Abstract
The SMAD proteins are responsible for transducing signals from activated transforming growth factor-beta. This is the first study assessing the expression of SMAD-1/8, SMAD-2/3, SMAD-4, and SMAD-7 in chronic lymphocytic leukemia cells with regard to their clinical significance and potential prognostic value. Overexpression of SMAD-1/8 was observed in 160 chronic lymphocytic leukemia patients compared to 42 healthy volunteers (p = 0.023) and was associated with a more progressive course of the disease (p = 0.016). Moreover, the high expression of SMAD-1/8 correlated with other, well-established prognostic factors, including clinical stage (p = 0.010) and lymphocyte doubling time (p = 0.021). The expression of SMAD-4 was lower in chronic lymphocytic leukemia patients compared with the control group (p = 0.003). Importantly, lower SMAD-4 levels correlated with longer progression-free survival (p = 0.009), progressive course of the disease (p = 0.002), advanced clinical stage (p = 0.0004), elevated beta-2-microglobulin and lactate dehydrogenase levels (p < 0.05), shorter lymphocyte doubling time (p = 0.009), and CD38 antigen expression (p = 0.039). In addition, lower SMAD-4 expression correlated with lower apoptotic index (p = 0.0007) and lower expression of receptors for vascular endothelial growth factors VEGFR-1 and VEGFR-2. A significant association was found between the low expression of inhibitory protein SMAD-7 and both zeta-chain-associated protein kinase 70–negative cells (p = 0.04) and lower apoptotic index (p = 0.004). No differences were observed in SMAD-2/3 expression. In conclusion, our results demonstrate a significant correlation between greater SMAD-1/8 and lower SMAD-4 expression in chronic lymphocytic leukemia cells, as well as more progressive outcome and poor prognosis. These data provide supporting evidence that the expression of SMAD proteins plays an important role in disease development and may be considered as a novel, biologic prognostic factor in this disease.
Keywords
Introduction
Chronic lymphocytic leukemia (CLL) still remains an incurable disease, in spite of the wide variety of novel drugs. It is an extremely heterogeneous disease with different clinical manifestation and overall survival (OS) among patients. As a result, it is necessary to stratify the patients according to prognostic risk. 1 Several novel prognostic parameters can be used besides such older, commonly used prognostic markers as age, Rai and Binet classification, or lymphocyte doubling time (LDT). 2 These include serum beta-2-microglobulin (β2-M) level; lactate dehydrogenase (LDH) activity; genomic aberrations such as 17p deletion, 13q deletion, trisomy 12, and 11q deletion; the mutational status of the B-cell receptor (BCR) and immunoglobulin variable heavy chain (IgVH); and expression of the CD38 antigen and zeta-chain-associated protein kinase 70 (ZAP-70).3,4 Recently, spontaneous in vitro apoptosis and accelerated angiogenesis have also been reported to have predictive value.5–7 Although significant progress has been made in the development of prognostic factors for CLL, still it is difficult to predict which patients will suffer from rapid progression and require early treatment. This study investigates the relationship between the expression of SMAD proteins and its potential prognostic value.
The SMAD proteins are a crucial element of one of the most versatile cytokine signaling pathways, the transforming growth factor-beta (TGF-β) pathway. 8 The term SMAD proteins derived from the combination of the Drosophila protein mothers against decapentaplegic (MAD) and the Caenorhabditis elegans protein SMA (from SMA gene for small body size). Based on the differences in their function, the mammalian SMAD family members are divided into three classes. The first group consists of receptor-associated SMADs (R-SMADs), including SMAD-1, SMAD-2, SMAD-3, SMAD-5, and SMAD-8. SMAD-1 and SMAD-5 are phosphorylated by activated type I receptor for TGF-β involved in the bone morphogenetic protein (BMP)-dependent pathway, while SMAD-2 and SMAD-3 are phosphorylated by the same receptor type but associated with activin (Act) pathway. SMAD-4 is the only molecule in the co-operating SMAD (Co-SMAD) group, which is responsible for forming complexes with R-SMADs. The third SMAD category, which has an inhibitory role (I-SMAD), consists of SMAD-6 and SMAD-7. 9 To initiate a particular TGF-β response, the activated R-SMADs bind to the SMAD-4 and form complexes, which enter into the cell nucleus, where they regulate transcription of the specific target genes. 10
TGF-β is a cytokine of great importance, involved in the regulation of major cellular processes, including proliferation, division, differentiation, and apoptosis. It regulates tissue fibrosis and regeneration in the liver, kidney, and lung, and it has many important functions during embryonic development. 11 Moreover, the TGF-β/SMAD pathway plays an important role in regulating apoptosis and angiogenesis. 12 As these processes have great prognostic importance in CLL patients, they are examined in this study together with several other well-known prognostic factors.
According to current knowledge, disturbances in SMAD expression have been noted in many different diseases. An imbalance in SMAD protein level is commonly observed in autoimmune diseases, especially rheumatoid arthritis and systemic lupus erythematosus, cardiovascular diseases, and in many types of cancer.13,14 Abnormalities in SMAD genes are frequent during cancerogenesis. Although the TGF-β/SMAD pathway is a potent tumor suppressor of normal cells, it acts as an enhancer of invasion and metastasis in more advanced carcinoma cells.15,16 Mutated or aberrant genes lead to the inactivation of SMAD proteins, causing the cells to become resistant to TGF-β growth inhibition. As a result, cancer cells may proliferate, invade, and metastasize beyond their tissue of origin. This phenomenon has been observed in many human cancers, most commonly in pancreatic, breast, and colon carcinomas.17–19 The most common mutation is being that of the SMAD-4 protein, which is observed in 50% of patients with pancreatic cancer, 20% with colon cancer, and 10% with lung cancer.20–24 Similar mutations have been observed in patients with hematological malignancies, including acute myeloid leukemia (AML). 25 Moreover, a low or undetectable level of SMAD-4 was related to tumor progression, metastasis, and unfavorable prognosis in many different types of cancer.26,27 Several studies have indicated that the overexpression of R-SMADs, especially SMAD-1 and SMAD-8, may be a valuable prognostic factor; however, further studies must be performed.28,29
This study represents the first investigation of the expression of SMAD proteins of all groups in CLL cells. In addition, they are evaluated as novel prognostic markers for disease outcome.
Materials and methods
A total of 160 treatment-naive patients with CLL, aged 52–85 (median: 69 years), were enrolled into the study. Patients were admitted to the Department of Hematology, Medical University of Lodz, Copernicus Memorial Hospital (Poland) during the years 2011–2014. The diagnosis was made based on the current guidelines. 30 The results were compared with those of 42 healthy volunteers. All participants signed an individual consent form. The study was approved by the Ethics Committee of the Medical University of Lodz.
Peripheral blood samples were obtained from untreated patients diagnosed with CLL. The clinical characteristics of the patients are shown in Table 1. At the time of diagnosis, all patients were evaluated for clinical stage according to Rai classification, LDH activity, β2-M level, and expression of CD38 and ZAP-70. LDT was evaluated after 12 months of observation. Information regarding the clinical and laboratory variables aforementioned were evaluated at local hematology centers. In addition, after 24 months of follow-up, patients were divided into groups with stable or progressive disease (SD or PD, respectively). The PD group included patients who required therapy during 2 years of follow-up visits.
The baseline characteristics of 160 patients with chronic lymphocytic leukemia.

ZAP-70: zeta-chain-associated protein kinase 70; LDH: lactate dehydrogenase; LDT: lymphocyte doubling time; β2-M: beta-2-microglobulin.
Peripheral blood mononuclear cells isolation
Peripheral blood mononuclear cells (PBMCs) were isolated from heparinized blood samples by centrifugation in Histopaque-1077 (Sigma Diagnostic, St Louis, MO, USA) density gradients. A 1:1 (v/v) mixture of blood and Hanks’ Balanced Salt Solution (HBSS; Biomed, Lublin, Poland) was layered on the top of the Histopaque media in centrifuge tubes and centrifuged for 30 min at 200g. The interphase region containing PBMCs was collected and then washed twice with HBSS and RPMI 1640 medium. Next, the cells were re-suspended in RPMI 1640 (0.5 × 106 cells/mL); 1 mL aliquots of cell suspensions were placed into 24 culture well dishes (Nunc, Roskilde, Denmark) for further cultures.
Determination of CD38 and ZAP-70 positivity of CLL cells by flow cytometry
Flow cytometric measurements were carried out on FACSCalibur (Becton-Dickinson Biosciences, San Jose, CA, USA). The antibodies used were CD19 (cytochrome-conjugated monoclonal antibody (MoAb) anti-CD19 clone) and CD5 (cytochrome-conjugated MoAb anti-CD5 clone), CD38-allophycocyanin (APC; clone HB7; Becton-Dickinson Biosciences, San Jose, CA, USA), and ZAP-70 (unlabeled, clone 2F3.2; BioLegend, San Diego, CA, USA) conjugated with Zenon reagent (Zenon™ Alexa Fluor 488 Mouse IgG2a Labeling Kit; Molecular Probes, Eugene, OR, USA). Cut-off points of 30% and 20% were used to define positivity for CD38 and ZAP-70, respectively.1,3
Assessment of apoptosis
In addition, SMAD protein expression correlated with the level of spontaneous in vitro apoptosis of CLL cells. Spontaneous in vitro apoptosis was assessed finally after 2 h of incubation in drug-free condition by the flow cytometry, using Annexin-V (Ann-V) assay. Cells were washed with phosphate-buffered saline (PBS) and then re-suspended in 100 µL of binding buffer containing 2 µL of fluorescein isothiocyanate (FITC)-conjugated Ann-V and 10 µg/mL of propidium iodide (PI; Becton-Dickinson Biosciences, San Jose, CA, USA). The samples were then incubated for 15 min (room temperature, at the dark). The fluorescence was measured by a flow cytometer using an FL1 (530 nm for Ann-V) standard emission filter. Apoptotic index (AI) was calculated as a percentage of Ann-V-positive (Ann-V+) cells.
Determination of plasma vascular endothelial growth factor receptor levels
Three vascular endothelial growth factor (VEGF) receptors—R1, R2, and R3—were assessed with the flow cytometry. MoAbs specific for VEGFR-1 were used: anti-human VEGFR-1/Flt1-phycoerythrin (PE; R&D Systems, Minneapolis, MN, USA), VEGFR-2: anti-human VEGFR-2/KDR-APC (R&D Systems), and VEGFR-3: anti-human VEGFR-3/Flt4-APC (R&D Systems). For each analysis, 100 µL of plasma was used.
SMAD protein expression evaluation
CLL cells isolated from peripheral blood were tested for the expression of SMAD-1/8, SMAD-2/3, SMAD-4, and SMAD-7 proteins. Before testing, the fixed cells were washed twice with PBS and then further permeabilized with 0.1% Tween-20 in PBS. Mouse MoAb anti-SMAD-1/8 (clone p463/pS467/p465/pS467), anti-SMAD-2/3 (anti-SMAD-1/8, p465/pS467/p423/pS425), and anti-SMAD-4 (EP618Y) were used at dilutions of 1:100–1:200; cells were incubated for 60 min in the dark at room temperature. Anti-SMAD-7 MoAb (pS463/pS465/pS465/pS467) was used at dilutions of 1:300. Cells were incubated for 60 min in the dark at room temperature. All MoAbs were from Becton-Dickinson Biosciences (Oxford, UK). Dilutions of all antibodies were prepared in 1% PBS–BSA (bovine serum albumin). The expression of the SMAD proteins was evaluated by the mean fluorescence intensity (MFI). An example of SMAD-1/8, SMAD-2/3, SMAD-4, and SMAD-7 expression is shown in Figure 1.

Exemplary differences in the expression of (b) SMAD-1/8, (c) SMAD-2/3, (d) SMAD-4, and (e) SMAD-7 proteins in flow cytometry. (a) The population of lymphocytes (SSC vs FSC axis; light scattering flow cytometry collected at two angles: side scatter (SSC) and forward scatter (FSC)) gated for further measurements is shown in blue frame. The differences between the control cells (green) and patients with CLL (red) are evaluated in mean fluorescence intensity (MFI; at the X axis).
Fluorescence in situ hybridization
Interphase fluorescence in situ hybridization (FISH) was performed on the cell suspension originated from peripheral blood according to the standard protocol. Cells were obtained by 72 h culture in the presence of 12-O-tetradecanoylphorbol-13-acetate (TPA; Sigma-Aldrich, Munich, Germany) or by direct preparation from uncultured peripheral blood. Fluorescent-labeled probes (Vysis, Downers Grove, IL, USA) were used in interphase cytogenetic analyses to detect prognostically relevant abnormalities of chromosomal regions 11q22.3 (LSI ATM), 13q14 (LSI D13S319), 17p13 (LSI p53), and centromere 12 (CEP12). The analysis was carried out as previously described. 31 Patients were categorized into high-risk (17p13.1 and ≥2 abnormalities), intermediate-risk (trisomy 12 and 11q22.3 deletions), and low-risk (13q14.3 deletion and normal) groups for subsequent analysis.
The secondary endpoints of the study: progression-free survival and OS
Median follow-up time and OS was 35 months (range: 11–45 months). All patients had follow-up visits every 3 months after SMAD assessment. Data pertaining to the types of treatment and death information for referred patients were obtained from electronic medical patient records (AMMS; Asseco Medical Management Solutions system). Our secondary endpoint was also to compare the expression of particular SMAD at the diagnosis, comparing this with progression-free survival (PFS; 27.5 months; range: 7–40 months).
Fluorescence measurements
All fluorescence measurements were performed by flow cytometry FACSCalibur (Becton-Dickinson Biosciences, San Jose, CA, USA), using standard emission filters: green (λ = 530λ20 nm; FL1), orange (λ = 560–600 nm; FL2), and red (λ > 600 nm; FL3), where necessary. A total of 10,000 cells were measured for each sample.
Statistics
The statistical analysis of data was performed using STATISTICA software v.12.0 (Tulsa, OK, USA). The range of the measured variable, means and standard deviations (SDs), and medians and ranges were calculated. The differences between values were evaluated with the Student t-test or by the Mann–Whitney U test, where necessary. The Spearman rank test was used for assessing correlations between some data sets. In addition, a log-rank test was performed to assess the potential correlation between SMAD expression and CLL patients. Values of p less than 0.05 were considered statistically significant.
Results
To elucidate the prognostic value of SMAD proteins in CLL, the expressions of SMAD-1/8, SMAD-2/3, SMAD-4, and SMAD-7 proteins of the patients were compared with those of healthy volunteers, and their correlation with established prognostic factors for the disease. In addition, the expression of SMAD proteins was investigated as a potential marker of disease progression and possibly disease onset.
Data according to SMAD-4 expression were the most valuable. Compared to the control group, the expression of SMAD-4 in CLL cells was significantly lower (p = 0.003; Figure 2; Table 1). In contrast, the level of SMAD-1/8 in CLL cells was significantly higher than that observed in the control group (p = 0.023). The expression of other SMAD proteins did not differ between CLL patients and healthy volunteers (Figure 2).

Expression of SMAD proteins in CLL cells compared to healthy lymphocytes. Mean ± standard deviation values are presented.
In addition, SMAD-4 expression varied significantly between patients with various courses of the disease. Namely, the average protein expression of SMAD-4 was significantly lower in patients with PD than in those with SD (p = 0.002; Figure 3). The expression of SMAD-4 protein negatively correlated with clinical stage according to the Rai classification (p = 0.0004; Figure 3). Furthermore, the level of SMAD-4 was found to be significantly lower in patients with shorter LDT (<12 months; p = 0.009; Figure 3; Table 2).

Comparison of SMAD-4 expression and classical prognostic factors for CLL. Mean ± standard deviation values are presented.
Comparison of SMAD-1/8, SMAD-2/3, SMAD-4, and SMAD-7 expression and classical prognostic factors in CLL.

CTRL: control; CLL: chronic lymphocytic leukemia; ZAP-70: zeta-chain-associated protein kinase 70; LDH: lactate dehydrogenase; LDT: lymphocyte doubling time; β2-M: beta-2-microglobulin; MFI: mean fluorescence intensity.
Median MFIs (Me) and/or p values are presented.
A correlation was found between SMAD proteins and novel prognostic markers for CLL patients. Serum markers such as LDH and β2-M were also tested at the time of the diagnosis. However, statistically significant differences were demonstrated only for SMAD-4 protein, whose expression was significantly lower in patients with high LDH activity in serum: equal to or more than 50% of the upper limit (p = 0.041) or equal to or greater than 100% of the upper limit of β2-M concentration (p = 0.033; Figure 4).The study also included other prognostic factors that have been previously validated, including ZAP-70 level and cell surface CD38: Positive CD38 (≥30% of cells) antigen expression on CLL cells correlated with a significantly lower SMAD-4 expression (p = 0.039; Figure 4). ZAP-70-positive (≥20%) cells were associated with a significantly higher expression of SMAD-7 protein (p = 0.040; Table 2).

Comparison of SMAD-4 expression and some other prognostic factors for CLL. Mean ± standard deviation values are presented.
Similar results were observed according to the expression of SMAD-8 protein. The levels of SMAD-1/8 significantly varied with regard to disease course, with the expression of SMAD-1/8 being significantly higher in the PD than in the SD group (p = 0.016; Figure 5). In addition, the expression of SMAD-1/8 positively correlated with higher (3 and 4) clinical stage according to Rai classification (p = 0.010; Figure 5). A relationship was found between LDT and SMAD-1/8 expression: The level of SMAD-1/8 was significantly higher in patients with LDT ≥ 12 months (p = 0.021; Figure 5; Table 2).
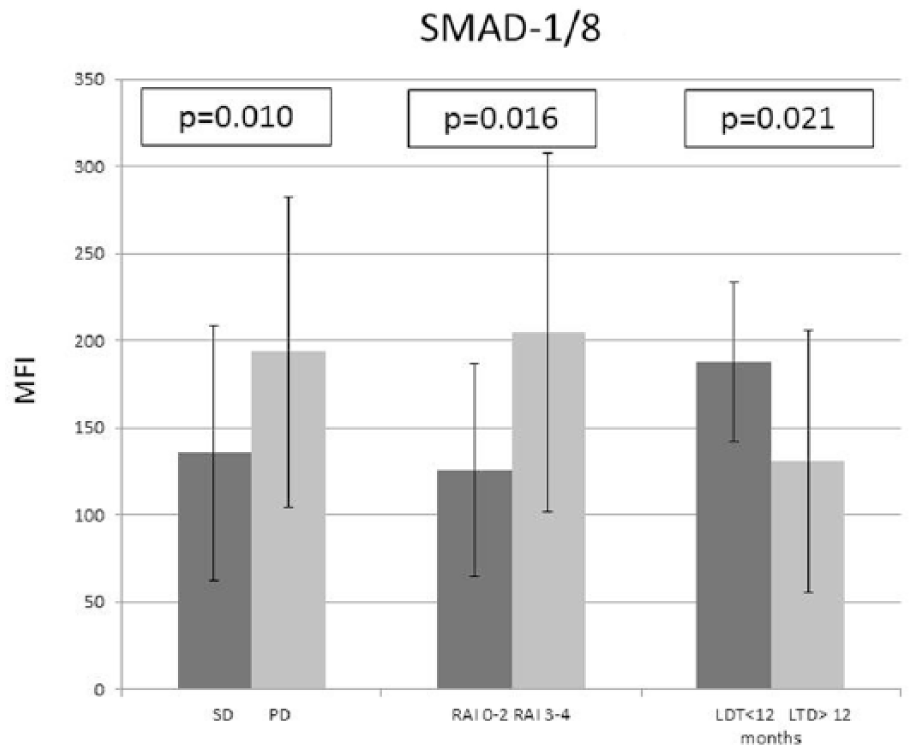
Comparison of SMAD-1/8 expression and prognostic factors for CLL. Mean ± standard deviation values are presented.
To estimate the prognostic impact of the SMAD signaling pathway, the dysregulation of angiogenesis by VEGFR-1, VEGFR-2, and VEGFR-3 expression and spontaneous in vitro apoptosis were examined. It has been shown that low SMAD-4 expression was associated with a high (above median) expression of VEGFR-1 (p = 0.009) and VEGFR-2 (p = 0.011; Figure 6; Table 3). No correlation was found between the expression of the other evaluated SMAD proteins and VEGF receptors. What is more, it has been found that high AI is observed in patients with significantly lower SMAD-4 level (p = 0.0007) and higher SMAD-7 expression (p = 0.004) (Figure 7).

Comparison of SMAD-4 and vascular endothelial growth factor expressions. Mean ± standard deviation values are presented.
Expression of vascular endothelial growth factor receptors 1–3 and apoptotic indices (AIs) of leukemic cells.

VEGFR-1: vascular endothelial growth factor receptor 1; VEGFR-2: vascular endothelial growth factor receptor 2; VEGFR-3: vascular endothelial growth factor receptor 3; MFI: mean fluorescence intensity.
The table shows comparison between cases with < versus ≥ median of particular SMAD protein expression. Median MFIs or AIs (in parentheses) and/or p values are presented.

Comparison of SMAD-4 and SMAD-7 expression and apoptosis of chronic lymphocytic leukemia cells.
Interestingly, statistical analysis revealed that the expression of SMAD proteins was not associated with any of the measured cytogenetic markers.
PFS and OS
PFS was found to be significantly longer in CLL patients with an MFI score of SMAD-4 protein lower than median (MFI < 401; p = 0.009; Figure 8(a)). This group of patients also demonstrated a distinct tendency to prolonged OS; however, these differences were not statistically significant (p = 0.089; Figure 8(b)).

(a) Correlation of SMAD-4 expression with progression-free survival (PFS) of patients with chronic lymphocytic leukemia. (b) Correlation of SMAD-4 expression with overall survival (OS) of patients with chronic lymphocytic leukemia.
No statistically distinct differences between PFS and OS were observed with regard to the levels of the remaining SMADs (p > 0.05).
Discussion
The natural history of CLL still remains controversial, although the current knowledge of its pathogenesis is improving every year. Therefore, identification of novel biomarkers will not only play a crucial role in the prediction of prognosis but also help to develop molecularly targeted therapies. To this end, the study examines the expression of SMAD proteins. Our results indicate that overexpression of SMAD-1/8 and, more importantly, underexpression of SMAD-4 are associated with a more aggressive course of the disease and may become a valuable prognostic factor for CLL patients.
Our findings indicate that SMAD-4 protein expression was significantly lower in cells from CLL patients than those from a healthy group of controls. This is the first study to make such an observation. Data from previous studies indicate that mutations in SMAD-4 or lack of expression of the corresponding genes is involved in carcinogenesis in multiple malignancies. The SMAD-4 mutation is detected in 50% of patients with pancreatic cancer, 30% of patients with breast and ovarian cancers, 20% of patients with colorectal and gastric cancers, and 10% of patients with lung cancer. 32 These statistics indicate that SMAD-4 expression may be as common as mutations in p53 or K-RAS.
Several studies have found the decreased expression of SMAD-4 protein to be associated with a worse prognosis and a shorter OS. In pancreatic cancer, low SMAD-4 level was responsible for promoting tumor cell migration and invasion and inducing tumor progression and metastasis.23,33 In future, the level of SMAD-4 protein could possibly become a marker for monitoring disease activity in patients with pancreatic cancer. Interestingly, similar observations apply to patients with gastrointestinal tract tumors, including stomach20,34 and colorectal tumors19,35,36 as well as ovarian cancer. 37 In addition to numerous reports of decreased SMAD-4 expression in solid tumors, studies were also carried out in patients with hematologic malignancies. One of the first studies was conducted by Imai et al. 25 in patients with AML, which found that a mutation in the gene encoding the SMAD-4 protein results in the reduced expression or complete absence of this molecule.
This study addresses not only the level of SMAD proteins but also their association with well-established prognostic factors for CLL. One novel aspect of our findings is that they indicate that a correlation exists between low levels of SMAD-4 and the more progressive course of the disease. In addition, a negative correlation was found between SMAD-4 expression and clinical stage according to Rai classification. It is well known that late stage (Rai 3 and 4) is an independent factor for a worse prognosis and is associated with both shorter OS and PFS. The exact relationship between clinical stage and the expression of SMAD-4 proteins has not been described before. In addition, we observed a statistically significant correlation between the level of SMAD-4 and other prognostic markers in CLL including LDT, biochemical markers such as LDH and β2-M, and CD38 expression on cancer cells. However, no correlation has been observed according to cytogenetic and ZAP-70. A negative correlation was found between low concentrations of SMAD-4 and PFS in patients with CLL (Figure 8(a)), as was a strong tendency to prolonged OS in patients with SMAD-4 protein below median MFI (Figure 8(b)).
One of the most important discoveries in the pathogenesis of CLL was the presence of defects in the process of apoptosis which were responsible for the prolongation of survival of leukemic cells. 38 It is a multifaceted process, which includes numerous mechanisms regulating various signaling pathways, including TGF-β.39,40 Therefore, this study goes on to examine the relationship between the expression of SMAD proteins and the level of spontaneous in vitro apoptosis. 5 Interestingly, a statistically significant correlation was found between lower percentages of apoptotic cells and lower SMAD-4 expression; these results are consistent with those of other reports comparing the expression of SMAD proteins with the rate of apoptosis.41–43
Another important aim of this study was to evaluate SMAD expression with regard to the process of angiogenesis. Clinical studies in CLL reported aggravation of angiogenesis in both the bone marrow and peripheral blood measured by an increased expression of proangiogenic factors, including VEGF. 6 In CLL patients, VEGFR-2 expression was significantly higher and was associated with a worse prognosis and more aggressive course of the disease. 7 Moreover, it was demonstrated that TGF-β plays an important role in the regulation of angiogenesis in the healthy body as well as in carcinogenesis. 44 Similar to previous studies, our findings indicate a correlation between lower SMAD-4 expression and overexpression of both VEGFR-1 and VEGFR-2. Therefore, it is reasonable to assume that angiogenesis and apoptosis are strongly associated with the SMAD signaling pathway.
Our results also demonstrate significantly higher SMAD-1/8 expression in CLL cells compared to healthy volunteers. To date, numerous studies have reported an increased expression of SMAD-1 and SMAD-8 proteins in different cancer types. In a study by Boeuf et al. 45 evaluating chondrosarcoma patients, significantly higher SMAD-1, SMAD-5, and SMAD-8 expression was observed when compared to the control group. In this study, overexpression of SMAD proteins mentioned above correlated with a shorter survival time, worse histological subtype, and a more progressive disease outcome. Similar results were reported by Karathanasi et al. 46 In 65 patients with malignant periodontitis tumors, the expression of SMAD-1, SMAD-5, and SMAD-8 proteins was found to be significantly higher than in healthy volunteers. The results of both Boeuf et al. 45 and Karathanasi et al. 46 are consistent with the results described in this study.
In previous reports, little attention was paid to the presence of SMAD abnormalities in patients with hematological malignancies. In a study by Bakkebø et al. 47 on different subtypes of non-Hodgkin lymphoma (NHL), a significantly increased expression of SMAD-1 was observed. Analogous results were described by Munoz et al. 48 This study found that significantly higher levels of SMAD-1/8 may be a significant independent prognostic factor in CLL patients and that the expression of SMAD-1/8 correlated with clinical stage according to Rai classification and elevated serum levels of β2-M. In addition, overexpression of SMAD-1/8 was connected with more progressive type of the disease.
Currently, few articles have reported differences in the expression of inhibitory SMAD-7 in patients with cancers. One of the earliest works was published by Papageorgis et al., 49 who note a significantly higher level of SMAD-7 in three different breast cancer cell lines compared with controls. Moreover, SMAD-7 overexpression was associated with inhibition of cell migration and had a protective effect to the formation of distant metastases. Similar results obtained from 142 patients with colorectal cancer were presented by Gulubova et al. 19 Interestingly, our findings include the first example of a correlation between increased SMAD-7 expression and apoptosis in cancer cells, as well as between the lower expression of SMAD-7 protein and the increased number of ZAP-70-positive cells.
Unfortunately, doubts about the expression of SMAD-2 and SMAD-3 proteins were not determined in this work. No statistically significant correlation was found between the CLL prognostic factors and SMAD-2 and SMAD-3 expression.
In conclusion, this study is the first examination of the clinical significance of the SMAD signaling pathway in CLL patients. Based on these findings, we suggest that abnormalities in the expression of SMAD-4 and SMAD-1/8 proteins may be part of the ethiopathogenetical chain and could even be considered an independent prognostic factor in patients with CLL; however, further studies including longer follow-up periods including PFS and OS are needed to determine this.
Footnotes
Acknowledgements
M.W. provided the study idea, collected the patients’ samples and their laboratory and clinical data, and wrote the paper, A.M. provided technical help and graphical elaboration. B.C.-O. provided technical help and performed statistics, E.W. provided technical help and performed cytogenetic assessment, T.R. assisted in writing and performed critical reading of the paper, and P.S., Chair of the Department, Grant Director, assisted in writing of the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by the grants (No. 502-03/8-093-01/502-64-069 and No. 503/8-093-01/503-01) from the Medical University of Lodz, Poland.
