Abstract
In this study, circulating microRNAs (miRNAs) are being investigated as non-invasive biomarkers for early diagnosis and prognosis of human cancers. Since the prognosis for pancreatobiliary subtype of periampullary carcinoma is poor, we assessed the prognostic relevance of miRNAs in combination with CA19-9 as noninvasive biomarker in periampullary carcinoma. Circulating miRNAs in plasma and serum CA19-9 were evaluated in periampullary carcinoma patients (n = 109) undergoing Whipple’s pancreaticoduodenectomy and in healthy volunteers (n = 92). Tumour tissues were subjected to staging and subtyping prior to determining differentially expressed miRNAs in them by quantitative real-time polymerase chain reaction (qRT-PCR). Statistical analysis involved correlation, receiver operating characteristic, logistic regression, survival analyses and Cox-proportional regression. Of the three differentially expressed circulating miRNA, miRNA192 was significantly increased both in circulation and in tumour tissue and correlated with tumour stage and aggressiveness (r = 0.96, P < 0.0001). Area under the curve of circulating miRNA192 + CA19-9 combination was 0.877 (95% confidence interval, 0.72 to 0.96) for stage III and 0.92 (95% confidence interval, 0.77 to 0.88) for tumour aggressiveness. The combination was associated with poor survival (median: 22 months, P = 0.0008) in stage III patients. Cox-proportional regression analysis revealed prognostic importance of combination of circulating miR192 and CA19-9 (HR = 1.005, P = 0.0001) in periampullary carcinoma. In conclusion, combination of circulating miRNA192 with serum CA19-9 is a better prognostic biomarker than CA19-9 alone.
Introduction
Periampullary carcinomas (PACs) are a heterogeneous group of neoplasms arising within 2 cm around ampulla of Vater. They may originate either from head of pancreas or distal common bile duct or duodenum or ampulla. Tumours arising from duodenum and intraduodenal region of ampulla are classified as intestinal subtype and those arising either from pancreas or distal bile duct are classified as pancreatobiliary subtype. 1 The clinical hallmark of these tumours is painless obstructive jaundice with surgery (Whipple’s pancreaticoduodenectomy) as the only curative treatment available. Although clinical presentation and treatment options for intestinal and pancreatobiliary types are similar, prognosis of pancreatobiliary type is poor. Tumour-specific 5-year survival rates are reported to vary with regard to the site, the rates being 15% from pancreatic region, 27% distal bile duct, 39% ampullary and 59% in duodenal tumours.2,3 This necessitates identifying circulating biomarkers to predict the prognosis and survival of PAC patients.
Several studies have demonstrated the importance of circulating microRNAs (miRNAs) in early diagnosis and prognosis of human cancers.4–7 miRNAs are novel, endogenously expressed noncoding RNAs (~20–25 nucleotide) performing basic cellular functions with distinct roles in inflammation, fibrosis and malignancy.8,9 Studies from various groups demonstrated differential miRNA expression patterns in pancreatic adenocarcinoma, ampullary adenocarcinoma and PAC tumour tissue.10,11 Studies from our own group demonstrated differential miRNA profiles in PAC with 20 miRNAs being upregulated and 9 being down-regulated in PAC tumour tissue, in comparison with adjacent non-tumour tissue. 12 Our results identifying common miRNA signatures between the subgroups of PAC suggested that they may have a definite role in disease pathogenesis as well as in disease prognosis. 12 In continuation of these results, we proposed to assess the relevance of miRNA in the prognosis of PAC. The specific objectives of this study were (a) to evaluate the expression of circulating miRNAs that were differentially expressed in PAC tumour tissue and (b) to assess the prognostic relevance of the identified miRNAs independently or in combination with serum CA19-9 in PAC.
Materials and methods
Study design
This is a prospective and observational study. Nine differentially expressed miRNAs identified in our previous study in PAC tumour tissues 12 were evaluated in circulation. Circulating miRNAs with significant differential expression in screening cohort were validated in the validation cohort and analyzed in combination with CA19-9 to predict tumour characteristics prior to surgery. Patients meeting the survival criteria of minimum survival of 12 months were considered for survival analysis (Figure 1).

Study design.
Patients and healthy participants
The study was conducted at the Asian Institute of Gastroenterology during January 2012 to June 2015 following the Reporting Recommendations for Tumour Marker Prognostic Studies (REMARK) guidelines. 13 Patients included in the study were newly diagnosed and treatment naive, histologically confirmed to have PAC on biopsy specimens collected during endoscopic retrograde cholangiopancreatography (ERCP; n = 109). Whole blood was collected from patients undergoing Whipple’s pancreaticoduodenectomy (n = 109). Tumour tissue and adjacent tumour-free tissue (2 cm away from tumour) were obtained for histological confirmation of malignancy and for tumour staging and aggressiveness. Classification of the tumours and TNM staging followed World Health Organization (WHO) guidelines. 14 Patients with concomitant acute and chronic pancreatitis and pancreatic adenoma, pancreatic adenocarcinoma, cholangiocarcinoma and duodenal carcinoma were excluded from the study. Tissue specimens were snap frozen and stored in TRIzol at −80°C. Whole blood (5 mL) was collected from all patients (prior to surgery and post surgery up to 3 years) and healthy volunteers willing to participate in the study as controls (n = 92) in paired box (PAX) gene blood RNA tubes (Qiagen). Informed consent was obtained from all the participants and the protocol was approved by the institutional review board (AIG/AHF IRB: 16/2012 dated 7 February 2012).
RNA isolation
Total RNA was isolated from plasma of PAC patients and healthy controls (n = 201) as well as from tumour and tumour-free tissues (n = 218) as required using miRNeasy Mini Kit (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions, and the quality was assessed as described earlier. 10
miRNA selection and detection
Nine miRNAs (miRNA-31, 375, 192, 196a, 215, 194, 203, 130b and 217), which were reported earlier 12 to be significantly differentially expressed in PAC tissues, were measured in the plasma of PAC patients and healthy participants. U6SnRNA (U6 small nuclear RNA) was used as internal standard for normalizations as described by earlier investigators. 15 To determine miRNA expression levels, 20 ng/µL of miRNA from plasma and tissue samples were reverse transcribed followed by quantitative real-time polymerase chain reaction (qRT-PCR) TaqMan assays according to manufacturer’s instructions (Applied Biosystems, USA). 10 Relative change in expression was calculated using mean threshold cycle (CT) values of target miRNA and reference internal control (ΔΔCt method).
Determination of circulating CA19-9 levels
Post stenting, serum CA19-9 levels were measured before surgery employing commercially available kits (Roche Cobas E601, Roche, Switzerland) involving electro-chemiluminescence immunoassay with the upper limit of normal reference value for this method being 27 U/mL.
Statistical analysis
The data (ΔCt values) used for the analysis were derived from the ΔΔCt method. Descriptive statistics, frequency distribution and Student’s t-test were used to test the significance. Receiver operating characteristic (ROC) curve analysis was used to assess the sensitivity and specificity and Pearson correlation was used to assess the correlation of miRNA expression between tissue and plasma and with tumour characteristics. Kaplan–Meier survival curve was used to assess the overall survival (OS), and statistical significance was assessed using the log-rank test. Cox proportional hazards regression model was used to determine whether plasma miRNAs serve as prognostic markers. Statistical package for Social Sciences (SPSS version 20) and MedCalc (Mariakerke, Belgium) were used for the analysis. P-value with <0.05 was considered as significant with a two-sided test.
Results
Patient characteristics
Clinical, demographic and tumour characteristics of patients are given in Table 1. All the Whipple’s surgeries were R0 resections. Out of 109 PAC patients recruited, good quality RNA could be obtained from both tumour tissue and plasma in 74 patients. Initially, tissue and plasma samples that were analyzed for the nine miRNAs from 40 patients were considered as the screening cohort. The identified differentially expressed miRNAs analyzed in the tissue and plasma of the 34 PAC patients were considered as the validation cohort.
Clinical, demographic and tumour characteristics of the PAC patients.

Circulating miRNA 192 showed increased expression and positive correlation with that of tumour
All the nine miRNAs that were identified to be differentially expressed in our previous study were detectable in plasma samples of the PAC patients. While all the nine miRNAs were confirmed to have differential expression in PAC tumour tissues in the screening cohort, only five (miR 375, 31, 192, 196a and 194) showed differential expression in the plasma of PAC patients in comparison to the controls. Of these five, miR375 and miR31 were significantly decreased (P < 0.0001), while miRNA192 showed significant increased expression (P < 0.0001) in circulation. Comparison of miRNA expression between tissue and plasma revealed similar pattern of expression of miRNA192 and miR375. While the expression of miRNA192 was increased in tumour tissue as well as in plasma (3.4 folds in circulation and 9.3 folds in tumour tissue), miR375 showed decreased expression (3.0 folds in tissue and plasma). miR194, 196a were dissimilar in their expression pattern between plasma and tissue (the expression increased in tumour tissues by 3.5 fold and decreased in circulation by 2.8 and 2.4 folds, respectively) as shown in Figure 2(a). Similarly, in the validation cohort, expression of plasma miR375 and miR31 was decreased (P = 4.12 × 10−13, P = 7.91 × 10−5), and miR192 was significantly increased (P = 2.27 × 10−7; Figure 2(b)).
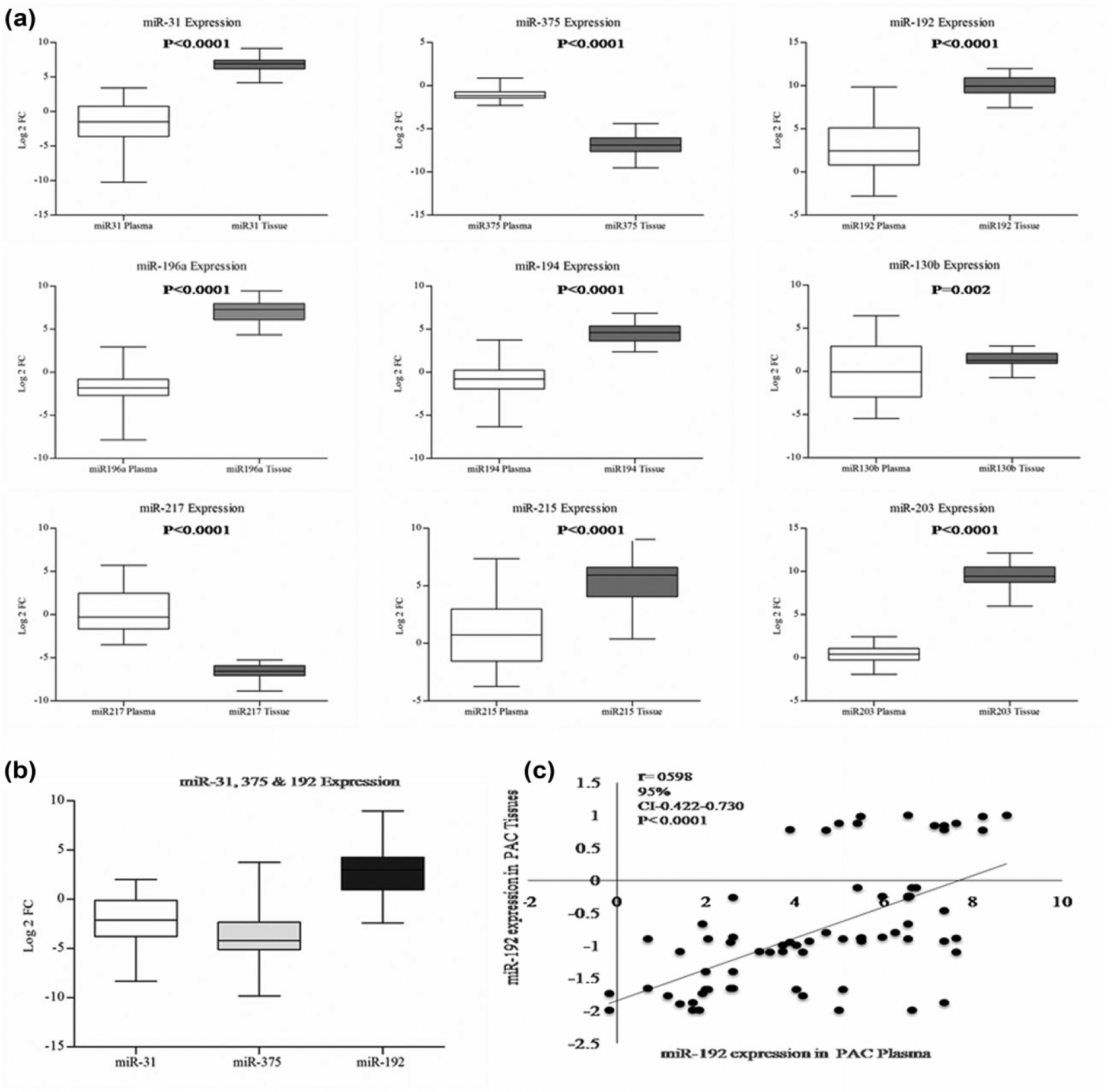
miRNA 192 is increased in plasma and tumour tissue in PAC. (a) miRNA expression in screening cohort: The miRNA expression patterns are represented in box plots with the ΔCt values. The ΔCt values are normalized expression values of miRNA threshold cycles of target miRNA versus reference housekeeping gene (U6SnRNA). The ΔCt values reflect opposite to actual expression patterns, that is, higher the ΔCt value, lower will be the expression and vice versa. The box plots of each miRNA comprised circulating miRNA expression in controls (light bar) and PAC Patients (dark bar); tissue miRNA expression from surgically resected tissues of PAC patients (dark bar) and adjacent tumour-free control (light bar). Upper panel shows differentially expressed miRNAs (miR31, 375, 192) with higher significance (P < 0.0001). Middle panel depicts miR194, 196a, which are dissimilar in plasma and tissue; while the expression increased in tumour tissues (3.5 fold), circulatory levels decreased (−2.8, −2.4, and −3.2). Four miRNAs (215, 203, 130b, 217) showed no significant change in circulation (patients vs controls). (b) Validation of miRNA 375, 31 and 192 expression in validation cohort: Box plots of miRNA 375, 31 and 192 shown as log-2-fold change in validation cohort confirm increased miR192 and decreased miR375 and 31 expression. (c) Scatter plot of miRNA 192: Expression between tissue and plasma (normalized miRNA expression values in 74 PAC patients) showed positive correlation.
In order to find a relation between tissue and plasma expressions, miRNAs were also assessed in tumour tissues of PAC patients and analyzed. Pearson correlation revealed highly significant positive correlation for miRNA 375 and 192 (r = 0.543, P < 0.0001; r = 0.780, P < 0.0001), and negative correlations for 31 and 196a (r = −0.590, P < 0.0001; r = −0.371, P < 0.019; Figure 2(c)) between tissue and plasma. miRNA192 alone showed increased and positive correlation between plasma and tumour tissue and was most promising as a non-invasive biomarker; hence miRNA192 was chosen for further analysis.
While Kaplan–Meier curve analysis showed area under the curve (AUC), sensitivity and specificity of miRNAs 375, 31 and 192 (Supplementary Figure 1), logistic regression analysis of miRNA panels ((miR31 and miR375), (miR375 and miR192), (miR31, miR375 and miR192) and (miR31, miR375, miR192 and miR196a)) significantly improved sensitivity and specificity (Supplementary Figure 2). Among all, the four miRNA combination miR 31 + 375 + 192 + 196a AUC was 99.5%.
Plasma miRNA192 and post-stent serum CA19-9 levels correlate with tumour characteristics in PAC
Correlation of plasma miRNA levels (miR 375, 31 and 192) and CA19-9 with tumour characteristics showed significant positive correlation of miRNA192 with node positivity (N1, r = 0.378, P = 0.016), tumour staging (pT3, r = 0.419, P = 0.04) and tumour aggressiveness–perineural invasion and lymphovascular embolization (r = 0.592, P = 0.0001). Circulating CA19-9 levels also correlated with tumour stage (pT3, r = 0.469, P = 0.02) and tumour aggressiveness (r = 0.476, P = 0.007). No significant correlation could be obtained between plasma levels of miR192 or CA19-9 with tumour differentiation (Supplementary Table 1).
Combination of miRNA192 with post-stent serum CA19-9 predicts tumour characteristics
Plasma miR192 and post-stent CA19-9 levels were evaluated in combination to assess the sensitivity and specificity to predict tumour characteristics such as node positivity, tumour stage and tumour aggressiveness employing binary logistic regression method. The combination (miR192 + CA19-9) derived logitP equations for node positivity, tumour stage and tumour aggressiveness are shown below.
LogitP = 1.455 − 0.3848 × miR192 + 0.0189 × CA19-9 (node positivity)
LogitP = −1.887 − 0.409 × miR192 + 0.1135 × CA19-9 (T3 stage)
LogitP = −0.3784 − 0.8028 × miR192 + 0.0738 × CA19-9 (tumour aggressiveness)
The combination had an AUC of 0.704 (95% CI, 0.525–0.883), 0.882 (95% CI, 0.783–0.982) and 0.900 (95% CI, 0.777–1.000) for node positivity, tumour stage and tumour aggressiveness, respectively (Supplementary Figure 3).
miRNA 192 expression was significantly higher in stage III PAC patients
miRNA192 expression was higher in stage III PAC patients (3.97 ± 1.02) in comparison to stage I and stage II PAC patients (1.28 ± 0.54) as shown in Figure 3(a).

(a) Comparison of miR192 expression levels in relation to the stage of the diseases of PAC patients: Left panel denotes expression in circulation, that is, in plasma, and right panel denotes expression in tumour tissues of PAC patients. Expression of miR192 in tumour tissue and plasma was significantly higher (P < 0.001) in stage III PAC in comparison to stage I and II and (b) higher levels of miR192 and CA19-9 combination are associated with poor survival: Left panel – stage I and II, right panel – stage III (a) miR192 and CA19-9 levels did not significantly correlate with survival in stage I and II patients. (b) Higher levels of miR192 and CA19-9 significantly correlated with lower survival in stage III PAC patients. The survival data were compared using the log rank test. miR192 expression levels in patients were defined as high or low relative to the interquartile range (IQR), and CA19-9 levels were defined as high or low relative to the reference range (27 U/mL).
Higher levels of circulating miRNA192 and CA19-9 are associated with poor survival in stage III patients
Kaplan–Meier survival curve analysis showed the OS rate to be 89.7% (n = 52/58). Stage I, II and III patients had 38, 32 and 22 months median survival, respectively. No significant association was found between miR192 and CA19-9 with OS. However, higher levels of miR192 in tumour tissue 6.87 ± 4.83 and plasma 3.97 ± 1.02 and serum CA19-9 64.5 ± 33.7 IU/L were significantly associated with poor survival (median: 22 months) in stage III PAC patients (P = 0.0008) as shown in Figure 3(b). Cox-proportional regression analysis revealed prognostic importance of circulating miR192 and CA19-9 (HR = 1.005, P = 0.0001) in PAC as shown in Table 2.
Cox regression analysis of PAC patients OS in relation to miR192 and CA19-9 levels.

OS: Overall survival.
Plasma miRNA192 levels decreased after surgery
Plasma miRNA192 levels increased in PAC patients prior to surgery (3.97 ± 1.02 in stage III and 1.28 ± 0.54 in stage I and II) in comparison to controls, while the levels decreased by the end of 3 months after surgery and continued up to 3 years in surviving patients. Pre- and post-operative expression were 3.3 ± 1.53 and 1.09 ± 0.48 log2 fold change, respectively, as shown in Figure 4.

Circulating pre- and post-operative miRNA expression: Post-operative expression levels of miRNA192 showed decreased expression. Preoperative levels were 3.33 ± 1.53 log2 fold change and post-operative levels were 1.09 ± 0.48 log2 fold change) at 1 year follow up (n = 58).
Discussion
Pancreatobiliary subtype of PAC is associated with poor prognosis in comparison to intestinal subtype, and serum CA19-9 is currently used as an independent prognostic biomarker, 16 the sensitivity and specificity of which is found to be low. In continuation of our earlier observation 12 indicating differential expression of miRNAs in tumours of PAC, we evaluated plasma miRNAs that were differentially expressed in PAC tumour tissues to assess if any of these miRNAs can be utilized as a better prognostic marker either alone or in combination with CA19-9. We demonstrate that only miRNA192 in plasma showed increased expression matching with that of PAC tumour tissue and the combination of plasma miRNA192 with CA19-9 significantly improved sensitivity and specificity of predicting tumour characteristics prior to surgery.
Differential expression of five miRNAs (miR 31, 375, 192, 196a, 194) observed in circulation suggests their suitability as non-invasive biomarkers in PAC. Further analysis revealing expression of two miRNAs 192 and 375 to be similar between tumour tissue and plasma identifies them as better candidate markers, since the level of expression in circulation is shown to reflect the tumour tissue expression. 17 Although both miRNA 192 and 375 showed similar expression patterns, miRNA 192 is considered to be a more suitable biomarker as its expression was increased in circulation and tumour tissue, while miRNA 375 showed decreased expression both in circulation and tumour tissue. Although earlier studies reported differential expression of miRNAs in tumour tissues of PAC,10–12 this is the first study demonstrating highly significant increased expression of plasma miRNA192. Previously, expression of miRNA192 was demonstrated to be up-regulated in gastric and pancreatic tumour tissues18,19 and down-regulated in lung and colon tumour tissues.20,21 Although, circulating miR192 was reported to be elevated in pancreatic ductal adenocarcinoma 19 (PDAC), the specificity was only 55% to detect PDAC. Since our results also demonstrated elevated circulating levels of miRNA192 in PAC, we further evaluated miRNA 192 for its utility as a prognostic biomarker in PAC.
Positive correlation of miRNA 192 between tissue and plasma (Figure 2(c)) and decrease in plasma miRNA192 levels after surgery (Figure 4) observed in this study indicate that plasma miRNA192 originates from the tumour tissue in PAC. Previous studies have also shown that circulating miRNAs in blood originate from sites of tissue injury such as tumours, 17 inflammatory loci 22 and endothelial cell damage. 23 Therefore, miRNA 192 was further assessed for its prognostic relevance in PAC patients. miR-192-5p belongs to a miRNA cluster (miR-194-2-192) localized at llql3.1, and its expression has been related to different cellular processes, in particular cell proliferation and apoptosis. Its transcription is regulated by p53, and in turn affects the mRNA expression of genes involved in cell-cycle progression. 24 miRNA 192 showed different patterns of expression in different types of malignancies and of their association with OS and recurrence-free survival. Feng et al. 20 showed miR192 to target retinoblastoma and suppress cell proliferation and induce cell apoptosis in lung cancer cells. Conversely, in pancreatic cancer and gastric cancer, ectopic expression of miR192 was shown to enhance cell proliferation, migration, reduced apoptosis and promoted cell cycle progression from the G1 to S phase.18,19 miRNA 192 differential pattern was not observed in inflammatory disease of pancreas such as in chronic pancreatitis. 25 miRNA 192 was shown to be decreased in colon cancer tissues 21 and Sandhu et al. 12 reported association of tissue miRNA192, 194 families with OS and recurrence-free survival. Although we could find an increase in circulating levels of miRNA192 in the pancreatobiliary subtype of PAC, we could not find significant difference in circulating levels of miRNA 192 in the intestinal subtype (n = 10), probably because intestinal subtype numbers are low as compared with pancreatobiliary subtypes. A majority of PACs are usually pancreatobiliary subtype as reported by Sandhu et al. 12 The main limitation of this study is that the recruitment rate was slow as the global incidence of PAC is low. Increased circulation levels of miRNA 192 in PAC found in this study is similar to that in PDAC, probably because PDAC and a majority of PACs arise from pancreas.
Tumour stage, tumour invasion, lymph node involvement, lymphatic invasion of the tumour and higher levels of CA 19-9 are accompanied by a reduced long-term prognosis in PAC.25,26 Since increased levels of miRNA192 and CA19-9 in circulation are found to be associated with tumour stage and reduced long-term prognosis, respectively, combination of miR192 and serum CA19-9 can be a better non-invasive biomarker to assess the tumour characteristics prior to surgery, in comparison to CA19-9 alone.
While earlier studies demonstrated a combination of miRNA panels to improve the sensitivity and specificity of predicting prognosis and survival in other malignancies,26–28 we tried to develop combination panels of differentially expressed miRNAs with an AUC > 75% and combination of miRNAs with CA19-9 in plasma to improve sensitivity and specificity of predicting tumour characteristics prior to surgery. All the combination panels significantly improved AUC, sensitivity and specificity. Since our objective was to evaluate the prognostic importance of CA19-9 in combination with identified miRNAs, we further tested the combinations with tumour characteristics and survival. Of all the combinations tested, miRNA192 in combination with CA19-9 showed significant improvement in AUC for tumour staging and aggressiveness (logitP model). Recently, combination of CA19-9 and miRNA panel was demonstrated to have 83.6% accuracy for early detection of pancreatic cancer.27,28 Our study assessing CA19-9 alone with tumour characteristics showed an AUC of 71.2% for tumour staging and 82.4% for tumour aggressiveness. Combined analysis of circulating CA19-9 and that of miR192 improved AUC and showed 87.7% for tumour staging and 92.4% for tumour aggressiveness. The clinical implication of this study is that the combination of miRNA192 with CA19-9 can be used to predict the tumour characteristics prior to surgery.
In this study, we observed a significant association of higher levels of plasma miRNA192 (3.97 ± 1.02) and CA19-9 (64.5 ± 33.7) with poor survival of 22 months in stage III PAC patients and not with survival in stage I and stage II PAC patients with 36 months median survival. The association of higher level of miRNA 192 only with stage III PAC could be due to the fact that the proliferating rate of tumour cells would be high in stage III and miRNA192 has a role in cell proliferation and apoptosis. 24 Higher AUC of 87.7% for tumour stage and 92.4% for tumour aggressiveness for the miRNA192 and CA19-9 combination in comparison to 71.2% for tumour stage and 82.4% for tumour aggressiveness for CA19-9 alone, as our results demonstrated, suggests that the combination of plasma miRNA192 with CA19-9 can be used as a predictive marker for assessing tumour characteristics prior to surgery. However, its clinical use needs to be assessed in a larger cohort before it can be used as a liquid biopsy. These results gain credence from a recent study demonstrating association of miRNA192 PAC tumour tissue expression levels with OS and recurrence-free survival. 12 Liu et al. 28 have shown the importance of miRNAs 16 and 196a combination with CA19-9 for early detection of pancreatic cancer, 27 while our results have shown the importance of combination of circulating miRNA192 with serum CA19-9 as a prognostic indicator in PAC. In conclusion, results of this study indicate that circulating miRNA192 in combination with CA19-9 is a better non-invasive biomarker to determine tumour stage, prognosis and survival in PAC in comparison to determination of serum CA19-9 alone.
Footnotes
Acknowledgements
The authors are thankful to Prof. C. Subramanyam for carefully editing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This research was funded by Asian Healthcare Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
