Abstract
In this study, the combined effect of doxorubicin with cucurbitacin B on survival of anaplastic thyroid carcinoma cells was evaluated. For experiments, 8505C and CAL62 human anaplastic thyroid carcinoma cells were used. Cell viability, the percentage of viable cells, and cytotoxic activity were measured using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, multiplexed cytotoxicity assay, and cytotoxicity assay, respectively. Reactive oxygen species production was measured. In experiments, doxorubicin and cucurbitacin B reduced cell viability in a dose- and time-dependent manner. Cotreatment of doxorubicin and cucurbitacin B, compared with treatment of doxorubicin alone, decreased the percentage of viable cells and increased cytotoxic activity. All of the combination index values were lower than 1.0, suggesting the synergism between doxorubicin and cucurbitacin B in induction of cytotoxicity. In cells treated with both doxorubicin and cucurbitacin B, compared with doxorubicin alone, the protein levels of cleaved poly(adenosine diphosphate-ribose) polymerase and cyclooxygenase 2 and reactive oxygen species production were enhanced. In contrast, the protein levels of B-cell chronic lymphocytic leukemia/lymphoma 2 and survivin and B-cell chronic lymphocytic leukemia/lymphoma 2/B-cell chronic lymphocytic leukemia/lymphoma 2–associated x protein ratio were diminished. The protein levels of Janus kinase 2 and signal transducer and activator of transcription 3 were reduced, while phospho-extracellular signal–regulated kinase 1/2 protein levels were elevated without change in total extracellular signal–regulated kinase 1/2 protein levels. These results suggest that doxorubicin synergizes with cucurbitacin B in induction of cytotoxicity in anaplastic thyroid carcinoma cells. Moreover, synergistic cytotoxicity of doxorubicin with cucurbitacin B is mediated by B-cell chronic lymphocytic leukemia/lymphoma 2 family proteins, survivin, and reactive oxygen species and modulated by Janus kinase 2/signal transducer and activator of transcription 3 and extracellular signal-regulated kinase 1/2 in anaplastic thyroid carcinoma cells.
Introduction
Anaplastic thyroid carcinoma (ATC) is a highly aggressive malignancy arising from thyroid gland characterized as extrathyroidal invasion, distant metastasis, and resistance to conventional treatment. 1 In ATC, multimodal therapeutic options including surgery, chemotherapy, and radiotherapy do not have potential effectiveness, and thus, new therapeutic strategies to improve efficacy against cancer cells are under consideration. 1
Doxorubicin (Dox), an anthracycline-based DNA intercalator, exerts antitumor activities by suppressing DNA polymerase, DNA topoisomerase II, and DNA methyltransferase, leading to cytotoxicity in cancer cells. 2 Dox has a cytotoxicity via modulation of B-cell chronic lymphocytic leukemia/lymphoma 2 (Bcl-2) family proteins in cancer cells. 2 Meanwhile, survivin, a member of inhibitor of apoptotic protein (IAP), represses cell death through interaction with IAPs, and thus, it is considered as a resistance factor to chemotherapeutic agents. 3 Survivin is upregulated in cancer cells resistant to Dox. 4 In regard to reactive oxygen species (ROS), Dox produces ROS which activate signaling cascades with changes in phosphorylation of proteins responsible for survival, growth, and invasion in cancer cells. 5 Artesunate and N,N-dimethyl phytosphingosine sensitize cancer cells to Dox-induced cytotoxicity by mediating ROS.6,7 In ATC patients, Dox has been approved for treatment in monotherapy, but it has shown a poor response rate.8–10 Considering that Dox does not get rid of cancer stem cells derived from ATC cells, 11 sensitization of ATC cells to Dox would be essential in treatment of ATC patients.
Cucurbitacins are a class of highly oxidized tetracyclic triterpenoids isolated from Cucurbitaceae and Cruciferae and are divided into 12 categories according to characteristics of their structures.12–14 In principle, cucurbitacins result in cytotoxicity via inhibition of signal transducer and activator of transcription 3 (STAT3) in cancer cells.12–14 Among cucurbitacins, cucurbitacin B (CuB) causes cell death through inactivation of Janus kinase 2 (JAK2)/STAT3 in pancreatic cancer cells. 15 CuB stimulates extracellular signal–regulated kinase (ERK) in laryngeal cancer cells, whereas it suppresses ERK in leukemia and brain cancer cells.16–18 CuB alone or in combination with chemotherapeutic agents has a cytotoxicity by modulating death-related proteins and ROS in cancer cells.14–22
Cucurbitacins augment Dox-induced cytotoxicity in cancer cells.23,24 In this regard, cucurbitacin D leads to cell death via repression of STAT3 in Dox-resistant human breast cancer cells. 23 Moreover, cucurbitacin E inhibits efflux of Dox and thereby enhances cell death in ovarian sarcoma cells. 24 With regard to the therapeutic implication of cucurbitacins in thyroid cancer cells, there is only one study reporting the result that cucurbitacin I potentiates cytotoxicity by Dox, cisplatin, 5-fluorouracil, and irradiation in CD133+ ATC cells, 25 but the impact of CuB alone or in combination with other chemotherapeutic agents on cell survival has not been identified in thyroid cancer cells.
The aim of this study was to elucidate the combined effect of Dox with CuB on survival of ATC cells. Our results demonstrate that Dox has a synergistic activity with CuB in induction of cytotoxicity through mediation of Bcl-2 family proteins, survivin, and ROS and regulation of JAK2/STAT3 and ERK1/2 in ATC cells.
Materials and methods
Materials
Dulbecco’s Modified Eagle’s Medium (DMEM), fetal bovine serum (FBS), and streptomycin/penicillin were purchased from Life Technologies (Carlsbad, CA, USA). Dox and CuB were obtained from Sigma (St. Louis, MO, USA). These were dissolved in dimethylsulfoxide (DMSO), which was provided to the control within permissible concentrations. The final concentration of the vehicle DMSO in the control did not exceed 0.1% in all treatments. The primary antibodies raised against B-cell leukemia/lymphoma x (Bcl-xL), Bcl-2, Bcl-2-associated x protein (Bax), BH3-interacting domain death agonist (Bid), cleaved poly(adenosine diphosphate (ADP)-ribose) polymerase (PARP), survivin, JAK2, STAT3, total and phospho-ERK1/2 (Thr402/Tyr404), and cyclooxygenase 2 (COX2) were purchased from Cell Signaling Technology (Danvers, MA, USA). The primary antibody raised against β-actin from Sigma was obtained. All other reagents were purchased from Sigma unless otherwise stated.
Cell culture
For experiments, 8505C and CAL62 human ATC cells were used. 8505C and CAL62 cells were purchased from Deutsche Sammlung von Mikroorganismen und Zellkulturen (DSMZ GmbH, Braunschweig, Germany) and grown in DMEM supplemented with 10% heat-inactivated FBS and 1% streptomycin/penicillin. Cells received fresh medium at regular intervals. Treatments and experiments were performed using cells that were 70% confluent.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
Cell viability was measured using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. Cells (5 × 103/100 µL) in each well on 96-well plates were incubated overnight and treated with Dox and CuB. Thereafter, MTT (1 mg/mL) was administered to medium for an additional 4 h at 37°C, and medium containing MTT was removed, and DMSO was added to dissolve formazan crystals developed by viable cells. After 30 min at room temperature, absorbance was measured using GloMax™ Discover System GM3000 (Promega, Madison, WI, USA). All experiments were performed in triplicate.
Multiplexed cytotoxicity assay
Cells (5 × 103/100 µL) were seeded in 96-well plates, and reagents of the MultiTox-Glo Multiplex Cytotoxicity Assay Kit (Promega) were added to cells after treatments as indicated by manufacturer’s protocol. 26 Fluorescent and luminescent values were measured using GloMaxTM Discover System GM3000 (Promega). Viability was calculated as a ratio of live/dead cells and expressed as percentage of untreated cells. All experiments were performed in triplicate.
Cytotoxicity assay
Cytotoxic activity was measured by the LDH Cytotoxicity Assay Kit (BioVision, Linda, CA, USA). Cells (5 × 103/100 µL) in each well on 96-well plates were incubated and centrifuged at 250g for 10 min. Supernatant of 100 µL was transferred in clear 96-well plates. After addition of reaction mixture (2.5 µL of catalyst solution in 112.5 µL of dye solution), cells were incubated for 30 min at room temperature. Absorbance was measured using GloMaxTM Discover System GM3000 (Promega). All experiments were performed in triplicate.
Measurement of ROS production
ROS production was measured by the ROS-Glo H2O2 Assay Kit (Promega). Cells (1 × 104/mL) in each well on 96-well plates were incubated and treated with H2O2 substrate solution (25 µM/well) and incubated at 37°C. After addition of ROS-Glo detection solution (100 µL/well), cells were incubated for 20 min at room temperature. Absorbance was measured using GloMaxTM Discover System GM3000 (Promega). All experiments were performed in triplicate.
Drug combination analysis
Combination index (CI) and isobologram were calculated by CalcuSyn program version 2.11 (Biosoft, Great Shelford, Cambridge, UK), and the effect of drug interactions was quantitatively determined. CI values less than 1.0, 1.0, and greater than 1.0 demonstrate synergism, additivity, and antagonism, respectively. The isobologram is formed by plotting the doses of each drug required for 50% inhibition (ED50) on the x- and y-axes and connecting them to draw a line segment, which is ED50 isobologram. Combination data points that fall on, below, and above the line segment indicate additivity, synergism, and antagonism, respectively. All experiments were performed in triplicate.
Western blotting
The total protein was extracted by radioimmunoprecipitation assay buffer (RIPA) buffer (Sigma) containing 1× protease inhibitor cocktail and 1× phosphatase inhibitor cocktail set V (Calbiochem, La Jolla, CA, USA). Western blotting was performed using specific primary antibodies and horseradish peroxidase–conjugated anti-rabbit and anti-mouse secondary antibodies. Bands were detected using ECL Plus Western Blotting Detection System (Thermo Fisher Scientific, Rockford, IL, USA). Target protein levels were quantified by densitometry using ImageJ software (National Institutes of Health (NIH)) and normalized to β-actin protein levels. The relative levels of target protein to β-actin protein were calculated. All experiments were performed in triplicate.
Statistical analysis
All data are expressed as mean ± standard error (SE). Data were analyzed by unpaired Student’s t-test or analysis of variance (ANOVA) as appropriate. A p value less than 0.05 was considered to be statistically significant. All analyses were performed using SPSS program version 23.0 (SPSS, Chicago, IL, USA).
Results
Dox has a synergistic activity with CuB in induction of death of ATC cells
While Dox alone is unable to eliminate cancer stem cells derived from ATC cells, CuB alone or in combination with chemotherapeutic agents causes cytotoxicity in cancer cells.11,14 In this study, the effect of Dox in combination with CuB on cell survival was evaluated in 8505C and CAL62 ATC cells.
First, to clarify the influence of each drug, cells were treated with Dox and CuB at different doses for 24 and 48 h, and cell viability was measured using MTT assay (Figure 1(a) and (b)). As a result of treatment, cell viability was reduced in a dose- and time-dependent manner.

The impact of Dox and CuB on survival of ATC cells. (a, b) 8505C and CAL62 cells were treated with (a) Dox at 0.25, 0.5, 1, 2, and 4 µM and (b) CuB at 37.5, 75, 150, 300, and 600 nM for 24 and 48 h, respectively. Cell viability was measured using MTT assay. All experiments were performed in triplicate. Data are expressed as mean ± SE.
Next, to verify the impact of combination of two drugs, cells were treated with both Dox and CuB, and the interactions were assessed by calculating the CI by Chou–Talalay equation, where CI < 1.0 shows synergism, and CI = 1.0 shows additivity, and CI > 1.0 shows antagonism (Figure 2(a), Table 1). After cotreatment, cell viability was measured using MTT assay, and death rate was calculated as 100-cell viability (%). All of the CI values were lower than 1.0 in combination of Dox with CuB. In the isobologram analysis, the combination data points were all located below the isobologram line at ED50, suggesting the synergism between Dox and CuB inducing cytotoxicity in ATC cells.

The influence of Dox in combination with CuB on survival of ATC cells. (a) 8505C and CAL62 cells were treated with both Dox at 0.25, 0.5, 0.75, and 1 µM and CuB at 37.5, 75, 112.5, and 150 nM for 48 h. Cell viability was measured using MTT assay, and death rate was calculated as 100-cell viability (%). Combination index (CI) and isobologram were calculated. The horizontal dashed lines at CI = 1.0 are drawn. (b, c) 8505C cells were treated with both Dox at 1 µM and CuB at 150 nM for 48 h, and (b) the percentage of viable cells was measured using multiplexed cytotoxicity assay and (c) cytotoxic activity was measured using cytotoxicity assay. All experiments were performed in triplicate. Data are expressed as mean ± SE. *p < 0.05.
Combination index (CI) values at combined doses determined by the median effect analysis method in ATC cells treated with both Dox and CuB.

Dox: doxorubicin; CuB: cucurbitacin B.
CI values less than 1.0, 1.0, and greater than 1.0 indicate synergism, additivity, and antagonism, respectively.
To confirm synergistic activity of Dox with CuB in induction of cell death, cells were treated with both Dox at 1 µM and CuB at 150 nM for 48 h, and the percentage of viable cells was measured using multiplexed cytotoxicity assay (Figure 2(b)), and cytotoxic activity was measured using cytotoxicity assay (Figure 2(c)). The percentage of viable cells was reduced, and cytotoxic activity was elevated as a result of cotreatment, compared with treatment of Dox alone. Taken together, these results indicate that Dox synergizes with CuB in induction of death of ATC cells.
The synergism between Dox and CuB in induction of cell death is mediated by Bcl-2 family proteins and survivin in ATC cells
Dox results in cell death through altered expression of Bcl-2 family proteins and diminution of Bcl-2/Bax ratio in cancer cells, whereas it enhances expression of survivin, thereby contributing to acquisition of resistance in thyroid cancer cells.2,27 Meanwhile, CuB leads to cell death by downregulating Bcl-2 and survivin in cancer cells.21,22 In regard to role of survivin in ATC cells, we previously reported that the heat shock protein 90 inhibitors NVP-AUY922 and 17-allylamino-17-demethoxygeldanamycin induced cytotoxicity via inhibition of survivin in ATC cells.28,29 In this study, Dox synergized with CuB in induction of death of ATC cells, and thus, the involvement of Bcl-2 family proteins and survivin in the synergism between Dox and CuB inducing cytotoxicity was identified.
First, cells were treated with both Dox at 1 µM and CuB at 150 nM for 48 h, and the protein levels of Bcl-xL, Bcl-2, Bax, Bid, and cleaved PARP were measured (Figure 3(a)). As a result of cotreatment, compared with treatment of Dox alone, cleaved PARP protein levels were enhanced, whereas Bcl-2 protein levels were diminished without change in the protein levels of Bcl-xL, Bax, and Bid. As depicted in Figure 3(b), Bcl-2/Bax ratio was diminished by cotreatment of Dox and CuB, compared with treatment of Dox alone.

The combined effect of Dox with CuB on expression of Bcl-2 family proteins and survivin in ATC cells. (a, b) 8505C cells were treated with Dox at 1 µM and CuB at 150 nM for 48 h, and (a) the protein levels of Bcl-xL, Bcl-2, Bax, Bid, and cleaved PARP were measured. (b) The protein levels of Bcl-2 and Bax were quantified by densitometry, and Bcl-2/Bax ratio was calculated. (c) 8505C cells were treated with Dox at 1 µM for 24 and 48 h, and at 0.5, 1, and 5 µM for 48 h. 8505C cells were treated with CuB at 200 nM for 24 and 48 h, and at 100, 200, and 1000 nM for 48 h. Survivin protein levels were measured. (d) 8505C cells were treated with both Dox at 1 µM and CuB at 150 nM for 48 h. Survivin protein levels were measured and quantified by densitometry. All experiments were performed in triplicate. The blots are representatives of independent experiments. Data are expressed as mean ± SE. *p < 0.05.
Next, when cells were treated with Dox at 1 µM for 24 and 48 h, and at 0.5, 1, and 5 µM for 48 h, survivin protein levels were enhanced (Figure 3(c)). In contrast, as cells were treated with CuB at 200 nM for 24 and 48 h, and at 100, 200, and 1000 nM for 48 h, survivin protein levels were diminished. After cotreatment of Dox at 1 µM and CuB at 150 nM for 48 h, compared with treatment of Dox alone, survivin protein levels were diminished (Figure 3(d)).
Taken collectively, these results manifest that synergistic activity of Dox with CuB is linked to the regulation of Bcl-2 family proteins and the repression of survivin in induction of death of ATC cells.
Dox synergizes with CuB through modulation of JAK2/STAT3 and ERK1/2 in induction of death of ATC cells
CuB causes cell death by suppressing JAK2/STAT3 in cancer cells. 15 Moreover, CuB alone or in combination with docetaxel exerts antitumor activities via mediation of STAT3 and ERK in induction of death of cancer cells.16–18 In this study, whether Dox has a synergistic activity with CuB in relation to JAK2/STAT3 and ERK in induction of death of ATC cells was explored.
Cells were treated with Dox and CuB according to various doses and times, and the protein levels of JAK2, STAT3, and total and phospho-ERK1/2 were measured (Figure 4(a)). As a result of treatment, STAT3 protein levels decreased, while phospho-ERK1/2 protein levels increased without alteration in total ERK1/2 protein levels by Dox and CuB. Intriguingly, JAK2 protein levels increased by Dox, but those decreased by CuB. After cotreatment of Dox at 1 µM and CuB at 150 nM for 48 h, compared with treatment of Dox alone, the protein levels of JAK2 and STAT3 decreased, whereas phospho-ERK1/2 protein levels increased without change in total ERK1/2 protein levels (Figure 4(b)).

The impact of Dox in combination with CuB on JAK2/STAT3 and ERK1/2 in ATC cells. (a) 8505C cells were treated with Dox at 1 µM for 24 and 48 h, and at 0.5, 1, and 5 µM for 48 h. 8505C cells were treated with CuB at 200 nM for 24 and 48 h, and at 100, 200, and 1000 nM for 48 h. The protein levels of JAK2, STAT3, and total and phospho-ERK1/2 were measured. (b) 8505C cells were treated with both Dox at 1 µM and CuB at 150 nM for 48 h, and the protein levels of JAK2, STAT3, and total and phospho-ERK1/2 were measured. All experiments were performed in triplicate. The blots are representatives of independent experiments. Data are expressed as mean ± SE.
All taken together, these results demonstrate that the synergism between Dox and CuB in induction of cell death is associated with the inactivation of JAK2/STAT3 and the activation of ERK1/2 in ATC cells.
Dox has a synergistic activity with CuB by ROS-dependent mechanism in induction of death of ATC cells
In cancer cells, artesunate and N,N-dimethyl phytosphingosine sensitize cells to Dox-induced cell death through ROS-mediated pathway, and CuB results in cell death by regulating ROS production.6,7,19,20 In this study, whether the synergism between Dox and CuB in induction of cell death is relevant to ROS in ATC cells was examined.
Cells were treated with both Dox at 1 µM and CuB at 150 nM for 48 h, and ROS production and COX2 protein levels were measured (Figure 5(a) and (b)). As a result of treatment, Dox elevated ROS production, while CuB did not. Dox in combination with CuB, compared with Dox alone, further elevated ROS production and COX2 protein levels.
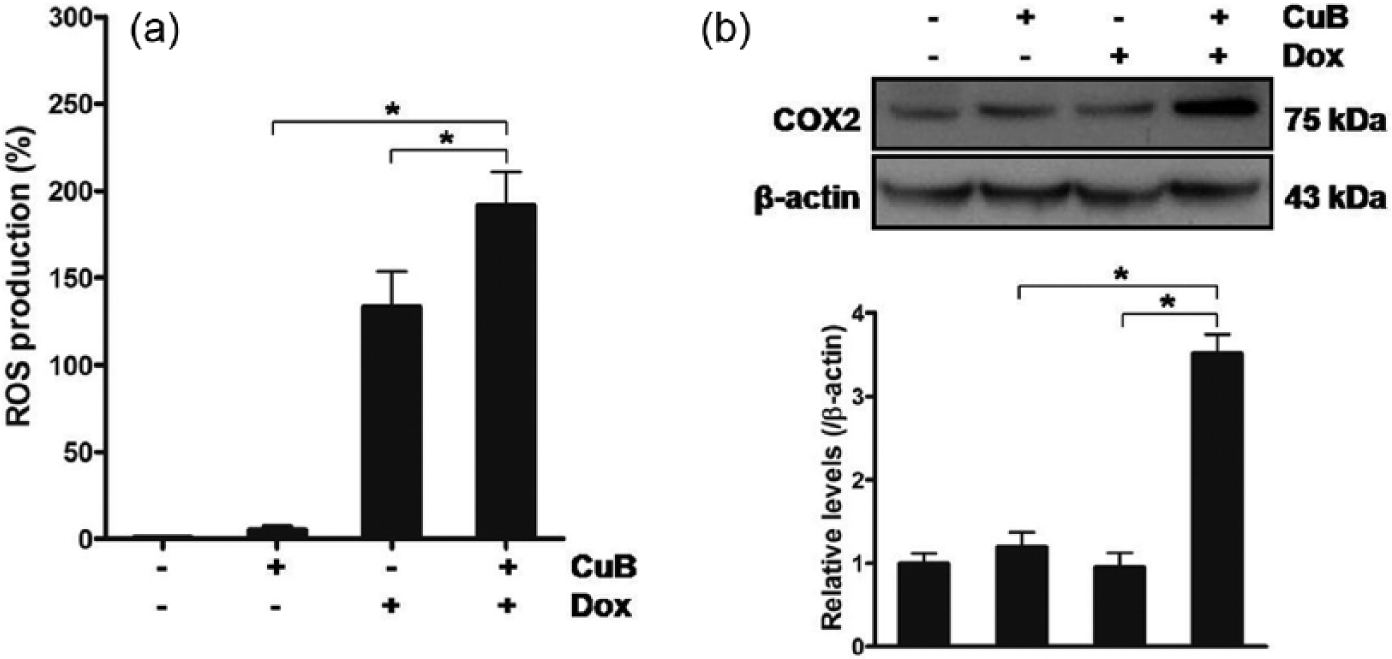
The influence of Dox in combination with CuB on ROS in ATC cells. (a, b) 8505C cells were treated with Dox at 1 µM and CuB at 150 nM for 48 h, and (a) ROS production was measured. (b) COX2 protein levels were measured and quantified by densitometry. All experiments were performed in triplicate. The blots are representatives of independent experiments. Data are expressed as mean ± SE. *p < 0.05.
In summary, these results reveal that Dox synergizes with CuB in involvement of ROS and COX2 in induction of death of ATC cells.
Discussion
This study for the first time demonstrates that Dox synergizes with CuB in induction of cytotoxicity by mediating Bcl-2 family proteins, survivin, and ROS and modulating JAK2/STAT3 and ERK1/2 in ATC cells.
Although Dox is currently used for treatment of ATC patients, its clinical efficacy and responsiveness are not satisfactory.8–10 Furthermore, it was reported that Dox failed to eradicate cancer stem cells derived from ATC cells. 11 Therefore, sensitization of ATC cells to Dox has become an urgent therapeutic challenge in treatment of ATC patients. In this study, when Dox was combined with CuB in ATC cells, all of the CI values in drug combination analysis were lower than 1.0, implying that Dox has a synergistic activity with CuB in induction of cytotoxicity in ATC cells. In addition, Dox in combination with CuB, compared with Dox alone, potentiated cytotoxicity, based on the data of the percentage of viable cells (multiplexed cytotoxicity assay) and cytotoxic activity (cytotoxicity assay), providing additional evidences for the synergism. All taken together, these data indicate that Dox synergizes with CuB in induction of death of ATC cells, suggesting that Dox plus CuB regimen may be an excellent therapeutic option in ATC.
Bcl-2 family proteins are crucial regulators for cellular process. 30 Relative expression of the pro-survival protein Bcl-2 and the anti-survival protein Bax, called Bcl-2/Bax switch, has an impact on the fate of cancer cells. 31 In malignant cells, Dox leads to cytotoxicity through modulation of Bcl-2 family proteins and reduction in Bcl-2/Bax ratio, whereas CuB results in cytotoxicity via underexpression of Bcl-2.2,21,22 Meanwhile, survivin counts as a chemoresistant factor and is overexpressed in cancer cells refractory to Dox.3,4 In addition, survivin inhibits CuB-induced cytotoxicity in pancreatic cancer and hepatoma cells.21,22 In this regard, it was shown that expression of survivin was higher in ATC than well-differentiated thyroid carcinoma and overexpressed by Dox, thereby contributing to procurement of resistance in thyroid cancer cells.27,32 Moreover, we previously reported that the heat shock protein 90 inhibitors NVP-AUY922 and 17-allylamino-17-demethoxygeldanamycin induced cytotoxicity by suppressing survivin in ATC cells.28,29 In this study, to elucidate a molecular mechanism for synergistic activity of Dox with CuB in ATC cells, whether Dox synergizes with CuB in involvement of Bcl-2 family proteins and survivin in ATC cells was explored. In cells treated with both Dox and CuB, compared with Dox alone, the protein levels of Bcl-xL, Bax, and Bid were not altered, whereas Bcl-2 protein levels and Bcl-2/Bax ratio were reduced. Furthermore, consistent with the previous study, 27 survivin protein levels were elevated by Dox, and the elevation was attenuated by combination with CuB. Taken together, these results connote that the synergism between Dox and CuB is related to regulation of Bcl-2 family proteins and repression of survivin in ATC cells (Figure 6).

A proposed model for mechanism underlying the synergism between Dox and CuB in ATC cells.
STAT3 and ERK have pivotal roles in survival, growth, and proliferation in thyroid cancer cells.33,34 In regard to the inhibitory effect of CuB on STAT3 and ERK in cancer cells, the effect on STAT3 is universe, while that on ERK is diverse. In this study, considering that CuB alone or in combination with chemotherapeutic agents exerts antitumor activities through modulation of JAK2/STAT3 and ERK in cancer cells, 14 whether Dox synergizes with CuB in association with JAK2/STAT3 and ERK in ATC cells was examined. Dox and CuB decreased STAT3 protein levels, whereas increased phospho-ERK1/2 protein levels without change in total ERK1/2 protein levels. Intriguingly, Dox increased JAK2 protein levels, while CuB decreased the protein levels. Cotreatment of Dox and CuB, compared with treatment of Dox alone, decreased the protein levels of JAK2 and STAT3 and increased phospho-ERK1/2 protein levels. These results denote that synergistic cytotoxicity of Dox with CuB is relevant to the suppression of JAK2/STAT3 and the stimulation of ERK1/2 in ATC cells.
ROS are highly reactive molecules that can cause oxidative damage in cancer cells.6,7,19,20 In malignant cells, artesunate and N,N-dimethyl phytosphingosine sensitize cells to Dox-induced cytotoxicity via regulation of ROS, and CuB induces cytotoxicity by ROS-dependent mechanism.6,7,19,20 In this regard, it was shown that overexpression of COX2 led to genomic instability, expression of Bcl-2, and resistance to Dox in breast cancer cells. 35 In this study, ROS production was enhanced by Dox, but not CuB. In cells treated with both Dox and CuB, compared with Dox alone, ROS production and COX2 protein levels were further enhanced. These results imply that the synergism between Dox and CuB is linked to ROS and COX2 in ATC cells.
In conclusion, our results suggest that Dox synergizes with CuB in induction of cytotoxicity in ATC cells. In addition, synergistic cytotoxicity of Dox with CuB is mediated by Bcl-2 family proteins, survivin, and ROS and modulated by JAK2/STAT3 and ERK1/2 in ATC cells. This study will provide the clinical implication of Dox in combination with cucurbitacins as an attractive therapeutic regimen in ATC patients refractory to conventional chemotherapeutic agents.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (no. 2015R1A2A2A01003589) to S.J. Lee, Republic of Korea.
