Abstract
Junctional adhesion molecules are important components of tight junctions, and Eph/ephrin proteins constitute the largest family of receptor tyrosine kinases. Both junctional adhesion molecules and Eph/ephrin are involved in normal tissue development and cancer progression. However, the expression levels and clinical significances of junctional adhesion molecule-A, a member of junctional adhesion molecules, and EphB2, a member of Eph/ephrin family, in lung adenocarcinoma patients are unclear. Therefore, in this study, we aimed to identify the expression and prognostic values of junctional adhesion molecule-A and EphB2 in lung adenocarcinoma patients’ cohort. In our study, 70 (55.6%) showed high expression of junctional adhesion molecule-A protein and 51 (40.5%) showed high expression of EphB2 protein in 126 lung adenocarcinoma tissues. Junctional adhesion molecule-A and EphB2 expressions were both significantly increased in tumor tissues compared with noncancerous lung tissues. Kaplan–Meier analysis and log-rank test indicated that low expression of junctional adhesion molecule-A and EphB2 proteins can predict better survival and low mortality rate of lung adenocarcinomas. In univariate analysis, high expression levels of junctional adhesion molecule-A and EphB2 were both found to be significantly correlated with poor overall survival of lung adenocarcinoma patients (hazard ratio = 1.791, 95% confidence interval = 1.041–3.084, p = 0.035; hazard ratio = 1.762, 95% confidence interval = 1.038–2.992, p = 0.036, respectively). The multivariate Cox proportional hazard model demonstrated that EphB2 expression is an independent prognosis parameter in lung adenocarcinoma patients (hazard ratio = 1.738, 95% confidence interval = 1.023–2.952, p = 0.016). Taken together, high expression of junctional adhesion molecule-A and EphB2 can predict poor overall survival and high mortality rate, and EphB2 is an independent prognostic biomarker in lung adenocarcinoma patients.
Introduction
Tight junctions (TJs) are adhesion structures between the membrane of epithelial cells which regulate solutes, ions, and other molecules’ permeability of cell layers. Additionally, TJs also form a paracellular fence and maintain cellular polarity.1,2 Increasingly, experimental evidences suggested that TJ proteins are players in initiation and progression of cancers. Loss of TJs’ barrier and fence function are involved in tumor cell proliferation and metastasis. 3 Junctional adhesion molecules (JAMs), members of immunoglobulin superfamily proteins, are located at the lateral membrane of endothelial and epithelial cells. 4 Recent studies showed that dysregulation of junctional adhesion molecule-A (JAM-A), an important component of JAMs as well as TJs, is associated with cancer progression, 5 demonstrating that potential clinical values in cancers. However, the exact roles and clinical significances of JAM-A in cancers remain unknown.
The Eph receptors and their corresponding ephrin ligands constitute the largest family of receptor tyrosine kinases in human genome and are primarily found to participate in cell–cell interactions in the nervous and vascular systems. 6 Eph receptor–ephrin complexes emanate their bidirectional signals in various diseases, including cancer. Its abnormal function has been implicated in many aspects of cancer development and progression.7,8 In recent years, studies in tumor systems revealed that Eph/ephrin is also involved in important signal transductions of carcinogenesis and progression.6,9–11 Particularly, the B-type Eph receptor 2 (EphB2) has been supposed to be a target of the Wnt signaling, which is often hyperactivated in cancer progression. 12 The prognostic values of EphB2 in different tumors remain diverse. For breast and ovarian carcinoma patients, increased EphB2 protein expression can predict shorter survival. 13 While loss of EphB2 expression is correlated with poorer survival in colorectal and gastric carcinoma.14,15
Eph systems have effects on cancer development through variant mechanisms. It is notable that Eph/ephrins promote epithelial cancers by inducing morphological changes. Eph receptors and ephrins’ expressions and phosphorylation disturb the integrity of TJs and cell–cell adhesion. 7 JAM-A is a major constituent protein of TJ; therefore, interplays between JAM-A and Eph receptors may lead to the dysregulation of intercellular junctions in tumorigenesis. However, the expression pattern and clinical significances of EphB2 in lung adenocarcinoma (AC) remain unknown. Further study is also needed to clarify the correlation between EphB2 and JAM-A, as well as their potential role in cancer progression and metastasis.
Quantum dots (QDs) are novel nanocrystals and have been used in a wide variety of applications. Compared with conventional organic dyes and fluorescent proteins, QDs are photostable and have extraordinary fluorescent properties including high brightness, wide excitation spectra as well as narrow, symmetrical emission spectra and can be conjugated to a wide range of biological targets.16,17 In this research, the expression and distribution of JAM-A and EphB2 in lung AC tissues were detected with QDs-based immunohistochemistry (QDs-IHC). The relationship between these two protein expression levels and the clinicopathological significance as well as survival data were analyzed in the lung AC patients.
Materials and methods
Tissue samples and follow-up
A total of 126 formalin-fixed, paraffin-embedded lung AC and corresponding noncancerous lung tissues were collected from the Department of Pathology, Zhongnan Hospital of Wuhan University. Lung AC patients were diagnosed in the period from July 2004 to December 2011. Written informed consent was obtained from all the patients preoperatively. The study was approved by the Ethics Committees of Medical College, Wuhan University.
The patients were composed of 70 men and 56 women with a mean age of 58 (range = 20–84) years. The follow-up began on the date of surgery and ended in August 2012. Clinicopathological features, including age, gender, survival status, depth of tumor invasion (T), lymph node metastasis (N), distant metastasis (M), TNM stage, and histological types, are shown in Table 1.
Patient characteristics (N = 126).

Two pathologists (H.C. and X.Z.) reconfirmed the histopathologic features of these samples independently. All of the lung AC tissue samples had been classified according to the 7th edition of TNM classification by International Association for the Study of Lung Cancer (IASLC). 18 The pathologic classification of the tumor cases was determined based on the 2011 IASLC/American Thoracic Society (ATS)/European Respiratory Society (ERS) classification. 19 Overall survival (OS) was defined as the period from initial diagnosis to death or the date of last follow-up. At the end of follow-up, 70 patients (55.6%) were alive and 56 patients (44.4%) died of lung ACs.
Tissue microarray construction and QDs-IHC
Hematoxylin and eosin–stained slides were screened for the most representative tumor tissues and matched adjacent noncancerous tissues. Two tissue microarray (TMA) slides with a diameter of 1.5 mm were constructed with a tissue manual arraying instrument. 20 Shanghai Outdo Biotech Co., Ltd provided technological support for TMA construction. The two TMA slides consisted of 126 lung AC tissues and each matched adjacent noncancerous tissues.
The expressions of JAM-A and EphB2 were detected by QDs-IHC staining according to the manufacturer’s instructions (Wuhan Jiayuan Quantum Dots Co., Ltd, Wuhan, China). Briefly, the TMAs were deparaffinized in xylene and rehydrated in graded alcohol washes. Antigen retrieval of JAM-A was performed in EDTA (1 mM, pH 8.0) at microwave oven for 25 min, while EphB2 was in EDTA (1 mM, pH 8.0) at autoclave for 4.5 min and then cooled to room temperature for 30 min. All dilution steps (antibodies and QDs) were performed in tris-buffered saline (TBS) containing 2% bovine serum albumin (BSA; Sigma, St. Louis, MO, USA). TMAs were first incubated in 2% BSA buffer at 37°C for 30 min. Then, TMAs were incubated with primary antibody for 2 h at 37°C. Primary antibody used for QDs-IHC staining was rabbit anti-JAM-A (diluted 1:100, ab52647; Abcam, Cambridge, MA, USA) and rabbit anti-EphB2 (diluted 1:100, ab5418; Abcam), respectively. Then, TMAs were washed three times with TBS-T (0.5% Tween in TBS) for 5 min each time and incubated in biotinylated goat anti-rabbit IgG (1:400 dilution; Jackson ImmunoResearch, West Grove, PA, USA) at 37°C for 30 min. For QDs conjugation, TMAs were incubated in 2% BSA buffer at 37°C for 10 min, incubated in QDs (605 nm) conjugated to streptavidin (1:300; Wuhan Jiayuan Quantum Dots Co., Ltd, Wuhan, China) for 1 h, and washed three times with TBS-T for 5 min each. Finally, TMAs were sealed with 90% glycerin (Sigma). TBS instead of two primary antibodies served as negative controls, only showing autofluorescence signal.
Scoring of QDs-IHC staining
The signals obtained from the labeling cells were detected via Olympus BX53 fluorescence microscopy (CCD DP80) at 605 nm. The QDs-IHC results were evaluated by two researchers, who were independent and blinded to the clinical parameters of the study. The scoring was according to the positive area and the staining intensity.
The area of positive (AP) was scored as 0 (no positive area or positive area < 5%), 1 (positive area 5%–25%), 2 (positive area 26%–50%), 3 (positive area 51%–75%), and 4 (positive area >75%). Intensity of staining (IS) was graded as 1 (weak), 2 (moderate), and 3 (strong). The final expression levels of proteins were determined by the following equation: intensity distribution (ID) = AP × IS. The total score ranged from 0 to 12. The cutoff point of high or low expression of JAM-A and EphB2 proteins was determined on the receiver operating characteristic (ROC) curve analysis with respect to OS.
Statistical analysis
SPSS 19.0 software (Chicago, IL, USA) was used to carry out all the statistical analyses. Statistical associations of protein expression and clinicopathological parameters were assessed using the chi-square test. Kaplan–Meier method and log-rank test were performed to analyze the survival data. Cox proportional hazard regression model was conducted to evaluate univariate and multivariate analyses of survival as well as the independent prognostic values. Two-tailed p values of <0.05 were considered statistically significant.
Results
Patient characteristics
Of 126 lung AC patients, 70 were males (55.6%) and 56 were females (44.4%), with the average age of 58 years (range = 20–84 years). According to the TNM stage, 26 patients (20.6%) were T1, 86 (68.3%) were T2, 10 (7.9%) were T3, and 4 (3.2%) were T4. In all, 78 (61.9%) patients were N0, 32 (25.4%) were N1, 6 (4.8%) were N2, and 10 (7.9%) were Nx. Furthermore, 123 patients (97.6%) were M0 and 3 (2.4%) were M1. In the aspect of pathological stage, 38 (30.2%) patients were classified as stage I, 70 (55.5%) as stage II, 15 (11.9%) as stage III, and 3 (2.4%) as stage IV. Other clinicopathological parameters are listed in Table 1.
Cutoff points of JAM-A and EphB2 expression
ROC curve analysis was used to determine the cutoff value of high or low JAM-A and EphB2 expression levels (Figure 1). Based on the optimal sensitivity and specificity of the ROC curve by OS status, 7.0 was defined as the cutoff point for JAM-A expression in lung AC (an ID score ⩾7.0 defined high expression and ID < 7.0 indicated low expression) and 3.5 was defined as the cutoff point of EphB2 expression (an ID score ⩾3.5 defined high expression and ID < 3.5 indicated low expression).

ROC analysis of JAM-A and EphB2 scores by OS of lung AC patients.
The cutoff values of JAM-A and EphB2 expression have optimal sensitivity and specificity. Blue line shows EphB2 expression level in lung AC (area under the curve was 0.570, 95% confidence interval (CI): 0.469–0.671; p = 0.180). Green line shows JAM-A expression level in lung AC (area under the curve was 0.563, 95% CI: 0.462–0.663; p = 0.229).
Expression of JAM-A and EphB2 in lung AC tissues
JAM-A was expressed in both lung AC tissues and corresponding adjacent noncancerous lung tissues including vascular endothelial cell (Figure 2(a)–(d)). Localization of JAM-A was primarily in cell membranes and cytoplasm. High expression of JAM-A was detected in 70 (55.6%) cases of lung AC specimens, which was higher than corresponding noncancerous lung tissues.
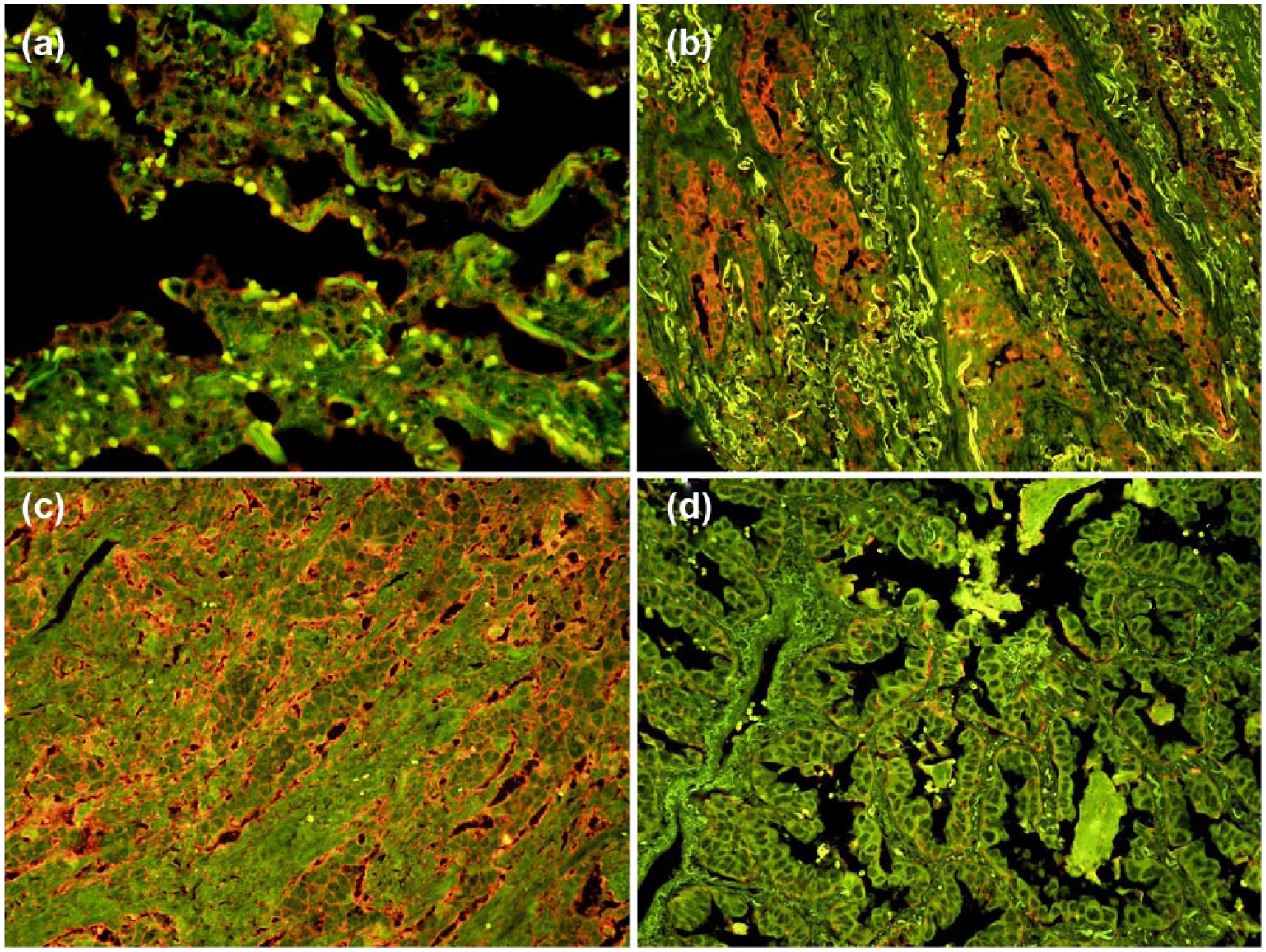
JAM-A protein expression in adjacent noncancerous lung tissues and lung AC tissues: (a) weak JAM-A expression in noncancerous tissue, (b) JAM-A positive expression in lung AC with acinar-predominant subtype, (c) JAM-A positive expression in lung AC with solid type, and (d) JAM-A negative expression in lung AC with papillary subtype (original magnification 200×).
EphB2 was slightly expressed in cytoplasm in the bronchial and alveolar epithelial cells of noncancerous lung tissues (Figure 3(a)). In tumor tissues, EphB2 was predominantly located in cell membranes and cytoplasm (Figure 3(b)–(d)). Compared with noncancerous lung tissues, EphB2 expression was significantly increased in cancer tissues. Of 126 lung AC tissues, 51 (40.5%) showed high EphB2 expression and 75 (59.5%) showed low EphB2 expression.

QDs-based IHC staining results of EphB2 in adjacent noncancerous tissues and lung AC tissues: (a) weak EphB2 expression in the bronchial epithelium, (b) EphB2 positive expression in lung ACs with acinar-predominant subtype, (c) EphB2 positive expression in lung ACs with lepidic-type, and (d) EphB2 negative expression in lung ACs with papillary subtype (original magnification 200×).
Clinical significances of JAM-A and EphB2 expressions
Table 2 shows the association between expression of two proteins and clinicopathological variables. JAM-A and EphB2 expressions were not significantly associated with sex, age, T, N, M, TNM stage, or pathological subtypes of lung AC. JAM-A and EphB2 expressions were both positive in 39 cases of lung AC and negative in 44 cases of lung AC tissues, which indicated that the dysregulation of JAM-A was significantly positively correlated with EphB2.
Correlations between JAM-A and EphB2 expression and clinicopathological parameters of lung adenocarcinoma.
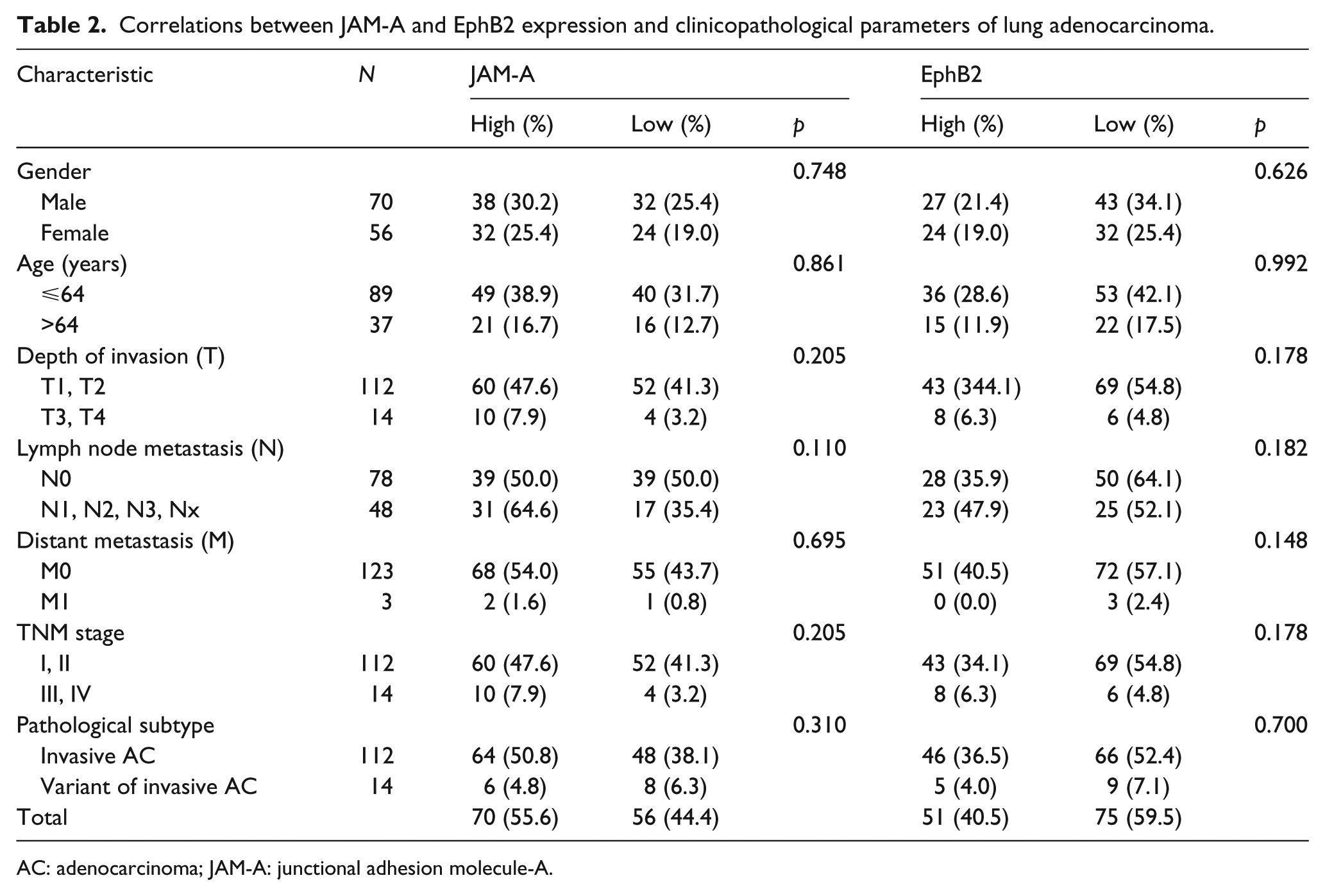
AC: adenocarcinoma; JAM-A: junctional adhesion molecule-A.
Subsequently, we investigated the prognostic value of JAM-A and EphB2 expressions in lung ACs. Kaplan–Meier analysis and log-rank test demonstrated that low JAM-A expression in lung ACs predicted better survival and low mortality rate (Figure 4(a), p = 0.032). Similarly, low expression of EphB2 also predicted better outcome of lung AC patients (Figure 4(b), p = 0.033). Furthermore, both negative and positive expressions of JAM-A and EphB2 also affect OS in lung ACs. Negative expressions of both JAM-A and EphB2 indicated better OS than positive expressions in lung AC patients (Figure 5, p = 0.009).

Cumulative survival curves of JAM-A and EphB2 in lung AC patients. The high level of (a) JAM-A and (b) EphB2 proteins in lung AC patients shows poor OS than the low levels (p1 = 0.032; p2 = 0.033).

Cumulative survival curves of JAM-A and EphB2 both negative and both positive in lung AC patients. JAM-A and EphB2 both negative show better OS than both positive in lung AC patients (p = 0.009).
In univariate analysis, both JAM-A and EphB2 expression levels were found to be significantly related to the OS of lung AC patients (hazard ratio (HR) = 1.791, 95% CI = 1.041–3.084, p = 0.035; HR = 1.762, 95% CI = 1.038–2.992, p = 0.036, respectively; Table 3). Simultaneously, depth of invasion and TNM stage were also significantly correlated with the OS of lung AC patients (Table 3). In order to analyze whether the above univariate was an independent prognostic factor, a multivariate Cox proportional hazard model on OS was performed. The clinicopathological characteristics and the two protein expressions were added in the multivariate analysis model. The results indicated that EphB2 expression was an independent prognostic parameter in OS of lung AC patients (HR = 1.738, 95% CI = 1.023–2.952, p = 0.041; Table 3). Concurrently, TNM stage was also an independent predictor regarding the patients OS. However, the expression of JAM-A was not an independent prognostic factor of lung AC patients in this research.
Cox proportional hazard models on OS of patients.

AC: adenocarcinoma; HR: hazard ratio; CI: confidence interval; JAM-A: junctional adhesion molecule-A.
Bold values represents significant difference.
Discussion
Lung cancer remains the first cause of death in the malignant carcinomas. 21 Although several biomarkers were identified as prognosis-related, more novel biomarkers are still highly required to elucidate the lung AC progression and predict the treatment responses and outcomes of lung AC patients. Here, we assessed the expression of JAM-A and EphB2 and examined their prognostic values in lung AC patients. Our data suggested that low JAM-A and EphB2 expressions in lung ACs could predict better survival and low mortality rate. What’s more, we first revealed that EphB2 is an independent prognostic biomarker in lung AC patients, which needs further prospective research.
Previous studies indicated that the expression of JAM-A seems to be tissue-specific and shows different expression levels in different kinds of tumors.22–24 JAM-A is expressed in many epithelial originated tumors and is complicated in cancer progression. 25 However, only a few studies reported that the expression of JAM-A protein in lung cancer specimens has been evaluated. Previous research showed that low levels of JAM-A in the normal lung tissues are detected, compared with cancer tissues, but prognostic analysis is not performed. 5 Therefore, our research fills the gaps in this field.
In this study, our results of JAM-A expression and clinical significances in lung AC patients were a little different from previous study regarding JAM-A and non–small cell lung cancer (NSCLC). 23 We both demonstrated that lung AC patients with high expression of JAM-A had a significantly lower survival. Differently, Zhang’s research showed that high expression of JAM-A was significantly correlated with TNM stage and lymph node metastases. But our data indicated that JAM-A expression was not significantly associated with those parameters. The cause of this difference may be due to sample size and different subtype of lung cancers. Our follow-up consists of 126 patients, while Zhang’s only contains 82 samples. Moreover, NSCLCs consist of squamous cell carcinoma, large cell carcinoma as well as lung AC, and other less common types. Zhang’s study focused on NSCLC patients, while our patients were only lung ACs. The above all may demonstrate that the expression of JAM-A and the relationship between clinicopathological characteristics are not the same in different subtypes of NSCLC patients.
EphB2, a receptor tyrosine kinase for ephrin ligands, is overexpressed in a variety of cancers and plays an important role in cancer progression.26–28 Before our research, only EphB2 messenger RNA (mRNA) transcription level in small cell lung carcinoma has been reported but no protein expression, distribution, or survival analysis is shown. 29 So far, we first reported that EphB2 was predominantly overexpressed in cell membranes and cytoplasm of lung AC tissues, and low EphB2 expression in lung AC predicted better survival and low mortality rate. Additionally, EphB2 has been proven to be an independent prognostic marker in lung AC patients. Besides the clinical significance, our results also pointed to the involvement of EphB2 in lung AC development, and further studies regarding EphB2 and lung AC biological features are needed.
Our study also found that JAM-A expression is positively correlated with EphB2 expression in lung AC samples. Negative expressions of both JAM-A and EphB2 indicated better OS than positive expressions in lung ACs. This may be explained as their dynamic synergic functions in cancer development. Both JAM-A and EphB2 are active participators in the process of epithelial phenotype7,26,30 in which epithelial-derived cancer cells lose their epithelial phenotypes, such as cell–cell adhesion and cell polarity, and acquire mesenchymal features. 31 Eph receptors and ephrin expressions and phosphorylation disturb the integrity of TJs and cell–cell adhesion. 7 Loss of cohesion of the TJ structure can lead to development and progression of cancer cells. As a new molecular target implicated in TJ formation, dysregulation of JAM-A involves cancer proliferation, invasion, and metastasis. Taken together, interplays between JAM-A and Eph receptors may facilitate the dysfunction of intercellular junctions in tumorigenesis. JAM-A and EphB2 may act as the targets or signaling molecules in the cancer progression and contribute to cancer invasion and metastasis. Moreover, the exact underlying mechanism of JAM-A and EphB2 interactions in lung ACs has not been well understood, which are worthwhile for further in vitro works to explore.
In conclusion, this is the first study that demonstrated the expression and distribution of EphB2 in lung AC patients. Our data elucidated that high expression of JAM-A and EphB2 proteins are correlated with poor OS of lung AC patients. Most notably, EphB2 is independently associated with the OS of lung AC, suggesting EphB2 may be a useful prognostic marker and potential therapeutic target of lung AC patients.
Footnotes
Acknowledgements
C.Z. and A.W. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
