Abstract
UHRF1 is a master regulator of epigenome as it coordinates DNA methylation and histone modifications. Compelling evidence suggests a strong link between UHRF1 overexpression and tumorigenesis, substantiating its ability to act as a potential biomarker for cancer diagnosis and prognosis. UHRF1 also mediates repair of damaged DNA that makes cancer cells resistant toward cytocidal drugs. Hence, understanding the molecular mechanism of UHRF1 regulation would help in developing cancer therapeutics. Natural compounds have shown applicability to downregulate UHRF1 leading to growth arrest and apoptosis in cancer cells.
Introduction
Cancer is among the biggest causes of mortality worldwide and arises from impairment in the intricate orchestration between genetic constitution and epigenetic regulation. Epigenetics refers to heritable alterations in gene expression without any underlying modification in the primary DNA sequence. DNA methylation and histone modifications cooperatively regulate the epigenetic code of every cell. Ubiquitin-like with PHD and RING Finger domains 1 (UHRF1), also known as inverted CCAAT box-binding protein of 90 kDa (ICBP90), acts as an epigenetic coordinator by allowing crosstalk between DNA methylation and histone code. 1 UHRF1, consisting of 793 amino acids was isolated while screening proteins which were able to bind to the inverted CCAAT box (ICB2) in the promoter of topoisomerase IIα (TopoIIα). 2 Involvement of UHRF1 in carcinogenesis emerged from the finding that it was highly expressed in proliferating cells and was inevitably required for G1/S phase transition. 3 Structurally, it is composed of five functionally recognizable domains which allow it to interact with several protein and nonprotein partners to actively regulate DNA methylation, chromatin modifications, cellular proliferation and DNA repair. 4
Aberrations in UHRF1 expression correlate with aggressiveness of many different human malignancies such as laryngeal squamous cell carcinoma, 5 breast cancer, 6 lung cancer, 7 hepatocellular carcinoma, 8 gastric cancer, 9 renal cell carcinoma, 10 cervical cancer, 11 bladder cancer, 12 colorectal cancer 13 and osteosarcoma. 14 Presence of high circulatory levels of UHRF1 DNA in blood samples of breast and gastric cancer patients underlined its importance to act as an independent diagnostic and prognostic marker.15,16 Its easy detection in tissue samples and urine sediments of bladder cancer patients by immunohistochemistry and enzyme-linked immunosorbent assay, respectively, also supports its diagnostic value. 17 The importance of UHRF1 as a potential cancer drug target emerged from several studies where knockdown or silencing of UHRF1 in cancer cells led to reduced proliferation and increased apoptosis.18–20 Its downregulation arrested colorectal cancer cells at G0/G1 phase by reducing the levels of cyclins A and D1, and induced p16INK4A-mediated apoptosis. 21 UHRF1 silencing arrested gall bladder cancer cells at G1/S phase in a p21-dependent pathway and triggered apoptosis by upregulating the expression of promyelocytic leukemia (PML) protein. 22 BALB/c nude mice injected with UHRF1 silenced gastric cancer cells (MKN45-shUHRF1 cells) produced smaller tumors and showed decrease in Ki-67 antigen, the marker of cell proliferation. 9
Recently, many studies have focused on the use of bioactive components of plants for the prevention and treatment of various human diseases, including cancer, due to their easy availability, low cost, oral bioavailability and lesser side effects. 23 Natural compounds have been shown to lower the proliferation of cancer cells by downregulating the expression of UHRF1 through p53- and p73-dependent signaling pathways.24,25 This review focuses on various dimensions of UHRF1 including its role in epigenetics, DNA repair and carcinogenesis. It also provides an insight into its molecular regulation and potential of natural compounds in cancer therapeutics.
UHRF1: the facilitator between DNA methylation and histone modifications
At the level of epigenome, DNA methylation coordinates with posttranslational modifications (PTMs) on histones such as methylation, acetylation and ubiquitination to regulate chromatin function. 26 UHRF1 acts as an epigenetic hub protein that allows crosstalk between DNA methylation and histone modifications. 1 Five functionally recognizable domains (Figure 1), namely, Ubiquitin-like Domain (UBL), Tandem Tudor Domain (TTD), Plant HomeoDomain (PHD), Set and RING Associated (SRA), and Really Interesting New Gene (RING) domain interdependently coordinate epigenetic functioning of UHRF1.

Schematic representation of UHRF1 domains. Ubiquitin-like domain (UBL) at the N terminal confers ubiquitination activity, Tandem Tudor domain (TTD) allows UHRF1 to bind to di-/tri-methylated H3K9, plant homeodomain (PHD) determines the binding specificity of UHRF1 to H3R2 and H3K9me2/3, Set and RING Associated (SRA) domain facilitates in maintaining DNA methylation and histone modifications by recruiting DNMT1 and HDAC1, respectively. Really Interesting New Gene (RING) domain at C terminal confers intrinsic E3 ligase activity toward histones and nonhistone proteins.
TTD is composed of two tightly packed subdomains TTDN and TTDC. A conserved aromatic cage formed by residues F152, Y188 and Y191 in TTDN allows UHRF1 to bind and read di- and tri-methylated lysine residues (H3K9me2/3) which act as transcription repressive chromatin marks. 27 Presence of a peptide binding groove between the subdomains of TTD ensures its specificity toward H3K9 by establishing a tight contact to the residues present both upstream and downstream of the methylated lysine. 28 PHD determines binding specificity of UHRF1 to histones via recognition of unmodified arginine (R2) and lysine (K4) present on H3. 29 PHD cooperates with TTD to read H3K9me3 mark and contributes to pericentric heterochromatin organization of UHRF1. 29 This was also supported by crystallographic studies on TTD–PHD bound to H3K9me3 which showed that UHRF1 possesses the ability to get engaged with both unmodified N terminus of H3 and H3K9me3 simultaneously. 30
SRA domain occurs only in UHRF family and helps in recognition of hemi-methylated DNA as well as recruitment of DNA (cytosine-5)-methyltransferase 1 (DNMT1) for ensuring faithful methylation. 31 The DNA interaction mediated by the crescent moon-shaped SRA domain could be explained by representation of a hand grasping the DNA helix with a conserved methyl cytosine binding pocket situated in the palm. Crystal structure of UHRF1 bound to hemi methylated DNA revealed that SRA domain contains two loops termed as thumb (444–449 residues) and NKR finger (483–496 residues) which mediates its interaction with the DNA duplex by penetrating through the minor and major grooves respectively. 32 R491 replaces 5mC in the DNA helix and pairs with the Hoogsteen edge of the orphaned guanine base. The flipped-out methylated cytosine gets positioned into the binding pocket by π-stacking interactions mediated by the presence of two aromatic conserved residues at the active site, namely, Y478 and Y466. By sterically clashing with the methylated cytosine base present on the opposite strand of DNA, N489 residue within the finger loop acts as selective filter and prevents symmetric C5 base from flipping out of the DNA duplex and therefore allows discrimination between fully and hemi-methylated DNA strands for ensuring truthful methylation patterns. 33
Earlier reports suggested that UHRF1 directly recruits DNMT1 to hemi-methylated DNA. SRA domain has been shown to interact with targeting sequence present within the replication foci targeting sequence (RFTS) domain of DNMT1. 34 However, recent studies suggest that interaction of UHRF1 with histone marks is essential for recruitment of DNMT1 to daughter strands, in addition to cooperative interaction between TTD and SRA domain for establishment and maintenance of methylation patterns.35,36 RING domain-mediated ubiquitination of histone H3 provides the docking site for DNMT1 and cooperative binding between TTD, PHD and SRA domains is prerequisite for ubiquitination.37,38 Ubiquitination of histones (H3K18/23) mediated by the RING domain targeted DNMT1 to its substrate by interacting with ubiquitin interacting motif (UIM) present in the targeting sequence of DNMT1 (Figure 2).38,39
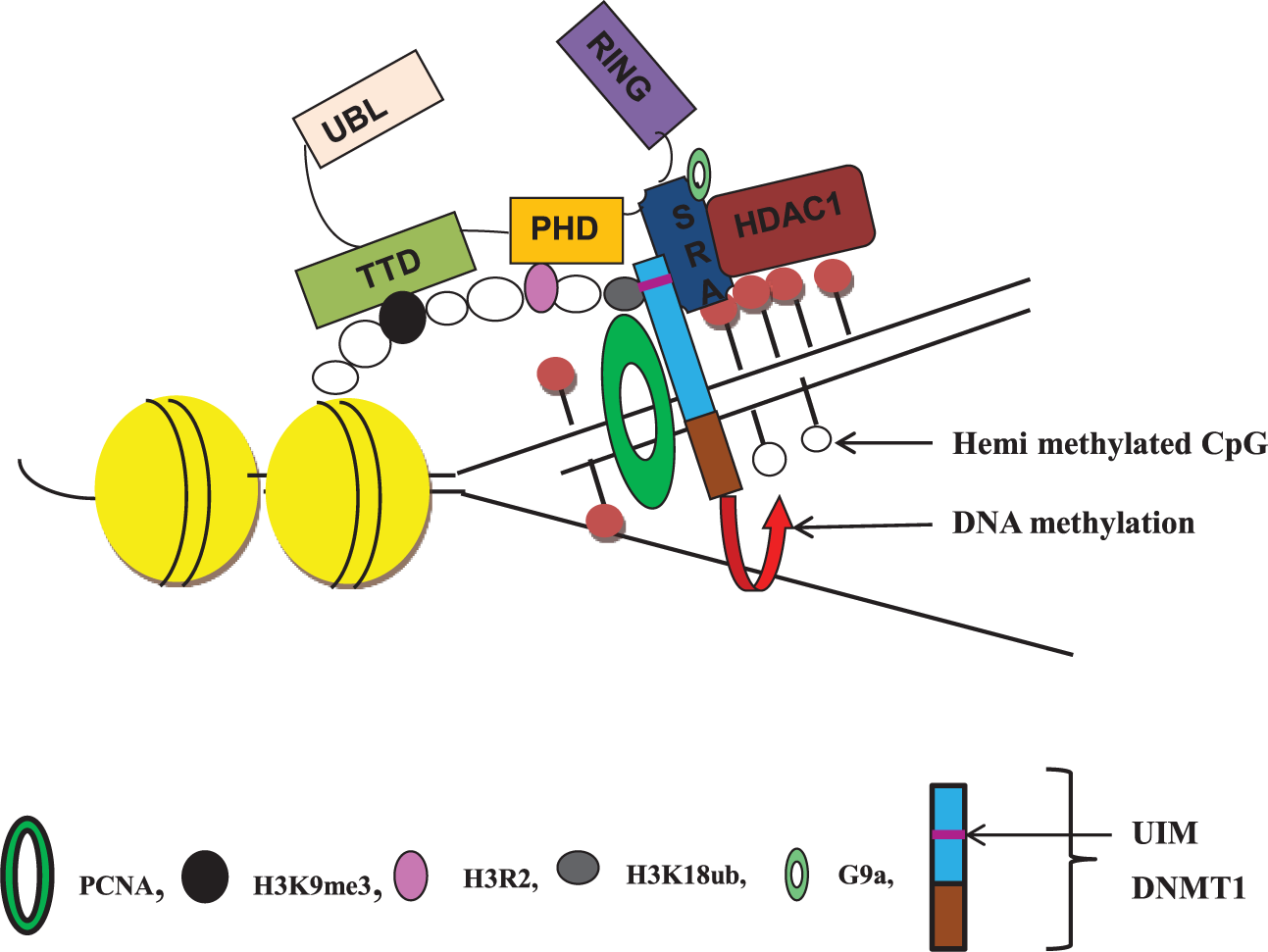
UHRF1 as adaptor protein for DNA methylation and histone modification. UHRF1 interacts with proliferating cell nuclear antigen (PCNA) at the replication fork and recognizes hemi-methylated CpG sites with subsequent recruitment of DNA (cytosine-5)-methyltransferase 1 (DNMT1) via its SRA domain for DNA methylation maintenance. Binding of ubiquitinated H3K18 (catalyzed by cooperative binding of TTD, PHD and SRA domains) to the ubiquitin interacting motif (UIM) present in the targeting sequence (TS) domain of DNMT1 is also essential for maintaining DNA methylation patterns. Recruitment of G9a methyltransferase to the SRA domain catalyzes di- and tri-methylation of H3 at lysine 9. Recognition of H3K9me3 by TTD and unmodified H3R2 by PHD further increases the chromatin binding ability of UHRF1. Further, histone deacetylase 1 (HDAC1) binding to SRA domain ensures histone deacetylation which represses transcriptional process by promoting heterochromatin formation.
The ability of UHRF1 to maintain the methylation status in neo-synthesized DNA strands during S phase has been explained on the basis of allosteric regulation which induces UHRF1 to adopt different conformational states. 30 In the absence of hemi-methylated DNA, UHRF1 attains an occluded conformation due to which recognition of histone marks is inhibited and recruitment of DNMT1 is obstructed. Mechanistically, in this closed state, polybasic region (PBR) acting as a spacer between SRA and RING domain binds within the peptide binding groove of TTD which inhibits interaction of TTD-PHD linker and therefore, recognition of H3K9me3 is prohibited. Binding of SRA domain to the PHD in this ground state also hinders recognition of unmodified R2 present at the N terminal of H3. 30 In the presence of hemi-methylated cytosine residues in a CpG dinucleotide, UHRF1 adopts an open-state conformation where intramolecular interactions within the protein are impaired and chromatin localization of UHRF1 is promoted due to recognition of H3K9me3 mark by TTD-PHD. 30 Binding of phosphatidylinositol 5-phosphate (PI5P) to the PBR located between SRA and RING domain also allowed UHRF1 to adopt an open state and facilitated SRA domain to recognize the hemi-methylated DNA with further recruitment of DNMT1 for faithful management of methylation patterns.31,40 The conformational dynamics of UHRF1 has also been explained by interaction of ubiquitin-specific-processing protease 7 (USP7), a deubiquitinase enzyme with PBR of UHRF1. Binding of USP7 to UHRF1 disturbs the interaction between TTD and PBR and results in its open conformation. 41
Multiple domains of UHRF1 also allow it to interact with several protein and nonprotein partners which are directly or indirectly involved in regulating the epigenome (Table 1).
Interacting partners of UHRF1 involved in epigenetics.

UHRF1-mediated silencing of tumor suppressor genes
Aberrant promoter methylation of tumor suppressor genes is an important epigenetic hallmark of carcinogenesis. 54 Since UHRF1 acts as an epigenetic adaptor protein, alterations in its expression disrupt the epigenome and mark cancer development and progression. 4 Localization of UHRF1 on methylated CpG islands of PAX1, 55 KiSS1, 56 CDKN2A, RASSF1, 57 p14AR and p16INK4A 6 maintained them in repressive state by inducing hypermethylation and histone deacetylation due to recruitment of DNMT1 and histone deacetylase 1 (HDAC1) by the SRA domain. 58 In breast cancer, UHRF1 led to hypermethylation and inhibition of BRCA1 by forming an inhibitory transcriptional complex comprising of HDAC1, DNMT1 and G9a over its promoter. 59 Modulation of DNMT1 by UHRF1 regulated the methylation of MEG3 and progression of hepatic cancer. 60 pRb2/p130, a tumor suppressor gene belonging to retinoblastoma (Rb) family which plays a vital role in regulating cell proliferation, cooperated with UHRF1 and DNMT1 for maintaining the methylation status of estrogen receptor α (ER-α) gene in MCF-7 breast cancer cells. 61 UHRF1 has been shown to interact with PRMT5, which catalyzes symmetric dimethylation of R8 residue on H3 for silencing tumor suppressor genes. 53 UHRF1-mediated H3R8 dimethylation on the promoter of SOCS3 and 3OST2 contributed to the development of endometrial carcinogenesis. 62 Similarly, alterations in the methylation of peroxisome proliferator-activated receptor (PPARγ) due to UHRF1-mediated DNMT3b recruitment on its promoter region resulted in progression of colorectal cancer. 63 In bladder cancer cells, UHRF1 suppressed regulator of G-protein signaling 2 (RGS2) gene expression via promoter hypermethylation resulting in cellular proliferation. 64
Role of UHRF1 in carcinogenesis was also supported by the observation that its downregulation in MKN45 gastric cancer cells resulted in reactivation of seven tumor suppressor genes (CDX2, CDKN2A, RUNX3, FOXO4, PPARG, BRCA1 and PML) by promoter demethylation and inhibited cell proliferation. 9 Knockdown of UHRF1 in A549 lung adenocarcinoma cells led to hypomethylation of otherwise frequently hypermethylated RASSF1, CYGB, and CDH13 tumor suppressor genes. 57 Exposure of BEL-7402, a hepatic cancer cell line, to trichostatin A (HDAC inhibitor) and 5-Aza-CdR (DNA methylation inhibitor) decreased binding of UHRF1 to the promoter of 3OST2 tumor suppressor gene, delineating the involvement of UHRF1 in the direct regulation of 3OST2. 65 These studies clearly indicate the involvement of UHRF1 in epigenetic silencing of key tumor suppressor genes which ultimately results in cancer development and progression.
Involvement of UHRF1 in DNA repair
Besides silencing tumor suppressor genes, UHRF1 has also been linked to DNA repair pathways which is responsible for cancer cells resistance toward chemo and radiotherapy. In NIH3T3 cells, interaction between RING finger of UHRF1 and Eme1 DNA repair enzyme actively rescued stalled DNA replication caused by camptothecin treatment (quinoline alkaloid which collapses DNA replication fork). 66 SRA domain recognizes and recruits UHRF1 to the interstrand crosslinks (ICLs) of DNA caused by various genotoxic agents such as mitomycin C and psoralen derivative 4,5′,8-trimethylpsoralen (TMP). 67 In vivo cell imaging demonstrated direct involvement of UHRF1 in triggering recruitment of Fanconi anemia group D2 (FANCD2) to ICL for initiation of DNA repair. 67 UHRF1 also acted as a scaffold for recruitment of endonucleases which are required for repairing ICL lesions. In HeLa nuclear extract, UHRF1 interacted and regulated the activity of Mus81-EME1 and ERCC1-XPF endonucleases which initiate incision and unhooking of ICLs. 68 Reduced accumulation of γH2AX and 53BP1 nuclear foci after exposure to genotoxic agents in UHRF1 mutant HCT116 cells also supported involvement of UHRF1 in DNA repair. 68
Co-immunoprecipitaion of UHRF1 with N-methylpurine DNA glycosylase (MPG), the first enzyme identified in base excision repair pathway, suggested its involvement in repairing single-strand DNA damage. 69 UHRF1 also acts as a deciding factor for the choice between homology-directed and nonhomologous end joining (NHEJ) double-strand DNA repair pathway. In HEK293T cells during S phase, an interaction between BRCT domain of BRCA1 (which promotes homologous recombination) and phosphorylated S674 residue of UHRF1 catalyzed polyubiquitination of replication timing regulatory factor 1 (RIF1). Subsequent dissociation of RIF1 from 53BP1, an antagonized effector of BRCA1, resulted in inhibition of NHEJ repair system and thus initiation of homologous recombination. 70 Involvement of UHRF1 in double-strand repair pathway was also shown in esophageal squamous cell carcinoma, where inhibition of UHRF1 downregulated endogenous levels of Ku heterodimer protein complex (Ku70/Ku80) which is well known to catalyze NHEJ DNA repair. 71
Molecular regulation of UHRF1
Phosphorylation is one of the main mechanisms responsible for regulation of UHRF1 which contains three cAMP-dependent protein kinase A (PKA) serine residue consensus sites (RRKS-298, KRKS-651, and RRTS-666). Since, PKA signaling has been implicated in cell cycle phase transitions, UHRF1 regulation by PKA could be a major event in forestalling cell cycle checkpoints which could result in unabated proliferation of cells. COS-1 cells treated with forskolin (a diterpene used to increase cAMP levels) showed enhanced UHRF1 phosphorylation at serine 298 resulting in efficient binding of UHRF1 to the ICB2 element present in the promoter region of TopoIIα and promoted G1 to S phase transition. 72 UHRF1 also contains consensus sequence motifs for protein kinase 2 (PK2), a ubiquitous serine/threonine kinase also known as casein kinase 2 (CK2) which is actively involved in cellular proliferation. COS-1 cells rapidly passed G1 phase due to enhanced phosphorylation by the catalytic subunit of CK2 on serine 354 present within the PHD of UHRF1. 73
Phosphorylation of UHRF1 at serine 661 by CCNA2/CDK2 complex in COS-7 cells regulated the nuclear entry and exit of UHRF1. The phosphorylated UHRF1 was detected in the cytosol while the nonphosphorylated form was restricted to the nucleus. 74 Activated extracellular signal-regulated kinase 1/2 (ERK1/2) signaling pathway is known to trigger entry of cells into S phase. Blocking of ERK1/2 pathway by using specific inhibitors (PD98059, LY294002 and AG490) in Jurkat T cells reduced UHRF1 expression, indicating the involvement of this pathway in UHRF1 regulation. 75 Peptide 4N1, agonist of cluster of differentiation 47 (CD47), a transmembrane integrin-associated protein also induced UHRF1 upregulation with subsequent downregulation of p16INK/4A in astrocytoma cells. 76
Presence of transcription factor binding sites in the promoter region of UHRF1 allows it to be regulated by several transcription factors. 3,3′,5-Triiodo-L-thyronine (T3)/thyroid receptor (TR) signaling which plays an important role in regulation of cellular proliferation and differentiation, downregulated UHRF1 in Hep G2 liver cancer cells by repressing the binding of transcription factor specificity protein 1 (SP1). This subsequently led to cell growth inhibition due to the induction of p21 expression. 77 Yin Yang 1 (YY1) transcription factor aided G9a (methyltransferase which catalyzes di-/tri-methylation at H3K9) to act as an upstream negative regulator of UHRF1. In H1299 lung cancer cells, both G9a and YY1 individually repressed UHRF1 transcription but the repression was higher when G9a was co-transfected with YY1, suggesting the ability of YY1 to function as a mediator for G9a recruitment to the UHRF1 promoter. 78 In cervical cancer also UHRF1 is inversely correlated with G9a. 78 E2F1 and E2F8 which are generally overexpressed in cancer cells have also been shown to control UHRF1 expression.6,79 In esophageal adenocarcinoma-derived OE33 cell line, UHRF1 was found to be the direct target of forkhead box M1 (FOXM1) transcription factor, which acts as a key regulator of periodic gene transcription at G2/M phase of cell cycle. 80
Different microRNAs (miRs) have been studied for their ability to regulate the expression of UHRF1 in cancer cells. The microRNA.org database provides an insight into the alignment score predicted for various miRNAs and UHRF1. 81 In bladder cancer, UHRF1 was regulated by miR-124 and miR-145.82,83 While in gastric cancer, overexpressed miR-146a/b repressed UHRF1 leading to reactivation of tumor suppressor genes (Slit3, CDH4, and RUNX3) through promoter DNA hypomethylation. 84 In renal cell carcinoma also miR-146a-5p targeted UHRF1. 85 miR-193a-3p has been shown to downregulate UHRF1 in a highly metastatic lung cancer cell line (SPC-A-1sci) resulting in reduced metastasis. 86 Blocking of miR-9 expression with antagonistic oligonucleotide in HCT116 and HT29 colorectal cancer cells resulted in enhanced expression of UHRF1 (Figure 3). 87

Molecular regulation of UHRF1.
Modulation of UHRF1 by natural polyphenols
Applicability of bioactive components from plant extracts as complementary and alternative medications for the prevention and treatment of cancer by targeting the epigenetic machinery has been documented in various studies. 88 In colorectal cancer cells, luteolin (3′,4′,5,7-tetrahydroxyflavone) caused apoptosis by downregulating UHRF1. 89 Limoniastrum guyonianum aqueous gall extract and luteolin arrested HeLa cells in G2 phase and induced apoptosis through inhibition of UHRF1/DNMT1 tandem expression, followed by an upregulation of p16INK4A. 90
Most of the natural plant extracts and compounds target UHRF1 through p53- or p73-dependent mechanisms. Induction of p53/p73 upregulates p21WAF1/CIP1 (CKI of cyclin E/CDK2 complex) resulting in downregulation of UHRF1 either directly or through deactivation of E2F1.6,24,25 Aronia melanocarpa juice (AMJ) upregulated p53 and p73 and downregulated UHRF1 resulting in caspase 3-dependent apoptosis in terato carcinomal cancer stem-like cells. 91 Epigallocatechin gallate (EGCG), 92 AMJ, 25 thymoquinone, 93 and red wine polyphenolic (RWP) extract 94 downregulated UHRF1 in Jurkat cells through p73-dependent mechanism and consequently upregulated the levels of caspase-3 leading to formation of apoptotic bodies. In BALB/c mice model, RWP treatment arrested tumor cells in G0/G1 phase involving p21-mediated reduction in UHRF1. 95
Thymoquinone and curcumin targeted UHRF1 by inhibiting calcium-/calmodulin-dependent 3′,5′-cyclic nucleotide phosphodiesterase 1A (PDE1A), an upstream repressor of p73 and a negative regulator of p21 as well as p27 (Figure 4).96,97 Bilberry extract (Antho 50) rich in polyphenols caused reactive oxygen species (ROS)-dependent but p53-/p73-independent UHRF1 downregulation in chronic lymphocytic leukemia cells. 98 UHRF1 has shown to regulate Keap-1-Nrf2 pathway which provides protection to cells from oxidative stress by inducing expression of antioxidant response element genes. 99 These studies underline the potential of natural compounds to control tumor growth by targeting UHRF1.

Downregulation of UHRF1 by natural polyphenols. Upregulation of p53 and p73 results in overexpression of p21 and p16 that downregulates UHRF1 with subsequent induction of cell cycle arrest and apoptosis.
Conclusion
UHRF1 acts as a master regulator of the epigenome with its ability to coordinate the methylation and histone modifications. Pentafunctional domains of UHRF1 allow it to interact with various protein and nonprotein molecules involved in epigenetics, DNA repair and cell proliferation. Overexpression of UHRF1 disrupts the harmony in the epigenetic code of a cell which results in silencing of key tumor suppressor genes. There is a growing body of evidence which indicates that upregulation of UHRF1 occurs in several types of cancers, indicating the potential of UHRF1 to act as a biomarker for cancer diagnosis and prognosis. Involvement of UHRF1 in repair of damaged DNA strands following radiation and chemotherapy makes cancer cells resistant toward cytocidal therapeutics with reduced survival of cancer patients. Hence, identification of direct upstream regulators of UHRF1 such as microRNAs, transcription factors and growth factors will aid in strategic planning for better regulation of UHRF1 in cancer cells. Natural polyphenols have shown the potential to significantly downregulate UHRF1 with subsequent arrest in cell cycle and apoptosis in cancer cells. Hence, targeting this multifactorial hub protein with natural dietary polyphenols might lead to improved therapeutics for cancer with lesser side effects.
Footnotes
Acknowledgements
Senior Research Fellowship to Harsimran Sidhu by Indian Council of Medical Research (ICMR), India, is duly acknowledged.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The study was supported by the University Grants Commission, India (no. 43-72/2014 (SR)).
