Abstract
Lung adenocarcinoma remains to be a high-mortality disease with few effective prognostic biomarkers. Novel biomarkers are urgently demanded to supplement the current prognostic biomarkers. Here, we explored the prognostic value of CYP3A4 and CYP3A5 in lung adenocarcinoma. The tissue microarray was made up of lung adenocarcinoma samples and corresponding normal lung tissues from Nanjing Medical University affiliated Cancer Hospital Tissue Bank. The expression of CYP3A4, together with CYP3A5, was detected by the chip data from Gene Expression Omnibus datasets and immunohistochemistry staining of the tissue microarray. Then, we assessed the relationships between CYP3A4 or CYP3A5 expression level and clinicopathological factors to estimate the clinical significance. Kaplan–Meier curves were applied to analyze the prognosis. Univariate and multivariate Cox analyses were subsequently applied to identify the independent prognostic factors. Immunohistochemistry staining results showed that by comparison with matched normal tissues, CYP3A4 was frequently hyper-expressed in lung adenocarcinoma tissues while CYP3A5 was hypo-expressed, which was consistent with the Gene Expression Omnibus analysis. Kaplan–Meier analysis indicated that high-CYP3A4 or low-CYP3A5 expression level predicted poor survival in lung adenocarcinoma patients. Multivariate Cox analysis found that hypo-expression of CYP3A5 was an independent prognostic factor. Further study revealed that combination of these two markers exhibited a more powerful predictor of poor prognosis, which could target to more accurate survival of lung adenocarcinoma. Our findings indicate that combination of CYP3A4 and CYP3A5 may serve as a novel prognostic biomarker in lung adenocarcinoma, which contribute to the precision of predicting the survival in lung adenocarcinoma.
Introduction
Lung cancer is still a major global health issue with high morbidity and mortality.1–3 Lung adenocarcinoma (LAC), a histological type of non-small-cell lung cancer, is becoming a major proportion of lung cancer.4,5 Despite recent advances in cancer therapy, the 5-year survival of LAC is still unsatisfied, which draws great attention.6,7 With the concept of precision medicine appears, molecular biomarkers and molecular drug targets become the hotspot of cancer investigations. 8 Therefore, it is a challenging and emerging task to discover novel molecular biomarkers to supplement the current prognostic predicting model.
CYP3A subfamily is one of the main subgroup of cytochrome P450 enzymes. CYP3A subfamily proteins, located in the endoplasmic reticulum, take part in many catalytic and synthetic reactions including drug metabolisms and synthesis of endogenous hormones.9,10 CYP3A4 and CYP3A5 are two major enzymes of CYP3A subfamily. Previous studies with regard to those two enzymes focused on the problems like acquired drug resistance, 11 gene polymorphisms, 12 pharmacogenomics, 13 exogenous or endogenous agent metabolism, 14 and carcinogens metabolism. 15 In cancer research, these two enzymes were widely reported to play critical roles in pharmacokinetics and pharmacogenomics of anticancer agents.16–18 Potential functional polymorphisms of CYP3A4 and CYP3A5 were observed to be correlated with cancer risks in cancer genomics research. 19 Additionally, CYP3A4 and CYP3A5, functioned as oncogenes or tumor suppressors, have aroused the attention of cancer researchers in recent years.16,20 Latest study shows that CYP3A5 functions as a tumor suppressor in hepatocellular carcinoma by regulating mTORC2/Akt signaling. 21 CYP3A4 also served as a potential oncogene to promote malignant behavior in breast, hepatocellular, and colon cancer.22–24 Nevertheless, few published studies investigated the correlation between expression of CYP3A5 or CYP3A4 and prognosis of cancer patients. 25 Therefore, the role of CYP3A4 and CYP3A5 in the lung cancer context remains puzzling and needs further clarification.
Our aim was to analyze CYP3A4 and CYP3A5 expression level in LAC tissues and to clarify the prognostic value of CYP3A4 and CYP3A5 in LAC patients.
Methods
Microarray dataset analysis
Eligible microarray datasets from Gene Expression Omnibus (GEO) database in the National Center for Biotechnology Information (NCBI) were downloaded to evaluate the messenger RNA (mRNA) expression of CYP3A4 and CYP3A5 in LAC. The screen criteria were listed as follows: (1) LAC tissues were included (>40 samples) and (2) normal tissues were collected for control. Student’s t-test was applied to analyze the different expressions between the tumor and normal tissues. Kaplan–Meier plotter, a survival analysis website, was used to analyze the impact of CYP3A4 and CYP3A5 on the survival of LAC patients (www.kmplot.com). 26
Patients and tissue microarray
A total of 92 pairs of specimens were retrospectively collected in tissue microarray (TMA) from Nanjing Medical University affiliated Cancer Hospital Tissue Bank. Informed consent was obtained from all individual patients included in the study. The criteria of cases selection were listed as follows: (1) adenocarcinoma specimens and adjacent normal tissues, (2) no neoadjuvant chemoradiotherapy before surgery, and (3) no history of other tumors. Histological diagnosis of each tumor specimen was independently validated by two pathologists. The tumor node metastasis (TNM) stages were classified according to the seventh edition of the American Joint Committee on Cancer manual. A total of 92 patients were regularly followed up to death or 60 months. The detailed clinical and pathological information of each patient is displayed in Table 1. This study was approved by the medical ethics committee of Nanjing Medical University affiliated Cancer Hospital. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Demographic and clinicopathological features of CYP3A4 and CYP3A5.

NSCLC: non-small-cell lung cancer; TNM: tumor node metastasis.
Immunohistochemistry and assessment
Paraffin-embedded tumor and normal tissues were punched into 1-mm-diameter cylinder and arrayed on TMA by a tissue array instrument (Shanghai Biochip Co., Ltd., Shanghai, China). The primary antibodies used in this study included anti-CYP3A4 antibody (rabbit monoclonal 1:200 dilution; Abcam, UK) and anti-CYP3A5 antibody (rabbit monoclonal 1:250 dilution; Abcam). Sections were prepared by routine procedures. Sections were dried at 63°C in stove for 1 h, deparaffinized and rehydrated in xylene and alcohol (100%, 90%, 80%, and 70%). Thereafter, antigen retrieval was performed by submerging the sections in citrate acid buffer and ethylenediaminetetraacetic acid (EDTA) buffer. Subsequently, two sections were independently incubated with anti-CYP3A4 and anti-CYP3A5 at 4°C overnight. Primary antibody was detected by incubating the TMAs with corresponding secondary antibody for 30 min. Immunoreactivity was detected by 3,3′-diaminobenzidine (DAB) staining kit based on the protocol. Finally, the sections were handled with hematoxylin counterstain, dehydrated and coated with coverslips.
Evaluating staining results of TMAs were completed by two independent investigators. Positive staining was determined as strong and continuous cytoplasmic staining. Negative staining was defined to be no staining, focally staining, and weakly staining. Scoring criteria of staining intensity were listed as follows: (1) percentage of positive tumor cells in the tumor tissue: 1 (0%−25%), 2 (26%–50%), 3 (51%–75%), and 4 (76%–100%); (2) staining intensity: 0 (no signal), 1 (weak), 2 (moderate), and 3 (strong). Final scores were calculated by multiplied percentage of positive tumor cells with staining intensity.
Statistical analysis
All statistical analyses were performed by SPSS version 21.0 software (SPSS Inc., Chicago, USA). Paired t-test was used to evaluate the expression level of CYP3A4 and CYP3A5 between tumor and adjacent tissues. Associations between clinical characteristics and expression of CYP3A4 and CYP3A5 were calculated by chi-squared test or Fisher’s exact test. Overall survival (OS) was defined from diagnostic date to death or loss of follow-up. Progression-free survival (PFS) was identified from diagnostic date to death or cancer recurrence. Kaplan–Meier method was applied to describe the OS and PFS. Significant discrepancies of OS or PFS were assessed by log-rank test. Univariate and multivariate Cox analyses were performed to identify the independent prognostic factors. p-value less than 0.05 indicated statistical significance.
Result
Expression patterns of CYP3A4 and CYP3A5 in LAC GEO database
Taking advantage of the online database, we accessed the mRNA expression level of CYP3A4 and CYP3A5 in GSE19804 and GSE19188 datasets.27,28 Baseline information of two datasets is shown in Table S1. Integrating the two datasets, we found that CYP3A4 was hyper-expressed in tumor tissues in comparison with normal tissues (p < 0.05). Conversely, CYP3A5 was identified to be significantly hypo-expressed in tumor tissues (Figure 1(a) and (b)). Furthermore, survival analysis, which was performed by Kaplan–Meier plotter, showed that high mRNA level of CYP3A4 predicted a poor survival in LAC (Figure 1(c)). Intriguingly, patients with hypo-expressed CYP3A5 exhibited a significantly shorter survival compared with those with hyper-expressed CYP3A5 (Figure 1(d)).
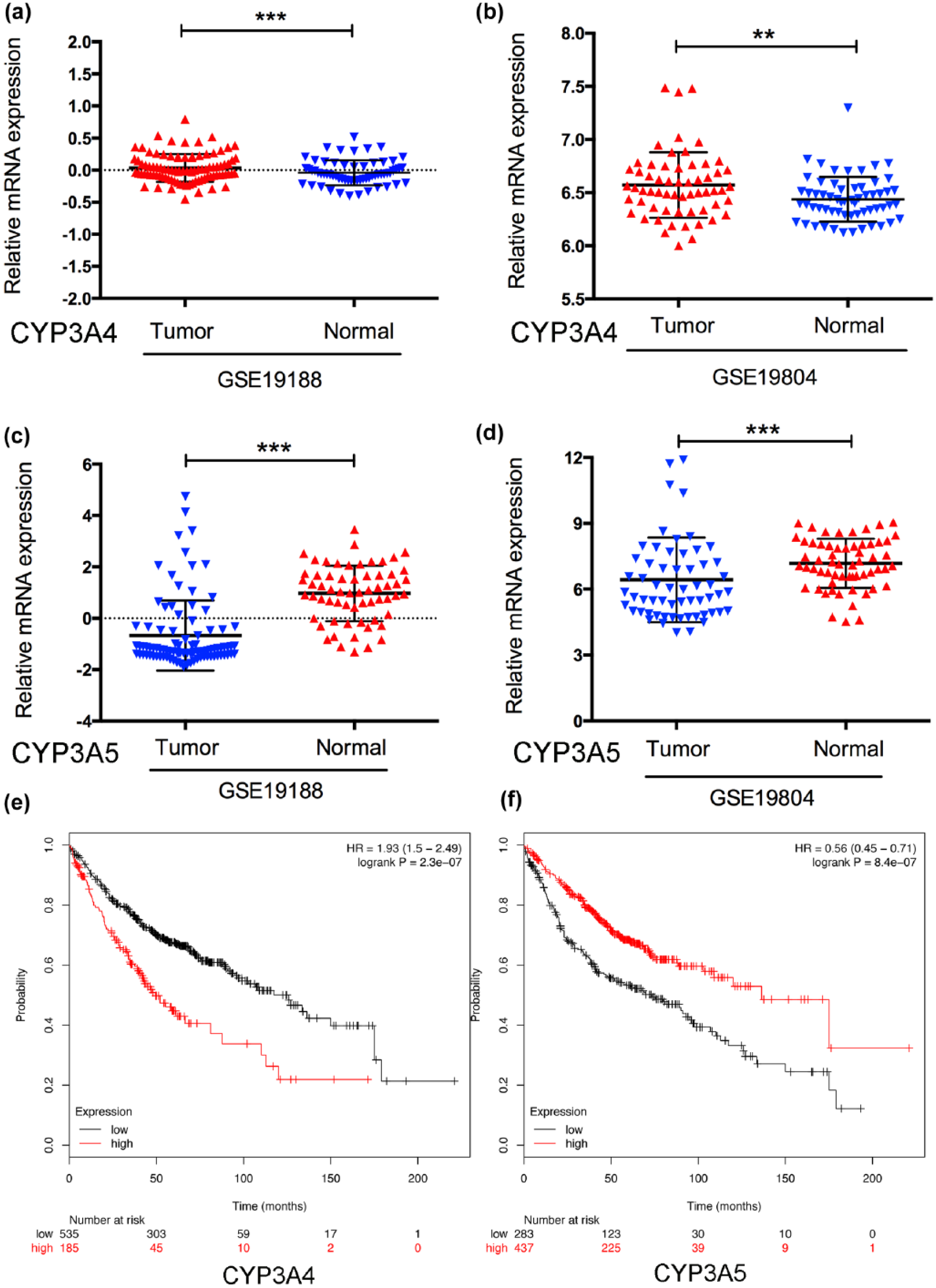
Expression patterns of CYP3A4 and CYP3A5 in LAC GEO database. (a, b) Expression of CYP3A4 in GSE 19188 and GSE19804. (c, d) Expression of CYP3A5 in GSE 19188 and GSE19804. (e) Hyper-expression of CYP3A4 mRNA predicts a poor survival in lung adenocarcinoma patients. (f) Patients with hypo-expression of CYP3A5 mRNA showed a short overall survival compared to those with hyper-expression of CYP3A5 mRNA.
Expression patterns of CYP3A4 and CYP3A5 in LAC TMA
After being scanned, 87 pairs of tissues in TMA were qualified to be scored and other tissues were eliminated for error pathological diagnosis or lack of essential information. Expression patterns and subcellular localization of CYP3A4 and CYP3A5 protein were detected by immunohistochemistry (IHC) staining. We observed that CYP3A4 and CYP3A5 were mainly distributed in cytoplasm of lung epithelial cells. Positive staining of CYP3A4 was mainly found in tumor tissues with a positive rate of 73.5% (Figure 2(a)). On the contrary, positive staining of CYP3A5 was rarely detected in tumor tissues and commonly observed in most adjacent tissues (Figure 2(b)). By analyzing the IHC score, we found that expression level of CYP3A4 was obviously up-regulated in LAC tissues (p < 0.001) (Figure 2(c)), while expression level of CYP3A5 tended to be down-regulated in tumor tissues (p < 0.001) (Figure 2(d)).

Immunohistochemical (IHC) analyses of CYP3A4 and CYP3A5 in LAC TMA. (a) CYP3A4 IHC staining in tumor tissues and adjacent normal tissues. (b) CYP3A5 IHC staining in tumor tissues and adjacent normal tissues. (c) IHC scores of CYP3A4. (d) IHC scores of CYP3A5.
Relationship between expression of CYP3A4 or CYP3A5 and clinicopathological factors
In this study, several key clinicopathological factors were collected to access the correlation with the expression level of CYP3A4 or CYP3A5. Detailed clinicopathological factors (including age, gender, and lymph node metastasis) are summarized in Table 1. Patients were separated to high- or low-expression cohort by IHC scores. The cut-off points between high- and low-expression group were defined as the mean of IHC scores. As a result, high CYP3A4 was significantly correlated with advanced TNM stages and poorly histological differentiation (p = 0.013 and p = 0.044, respectively), but not with other factors like age, gender, or lymph node metastasis (Table 1). Additionally, we observed that low CYP3A5 was statistically associated with poorly histological differentiation (p = 0.017), while no significant relationship was found between expression of CYP3A5 and other clinicopathological factors (Table 1).
Impact of CYP3A4 or CYP3A5 on OS and PFS of LAC patients
The follow-up information of 87 patients was obtained from our tissue bank system. The mean OS and PFS of all patients were 41.9 and 36.9 months, respectively. Kaplan–Meier curve analysis revealed that patients with hyper-expression of CYP3A4 showed a significantly shorter OS and PFS in comparison with those with hypo-expression of CYP3A4, which indicated that hyper-expression of CYP3A4 appeared to be a poor survival predictor (Figure 3(a) and (b)). Univariate Cox analysis showed that age, lymph node metastasis, TNM stage, histologic grade, and CYP3A4 were significantly correlated with OS and PFS (Table 2). After adjusting these risk variables, multivariate Cox analysis unveiled that high CYP3A4 was not an independent prognostic risk factor of OS or PFS in LAC patients with a borderline significance (p = 0.087). In addition, subgroup analysis showed that patients with hyper-expression of CYP3A4 had poorer OS and PFS compared to those with hypo-expression of CYP3A4 in none lymph node metastasis group (p = 0.02). However, no significant relationship was found in lymph node metastasis.
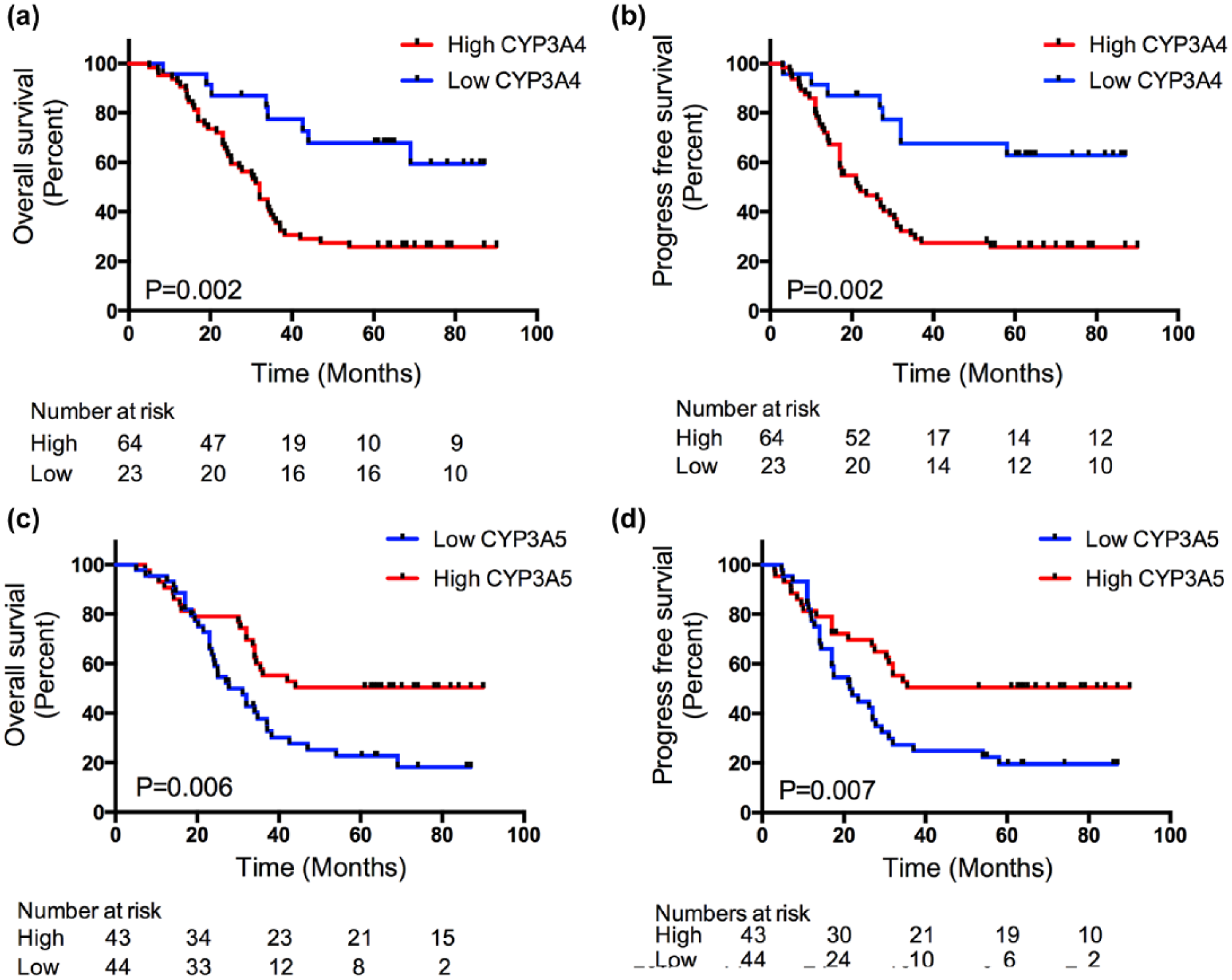
Impact of CYP3A4 or CYP3A5 on OS and PFS of LAC patients. (a, b) Overall survival (OS) and progression-free survival (PFS) of CYP3A4. (c, d OS and PFS of CYP3A5.
Cox regression analyses of CYP3A4.

CI: confidence interval; HR: hazard ratio; TNM: tumor node metastasis.
Next, we investigated the prognostic value of CYP3A5 in LAC patients. Kaplan–Meier analysis found that patients with hypo-expression of CYP3A5 were related to worse OS (35.3 months vs 48.7 months; p = 0.005). Similar tendency was observed in PFS (29.4 months vs 44.7 months; p = 0.0038). It indicated that patients with hypo-expression of CYP3A5 tended to be a worse prognosis compared to those with hyper-expression of CYP3A5 (Figure 4(c) and (d)). Subgroup analysis revealed that patients with hypo-expression of CYP3A5 exhibited much worse OS and PFS in lymph node metastasis and advanced stage subgroup, respectively. Univariate Cox analysis identified several prognostic risk factors including age, lymph node metastasis, TNM stage, and histologic grade. We found that hypo-expression of CYP3A5 retained an independent prognostic factor with a hazard ratio of 1.797 (p < 0.05) in OS and PFS by multivariate Cox analysis (Table 3).
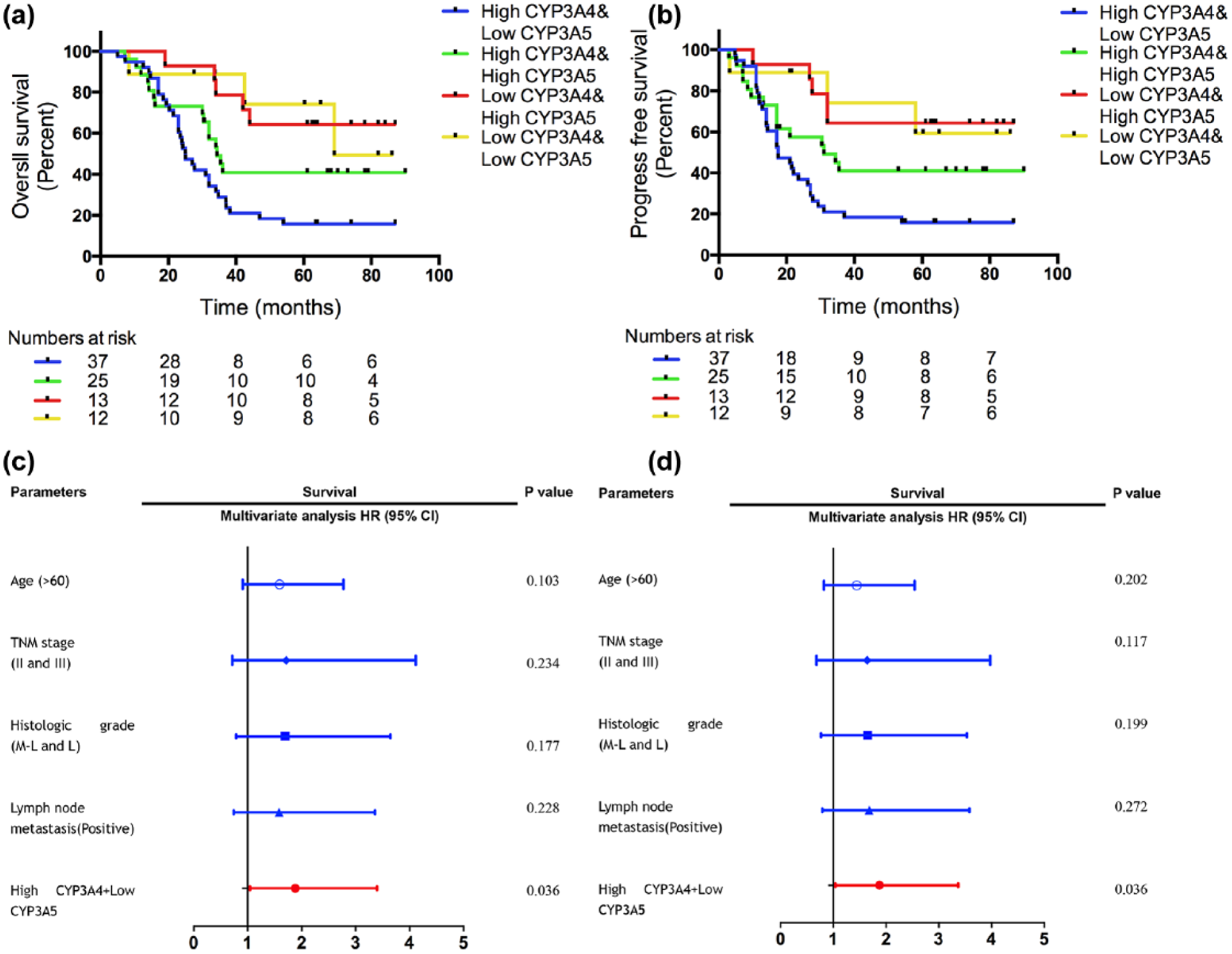
Prognostic value of combining CYP3A4 and CYP3A5 in LAC patients. (a, b) OS and PFS of combining CYP3A4 and CYP3A5. (c, d) Multivariate analysis of combining CYP3A4 and CYP3A5 in OS and PFS.
Cox regression analyses of CYP3A5.

CI: confidence interval; HR: hazard ratio; TNM: tumor node metastasis.
Combination of CYP3A4 and CYP3A5 exhibits improved prognostic accuracy for LAC
Based on the above-mentioned significant prognostic discrepancies by CYP3A4 or CYP3A5, we further investigated the impact on OS and PFS by combining molecular phenotypes of CYP3A4 and CYP3A5. The patients were classified into four subgroups (high-CYP3A4/low-CYP3A5, high-CYP3A4/high-CYP3A4, low-CYP3A4/high-CYP3A5, and low-CYP3A4/low-CYP3A5). As a result, we observed that individuals with different phenotypes showed different prognosis, and phenotype of high-CYP3A4/low-CYP3A5 predicted the worst prognosis among four phenotypes (Figure 4(a)). More importantly, further analysis unveiled that the phenotype of high-CYP3A4/low-CYP3A5 predicted a worse prognosis than high-CYP3A4 or low-CYP3A5 alone. In addition, phenotype of high-CYP3A4/high-CYP3A5 showed an optimistic 5-year OS with a unit of 20% increment than phenotype of high-CYP3A4/low-CYP3A5. Furthermore, the cases with other two phenotypes (low-CYP3A4/low-CYP3A5 or low-CYP3A4/high-CYP3A5) exhibited a similar and more favorable survival. Similar trends were observed in PFS (Figure 4(b)). These results indicated that combining CYP3A4 and CYP3A5 could accurately and effectively discriminate different prognosis in different LAC patients. Subsequently, we assessed the correlation between four phenotypes and clinicopathological factors. This analysis revealed that four phenotypes were significantly associated with LAC differentiation and TNM stages (p < 0.05). But other factors (such as age, gender, and lymph node metastasis) showed no statistical significance. By univariate analysis, we found that age, TNM stage, differentiation, and lymph node metastasis played an important role in OS and PFS in LAC patients (Table S2). Multivariate Cox analysis adjusting for other risk factors indicated that high-CYP3A4/low-CYP3A5 was an independent prognostic factor in either OS or PFS (Figure 4(c) and (d)).
Discussion
LAC is a malignant behavior disease with poor clinical outcome. 29 Great advances have been achieved in diagnosis and treatment, which notably improve the prognosis of LAC patients. Large amounts of molecular biomarkers have been identified to predict the survival of LAC patients. 30 However, existing prognostic predictors cannot comprehensively and accurately predict the survival of LAC patients. Discovery and validation of useful molecular biomarkers to supplement the current group of molecular biomarkers and guide the treatment decisions are urgently needed.
A key discovery of this investigation was that combination of CYP3A4 and CYP3A5 could accurately and effectively predict the prognosis in LAC patients. This result indicated that combination of CYP3A4 and CYP3A5 could be a novel and useful molecular biomarker to predict the clinical outcome of LAC patients.
In this study, we first explored the expression patterns of CYP3A4 and CYP3A5 in available online datasets and found completely opposite expression profiles of CYP3A4 and CYP3A5 in LAC tissues. Analysis of two GSE datasets revealed a significant hyper-expression of CYP3A4 mRNA level and hypo-expression of CYP3A5 mRNA level in LAC tissues. By IHC technology, we validated this different expression profiles in protein level. We also used the Kaplan–Meier plotter to explore the impact of these two genes on survival. We identified that patients with hyper-expression of CYP3A4 showed a worse survival compared to those with hypo-expression of CYP3A4 in mRNA expression level, while patients with hypo-expression of CYP3A5 exhibited a shorter survival in comparison with those with hyper-expression of CYP3A5. These different prognostic trends were consistent with previous studies which were conducted in different types of cancer.24,26 Mutation of CYP3A4 and CYP3A5 might impact their expression in LAC, which was correlated with cancer risk.31,32 Various important clinicopathological factors for LAC had been collected in our patients’ information, including TNM stage, histological grade, lymph node metastasis, and age. We found that expression of CYP3A4 was correlated with TNM stage and histological grade, whereas expression of CYP3A5 was only related with histological grade. These results revealed that their expression discrepancy might have the clinical significance. Consequently, we analyzed the impact of CYP3A4 and CYP3A5 on OS and PFS of LAC. We identified that high-CYP3A4 exhibited a worse survival in adenocarcinoma patients, but it was not an independent prognostic factor after we adjusted for other risk factors. On the contrary, low CYP3A5 demonstrated a significantly worse prognosis in adenocarcinoma patients and retained its independent significance in multivariate analysis. These findings demonstrated two novel molecular biomarkers which could predict the OS and PFS of LAC. Reviewing previous articles, we only found that “hypo-expression of CYP3A5 could predict poor survival of hepatocellular carcinoma” this conclusion was consisted with our result. 21 Therefore, we reported CYP3A4 and CYP3A5 to be functioned as a LAC prognostic predictor for the first time. As reported by previous articles, CYP3A4 and CYP3A5 belonged to CYP3A subfamily together. However, the reverse expression patterns of CYP3A4 and CYP3A5 might indicate their different biological functions in LAC.
Accumulating evidence also indicates that combining multiple markers could be more informative and accurate than single biomarker to predict the prognosis. We combined the CYP3A4 and CYP3A5 expression together to get increased prognostic accuracy in LAC. By combining them, we classified patients into four phenotypes (high CYP3A4/high CYP3A5, high CYP3A4/low CYP3A5, low CYP3A4/high CYP3A5, and low CYP3A4/low CYP3A5). As a result, we identified that the patients with high CYP3A4/low CYP3A5 phenotype showed significantly shorter OS and PFS compared to those with high CYP3A4 or low CYP3A5, respectively. These results indicated that high CYP3A4/low CYP3A5 phenotype was a more robust predictor, which predicted the worst survival of LAC. Additionally, high CYP3A4/high CYP3A5 performed a better survival than high CYP3A4/low CYP3A5 phenotype. Phenotype of low CYP3A4/high CYP3A5 and low CYP3A4/low CYP3A5 showed a favorable survival. Based on these findings, we could predict the survival of LAC patients more accurately and effectively by different phenotypes. This predictive model with different phenotypes made its clinical application to predict the survival.
However, there were still some limitations that should be mentioned. First, there were totally 92 pairs of tissues collected in this study. The number of samples was limited. There was lack of multi-center samples to validate our findings in particular. Second, the precise role of two genes in carcinogenesis remained unclear. Further validation with a larger number of samples was warranted to strengthen our results.
Conclusion
In summary, we determined two novel prognostic biomarkers (CYP3A4 and CYP3A5) by TMA technology. Furthermore, our findings designated combining CYP3A4 and CYP3A5 as an accurate and powerful predictor of prognosis in LAC patients, which could supplement the prognostic model of LAC.
Footnotes
Acknowledgements
Mao Qixing, Xu Juqing, and Wang Yajing have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This was a retrospective study approved by Nanjing Medical University affiliated Cancer Hospital.
Funding
This study was funded by the Natural Science Foundation of China (81372321 to Xu Lin; 81472702 to Jiang Feng) and the Nanjing Medical University affiliated Cancer Hospital Tissue Bank (BM2015004).
Informed consent
Informed consent was obtained from all individual participants included in the study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
