Abstract
Global burden of breast cancer is expected to increase to >2 million new cases every year by 2030 and 10% of these are likely to have hereditary breast and ovarian cancer syndrome. Identifying these individuals by pedigree and BRCA1/2 mutation analyses will enable us to offer targeted mutation testing and appropriate counseling. This study from a tertiary care hospital showed that of the 127 breast cancer patients on treatment during 2014–2015, 24 of them fulfilled the criteria of hereditary breast and ovarian cancer syndrome after detailed verbal autopsy and pedigree analysis, and BRCA1 and 2 next-generation sequencing done after pre-test counseling revealed mutations in 13 cases (54%), these included 9 BRCA1 mutations (69%) and 4 BRCA2 mutation (31%). Subsequent post-test counseling recommended targeted mutation analysis for 64 high-risk members in these 13 families with pathogenic mutations, which will help in surveillance for early detection, appropriate management, and prevention of the disease by decreasing the burden to both family and nation. Results from this preliminary study highlight the importance of genetic counseling, pedigree analysis, and genetic testing. It can be recommended that all oncology units should have a genetic counseling service for providing appropriate support to oncologists, patients, and families to prevent unnecessary testing; however, breast cancer screening program is incomplete without evaluating for hereditary breast and ovarian cancer syndrome.
Keywords
Introduction
Breast cancer is the most commonly diagnosed malignancy in women worldwide, and in India, it ranks second to cervical cancer. It is the leading cause of female cancer mortality and hence constitutes a serious public health problem. Global burden of breast cancer is expected to increase to over 2 million new cases every year by 2030. 1 The age-standardized incidence of breast cancer in the world is 43.3 per 100,000 females, while in India, it is 25.8 per 100,000 females. 2 Despite having a relatively low incidence, the survival rates in India are about 20% lower than what is observed for breast cancer in developed countries, mostly due to late diagnosis and the fact that 46% of them are triple negative cases, known for poor prognosis. 3
Several conventional risk factors for breast cancer are documented, which include female sex, age, exposure to radiation, nulliparity or having a first child after age 35 years, failure to lactate/breast feed, older age at menopause (>55 years), younger age at first period (<12 years), alcohol consumption, current or recent use of birth control pills, and overweight or weight gain as an adult. But, a positive family history is one of the most important risk factors for developing breast cancer and it is currently estimated that approximately 10% of all breast cancers have a hereditary basis and these families have a dominant inheritance pattern. Hereditary breast and ovarian cancer syndrome (HBOCS) is characterized by an early age of onset, overrepresentation of bilateral breast cancers and male breast cancer.4,5 Breast and Ovarian cancer (BOC) occurring simultaneously and occurrence of pancreatic, colon cancers, prostate, and male breast in the family are associated with Breast Cancer 1 and 2 (BRCA1 and 2) gene mutations.6,7
BRCA1 and 2 genes function as tumor suppressors and pathogenic mutations in these appear to account for ~30% of high-risk breast cancer families and explain ~15% of familial breast cancer risk. 8 Carriers of germline mutations in BRCA1 are believed to have a 57% lifetime risk of developing breast cancer, while those with BRCA2 mutation have 49% by the age of 70 years. The average risk of ovarian cancer in women with a BRCA1 mutation is 40% and with a BRCA2 mutation is 18%. While these are rare syndromes that cause an estimated 5%–10% of all breast and 23% of ovarian cancers, individuals carrying these mutations have the highest cancer incidence of any known group. 9 For males, mutations in BRCA2 gene imply approximately 6% increased risk of prostate cancer. 10 Founder mutations in BRCA1 and BRCA2 have been useful in testing specific populations. 11 Such founder mutations have been identified in Icelandic, Polish, French-Canadian, and Ashkenazi Jewish populations.12–15 No such specific mutations have been identified in the Indian population(s) yet.
The aim of this study was to evaluate the feasibility of using pedigree analysis and subsequent BRCA1 and BRCA2 testing in selected breast cancer patients undergoing treatment, as a means of identifying HBOCS. With cost of sequencing being reduced and the advent of next-generation sequencing (NGS), instead of specific mutation analysis, this study carried out BRCA1 and BRCA2 whole gene sequencing. The variations identified were also correlated with conventional risk factors to assess which could be used in subsequent surveillance programs to reduce morbidity and mortality caused by this pathology.
Materials and methodology
Patient selection
All the breast cancer patients who were undergoing treatment at the oncology unit, Kamineni Hospital, Hyderabad, during a period of 1 year (September 2014 to August 2015) were screened by a genetic counselor, for the following criteria: for patients, who appeared to have characteristics of HBOCS: (1) age of onset <40 years, (2) male breast cancer patients, (3) women with self-proclaimed positive family history, and (4) triple negative breast cancer and were referred to Department of Genetics and Molecular Medicine for detailed evaluation. Clinical history, family history with verbal autopsy, and detailed pedigree were recorded in a well-designed proforma. Representative pedigrees of those with familial BOC are shown in Figure 1(a) and (b).
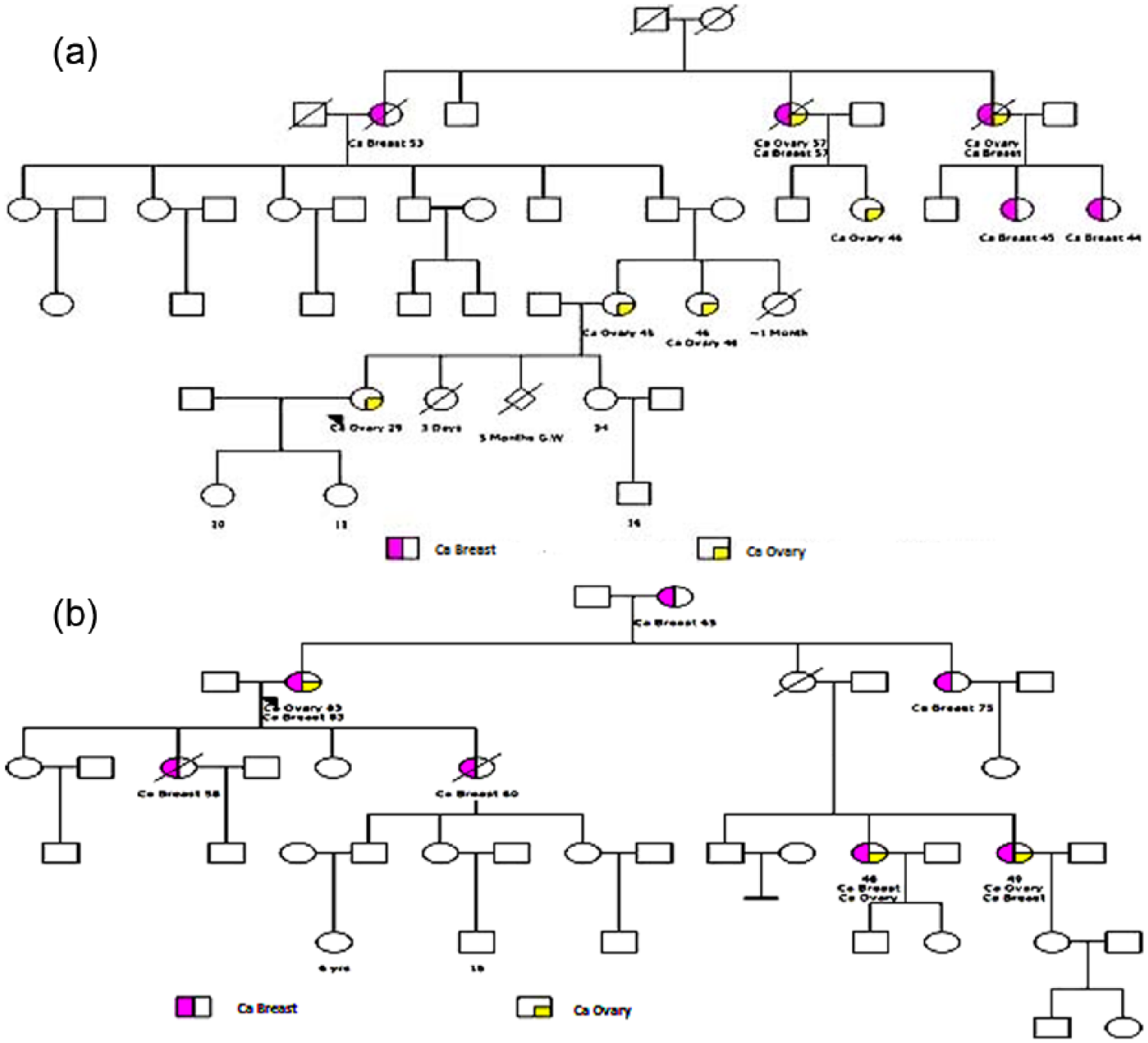
(a and b) Representative pedigrees with multiple affected family members in different generations indicating HBOCS.
Methodology
All individuals, who fulfilled the criteria, were provided with pre-test genetic counseling prior to obtaining 3 mL of peripheral ethylenediaminetetraacetic acid (EDTA) blood sample. Informed consent from the subjects was obtained as suggested by the institutional ethics committee before sending samples for complete gene sequencing of BRCA1 and BRCA2 to Centogene, Germany. BRCA1 and BRCA2 genes were amplified using the Ion AmpliSeq BRCA1 and BRCA2 Panel (Life Technologies, USA). The panel consisted of three primer pools (167 amplicons) which targeted the entire coding region, including 10–20 bp of intronic flanking sequences around all coding exons, of both genes.
The samples were processed on the Ion Proton Platform (Life Technologies). Approximately 33 Mb of coding exons were covered as described by consensus coding sequences for BRCA1 and 2 genes. Highly multiplexed primer pools were used to construct the amplicon library using polymerase chain reaction (PCR)-based targeted amplification. Following the base calling and primary filtering of low-quality reads, standard bioinformatics pipeline was implemented to annotate detected variants and to filter out probable artifacts. Any clinically relevant variant identified by NGS was validated by Sanger sequencing to confirm it as a true positive. Sanger sequencing system is optimized to validate and interpret relevant variants identified by NGS. Sanger sequencing was performed in both the forward and reverse directions. BRCA1 and BRCA2 whole gene sequencing was done at Centogene, Germany.
Mutations detected by NGS were confirmed by target direct Sanger sequencing. Sanger sequencing was performed using the BigDye Terminator v3.1 Cycle Sequencing kit (Applied Biosystems, Thermo Fisher Scientific, Inc., USA). The purification of sequencing reactions was performed using the Sephadex® (Sigma-Aldrich, USA) method. The samples were run on an ABI 3130 Genetic Analyzer (Applied Biosystems, Thermo Fisher Scientific, Inc.). All the samples for Sanger sequencing were out sourced to a company Bioartis, India. The chromatograms of the Sanger sequencing results were analyzed using Chromas software.
Results
Of the 127 breast cancer patients who were undergoing treatment in the Department of Oncology, 33 were selected for detailed evaluation in the Department of Genetics and Molecular Medicine. In total, 24 of these unrelated breast cancer patients fulfilled criteria of HBOCS (18.89%) and consented for BRCA1 and BRCA2 sequence analysis after pre-test genetic counseling.
Demographic details are given in Table 1. In total, 2 of the 24 patients were males. Mean age at menarche for rest was 12.6 ± 1.29 years, none of them was obese, and all of them had children and had breast fed for more than 1 year. None of them had used oral contraceptives or/and hormone replacement therapy. None of the patients in our study had children after 35 years of age or was nulliparous. The subjects included in this study were from different socioeconomic groups. Hence, the accepted conventional risk factors for breast cancer did not appear to be associated with this cohort of patients.
Demographic details of patients.

NGS was carried out for BRCA1 and 2 sequence analysis and 13/24 cases (54%) were found to have a sequence variation, 69% (9 out of 13 cases) were identified in BRCA1 gene and 31% (4 out of 13 cases) in BRCA2 gene (Table 2). Seven known BRCA1 pathogenic mutations identified in seven patients. Among them: (1) intronic 16 c.5074+1G>A mutation observed in two patients (BC-02 and BC-18), (2) c.68_69del (185delAG) p.Glu23Val in exon 2 in another two patients (BC-07 and BC-15), (3) c.4224_4231del (p.Gln1408His) in exon 12, (4) c.1504_1508del (p.Leu502Ala) in exon 10, and (5) g.50962G.GT (IVS14-1 G>GT) in intron 14 in one patient each. While in BRCA2, only one known pathogenic mutation, c.8587dup p.Glu 2863 Gly in exon 20, was identified.
Sequence variations in BRCA1 and BRCA2 genes.

VUS: variant of unknown significance.
Apart from the known pathogenic mutations, five variants of unknown significance (VUSs) were identified, two in BRCA1 gene: (1) c.2426A>G (p.Glu809Gly) in exon 10 and (2) c.4286A>G (p.Tyr1429Cys) in exon 12 and three in BRCA2: (1) c.1792A>G (p.Thr598Ala) in exon 10, (2) c.5986G>A (p.Ala1996Thr) in exon 11, and (3) c.7445C>T (p.Thr2482Ile) in exon 15.
Extended family screening
On pedigree analysis, we found 64 high-risk individuals in these 13 families. Targeted mutation analysis was recommended for the high-risk family members in those families where pathogenic mutations were detected in the index cases. Six families consented for targeted mutation analysis after post-test genetic counseling and 15 family members were tested, 12 of them were identified as carriers of the targeted mutations and were offered to be part of the cancer surveillance program. High-risk members who were below the age of 18 years were not offered targeted mutation testing as per the current recommendation.
In patients where a VUS was identified, targeted analysis was recommended for all other affected individuals in the family to establish whether the VUS has pathogenic implications. A larger panel of hereditary cancer genes associated with familial BC was recommended to the 11 breast cancer patients, who were not detected with BRCA1 or BRCA2 mutations.
Discussion
The global burden of breast cancer is expected to cross 2 million by the year 2030, with growing proportions from developing countries. 16 Gupta et al. 1 indicated that, breast cancer incidence rates within India display a three–fourfold variation across the country, with the highest rates observed in the Northeast and in major metropolitan cities. This indicates that familial cancer may be more prevalent in these regions due to founder mutations in BRCA genes, which are strongly incriminated in the predisposition to HBOCS. 17 Genetic cancer risk assessment and genetic testing for inherited breast cancer susceptibility have become standard clinical management techniques for patients and families in developed countries. But the situation in India is different, with only a very limited number of patients being offered the test either because the treating clinician does not recommend it or due to the high cost of the test. In some cases, affording patients opt for the test due to anxiety although they do not fit the HBOCS criteria and may not benefit from the test. It is well established that most breast cancers are sporadic and several conventional risk factors have been implicated in the etiology of this pathology, but in this study, none of the documented factors apart from family history appears to play a role in this cohort of breast cancer patients (Table 1).
It is believed that 10% of breast cancers belong to hereditary syndromic types, but this preliminary study from an oncology unit of a tertiary care hospital indicates that 18.89% of breast cancer cases have a positive family history,
In total, 10.23% of them exhibited a sequence variation in either BRCA1 or 2 and 6.29% had a confirmed pathogenic mutation. Similar study from China showed 8.9% breast cancer patients with BRCA1 mutations. 18 A recent paper from a company offering sequencing in India showed that occurrence of BRCA mutations in their cases was 36.2%, but this high percentage may be due to the reason that the cases sent for BRCA1 and 2 analysis to the company were mostly of familial type, unlike this study where all cases presenting with BC were used for percentage analysis. 19
Founder mutations are specific sequence variations which appear repeatedly in ethnically defined groups because of shared common ancestry. This effect explains the high frequencies of disease-associated mutations in specific human populations. Founder mutations in BRCA1 and BRCA2 have been useful in defining mutation-associated risks in specific population. The BRCA variant c.68-69delAG (185delAG) reported as founder mutation in Ashkenazi Jewish population occurs with a frequency of 18% among BOC families.20,21
Indian studies reported a frequency of this mutation in 0.5%–4.1% BC cases in different states.22–24 This was also reported in two south Indian families by Rajkumar et al. 22 in 2015. We also found this mutation in two of our breast cancer patients, who hailed from the coastal region of India (i.e. Andhra and Karnataka). A previous study had suggested that the origin of the c.68-69delAG (185delAG) mutation in Indian population is independent of that of Ashkenazi Jews based on haplotype analysis. 25 However, reflecting on the fact that this mutation is present in two opposite coastal regions, it indicates that it may be the same mutation coming to India with people who migrated from the sea route.
Another BRCA1 splice site mutation in intronic 16 c.5074+1G>A which was earlier reported as an Icelandic founder mutation was found in two of our patients. 26 The same mutation was reported in three patients by Mannan et al. 19 suggesting that there may be a common ancestry or they migrated from the same place.
Two other known pathogenic mutations are the following: (1) c.1504_1508del, p.Leu502Ala in exon 10 of BRCA1 gene, which was previously identified as deleterious mutation in two different populations of non-Afrikaner breast cancer patients of the Western Cape of South Africa 27 and breast cancer patients in United States. 28 (2) The BRCA1 IVS14-1 G>A was the other pathogenic mutation reported earlier in breast cancer families from Pakistan. They identified this mutation in two unrelated patients, one with Punjabi background and the other of Pashtun ethnicity that suggest this mutation to have a founder effect. 29
In our cohort, we found two novel mutations: (1) c.4224_4231del (p.Gln1408His) in exon 12 of the BRCA1 gene, which is creating a frame shift mutation and (2) c.8587dup (p.Glu2863Gly) in exon 20 of the BRCA2 gene, which creates a frame shift starting at codon Glu2863 resulting in a truncation. To the best of our knowledge, this is the first time these have been identified in breast cancer patients.
A VUS is an alteration in the gene sequence with unknown consequences on the function of the gene product or risk of causing disease. In total, 10%–20% of reported mutations are VUSs. 30 A class 3 variant (VUS) has a 0.05–0.949 probability of being pathogenic, evidence may be limited or conflicting and the closer to 95% probability a variant reaches, the more useful additional family and functional studies are to improve the classification according to Eccles et al. 31 In this study, five BRCA VUSs were identified, two in BRCA1 and three in BRCA2. The BRCA1 c.2426A>G (p.Glu809Gly) VUS in exon 10 has been reported thrice earlier in ClinVar database associated with HBOCS. A paper from China indicates that breast cancer patients from Guangxi region have mutations only in exon 10; hence, this may be a significant finding. 18 The VUS c.4286A>G (p.Tyr1429Cys) in exon 12, which was found in a male breast cancer patient, has been reported once earlier in a tumor predisposition syndrome in ClinVar.
Three VUSs in BRCA2 were also identified; the c.1792A>G (p.Thr598Ala) in exon 10 was reported 11 times in ClinVar and has conflicting interpretations of pathogenicity. This was also previously described as a polymorphism, rs28897710, in Slovenian population. 32 The second VUS was c.5986G>A (p.Ala1996Thr) in exon 11, which has been previously described as a harmful variant in a Croatian BC study 33 and is reported in ClinVar five times in HBOCS patients, with conflicting interpretation of pathogenicity. The third one was c.7445C>T (p.Thr2482Ile) in exon 15. Based on this evidence, VUS-targeted testing was recommended for other affected individuals in the family to understand the association of the mutation with disease to further offer predictive testing. Hereditary cancer gene panel and evaluation of larger deletions were advised to the five individuals who were found to have no BRCA1 and 2 mutations, but had a strong family history of the disease.
In conclusion, a basic simple pedigree analysis, with information on age at clinical diagnosis along with the immunohistochemistry (IHC), report would be useful in identifying hereditary cancer cases. This will enable us to offer genetic testing and subsequent predictive targeted mutation analysis for the high-risk members in the family after genetic counseling. This will reduce the number of unnecessary genetic tests for breast cancer patients. Individuals who are carriers of BRCA1 and 2 mutations in the family after targeted mutation analysis can be put on age appropriate surveillance to reduce morbidity, mortality, and emotional and financial burden. In developing countries like India where the cost of treatment is borne either by the patient or by the government (for individuals below the poverty line), offering the genetic test to identified cases/families after a simple effective genetic counseling session will help patients, families, and the government in healthcare management of this vulnerable group. Including a genetic counselor in the health management faculty of oncology units is a simple cost-effective way of preventive management of breast cancer.
Footnotes
Acknowledgements
The authors thank Kamineni Academy of Medical Sciences and Research Centre for providing infrastructure and necessary equipment to carry out their experimental work. Authors also extend their gratitude toward Centogene company for performing sequence analysis. They thank all patients who participated in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
