Abstract
Pemetrexed is an antifolate cytostatic agent targeting several folate-dependent enzymatic pathways, widely used in the treatment of locally advanced or metastatic stage non-small cell lung cancer. Aside from the non-squamous histology, there is still no available molecular biomarker predicting treatment efficacy of pemetrexed-based chemotherapy. The aim of our retrospective study was to evaluate the association of thyroid transcription factor 1 expression with outcome of a large cohort of patients with non-squamous non-small cell lung cancer treated with pemetrexed. We retrospectively analysed clinical data of 463 patients with advanced-stage non-small cell lung cancer (IIIB or IV) treated with pemetrexed-based chemotherapy. Thyroid transcription factor 1 expression was assessed using indirect immunohistochemical detection in formalin-fixed paraffin-embedded tumour tissue at the time of diagnosis. Thyroid transcription factor 1 expression was detected in the tumour tissue from 76.0% of patients, and tumours from 24.0% of patients were thyroid transcription factor 1 negative. The median progression-free survival and overall survival for patients with thyroid transcription factor 1 positive tumours were 4.8 and 11.8 months compared to 2.8 and 8.3 months for those with thyroid transcription factor 1 negative tumours (p = 0.001 and p < 0.001). The multivariable Cox proportional hazards model revealed that thyroid transcription factor 1 expression was significantly associated with progression-free survival (hazard ratio = 1.57, p < 0.001) and also with overall survival (hazard ratio = 1.73, p < 0.001). In conclusion, the results of the conducted retrospective study suggest that the thyroid transcription factor 1 expression was independently associated with progression-free survival and overall survival in patients with advanced-stage non-squamous non-small cell lung cancer treated with pemetrexed-based chemotherapy.
Keywords
Introduction
Lung cancer is one of the leading cancer-related causes of morbidity and mortality in developed countries.1,2 Non-small cell lung cancer (NSCLC) is the most common histological type constituting more than 80% of all lung carcinomas. 3 Pemetrexed represents one of the novel effective agents, which have been approved for the treatment of patients with advanced-stage NSCLC in recent years. It has been previously demonstrated that the superiority of pemetrexed is limited to patients with non-squamous histology.4,5 However, so far, no reliable molecular or genetic biomarker predicting response to pemetrexed in the subgroup with non-squamous NSCLC has been established.
The prognostic role of thyroid transcription factor 1 (TTF-1) expression in NSCLC patients was previously assessed in several studies with conflicting results.6–13 The meta-analysis performed by Berghmans et al. 14 including 1101 NSCLC patients suggested that TTF-1 is a prognostic factor for survival in NSCLC and that its effect appears also significant in the subgroup of patients with adenocarcinoma. However, the prognostic role of TTF-1 expression in patients with advanced or metastatic stage seems to be controversial. The previous studies mostly included patients with early-stage NSCLC and most of them did not receive any adjuvant therapy, and subsequently the survival was less likely to be affected by other treatments, as compared with surgical resection.
The aim of our retrospective study was to evaluate the association of TTF-1 expression with outcome of a large cohort of patients with non-squamous NSCLC treated with pemetrexed-based chemotherapy.
Patients and methods
Patients and treatment
We retrospectively analysed clinical data of 463 adult patients with histologically confirmed locally advanced (IIIB) or metastatic stage (IV) non-squamous NSCLC treated with pemetrexed-based therapy between 2008 and 2015. Pemetrexed was administered intravenously at the standard approved doses (500 mg/m2 every 3 weeks) in combination with platinum derivative (i.e. cisplatin or carboplatin) as the first-line treatment or in monotherapy as the second- or third-line treatment. Dose interruption or reduction was permitted in the event of treatment-related toxicity.
Data source
The clinical registry TULUNG (http://tulung.registry.cz/), in which Faculty Hospital Pilsen, Faculty Hospital Brno, Faculty Hospital Olomouc and Faculty Hospital Hradec Kralove are participating since its creation, is a non-interventional post-registration database of epidemiological and clinical data of patients with advanced-stage NSCLC treated with targeted or novel therapies in the Czech Republic. The registry contains anonymized individual patient data including demographic parameters, initial staging and disease characteristics, baseline patient information at the start of targeted therapy, as well as data on survival and adverse events, which is updated at least twice a year. Data on TTF-1 expression findings were extracted from the hospital information system and merged to the registry data. The protocol was approved by the independent ethics committee of the University Hospital Pilsen, Faculty Hospital Brno, Faculty Hospital Olomouc and Faculty Hospital Hradec Kralove and complied with the International Ethical Guidelines for Biomedical Research Involving Human Subjects, Good Clinical Practice guidelines, the Declaration of Helsinki and local laws.
Clinical monitoring
The treatment was prospectively monitored and the clinical course of patients was continuously assessed at specific time points. Clinical follow-up controls including physical examination, plain chest X-ray and routine laboratory tests were performed every 3–4 weeks; computed tomography (CT) or positron emission tomography (PET)-CT was performed after 2 or 3 months of the treatment. The objective tumour response was assessed by the attending physician using Response Evaluation Criteria in Solid Tumours (RECIST). 15
Assessment of TTF-1 expression
The assessment of TTF-1 expression status was performed at the time of diagnosis. As it is a standard practice in the Czech Republic, immunohistochemical testing of markers characteristic for glandular (TTF1) and squamous (p63/p40) differentiation is performed as an integral part of the final histological diagnosis in vast majority of NSCLC samples.
The TTF1 immunohistochemistry was performed using standardized protocols of pathology department in each participating centre. Generally, all laboratories use indirect immunohistochemical detection of TTF1 in formalin-fixed paraffin-embedded tumour tissue. Of four participating centres, two use primary antibody SPT24 (Leica/Novocastra) and the other two use 8G7G3/1 monoclonal antibody (BioLegend/Dako). As a detection system, either one of the following kits is used: Leica BondMax, Dako Real Envision and Ventana UltraView. A brief analysis (unpublished results) focused on correlation of TTF1 expression depending on the particular method with the herein reported clinical data did not show any significant difference.
Statistics
Standard frequency tables and descriptive statistics were used to characterize sample data set. Progression-free survival (PFS) and overall survival (OS) were estimated using Kaplan–Meier method and all point estimates were accompanied by 95% confidence intervals. PFS was determined from the date of pemetrexed treatment initiation until the date of first documented progression or death. OS was determined from the date of pemetrexed treatment initiation until the date of death due to any cause. Patients who had not progressed or died were censored at the date of last follow-up. Statistical significance of the differences in Kaplan–Meier estimates was assessed using the log-rank test. Finally, multivariable Cox proportional hazards model was used to evaluate the effect of all potential prognostic factors on the survival measures. As a level of statistical significance, α = 0.05 was used.
Results
Patient characteristics
Records of 463 patients were analysed. The study included 264 patients treated with pemetrexed in the first-line treatment and 199 patients treated in the second- or third-line treatment. The baseline patient characteristics are summarized in Table 1. TTF-1 expression was detected in the tumour tissue from 76.0% (352/463) of patients and tumours from 24.0% (111/463) of patients were TTF-1 negative.
Baseline patient’s characteristics.

NSCLC: non-small cell lung cancer; TTF-1: thyroid transcription factor 1; ECOG PS: Eastern Cooperative Oncology Group performance status; CR: complete response, PR: partial response; SD: stable disease; PD: progressive disease.
Survival of patients according to TTF-1 expression
The median PFS and OS for patients with TTF-1 positive tumours was 4.8 and 11.8 months compared to 2.8 and 8.3 months for those with TTF-1 negative tumours (p = 0.001 and p < 0.001). The PFS and OS data are summarized in Table 2 and survival curves are shown in Figure 1. Baseline clinical parameters were assessed together with TTF-1 expression in the multivariable model. The multivariable Cox proportional hazards model revealed that TTF-1 expression (hazard ratio (HR) = 1.57, p < 0.001), Eastern Cooperative Oncology Group performance status (ECOG PS) (HR = 1.71, p < 0.001) and line of therapy (HR = 1.26, p = 0.029) were significantly associated with PFS, whereas TTF-1 expression (HR = 1.73, p < 0.001), smoking (HR = 1.41, p = 0.019) and ECOG PS (HR = 1.94, p < 0.001) were significantly associated with OS (Table 3).
Progression-free and overall survival according to TTF-1 expression.

TTF-1: thyroid transcription factor 1; PFS: progression-free survival; OS: overall survival; CI: confidence interval.

(a) Progression-free and (b) overall survival according to TTF-1 expression.
Multivariable Cox proportional hazards model for progression-free and overall survival.
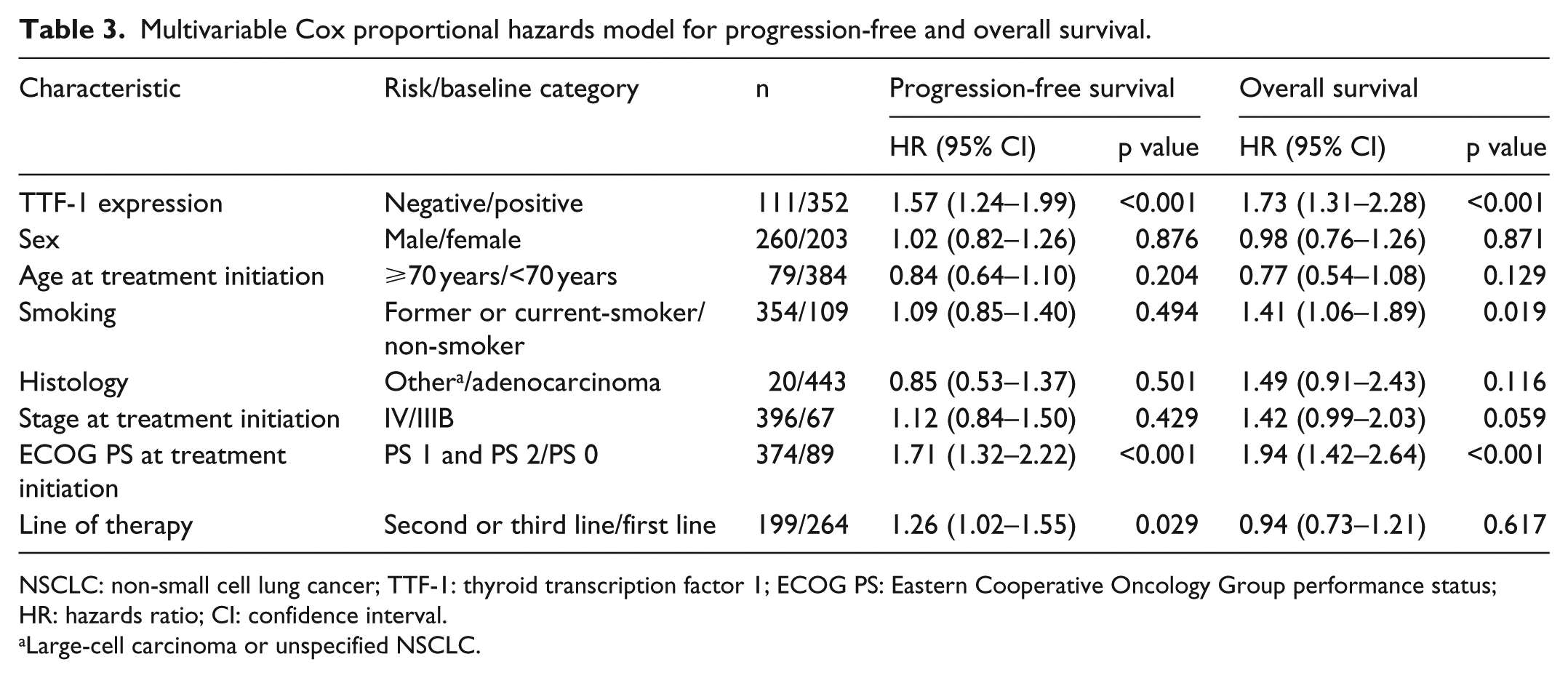
NSCLC: non-small cell lung cancer; TTF-1: thyroid transcription factor 1; ECOG PS: Eastern Cooperative Oncology Group performance status; HR: hazards ratio; CI: confidence interval.
Large-cell carcinoma or unspecified NSCLC.
Among patients treated with pemetrexed in the first-line treatment (n = 264), the median PFS and OS for those with TTF-1 positive tumours was 5.4 and 11.6 months compared to 3.0 and 8.5 months for those with TTF-1 negative tumours (p = 0.007 and p = 0.006) (Figure 2(a) and (b)).

Progression-free and overall survival according to TTF-1 expression for patients treated in the (a, b) first-line treatment and (c, d) second- or third-line treatment.
Among patients treated with pemetrexed in the second- or third-line treatment (n = 199), the median PFS and OS for those with TTF-1 positive tumours was 3.2 and 12.4 months compared to 2.4 and 8.1 months for those with TTF-1 negative tumours (p = 0.024 and p = 0.012) (Figure 2(c) and (d)).
Discussion
Pemetrexed is an intravenously administered antifolate cytostatic agent targeting several folate-dependent enzymatic pathways. Randomized phase III clinical trials as well as observational studies have provided evidence for the efficacy and safety of pemetrexed in combination with cisplatin in the first-line and also as a single-agent in the second- or third-line treatment for patients with advanced-stage non-squamous NSCLC.4,5,16 Recently, the phase III clinical trial PARAMOUNT proved efficacy of pemetrexed in the maintenance setting. 17 Aside from the non-squamous histology, there is still no available molecular biomarker predicting treatment efficacy of pemetrexed-based chemotherapy. Several candidate predictive biomarkers including expression of thymidylate synthase, miR-25, miR-145, miR-210 and serum leptin have been studied but not sufficiently validated for the routine clinical use.18–21 In this study, we focused on the association between TTF-1 expression and the efficacy of pemetrexed-based chemotherapy in patients with advanced-stage non-squamous NSCLC.
TTF-1, also known as NKX2-1, is a tissue-specific member of the homeodomain-containing transcription factor family expressed in thyroid grand, lung and diencephalon during embryogenesis. 22 Homeodomain-containing transcriptional factors play a key role in the control of embryonic development and differentiation.23,24 In the lung, TTF-1 controls the expression of several important lung-specific genes including surfactant protein (SP)-A, SP-B, SP-C, Clara cell secretory protein (CCSP), ATP binding-cassette transporter A3 (ABCA3), lysosomal-associated membrane protein 3 (LAMP3) and carcinoembryonic antigen related cell adhesion molecule 6 (CEACAM6).25–34 The role of TTF-1 in lung carcinogenesis is not fully explained. It was suggested that TTF-1 could promote carcinogenesis by regulating the activity of proliferating cells and the formation of new vessels, at least in adenocarcinoma, and by increasing the rate of cell proliferation.6,35 In primary NSCLC, TTF-1 expression was significantly correlated with adenocarcinoma and small-cell lung cancer, rather than squamous cell carcinoma.7,8
However, the prognostic role of TTF-1 expression in advanced-stage NSCLC patients treated with pemetrexed has been recently assessed in several retrospective studies, data on a large patient cohort are still missing.12,13
In our study, we observed a significantly longer PFS (4.8 vs 2.8 months; p = 0.001) and also OS (11.8 vs 8.3 months; p < 0.001) for patients with tumours expressing TTF-1 compared to those with tumours not expressing TTF-1. The multivariate Cox proportional hazards model confirmed that TTF-1 expression was independently associated with longer PFS (HR = 1.57, p < 0.001) and longer OS (HR = 1.73, p < 0.001). The results are in agreement with previously published study by Sun et al. 13 including 284 advanced-stage non-squamous NSCLC patients; however, the study included only 60 patients treated with pemetrexed-based therapy in the first line. In our study, the difference in survival was more evident for patients treated with pemetrexed in the first line (PFS: 5.4 vs 3.0 months; p = 0.007 and OS: 11.6 vs 8.5 months; p = 0.006) than in those treated in the second or third line (PFS: 3.2 vs 2.4 months; p = 0.024 and OS: 12.4 vs 8.1 months; p = 0.012) suggesting that there could be a significant change in the tumour’s biology or structure before and after the first-line treatment. Thus, the predictive or prognostic factors for the first-line treatment might not be the same as for second- or third-line treatment, particularly when the expression of biomarkers is concerned.36,37 Moreover, the results could be also affected by the use of pemetrexed maintenance in the first-line treatment in those who achieved objective response.
The principal limitations of this study are its retrospective design and relative heterogeneity of the study population including patients treated with combination of pemetrexed with platinum derivative in the first line and those treated with pemetrexed monotherapy in the second or third line; however, separate subgroup analysis is included. This study also did not include control group not treated with pemetrexed; therefore, it cannot be concluded with certainty that patients with tumours not expressing TTF-1 will not benefit from pemetrexed therapy. This question should be answered in prospective randomized trials in the future. Nevertheless, this is the largest study published so far evaluating the prognostic role of TTF-1 expression in patients with advanced-stage NSCLC treated with pemetrexed-based chemotherapy.
In conclusion, the results of the conducted retrospective study suggest that the TTF-1 expression was independently associated with PFS and OS in patients with advanced-stage non-squamous NSCLC treated with pemetrexed-based chemotherapy. Given the poor survival for patients with TTF-1 negative tumours, early restaging should be considered in these patients to identify disease progression, spare them unnecessary therapy with pemetrexed and perhaps enable them to participate in high-risk population clinical trials. Prospective studies on the predictive role of TTF-1 expression should be performed to confirm these results.
Footnotes
Acknowledgements
The authors would like to thank all patients voluntarily taking part in the observational, population-based registry TULUNG.
Declaration of conflicting interests
JF has received honoraria from Astra Zeneca, Roche and Novartis for consultations and lectures unrelated to this project. MS has received honoraria from Astra Zeneca, Roche and Boehringer Ingelheim for consultations and lectures unrelated to this project. AR has received unrestricted grant support for research from Astra Zeneca and Pfizer and honoraria from Astra Zeneca, Roche, Pfizer and Novartis for consultations and lectures unrelated to this project. OF, MP, JS, VK, FS, MT, MS, JK, JK, MH, KH, RC, OT, HH and MN declare that they have no actual or potential conflict of interest including any financial, personal or other relationships with other people or organizations that could inappropriately influence this work.
Funding
This study was supported by the National Sustainability Program I (NPU I) Nr. LO1503 provided by the Ministry of Education Youth and Sports of the Czech Republic.
