injecting the drug-eluting devices inside or in near contact with the tumors allows the drug delivered to diffuse immediately into the lesion to be treated. Thus, we demonstrated that doxorubicin eluted from the microspheres penetrated in the surrounding tissue over several millimeters. The concentration was higher than the in vitro cytotoxic concentration up to a distance of 1.4 mm from the microspheres.18,19 Interestingly, this distance corresponds to the thickness of the necrotic tissue around the beads. This finding suggests that the microspheres have released doxorubicin in the tissue and that the levels of drug achieved are responsible for the necrosis observed around the DEM-DOX.
Other preclinical works have demonstrated that at shorter time point, the drug may penetrate up to 5 mm around the drug-loaded deposit. 27 Accordingly, the local tissue concentration obtained with doxorubicin-eluting microspheres is maximum in the hours following the injection and decreases afterward.28,29 The impregnation of surrounding tissue and tumor border extended much further in the first days after injection that the distance measured at 7 days. Thus, the reduction of tumor growth could be explained by the burst release and high levels of doxorubicin achieved at tumor border in the first hours after the treatment. Another hypothesis would be that subtoxic concentrations of the doxorubicin have also acted as a barrier around the site of injection and limited the proliferation of tumoral cells as demonstrated in vitro 30 and in vivo. 31
Altogether, our data suggest that the local delivery of doxorubicin by DEM-DOX is associated with both antiproliferative and cytotoxic effects on the tumor.
Limitations
Particles were not found at pathology examination for five of 24 treated tumors. Several hypotheses could explain the absence of microspheres near the tumor. A leak of microspheres after the injection could be one of the hypotheses, although no leak was noticed at the moment of treatment. DEM-BLAND are transparent so another hypothesis could be that the deposit was missed when cutting the tumor for pathology analysis.
The tumor volume was not measured quantitatively before treatment but estimated by the operator at the moment of injection. No major discrepancy between tumors was noticed. Moreover, the treatment assigned to each animal was randomized to avoid a potential bias in the size of treated tumors.
Conclusion
The laparoscopic subperitoneal injection of chemo-loaded particles is safe and efficient to treat PC in a rabbit model of VX2 tumor. After 1 week, no evidence for general and local toxicity was observed, the tumor growth was reduced by 65%, and the viability of tumoral cells was reduced by 42% when compared with the control group. In future preclinical studies, several doses of DEM-DOX and other type of drug delivery devices should be tested. Subperitoneal and intratumoral routes should also be compared.
Footnotes
Acknowledgements
The authors thank Dr Laurent Bédouet from Occlugel for the manuscript revision and writing. They also thank Nathalie Guatto from Anatomopathology Laboratory of Lariboisiere Hospital for technical support in histology. The results of this study were partially presented at the 39th Global Congress of Minimally Invasive Gynecology, American Association of Gynecologic Laparoscopists ( AAGL), Las Vegas, Nevada, USA, in November 2010.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
International, national, and/or institutional guidelines for the care and use of animals were followed.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
