Abstract
Inositol polyphosphate 4-phosphatase type II emerges as a tumor suppressor in prostate cancer, and its loss of expression is associated with poor prognosis for prostate cancer. However, the mechanism of downregulation of inositol polyphosphate 4-phosphatase type II in prostate cancer development has not yet been fully clarified. In this study, microRNA-590-3p was found to be upregulated in both prostate cancer tissues and cell lines. Overexpression of microRNA-590-3p by microRNA-590-3p mimics promoted prostate cancer cell proliferation and invasion and accelerated the growth of xenografted tumors, while microRNA-590-3p inhibitors contributed to inhibition of cellular proliferation and invasion as well as tumor growth. A dual-luciferase reporter assay and expression analysis further confirmed that inositol polyphosphate 4-phosphatase type II was a direct target of microRNA-590-3p. Enforced expression of microRNA-590-3p led to repression of inositol polyphosphate 4-phosphatase type II messenger RNA and protein expression, as well as upregulation of p-Akt, p-FoxO3a, and cyclin D1 and downregulation of p21 expression in prostate cancer cell lines. Overexpression of inositol polyphosphate 4-phosphatase type II could reduce microRNA-590-3p-induced cell proliferation and invasion as well as tumor growth, and decrease microRNA-590-3p-mediated upregulation of cyclin D1 and downregulation of p21 expression in prostate cancer cells. Taken together, our findings reveal that microRNA-590-3p is a potential onco-microRNA that participates in carcinogenesis of human prostate cancer by suppressing inositol polyphosphate 4-phosphatase type II expression and involving the Akt/FoxO3a pathway. MicroRNA-590-3p may represent a potential therapeutic target for prostate cancer patients.
Introduction
Prostate cancer (PCa) is one of the most prevalent solid malignancies in humans worldwide, and its prognosis is generally poor even after surgery. Despite the considerable advances that have been achieved in the diagnosis and treatment of PCa in the past few decades, almost all patients will relapse and eventually progress to metastatic castration-resistant prostate cancer (CRPC), which is currently incurable and accounts for most PCa mortality. 1 Therefore, identifying novel targets responsible for the pathogenesis of PCa is necessary to improve the quality of life for PCa patients and to prolong their lives.
Inositol polyphosphate 4-phosphatase type II (INPP4B), a lipid phosphatase, has been described as a potential tumor suppressor in many human malignant tumors, 2 such as breast cancer, 3 ovarian cancer, 4 and melanocytic neoplasms. 5 INPP4B is a dual-specificity phosphatase that preferentially dephosphorylates phosphatidylinositol polyphosphates on the fourth position of the inositol ring and has phosphotyrosine phosphatase activity. 6 It is reported that INPP4B is highly expressed in prostate intermediate cells 7 but decreased in PCa tissues. 6 Cumulative evidences have demonstrated that loss of INPP4B expression correlates with disease recurrence and poor clinical outcome for PCa patients. 7 Moreover, INPP4B was proved to be an androgen responsive gene, 6 and overexpression of INPP4B contributes to an antitumor effect in therapy for CRPC. 8 All of these lines of evidence suggest that INPP4B may play a crucial role in the progression of PCa. However, the mechanism of INPP4B regulation in PCa development has not yet been fully clarified.
MicroRNAs (miRs) are important regulators of gene expression and protein production. Dysregulated expression of miRs implicates their role in diverse physiological and pathological processes, including human cancers. 9 MiR-937 is reported to involve in the regulation of lung cancer cell proliferation by targeting INPP4B. 10 MiR-765 can promote cell proliferation in hepatocellular carcinoma by downregulating INPP4B expression. 11 However, it remains unclear whether and how miRs involve in the regulation of INPP4B expression and PCa development.
In this study, miR-590-3p was found to be upregulated in both PCa cell lines and tissues, and exerted promoting effects on the proliferation and invasion of PCa cells. Moreover, INPP4B was identified as a direct target of miR-590-3p and shown to mediate the effects of miR-590-3p on the proliferation and invasion of PCa cells. Further investigation demonstrated that the effect of the miR-590-3p/INPP4B axis on PCa function is mediated by a mechanism involving the Akt/FoxO3a pathway.
Materials and methods
Tissue samples
A total of 28 pairs of human PCa tissue samples and adjacent noncancerous tissues were collected from PCa patients during biopsy before any therapeutic intervention, obtained from the Second Affiliated Hospital of Xi’an Jiaotong University (Xi’an, China). Written informed consent was obtained from each patient before surgery, and the study was performed with approval from the ethics committee of the Second Affiliated Hospital of Xi’an Jiaotong University.
Cell culture
Normal human prostate epithelial cell line RWPE-1, human PCa cell lines (LNCaP, 22RV1, VCaP, C4-2, PC3, and DU-145), and human embryonic kidney (HEK) 293T cells were purchased from the American Type Culture Collection (ATCC, Manassas, VA, USA). RWPE-1 cells were cultured in keratinocyte serum-free medium (K-SFM; Invitrogen, Carlsbad, CA, USA). The PCa cell lines were cultured in RPMI 1640 medium (Invitrogen) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Invitrogen). HEK293T cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Invitrogen) containing 10% FBS. All cells were routinely maintained at 37°C in a humidified 5% CO2 atmosphere.
RNA isolation and real-time polymerase chain reaction
Total RNA extraction was performed with Trizol Reagent (Invitrogen) according to the manufacturer’s instructions. Complementary DNAs (cDNAs) were synthesized using the TaqMan miR reverse transcription kit (Applied Biosystems, Foster City, CA, USA). Quantitative real-time polymerase chain reaction (PCR) was performed using an ABI PRISM 7900 HT Sequence Detection System (Applied Biosystems). Sequence-specific primers (miR-590-3p: forward 5′-TAGCCAGTCAGAAATGAGCTT-3′, reverse 5′-TGCTGCATGTTTCAATCAGAGA-3′; INPP4B: forward 5′-CCCCGGGTACTGAGGCTTCG-3′, reverse 5′-CTTTGTATTCTCTCCCGGAGGCG-3′; cyclin D1: forward 5′-AACTACCTGGACCGCTTCCT-3′, reverse 5′-CCACTTGAGCTTGTTCACCA-3′; p21: forward 5′-CTGTGGGTCTCTGCCAGCTGC-3′, reverse 5′-GAGGCCTGTCTCACCACCAAG-3′; GAPDH: forward 5′-AACTTTGGCATTGTGGAAGG-3′, reverse 5′-TGTGAGGGAGATGCTCAGTG-3′) were synthesized by GenePharma (Shanghai, China). Expression levels of these genes were normalized to GAPDH and quantified by the 2−ΔΔCt method.
Cell transfection
The miR-590-3p mimics (5′-UAAUUUUAUGUAUAAGCUAGU-3′), miR-590-3p inhibitors (5′-ACUAGCUUAUACAUAAAAUUA-3′), and their negative controls (miR-NC) were synthesized by GenePharma and transfected into prepared cells at a final concentration of 50 nM. Cell transfections were performed with Lipofectamine™ 2000 (Invitrogen) according to the manufacturer’s instructions. For INPP4B overexpression, the pcDNA3.1(+)/INPP4B expression vector was constructed by cloning INPP4B fragment into pcDNA3.1(+) plasmid (Invitrogen). Thereafter, the recombined plasmids were transfected into PCa cells, and empty pcDNA3.1(+) plasmid was taken as a negative control.
Cell proliferation assay
After 48-h transfection, LNCaP and C4-2 cells were harvested, seeded at a density of 1 × 104 per well onto 96-well plates, and incubated at 37°C. Cell proliferation was measured by cell counting kit-8 (CCK-8; Sigma, St Louis, MO, USA). At specific time points, each well was added 10 µL of CCK-8 solution for another 4-h incubation, and the absorbance at 450 nm was then measured using a microplate reader.
Transwell invasion assay
After 48-h transfection, LNCaP and C4-2 cells were resuspended in serum-free RPMI-1640 medium. Then, 1 × 105 prepared cells were placed in the upper chamber of a 8-µm pore size transwell (Millipore, Billerica, MA, USA), which was pre-coated with Matrigel (BD Biosciences, San Jose, CA, USA). RPMI 1640 medium containing 10% FBS in the lower chamber served as the chemoattractant. After 24 h of incubation at 37°C, the invaded cells were fixed with 100% methanol, stained with 1% crystal violet for 20 min, and counted under a microscope.
Xenograft tumor model
A total of 24 female BALB/c nude mice (6 weeks old), purchased from the Laboratory Animal Center of Chinese Academy of Sciences (Shanghai, China), were used to establish the xenograft PCa model. All animal experiments were performed with the guidance and approval of the Institutional Animal Care and Use Committee of the Second Affiliated Hospital of Xi’an Jiaotong University. The mice were randomly divided into three groups, and C4-2 cells (2 × 106) stably transfected with miR-NC, miR-590-3p inhibitors, and miR-590-3p mimics with or without pcDNA3.1(+)/INPP4B were inoculated subcutaneously into the flanks of nude mice. Tumor size was measured every 5 days and calculated using the formula: volume = 0.5 × length × width 2 . Thirty days after inoculation, the mice were sacrificed and the tumors were weighed.
Luciferase reporter assay
The fragment of the human INPP4B with (wild type) or without (mutant) the miR-590-3p binding site at the 3′-untranslated region (3′-UTR) was cloned and inserted into the pGL3-basic luciferase report plasmid (Promega, Madison, WI, USA) to generate luciferase reporter vectors, INPP4B 3′-UTR-wt and INPP4B 3′-UTR-mut. HEK293T cells were seeded in 96-well plates at 5000 cells per well and allowed to settle for 24 h prior to transfection. Then, miR-590-3p mimics or miR-NC were, respectively, transfected into HEK293T cells using Lipofectamine 2000 with 100 ng of INPP4B 3′-UTR-wt or INPP4B 3′-UTR-mut, plus 10 ng of pRL-TK Renilla plasmid (Promega). Following 48-h incubation, the luciferase activities were determined using a Dual Luciferase Reporter System (Promega).
Western blot analysis
Total protein was extracted using radioimmunoprecipitation assay (RIPA) buffer (Abcam, Cambridge, MA, USA) and quantitated by the BCA Protein Assay Kit (Pierce, Rockford, IL, USA). Equal amount of proteins were subjected to western blot analysis according to the standard protocols. The primary antibodies used were anti-INPP4B, anti-Akt, anti-p-Akt (Ser473), anti-FoxO3a, anti-p-FoxO3a (Ser253), anti–cyclin D1, anti-p21, and anti-β-actin (Abcam). After incubation with horseradish peroxidase (HRP)-conjugated secondary antibodies (Abcam), the resultant protein bands were visualized using an enhanced chemiluminescence (ECL) detection system (Amersham Biosciences, San Francisco, CA, USA). Densitometric analysis of western blot bands was performed with ImageJ software, and protein levels were quantified by normalization to β-actin levels.
Statistical analysis
Numerical data were expressed as the mean ± standard deviation (SD) of results derived from three independent experiments performed in triplicate. Statistical analyses were performed using Student’s t test and analysis of variance (ANOVA). Values of p <0.05 were considered statistically significant compared to the respective control.
Results
MiR-590-3p is upregulated in PCa tissues and cell lines
Real-time PCR analysis showed that miR-590-3p was significantly upregulated in PCa tissues compared to adjacent noncancerous tissues (Figure 1(a)). Further detection revealed that miR-590-3p expression was notably increased in all PCa cell lines, especially in androgen receptor (AR)-positive cells (LNCaP, 22RV1, C4-2, and VCaP; Figure 1(b)). These data suggested that miR-590-3p was upregulated in PCa tissues and cells, indicating that increased miR-590-3p expression might promote PCa development.

The expression levels of miR-590-3p in PCa tissues and cell lines. (a) Real-time PCR analysis of miR-590-3p expression in PCa tissues and their matched adjacent noncancerous (non-PCa) tissues. (b) Real-time PCR analysis of miR-590-3p expression in six human PCa cell lines (LNCaP, 22RV1, C4-2, VCaP, PC3, and DU-145) compared with normal human prostate epithelial cells (RWPE-1). The average miR-590-3p expression was normalized by U6 expression and represented as the mean ± SD of all three experiments.
MiR-590-3p promotes the proliferation and invasion of PCa cells
To further assess the effect of miR-590-3p on PCa progression, miR-590-3p mimics, miR-590-3p inhibitors, or miR-NC oligonucleotides were transfected into two PCa cell lines, LNCaP (androgen dependent) and C4-2 (androgen independent), respectively. As shown in Figure 2(a), miR-590-3p mimics significantly upregulated, while miR-590-3p inhibitors markedly downregulated the expression of miR-590-3p. MiR-590-3p mimics significantly accelerated, while miR-590-3p inhibitors notably reduced cell proliferation in both LNCaP (Figure 2(b)) and C4-2 cells (Figure 2(c)). Furthermore, miR-590-3p mimics lead to a significant promotion of invasive properties, whereas miR-590-3p inhibitors result in a suppression of invasive activities in both LNCaP and C4-2 cells (Figure 2(d)). Moreover, miR-590-3p mimics markedly accelerated, while miR-590-3p inhibitors notably suppressed the growth of xenografted tumors (Figure 2(e) and (f)). These results indicated that increased miR-590-3p can promote the proliferation and invasion of PCa cells.
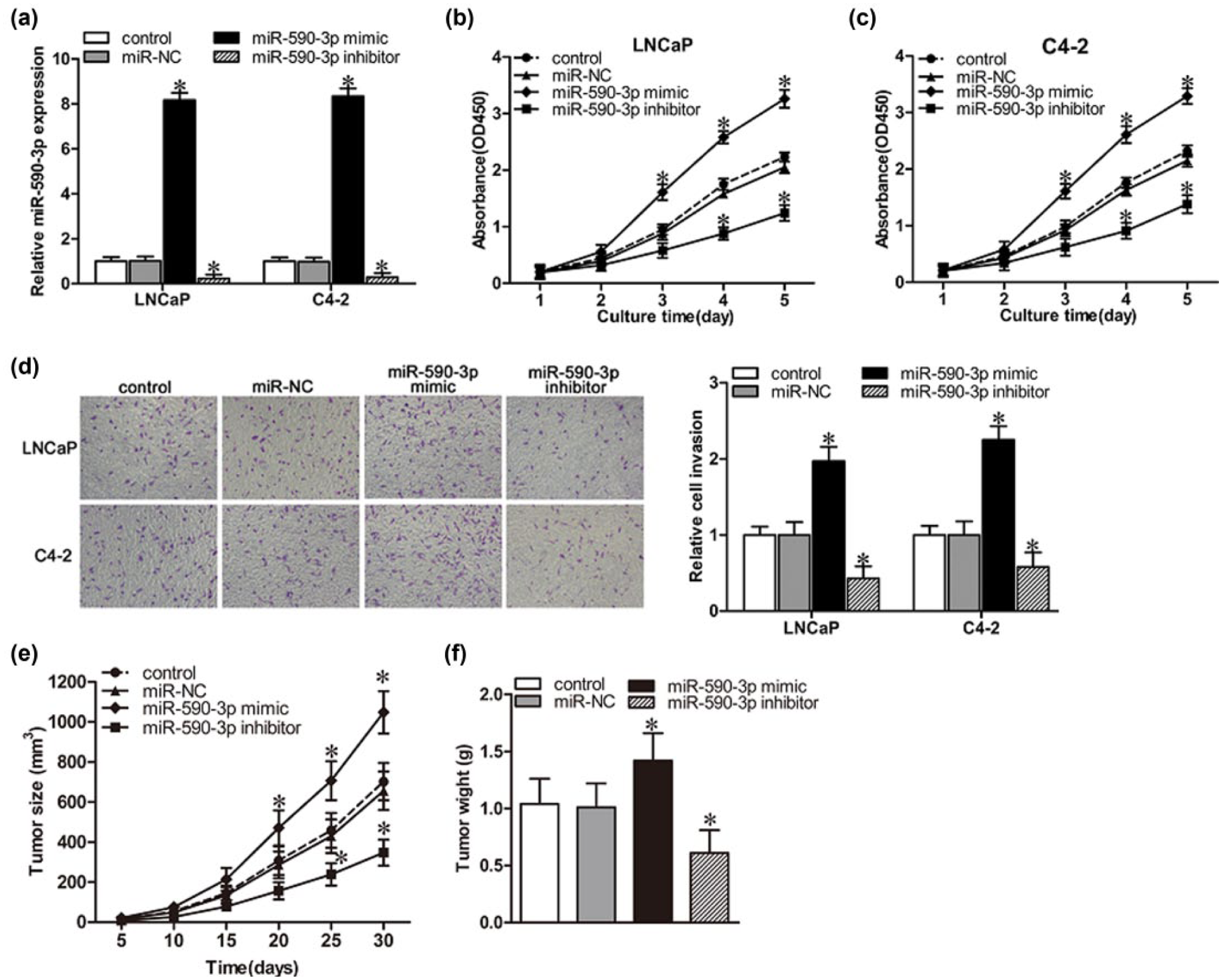
MiR-590-3p affects PCa cell proliferation and invasion in vitro. LNCaP and C4-2 cells were transfected with miR-590-3p mimics, miR-590-3p inhibitors, or miR-NC. Various indexes were measured after 48-h incubation. (a) Expression of miR-590-3p in transfected LNCaP and C4-2 cells was quantified by real-time PCR. (b and c) CCK-8 analysis of cell proliferation rates of transfected (b) LNCaP and (c) C4-2 cells. (d) Representative micrographs (left panel) and quantification (right panel) of invasiveness of LNCaP and C4-2 cells, as determined by a transwell assay. (e and f) In vivo tumorigenesis assay was used to measure (e) tumor volume and (f) tumor weight. All data are expressed as mean ± SD in at least three experiments.
INPP4B is a direct target of miR-590-3p
As we know, miRNAs exert biological functions by regulating specific target genes. To explore the molecular mechanism of miR-590-3p function in the proliferation and invasion of PCa cells, algorithm programs (microRNA.org, TargetScan, miRDB, etc.) were used to pick out the potential target genes of miR-590-3p in humans, and INPP4B was found to be one of the theoretical targets of miR-590-3p (Figure 3(a)). Luciferase reporter assay showed that miR-590-3p mimics significantly repressed the luciferase activity in INPP4B 3′-UTR-wt-transfected cells but not in INPP4B 3′-UTR-mut-transfected cells (Figure 3(b)). Contrary to higher expression of miR-590-3p, INPP4B showed lower levels in PCa tissues compared to adjacent noncancerous tissues (Figure 3(c)). Spearman correlation analysis indicated a negative correlation between miR-590-3p and INPP4B messenger RNA (mRNA) expression in PCa specimens (Figure 3(d)). Moreover, miR-590-3p mimics significantly downregulated, while miR-590-3p inhibitor remarkably upregulated the protein expression of INPP4B in both LNCaP and C4-2 cells (Figure 3(e)). As INPP4B acts as a tumor suppressor in PCa and its loss of expression is associated with poor prognosis for PCa patients, 7 we investigated the effects of miR-590-3p-mediated downregulation of INPP4B on cell proliferation and invasion in PCa cells. INPP4B was overexpressed by using pcDNA3.1(+)/INPP4B (Figure 3(f)), and as a result, miR-590-3p mimic–induced cell proliferation and invasion were significantly abrogated in both LNCaP and C4-2 cells (Figure 3(g) and (h)). Overexpression of INPP4B also notably repressed the growth of xenografted tumors (Figure 3(i)). These data suggested that INPP4B is a direct target of miR-590-3p, and miR-590-3p-mediated suppression of INPP4B is important for cell proliferation and invasion in PCa cells.

MiR-590-3p regulates PCa cell proliferation and invasion by targeting INPP4B expression. (a) Predicted miR-590-3p target sequence in the 3′-UTR of INPP4B (INPP4B 3′-UTR-wt), and a mutant containing a mutated miR-590-3p binding site seed sequence in the 3′-UTR of INPP4B (INPP4B 3′-UTR-mut). (b) Luciferase assay of INPP4B 3′-UTR-wt and INPP4B 3′-UTR-mut luciferase reporter co-transfected with the miR-590-3p mimic or miR-NC. (c) mRNA expression of INPP4B in PCa tissues and their matched non-PCa tissues. (d) Spearman’s correlation analysis between miR-590-3p and INPP4B expression in PCa specimens. (e) Western blotting analysis of INPP4B protein levels in LNCaP and C4-2 cells transfected with miR-590-3p mimic or miR-590-3p inhibitor, compared with miR-NC. β-actin served as the loading control. (f) mRNA expression of INPP4B in LNCaP and C4-2 cells transfected with pcDNA3.1(+)/INPP4B, compared with empty pcDNA3.1(+) plasmid. (g) CCK-8 analysis of cell proliferation of LNCaP and C4-2 cells transfected with miR-NC, miR-590-3p mimic, miR-590-3p, and pcDNA3.1(+)/INPP4B. (h) Transwell invasion analysis of LNCaP and C4-2 cells transfected with miR-NC, miR-590-3p mimic, miR-590-3p, and pcDNA3.1/INPP4B. (i) Changes of tumor weights after different transfection. All data are expressed as mean ± SD in at least three experiments.
MiR-590-3p exerts its function partly by regulating INPP4B/Akt/FoxO3a pathway
Since it was previously reported that loss of INPP4B drives localized activation of the phosphatidylinositol 3-kinase (PI3K)/Akt signaling pathway, 12 we next evaluated whether the activity of PI3K/Akt signaling and the expression of its downstream genes would be modulated by miR-590-3p expression in PCa cells. There was a significant increase in the expression of phosphorylated AKT (p-Akt) and phosphorylated FoxO3a (p-FoxO3a) in miR-590-3p-mimic-transduced cells (Figure 4(a)). Moreover, miR-590-3p mimics significantly upregulated, while miR-590-3p inhibitors markedly downregulated the protein and mRNA levels of cyclin D1 in both LNCaP and C4-2 cells (Figure 4(a) and (b)). Simultaneously, miR-590-3p mimics dramatically downregulated, while miR-590-3p inhibitors markedly upregulated the protein and mRNA levels of p21 in both LNCaP and C4-2 cells (Figure 4(a) and (c)). Interestingly, transfection with pcDNA3.1(+)/INPP4B or pre-treatment with the PI3K/Akt kinase inhibitor LY294002 in miR-590-3p-overexpressing cells partly prevented these changes (Figure 4(d)–(f)). These consequences indicated that miR-590-3p promotes PCa progression partly through the INPP4B/Akt/FoxO3a pathway.
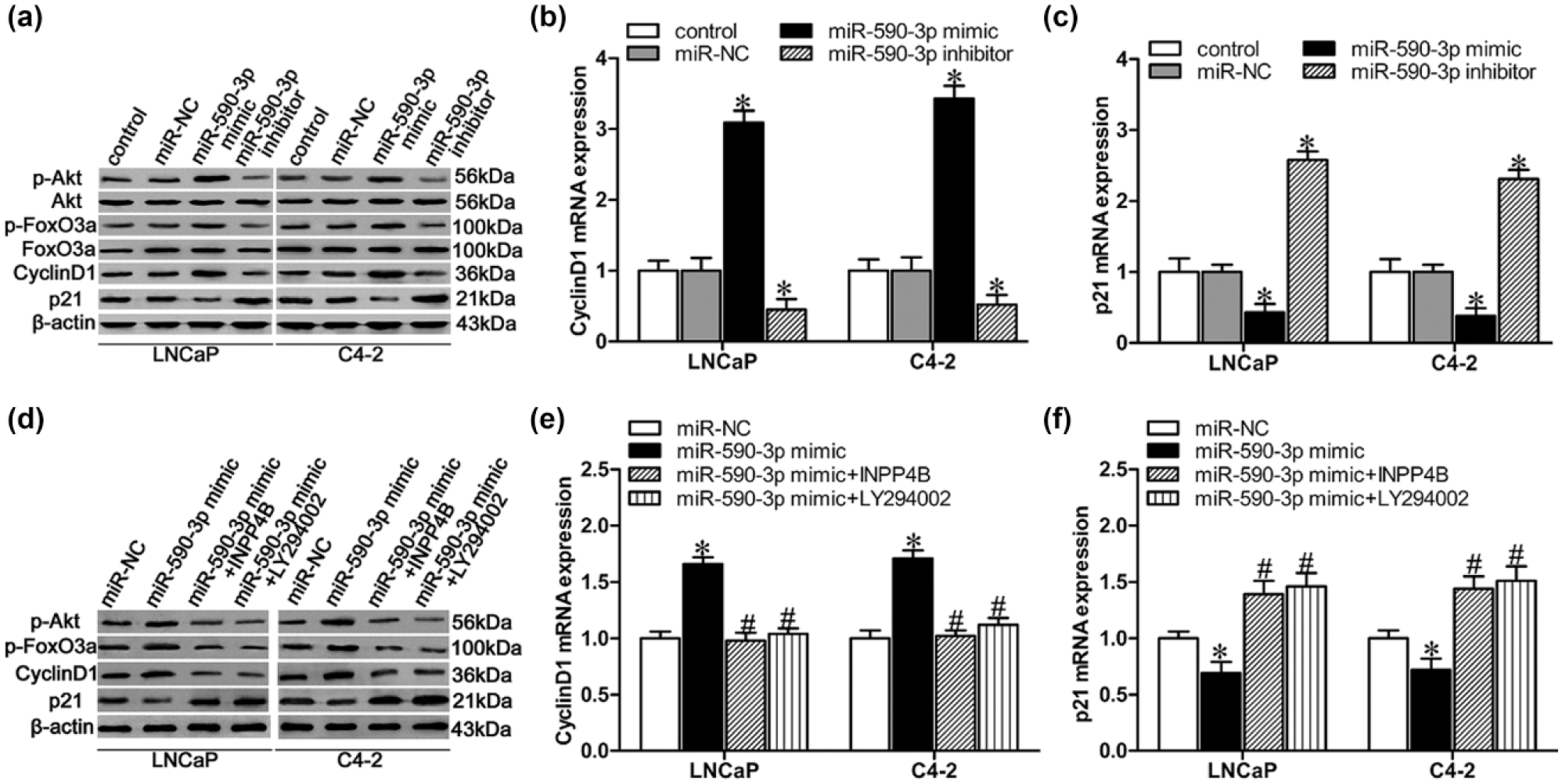
MiR-590-3p regulates PCa progression via the INPP4B/Akt/FoxO3a pathway. (a) Western blot analysis to measure p-Akt, Akt, p-FoxO3a, FoxO3a, cyclin D1, and p21 protein levels in LNCaP and C4-2 cells after transfection with either miR-590-3p mimic, miR-NC, or miR-590-3p inhibitor, with β-actin as loading control. (b and c) Real-time PCR analysis of (b) cyclin D1 mRNA and (c) p21 mRNA expression levels in LNCaP and C4-2 cells after transfection with miR-590-3p mimic, miR-NC, or miR-590-3p inhibitor. (d) Western blot analysis to measure p-Akt, p-FoxO3a, cyclin D1, and p21 protein levels in LNCaP and C4-2 cells after transfection with miR-590-3p mimic, cotransfection with pcDNA3.1(+)/INPP4B, or pre-treatment with the PI3K/Akt kinase inhibitor LY294002. (e and f) Real-time PCR analysis of (e) cyclin D1 mRNA and (f) p21 mRNA expression levels in LNCaP and C4-2 cells after transfection with miR-590-3p mimic, cotransfection with pcDNA3.1(+)/INPP4B, or pre-treatment with the PI3K/Akt kinase inhibitor LY294002.
Discussion
MiRs are regarded as one of the important modulators in the pathogenesis and progression of various cancers by affecting the expression of multiple genes which play key roles in tumorigenesis, chemoresistance, or other pivotal processes in cancer development.13–15 INPP4B can function as a carcinostatic factor through negatively regulating PI3K/Akt signaling in PCa development.7,16 Here, we demonstrated for the first time that miR-590-3p is markedly elevated in both PCa cells and clinical tissues and confirmed that miR-590-3p served as a tumor promoter by regulating cell proliferation and invasion in PCa development by involving the INPP4B/Akt/FoxO3a pathway.
Among types of miRs involved in human cancer, miR-590-3p is an active epigenetic regulator, acting as either an oncogene or a tumor suppressor in tumorigenesis.17,18 As previously discovered, overexpression of miR-590-3p can promote cell proliferation in hepatocellular cancer 17 but suppress cell migration, invasion, and epithelial–mesenchymal transition in glioblastoma multiforme. 18 MiR-590-3p can also inhibit cell proliferation, migration, and colony-forming ability in bladder carcinoma. 19 However, the expression and function of miR-590-3p in PCa development remains unknown. For the first time, we have demonstrated that miR-590-3p expression is upregulated in PCa cells. Previous research reported that miR-590-5p, another arm of hsa-miR-590, 20 was significantly overexpressed in CRPC. 21 Further studies indicated that ectopic expression of miR-590-3p dramatically promoted, whereas suppression of endogenous miR-590-3p attenuated the proliferation and invasion of LNCaP and C4-2 cells. Moreover, miR-590-3p mimics accelerated the growth of xenografted tumors. Collectively, these findings suggest that miR-590-3p is correlated with PCa progression and might play an important part during PCa development.
INPP4B was identified as one of the potential targets of miR-590-3p in bioinformatics analysis. Western blot and luciferase analysis confirmed that miR-590-3p can suppress INPP4B expression by directly targeting its mRNA 3′-UTR. Recent research showed that INPP4B is highly expressed in the luminal epithelium of normal prostate, and its loss of expression contributes to tumor recurrence and poor long-term survival in prostate carcinoma. 7 Our study demonstrated that overexpression of INPP4B can repress cell proliferation and invasion in miR-590-3p-overexpressing cells, suggesting that INPP4B downregulation is critical for miR-590-3p-induced cell proliferation and invasion in PCa cells. This result is inconsistent with a previous report that INPP4B expression did not affect cell proliferation in PC3 cells. 16 However, another study suggested that depletion of INPP4B can increase the proliferation of LNCaP cells in vitro. 6 We note that PC3 is an AR-negative cell lines, while LNCaP and C4-2 are both positive for AR. As we all known, AR plays a critical role during the development and progression of PCa.22,23 INPP4B was proved to be an androgen responsive gene, and there is a feedback loop between AR and INPP4B. 6 Moreover, miR-590-5p has been identified as androgen-regulated miRNAs; 20 miR-590-3p shared the same pre-miRNA with miR-590-5p. Together, these data allow us to suggest that miR-590-3p promotes cell proliferation and invasion in PCa development by directly targeting INPP4B, and these processes may be linked to AR signaling. Whether and how AR signaling influence miR-590-3p and INPP4B expression so as to affect PCa progression may be our next research in the future.
INPP4B has been defined as a tumor suppressor in breast cancer and melanocytic neoplasms, with negative regulation on PI3K/Akt signaling output.2,5 Here, in this study, the downregulation of INPP4B induced by miR-590-3p was accompanied by the activation of Akt and FoxO3a signaling, upregulation of the G1/S transitional promoter cyclin D1, and downregulation of the cyclin-dependent kinase (CDK) inhibitor p21. Several reports have demonstrated that INPP4B exerts its effects on cell proliferative, invasive, and tumorigenic capacity by inhibiting PI3K/Akt signaling.12,24 FoxO3a, a forkhead transcription factor with antagonistic roles in various cancers, is one of the major targets of the PI3K/Akt pathway, 25 and has been reported to be involved in cell proliferation and invasion.26,27 Cyclin D1 is a cell-cycle regulator that participates in cancer progression, and its silencing can inhibit proliferation, induce apoptosis, and attenuate the invasive capacity of human glioblastoma cells. 26 As an important member of the CDK inhibitors, p21 is also involved in regulating cell invasion and proliferation. 28 Thus, expression changes of these proteins maybe jointly contribute to miR-590-3p-induced cell proliferation and invasion in human prostate carcinoma.
In conclusion, our study demonstrated that miR-590-3p plays an important role during PCa carcinogenesis. MiR-590-3p upregulation dramatically induces PCa cell proliferation and invasion via the INPP4B/Akt/FoxO3a pathway. This report may provide a novel target for PCa diagnosis and therapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
