Abstract
The chemotherapy with fluorouracil is not always effective, in which some breast cancer cells may survive the fluorouracil treatment through enhanced autophagy. Crocetin is the major constituent of saffron, a Chinese traditional herb, which has recently found to have multiple pharmacological effects, including anticancer. However, the effects of Crocetin on the outcome of fluorouracil therapy for breast cancer have not been studied. Here, we showed that fluorouracil treatment inhibited the growth of breast cancer cells, in either a Cell Counting Kit-8 assay or an MTT assay. Inhibition of autophagy further suppressed breast cancer cell growth, suggesting that the breast cancer cells increased autophagic cell survival during fluorouracil treatment. However, Crocetin significantly increased the suppressive effects of fluorouracil on breast cancer cell growth, without affecting either cell apoptosis or autophagy. Inhibition of autophagy at the presence of Crocetin partially abolished the suppressive effects on breast cancer cell growth, suggesting that Crocetin may increase autophagic cell death in fluorouracil-treated breast cancer cells. Furthermore, Crocetin decreased Beclin-1 levels but increased ATG1 levels in fluorouracil-treated breast cancer cells. Together, these data suggest that Crocetin may shift autophagic cell survival to autophagic cell death in fluorouracil-treated breast cancer cells, possibly through modulation of the expression of ATG1 and Beclin-1.
Introduction
Breast cancer is a type of malignant neoplasms that are fast growing and usually insensitive to radiation therapy.1–5 Currently, surgical treatment is the main therapy of breast cancer, although the outcome of the treatment is far from satisfaction.6–12 Chemotherapy, especially fluorouracil (5-FU), is used as a supplementary treatment for breast cancer and has significantly improved the prognosis of breast cancer patients.13–18 However, the chemotherapy with 5-FU is not always effective, in which some breast cancer cells may survive the 5-FU treatment through enhanced autophagy. Hence, there is a need for suppressing breast cancer cell autophagy during 5-FU treatment.
Crocetin is the major constituent of saffron, a Chinese traditional herb, which has recently found to have multiple pharmacological effects, including anticancer. 19 However, the effects of Crocetin on the outcome of 5-FU therapy for breast cancer have not been studied.
Autophagy is a catabolic pathway to degrade and recycle cellular compartments in order to improve cell survival in the harsh environment, for example, nutrient deprivation and deadly insults. Autophagy often leads to cell death, which is distinguishable from apoptotic cell death. 20 Autophagy also plays a critical and complicated role in tumorigenesis, since it not only significantly reduces tumor growth but also increases tumor cell survival upon therapeutic treatments like chemotherapy.21–34 Microtubule-associated protein 1A/1B-light chain 3 (LC3) is a soluble cellular protein and a marker for autophagy. During autophagy, autophagosomes engulf cytoplasmic components, resulting in changes of the cytosolic LC3-I into LC3-II. Thus, the ratio of LC3-II to LC3-I has been widely used as a marker of the autophagic activity.20–22 Autophagy-associated protein 6 (ATG6 or Beclin-1) and ATG1 are two other autophagy-associated proteins, and their roles in autophagic cell survival and autophagic cell death have not been acknowledged. 35
Here, we showed that 5-FU treatment inhibited the growth of breast cancer cells, in either a Cell Counting Kit-8 (CCK-8) assay or an MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay. Inhibition of autophagy further suppressed breast cancer cell growth, suggesting that the breast cancer cells increased autophagic cell survival during 5-FU treatment. However, Crocetin significantly increased the suppressive effects of 5-FU on breast cancer cell growth, without affecting either cell apoptosis or autophagy. Inhibition of autophagy at the presence of Crocetin partially abolished the suppressive effects on breast cancer cell growth, suggesting that Crocetin may increase autophagic cell death in 5-FU-treated breast cancer cells. Furthermore, Crocetin decreased Beclin-1 levels but increased ATG1 levels in 5-FU-treated breast cancer cells. Together, these data suggest that Crocetin may shift autophagic cell survival to autophagic cell death in 5-FU-treated breast cancer cells, possibly through modulation of the expression of ATG1 and Beclin-1.
Materials and methods
Breast cancer cell line culture
A human breast cancer cell line MCF-7 was purchased from American Type Culture Collection (ATCC, Rockville, MD, USA) and was cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 15% fetal bovine serum (FBS; Sigma-Aldrich, St Louis, MO, USA) in a humidified chamber with 5% CO2 at 37 C.
Reagents
5-FU (Sigma-Aldrich) was prepared in a stock of 1 mmol/L and applied to the cultured breast cancer cells at 10 µmol/L. Crocetin (C20H24O4, molecular weight: 328.4; MP Biomedicals, Santa Ana, CA, USA) was prepared in dimethyl sulfoxide (DMSO) and given to cultured cells at a dose of 50 µmol/L. 3-Methyladenine (3-MA; Sigma-Aldrich) was prepared in DMSO and given to cultured cells at a dose of 5 mmol/L.
MTT assay
For assay of cell viability, cells were seeded into 24-well plate at 10,000 cells per well and subjected to a Cell Proliferation Kit (MTT; Roche, Indianapolis, IN, USA), according to the instruction of the manufacturer. The MTT assay is a colorimetric assay for assessing viable cell number, taking advantage that nicotinamide adenine dinucleotide phosphate (NADPH)-dependent cellular oxidoreductase enzymes in viable cells reduce the tetrazolium dye MTT to its insoluble formazan in purple readily being quantified by absorbance value (optical density (OD)) at 570 nm. Experiments were performed five times.
CCK-8 assay
The CCK-8 detection kit (Sigma-Aldrich) was used to measure cell viability according to the manufacturer’s instructions. Briefly, cells were seeded in a 96-well microplate at a density of 5 × 104/mL. After 24 h, cells were treated with resveratrol. Subsequently, CCK-8 solution (20 mL/well) was added, and the plate was incubated at 37°C for 2 h. The viable cells were counted by absorbance measurements with a monochromator microplate reader at a wavelength of 450 nm. The OD value was reported as the percentage of cell viability in relation to the control group (set as 100%).
Western blot
Protein was extracted from the cultured cells with radioimmunoprecipitation assay (RIPA) lysis buffer (1% NP40, 0.1% sodium dodecyl sulfate (SDS), 100 µg/mL phenylmethylsulfonyl fluoride, and 0.5% sodium deoxycholate in phosphate-buffered saline (PBS)) on ice. The supernatants were collected after centrifugation at 12,000g at 4°C for 20 min. Protein concentration was determined using a BCA Protein Assay Kit (Bio-Rad, China, Shanghai), and whole lysates were mixed with 4× SDS loading buffer (125 mmol/L Tris–HCl, 4% SDS, 20% glycerol, 100 mmol/L dithiothreitol (DTT), and 0.2% bromophenol blue) at a ratio of 1:3. Samples were heated at 100°C for 5 min and were separated on SDS–polyacrylamide gels. The separated proteins were then transferred to a polyvinylidene difluoride (PVDF) membrane. The membrane blots were first probed with a primary antibody. After incubation with horseradish peroxidase (HRP)-conjugated secondary antibody, autoradiograms were prepared using the enhanced chemiluminescent system to visualize the protein antigen. The signals were recorded using X-ray film. Primary antibodies were rabbit anti-LC3, anti-Beclin-1, anti-ATG1, and anti-α-tubulin (Cell Signaling, San Jose, CA, USA). Secondary antibody is HRP-conjugated anti-rabbit (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA, USA). α-Tubulin was used as a protein loading control. The protein levels were first normalized to α-tubulin and then normalized to the experimental control. Images shown in the figures were representative of five individuals. Densitometry of western blots was quantified with National Institutes of Health (NIH) ImageJ software (Bethesda, MA, USA).
Apoptosis assay by flow cytometry
The cultured cells were re-suspended at a density of 106 cells/mL in PBS. After double staining with fluorescein isothiocyanate (FITC)-Annexin V and propidium iodide (PI) from an FITC-Annexin V Apoptosis Detection Kit I (Becton-Dickinson Biosciences, San Jose, CA, USA), cells were analyzed using FACScan flow cytometer (Becton-Dickinson Biosciences) for determination of Annexin V+/PI− apoptotic cells, with FlowJo software (FlowJo LLC, Ashland, OR, USA).
Statistical analysis
All statistical analyses were carried out using GraphPad Prism software (GraphPad Software, Inc., La Jolla, CA, USA). All data were statistically analyzed using one-way analysis of variance (ANOVA) with a Bonferroni correction, followed by Fisher’s exact test. All values are depicted as mean ± standard deviation and are considered significant if p < 0.05.
Results
5-FU inhibits breast cancer cell growth and increases autophagy
We examined the effects of 5-FU on the viability of breast cancer cells. We gave 5-FU at 10 µmol/L to a human breast cancer cell line, MCF-7. We found that 5-FU deceased the cell viability of MCF-7 cells in either a CCK-8 assay (Figure 1(a)) or an MTT assay (Figure 1(b)). Thus, 5-FU dose-dependently inhibits breast cancer cell growth. In addition, 5-FU significantly increased the ratio of LC3-II versus LC3-I in breast cancer cells (Figure 1(c)), suggesting that 5-FU enhanced breast cancer cell autophagy.
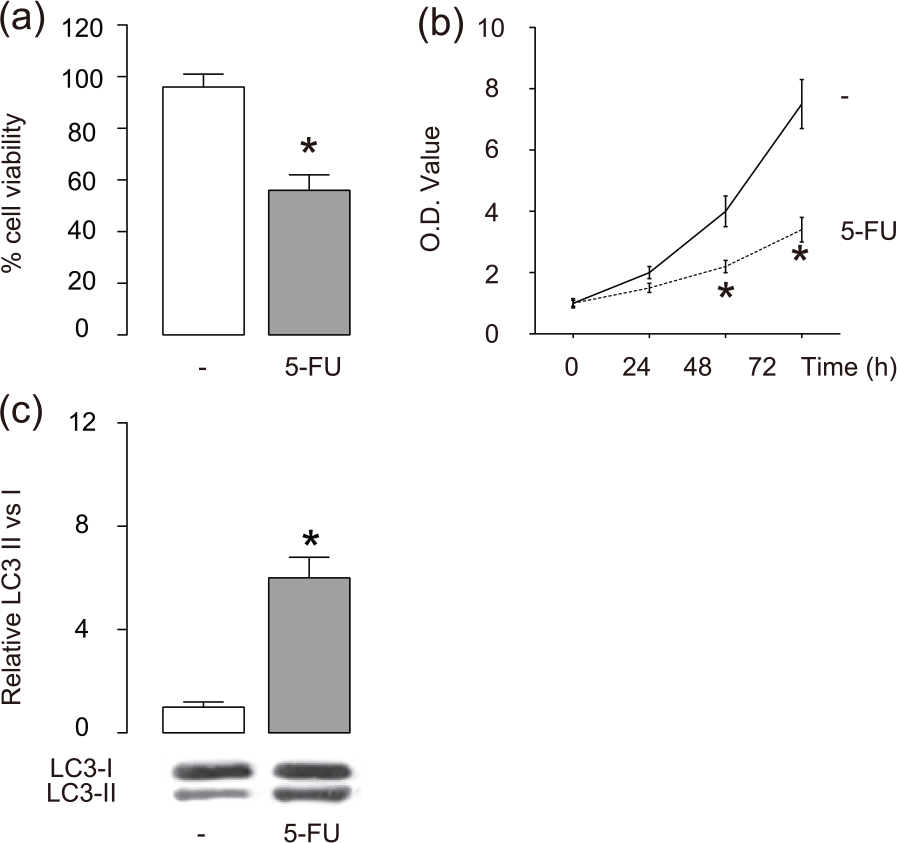
5-FU inhibits breast cancer cell growth and increases autophagy. We gave 5-FU at 10 µmol/L to the cultured breast cancer cells MCF-7. (a and b) Cell viability of MCF-7 cells in either (a) a CCK-8 assay or (b) an MTT assay, and (c) western blot for LC3 in 5-FU-treated MCF-7 cells.
5-FU-induced autophagy attenuates the suppression of breast cancer cell growth
Then, we examined the role of autophagy in the 5-FU-induced breast cancer cell growth suppression. We gave the 5-FU-treated cells with an autophagy inhibitor, 3-MA, at a dose of 5 mmol/L. We found that blocking of cell autophagy by 3-MA further augmented the suppressive effects of 5-FU on breast cancer cell growth, in either a CCK-8 assay (Figure 2(a)) or an MTT assay (Figure 2(b)). Moreover, the suppressive role of 3-MA in autophagy was confirmed by analyzing the ratio of LC3-II versus LC3-I in breast cancer cells (Figure 2(c)). In addition, the application of 3-MA did not alter the levels of apoptosis (Figure 2(d)). These data suggest that the 5-FU-induced autophagy may contradict the apoptotic effects by 5-FU in breast cancer cells, and the outcome of autophagy here may mainly lead to autophagic cell survival.

5-FU-induced autophagy attenuates the suppression of breast cancer cell growth. We gave the 5-FU-treated cells with an autophagy inhibitor, 3-methyladenine (3-MA) at a dose of 5 mmol/L. (a and b) Cell viability of MCF-7 cells in either (a) a CCK-8 assay or (b) an MTT assay, (c) western blot for LC3 in MCF-7 cells, and (d) cell apoptosis assay.
Crocetin enhances the suppressive effects on 5-FU-treated breast cancer cell growth without affecting apoptosis and autophagy levels
We then examined the effects of Crocetin on the viability of 5-FU-treated breast cancer cells. We found that Crocetin further deceased the cell viability of MCF-7 cells in either a CCK-8 assay (Figure 3(a)) or an MTT assay (Figure 3(b)). However, either autophagy levels (Figure 3(c)) or apoptosis levels (Figure 3(d)) in 5-FU-treated breast cancer cells were not altered by Crocetin treatment.

Crocetin enhances the suppressive effects on 5-FU-treated breast cancer cell growth without affecting apoptosis and autophagy levels. We then examined the effects of Crocetin (CRO) on the viability of 5-FU-treated breast cancer cells. (a and b) Cell viability of MCF-7 cells in either (a) a CCK-8 assay or (b) an MTT assay, (c) western blot for LC3 in MCF-7 cells, and (d) cell apoptosis assay.
Crocetin shifts the autophagic cell survival to autophagic cell death in 5-FU-treated breast cancer cells
Since autophagy may lead to different outcome in terms of cell viability, autophagic cell survival to autophagic cell death, we thus analyzed the effects of autophagy in the setting of Crocetin and 5-FU combination. We gave the Crocetin and 5-FU-treated cells with 3-MA at a dose of 5 mmol/L. We found that blocking of cell autophagy by 3-MA abolished the effects of Crocetin on the growth of 5-FU-treated breast cancer cells, in either a CCK-8 assay (Figure 4(a)) or an MTT assay (Figure 4(b)). Moreover, the suppressive role of 3-MA in autophagy was confirmed by analyzing the ratio of LC3-II versus LC3-I in breast cancer cells (Figure 4(c)). In addition, the application of 3-MA did not alter the levels of apoptosis (Figure 4(d)). These data suggest that the 5-FU-induced autophagy may shift the autophagic cell survival to autophagic cell death in 5-FU-treated breast cancer cells.

Crocetin shifts the autophagic cell survival to autophagic cell death in 5-FU-treated breast cancer cells. We gave the Crocetin (CRO) and 5-FU-treated cells with 3-MA at a dose of 5 mmol/L. (a and b) Cell viability of MCF-7 cells in either (a) a CCK-8 assay or (b) an MTT assay, (c) western blot for LC3 in MCF-7 cells, and (d) cell apoptosis assay.
Crocetin decreases Beclin-1 levels but increases ATG1 levels in 5-FU-treated breast cancer cells
We then analyzed the differences in the autophagy-associated proteins in our experimental settings. We found that Crocetin decreased Beclin-1 levels but increased ATG1 levels in 5-FU-treated breast cancer cells (Figure 5(a)–(c)). These data suggest that Crocetin may shift autophagic cell survival to autophagic cell death in 5-FU-treated breast cancer cells, possibly through modulation of the expression of ATG1 and Beclin-1.

Crocetin decreases Beclin-1 levels but increases ATG1 levels in 5-FU-treated breast cancer cells. (a) Representative western blots for Beclin-1 and ATG1 levels in 5-FU/Crocetin (CRO)-treated MCF-7 cells, and (b and c) quantification for protein levels of (b) Beclin-1 and (c) ATG1.
Discussion
Natural carotenoids are highly pigmented compounds that possess anti-carcinogenic, anti-mutagenic, and immunomodulating effects. Retinoids, like vitamin A, have been shown to inhibit the growth of some types of tumor cells. However, the toxicity of carotenoids is an important limiting factor that prevents them to be widely used in therapeutic approaches. There are many compounds from natural products with demonstrated tumor-suppressing potential. Saffron is a spice and food colorant present in the dry stigmas of the plant Crocus sativus L. and has been used to treat various diseases, including malignant cancer. Crocetin is a major commercially available constituent of saffron, with significant potential as an anti-tumor agent and reduced toxicity.
In this study, we analyzed the effects of Crocetin on the tumor cell viability of 5-FU-treated breast cancer cells. We found that 5-FU significantly decreased breast cancer cell viability, in two independent assays for measuring cell viability. In addition, 5-FU appeared to induce autophagy of breast cancer cells. Cancer cells use autophagy as a survival strategy to provide essential biomolecules that are required for cell viability under harsh environment, whereas autophagy can also lead to autophagic cell death as an alternative pathway for cell death in chemotherapy. 36 Here, we found that Crocetin did not alter cell apoptosis but further suppressed breast cancer cell growth and increased cell death. Hence, this alteration in cell death seemed to result from the outcome of cell autophagy. Indeed, blocking of autophagy in 5-FU-only-treated breast cancer cells and combined 5-FU- and Crocetin-treated breast cancer cells gave very different results, showing that upon 5-FU-treatment alone, autophagy appeared to be a natural response of breast cancer cells to antagonize the lethal effects of 5-FU to improve survival-autophagic cell survival. However, when Crocetin was given, it seemed to alter the outcome of autophagy to autophagic cell death to further augment the effects of 5-FU. Hence, Crocetin appeared to be a useful supplementary reagent to enhance the chemotherapeutic effects of 5-FU.
Since we showed that the levels of Beclin-1 and ATG1 were counterbalanced in the Crocetin treatment, we proposed that the adaption of autophagic cell survival to autophagic cell death might result from a delicate control of these key autophagy-associated proteins. Future studies may be applied to further identify the underlying molecular mechanisms.
Of note, here we used a breast cancer line MCF-7 to draw the conclusion; future studies may be applied using cell lines other than MCF-7 to confirm this finding. Together, our data shed light on a previously unrecognized role of Crocetin and a signaling regulatory pathway that modulate the chemotherapeutic effects of 5-FU against breast cancer cells through autophagy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
