Abstract
Background
Exosomes, key mediators of intercellular communication, transport functional biomolecules, making them promising tools for diagnostics and therapeutics across various human diseases.
Objectives
To systematically review and analyze the clinical applications of exosomes, focusing on their sources, functions, therapeutic roles, and research trends through a bibliometric approach.
Methodology
A comprehensive PubMed search was conducted using the term “exosomes” in titles and abstracts, limited to clinical studies, including Clinical Trials, Phase I–IV studies, and Randomized Controlled Trials. Ninety clinical trials were included. Bibliometric analysis using VOSviewer identified key terms, conceptual clusters, and emerging research topics.
Results
Exosomes demonstrated diverse clinical applications in oncology, neurodegenerative diseases (e.g., Alzheimer’s), cardiovascular conditions, osteoarthritis, COVID-19, spinal cord injuries, transplant rejection, and skin regeneration. Predominant exosome sources included plasma, serum, urine, saliva, tears, ascites, adipose tissue, and stem cells. Functionally, exosomes served as diagnostic biomarkers, therapeutic agents, and molecular delivery systems. Key terms included miRNA, plasma, safety, efficacy, and trial, with conceptual clusters reflecting a shift from molecular exploration to clinical implementation. Emerging themes included precision medicine, biobank integration, and regenerative therapies using mesenchymal stem cell-derived or herbal-modified exosomes.
Conclusion
Exosome-based interventions show promising safety and efficacy but face challenges in standardizing isolation methods, conducting large-scale randomized trials, and ensuring long-term follow-up. Future research should focus on integrating exosome platforms into precision medicine with robust clinical frameworks.
Introduction
Extracellular vesicles (EVs) are released by all cells, both prokaryotic and eukaryotic, both normally and during acquired disorders. 1 Ectosomes and exosomes are the two general types into which EVs fall. Ectosomes, which comprise microvesicles, microparticles, and big vesicles with a diameter of around 50 nm to 1 µm, are vesicles that pinch off the plasma membrane surface by outward budding. 2 Exosomes (Figure 1) are endosomal-derived EVs that range in size from 40 to 160 nm (average diameter of 100 nm). 3 Multivesicular bodies are eventually formed by sequential invagination of the plasma membrane. These bodies can intersect with different intracellular vesicles and organelles, adding to the diversity of exosome contents.4–7 Many components of a cell, including DNA, RNA, lipids, metabolites, and cytosolic and cell-surface proteins, can be found in EVs, including exosomes, depending on the cell of origin.4, 8, 9 There is still much to learn about the physiological function of exosome production. Exosomes are thought to play a part in preserving cellular homeostasis by removing extraneous or surplus components from cells.2, 10 Additionally, recent research examined here shows that some cellular components accumulate in exosomes in a functional, targeted, mechanism-driven manner, indicating that these exosomes may play a part in controlling intercellular communication.11–13
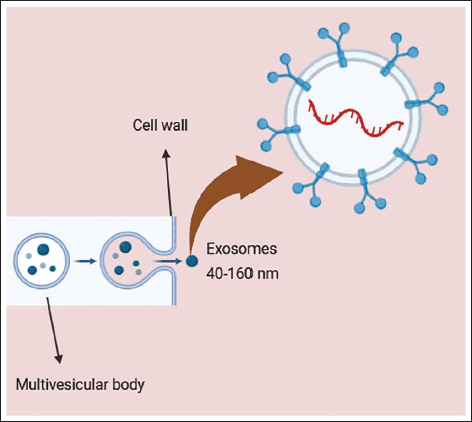
Exosomes are increasingly recognized for their multifaceted roles in health and disease, with implications spanning cancer progression, cardiovascular diseases, central nervous system (CNS) disorders, pregnancy, immune modulation, and viral infections. 14 These nanoscale vesicles mediate intercellular communication by transferring proteins, metabolites, and nucleic acids to recipient cells, thereby modulating their biological responses. 15 Depending on context, exosome-mediated signaling may either promote or suppress disease pathogenesis.4, 16 Their unique ability to influence complex intracellular pathways has positioned exosomes as promising candidates for therapeutic interventions, particularly in cancer and neurological diseases. 17 Exosomes can be engineered to carry diverse therapeutic agents, such as chemotherapeutics, immune modulators, antisense oligonucleotides, and siRNAs, while their intrinsic lipid and protein composition enhances pharmacokinetics, bioavailability, and reduces toxicity.6, 18, 19 Beyond therapeutics, exosomes offer powerful diagnostic potential. As they are present in all biological fluids, liquid biopsies allow for non-invasive access to their cargo, enabling real-time disease monitoring. For example, pediatric glioma patients are undergoing exosome-based liquid biopsies to assist with diagnosis and prognosis. Furthermore, multicomponent analyses of exosomes provide insights into disease progression and therapeutic responses.2, 20–23 The functional significance of exosomes is further illustrated in Table 1, which summarizes their key membrane markers, diverse cargo, and broad biological roles—from gene regulation and immune balance to angiogenesis and metastatic potential—underscoring their central role in intercellular communication and disease modulation. This review provides a comprehensive overview of the clinical applications of exosomes based solely on data from clinical trials to offer an accurate and up-to-date picture of their real-world translational relevance. Across diverse studies, exosomes have demonstrated significant promise as diagnostic and prognostic biomarkers. However, a clear research gap remains in the systematic clinical validation of these findings, especially across larger, multicenter trials and in standardized protocols for exosome isolation, characterization, and dosing. Additionally, there is a need for greater clarity on exosome pharmacokinetics, biodistribution, and long-term safety. This review addresses this gap by focusing strictly on clinical trial data, moving beyond preclinical and in vitro findings, to provide a grounded and realistic evaluation of the current and potential future roles of exosomes in clinical medicine.
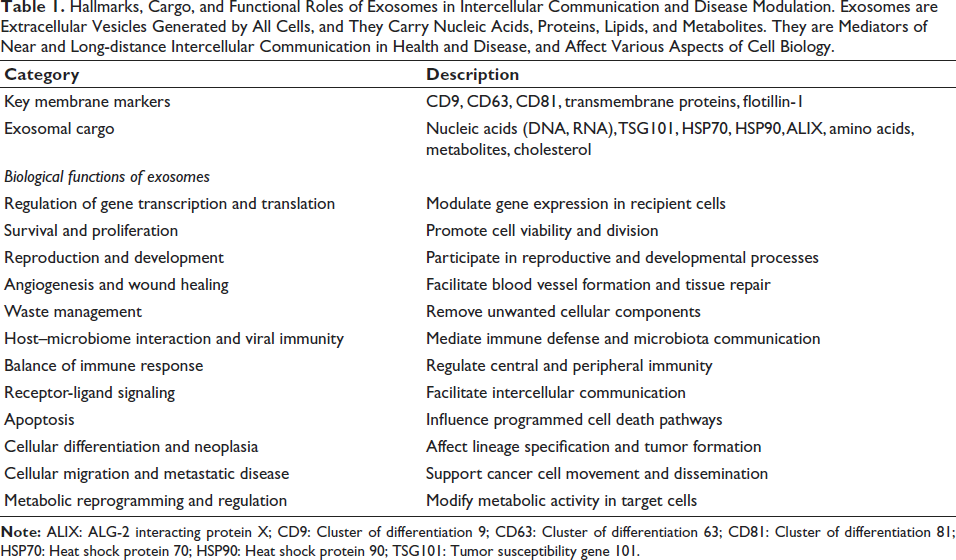
Hallmarks, Cargo, and Functional Roles of Exosomes in Intercellular Communication and Disease Modulation. Exosomes are Extracellular Vesicles Generated by All Cells, and They Carry Nucleic Acids, Proteins, Lipids, and Metabolites. They are Mediators of Near and Long-distance Intercellular Communication in Health and Disease, and Affect Various Aspects of Cell Biology.
Methodology
Search Strategy and Data Sources
A systematic PubMed search was conducted on March 28, 2025, using the keyword “exosomes” in the title and abstract. Filters included Clinical Trial, Protocol, Phase I–IV, and Randomized Controlled Trial, limited to human studies in English. Only interventional studies indexed in PubMed were included; observational studies and external registries (e.g., ClinicalTrials.gov) were excluded. The Boolean string used was: (“exosomes”[Title/Abstract]) AND (Clinical Trial OR Clinical Trial Protocol OR Clinical Trial Phase IOR Clinical Trial Phase IIOR Clinical Trial Phase IIIOR Clinical Trial Phase IVOR Randomized Controlled Trial) AND (humans) AND (english[lang]). A total of 90 clinical trials met the inclusion criteria and were selected for in-depth review and analysis.
Inclusion and Exclusion Criteria
Only interventional studies were included in this analysis, as defined by the applied PubMed filters (Clinical Trial, Clinical Trial Protocol, Phase I–IV trials, and Randomized Controlled Trials). Observational studies were excluded to maintain consistency in evaluating therapeutic and diagnostic interventions. No external clinical trial registries (e.g., ClinicalTrials.gov, EU Clinical Trials Register) were consulted in this study; all included records were indexed in PubMed and published in peer-reviewed journals. The selection was limited to English-language articles that reported original clinical trial data involving human participants.
Data Extraction and Categorization
Each clinical trial was reviewed to extract key data, including the disease studied, exosome source (e.g., plasma, urine, stem cells), application type (diagnostic, therapeutic, or drug delivery), intervention details, trial phase, and sample size. Clinical outcomes such as efficacy, safety, biomarkers, and therapeutic responses were also recorded to assess the role of exosomes across various conditions.
Bibliometric Analysis
To investigate research trends and thematic structures within clinical exosome research, a bibliometric analysis was performed using VOSviewer (version 1.6.20). 24 Bibliographic data were exported from PubMed in RIS format and uploaded into the software for analysis. Terms were extracted from the titles and abstracts using the full counting method, with a minimum occurrence threshold applied based on relevance criteria (e.g., appearing at least 10 times). Several visualizations were generated to support the analysis, including a Term Co-occurrence Density Map to highlight frequently used keywords, a Conceptual Mapping diagram to identify clusters of thematically related terms, and an Overlay Visualization to illustrate the emergence and evolution of recent research topics over time. This integrated approach enabled both qualitative insight and quantitative mapping of the clinical research landscape surrounding exosomes.
Results and Discussion
Trajectory
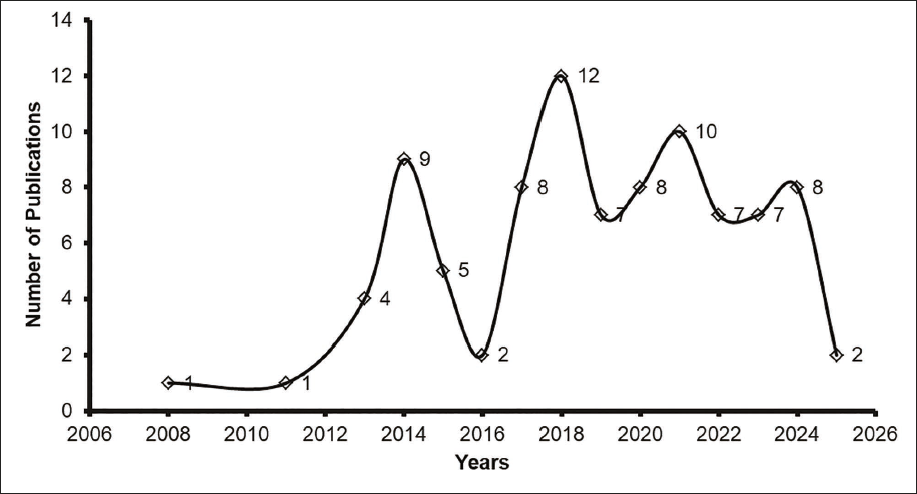
Clinical research on exosomes has progressed significantly over the past decade, as illustrated in Figure 2. The figure depicts the number of clinical exosome-related publications per year from 2008 to 2025. Initial research between 2011 and 2014 focused on biomarker discovery and exosome profiling. From 2015 onward, publications increased, reflecting the growing use of exosomes in oncology, neurology, nephrology, and infectious disease. A major surge is seen in 2018, with continued high output through 2024. The drop in 2025 may be due to incomplete data. The overall trend highlights a shift from foundational research to translational and clinical applications such as regenerative medicine, immunotherapy, and diagnostics. This finding aligns with prior reports indicating an exponential surge in research focusing on the biology and clinical properties of exosomes.14, 15
Trends in Clinical Research on Exosomes (2008–2025). The figure Shows the Annual Number of Clinical Studies on Exosomes From 2008 to 2025. Each Point Marks the Number of Publications per Year, With a Peak in 2018 (12 Studies) and Steady Output From 2020 to 2024. The Drop in 2025 May Reflect Incomplete Data. The Trend Highlights Rising Clinical Interest in Exosome Applications.
Descriptive Summary (n = 90 Trials)
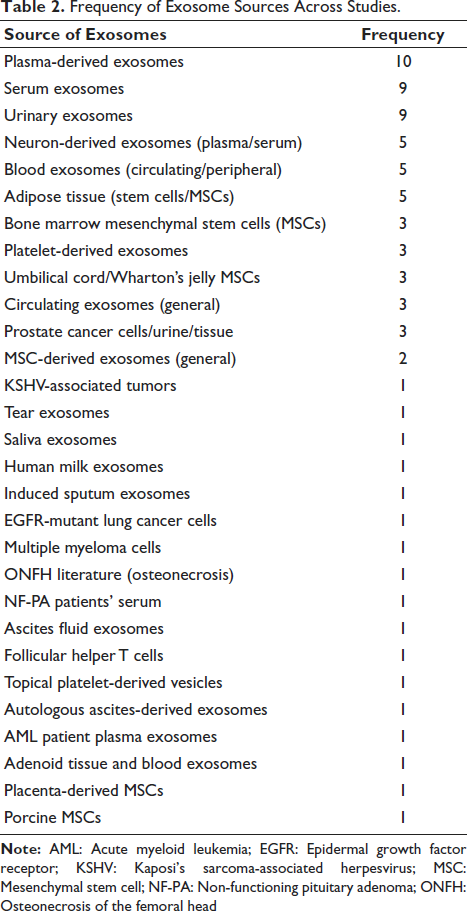
This analysis of 90 clinical trials highlights the diverse clinical applications of exosomes. Commonly sourced from plasma, serum, and urine, exosomes were mainly used as diagnostic and prognostic biomarkers, especially in cancers like breast, prostate, and liver. Non-oncology uses included nephrology, infectious diseases, and endocrine disorders. About 21% of trials explored therapeutic roles, particularly stem cell-derived exosomes for coronavirus disease 2019 (COVID-19), neurodegenerative diseases, and drug delivery. Around 7% focused on treatment monitoring, with some studying how physiological or pharmacological factors influence exosomes. Overall, the findings underscore exosomes’ promise in diagnostics, therapy, and precision medicine, though standardization and larger trials are still needed.
Exosomes are released by all cells and present in all bodily fluids, making them suitable for non-invasive liquid biopsies. Their molecular cargo allows for detailed diagnostic evaluation, with surface proteins aiding immunological capture. Exosomes have shown diagnostic utility in cardiovascular diseases, 25 CNS disorders, 21 and cancer.15, 26, 27 This initiative is swiftly extending to additional disorders affecting the liver,28–31 the kidney,32, 33 and the lung.34–37
Therapeutic Applications
Several clinical trials (Table 2) have investigated mesenchymal stem cell (MSC)-derived exosomes in COVID-19,1, 34, 38–40 showing safety and efficacy. Chu et al. used nebulized umbilical cord mesenchymal stem cell (UC-MSC) exosomes in seven patients, showing no adverse events and reduced lesion burden. 34 Zhu et al. tested aerosolized adipose MSC exosomes in a Phase 2a trial, showing immune modulation and lung repair. 40 Sengupta et al. reported improved oxygenation and reduced inflammatory markers in 24 acute respiratory distress syndrome (ARDS) patients treated with ExoFlo™, achieving 83% survival. 39 Zamanian et al. found reduced mortality (19% vs. 54%) in a randomized controlled trial using human placental mesenchymal stromal cell-derived small extracellular vesicles (hPMSC-sEVs) 1 (Table 3). Salomon-Zimri et al. applied exosomes in amyotrophic lateral sclerosis (ALS) patients to monitor biomarkers like TAR DNA-binding protein 43 (TDP-43) and microtubule-associated protein 1 light chain 3 (LC3), underscoring diagnostic and therapeutic roles. 38 MSC-derived exosomes in COVID-19 show immunomodulatory effects by reducing inflammation (C-reactive protein (CRP), ferritin, D-dimer), reversing lymphopenia (cluster of differentiation 3 positive (CD3+), cluster of differentiation 4 positive (CD4+), cluster of differentiation 8 positive (CD8+)), improving oxygenation (192% increase in partial pressure of oxygen/fraction of inspired oxygen (PaO2/FiO2)), and supporting tissue repair through bioactive molecule delivery. Delivery methods include nebulization and intravenous infusion, with consistent safety.
Frequency of Exosome Sources Across Studies.
Therapeutic Applications.
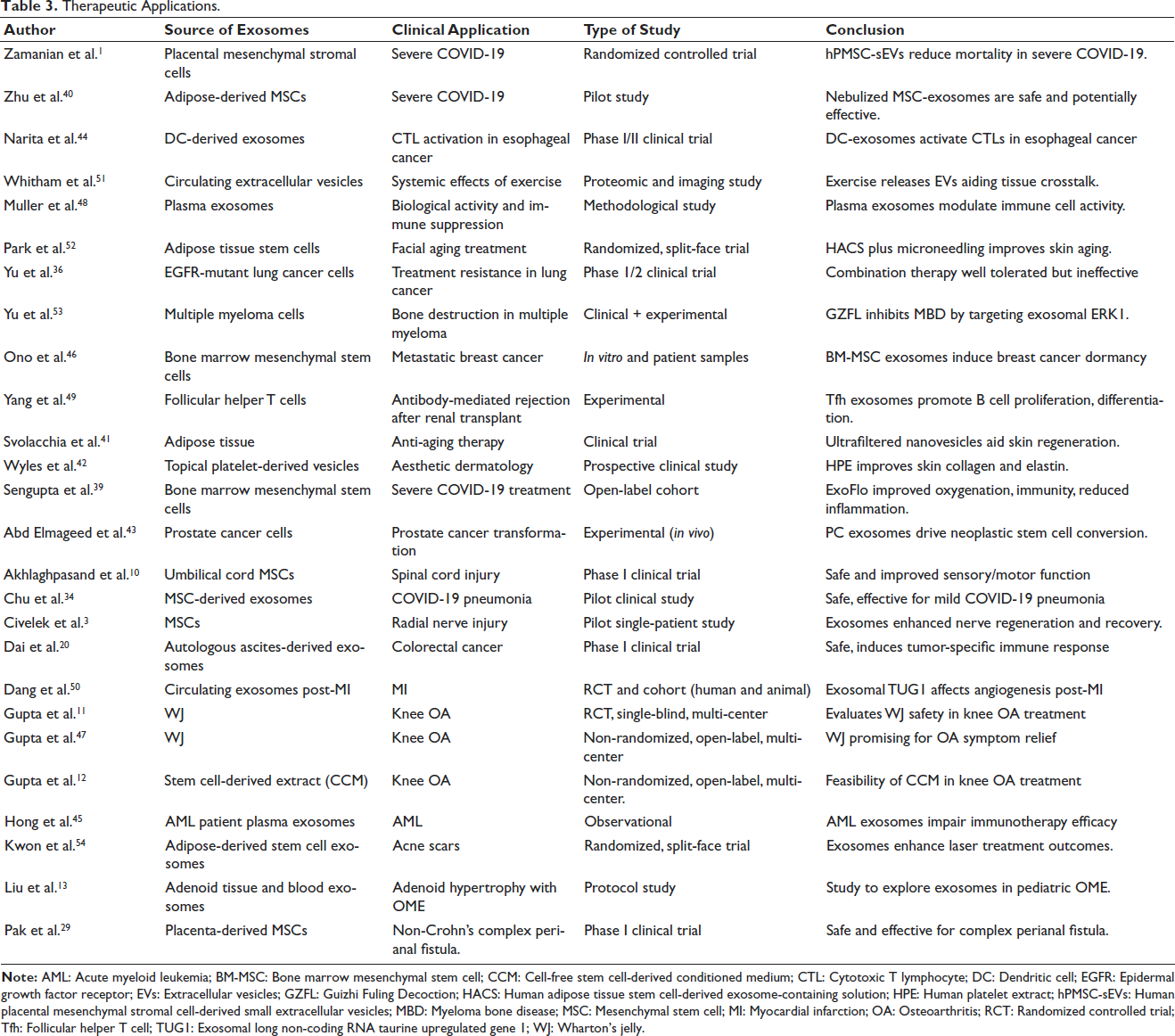
In skin regeneration, Pak et al. used placenta-derived exosomes for perianal fistulas with significant improvement and no adverse effects. Park et al. showed improved hydration, elasticity, and pigmentation using human adipose tissue stem cell-derived exosome-containing solution (HACS) with microneedling. 29 Svolacchia et al. demonstrated dermal regeneration using filtered nanofat vesicles. 41 Wyles et al. found increased collagen and elastin from topical human platelet extract (HPE). These studies confirm exosome safety and efficacy in dermatology. 42 Exosomes in cancer therapy include immunomodulation, monitoring drug resistance, and reducing tumor progression. Exosomes reprogram cells, 43 induce cytotoxic T lymphocyte (CTL) responses, 44 suppress NK cells, 45 promote dormancy, 46 and track therapy response. 36 Guizhi Fuling Decoction (GZFL) exosomes reduce osteoclast activity in myeloma. 36 Gupta et al. explored umbilical cord Wharton’s jelly (UC-WJ) exosome therapy in knee osteoarthritis (OA). A randomized trial (n = 168) compared UC-WJ to hyaluronic acid and saline. A non-randomized open-label study (n = 12) used clinical/imaging assessments. Another study used cell-free stem cell-derived cell-free stem cell-derived conditioned medium (CCM) in a 24-month follow-up. These trials show promise for minimally invasive OA therapy.11, 12, 47
Exosomes’ immunomodulatory roles were shown by Müller et al., who isolated plasma exosomes that downregulated cluster of differentiation 69 (CD69) on CD4+ T cells, indicating immune suppression in cancer. Yang et al. demonstrated that follicular helper T cell (Tfh)-derived exosomes from antibody-mediated rejection (AMR) patients expanded B and plasma cells, implicating them in graft rejection.48, 49 In tissue injury, Akhlaghpasand et al. showed that intrathecal human umbilical cord mesenchymal stem cell (HUC-MSC) exosomes improved neurological function, bowel control, and autonomy in spinal cord injury. 10 Civelek et al. reported sensory and motor recovery in a patient with radial nerve injury treated with MSC exosomes. These trials support exosomes’ regenerative potential. 3 Dang et al. showed that exosomal long non-coding RNA taurine upregulated gene 1 (exo-LncRNA TUG1) from MI patients inhibits angiogenesis via hypoxia-inducible factor 1-alpha/vascular endothelial growth factor-alpha (HIF-1α/VEGF-α) suppression, reversible by remote ischemic conditioning (RIC). Exosomal TUG1 reduction restored endothelial function, positioning it as a target for MI recovery, especially post-percutaneous coronary intervention (PCI). 50
Applications of Exosomes as Biomarkers
Table 4 summarizes clinical studies using exosomes as biomarkers across diverse diseases. Exosomal microRNAs (miRNAs), proteins, and other molecules are widely studied for diagnostic, prognostic, and monitoring purposes. Neurodegenerative disorders, particularly Alzheimer’s disease and mild cognitive impairment (MCI), were frequently assessed using neuronal-derived plasma exosomes, examining markers like insulin signaling and synaptic proteins.21, 55–57 Parkinson’s disease was evaluated using protein kinase B/mammalian target of rapamycin (Akt/mTOR) markers. 16
Applications of Exosomes as Biomarkers.
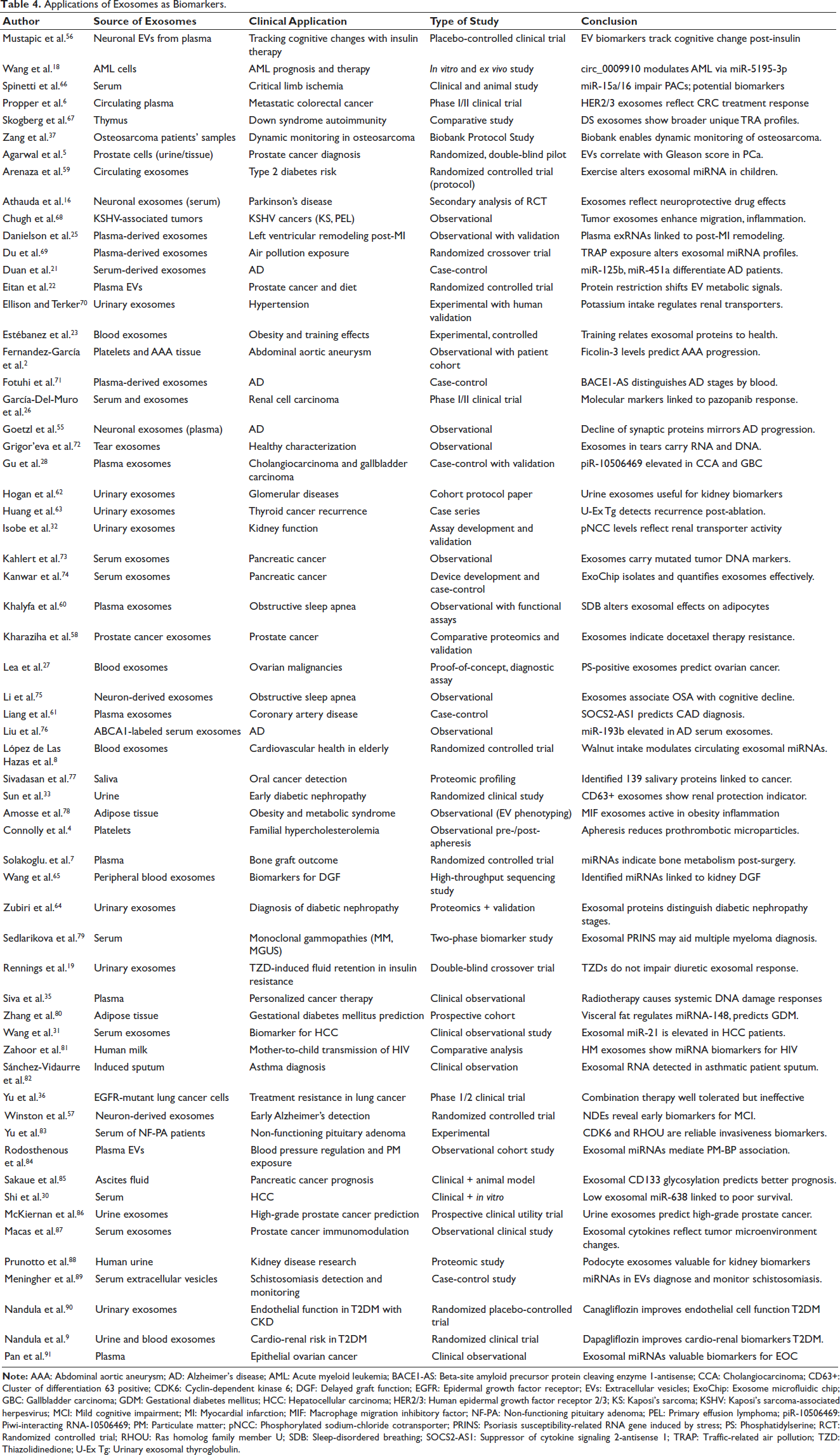
Cancer applications included detecting exosomal miRNAs, proteins, and mutations in prostate, pancreatic, ovarian, hepatocellular, renal, and pituitary cancers. Pan et al. studied exosomal miRNAs in ovarian cancer; Kahlert et al. and Kanwar et al. identified KRAS and tumor protein 53 (p53) mutations in pancreatic cancer. Resistance markers such as cluster of differentiation 133 (CD133) and docetaxel-resistance proteins were studied in prostate and colorectal cancers.6, 58
Exosomal RNA and protein markers were explored in inflammatory and infectious diseases like schistosomiasis, asthma, and maternal HIV via serum, sputum, and milk samples.59–61 Novel uses included autoimmune profiling in Down syndrome and osteosarcoma biobanking.62–65 Overall, Table 3 underscores exosomes’ biomarker versatility and potential in personalized medicine.
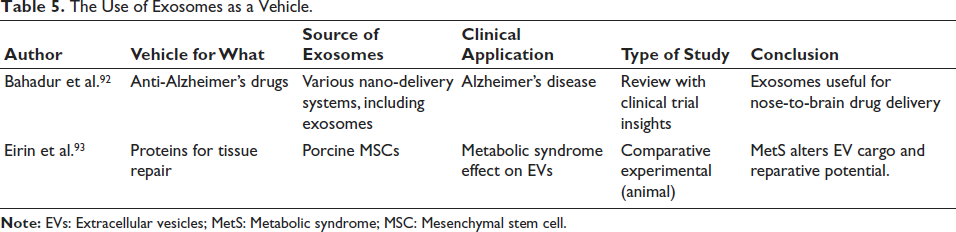
The Use of Exosomes as a Vehicle
The therapeutic potential of exosomes as delivery vehicles is highlighted in two key studies. Bahadur et al. identified exosomes as effective nanocarriers for intranasal delivery of anti-Alzheimer’s drugs, noting their biocompatibility, ability to cross the blood–brain barrier, and enhanced bioavailability over conventional systems. 92 Eirin et al. analyzed MSC-derived exosomes from pigs with and without metabolic syndrome, revealing that while healthy MSC exosomes support cell repair, metabolic syndrome (MetS)-derived exosomes carry pro-inflammatory proteins that may reduce regenerative efficacy. This underscores the impact of donor health on exosome function. 93 Both studies support exosomes as smart delivery platforms across neurological and metabolic diseases (Table 5).
The Use of Exosomes as a Vehicle.
Bibliometric and Visualization Study
Most Frequent Terms
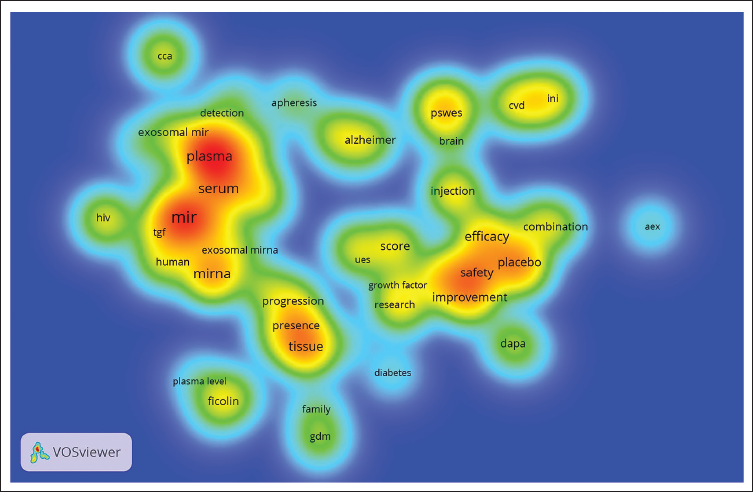
A bibliometric analysis of 3,693 terms from clinical trials on exosomes revealed key research focuses. Frequent terms like “mir,” “mirna,” “plasma,” and “serum” reflect a strong emphasis on exosomal miRNAs as diagnostic biomarkers. Terms such as “efficacy,” “placebo,” and “safety” indicate a clinical focus on therapeutic evaluation. Disease-specific mentions like “Alzheimer” and “COVID” highlight growing interest in neurodegenerative and infectious conditions. Heatmaps show dense clusters around plasma/serum studies and therapeutic applications, underscoring the translational potential of exosome-based diagnostics and treatments.
Figure 3 shows a VOSviewer density map of terms from clinical trial abstracts on exosomes. Warmer colors (red/yellow) indicate more frequent terms, such as mir, plasma, serum, and mirna, highlighting biomarker-related research. Medium-frequency terms like efficacy, safety, and placebo cluster in yellow-green areas, reflecting clinical focus. Blue areas show emerging or less frequent topics. This map reveals key research themes and evolving trends in exosome-related clinical trials.
Term Density in Exosome Clinical Trials.
Conceptual Mapping
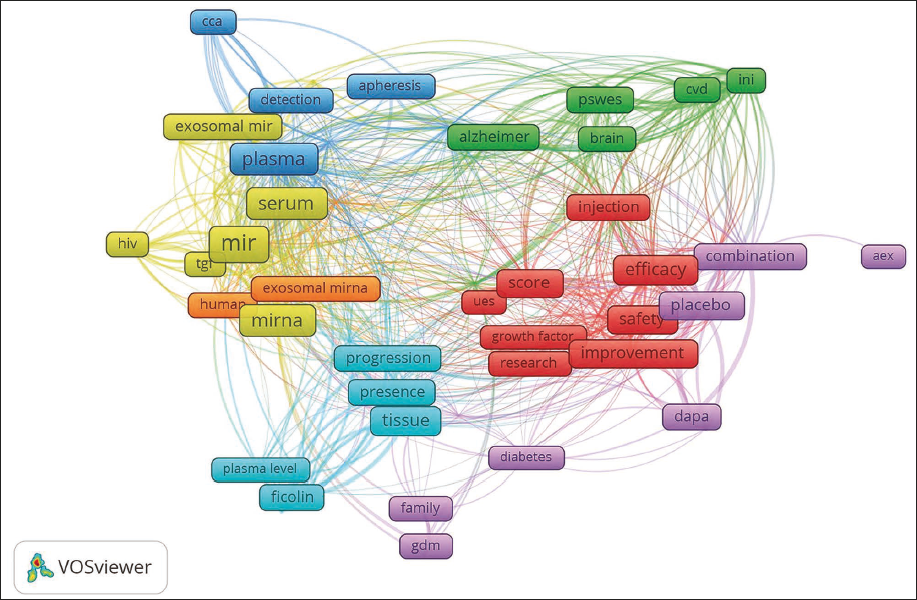
Figure 4 illustrates seven research clusters in exosome clinical trials. The yellow and orange clusters focus on biomarkers and RNA-based cargo. The red cluster centers on therapeutic outcomes (efficacy, safety, placebo). Green highlights neurological studies (e.g., Alzheimer’s), while blue relates to biofluid sources and isolation methods. Light blue addresses disease monitoring, and purple explores emerging therapies like diabetes and drug combinations. These clusters reflect the diverse and evolving clinical applications of exosomes.
Conceptual Mapping. The Figure Shows the Conceptual Mapping of Exosome-based Clinical Trials Clustered into Seven Thematic Groups. The Red Cluster Focuses on Clinical Outcomes like Efficacy, Safety, and Placebo. The Green Cluster Highlights Disease-specific Terms Such as Alzheimer and Cardiovascular Disease (CVD). The Blue Cluster Represents Sample Sources and Methods like plasma, serum, and detection. The Yellow Cluster Centers on Molecular Markers such as miR and miRNA. The Orange Cluster Relates to Exosomal miRNA and Human Studies. The Purple Cluster Involves Metabolic Conditions like Diabetes and Gestational Diabetes Mellitus (GDM), While the Cyan Cluster Includes Terms like Progression and Ficolin, Reflecting Immune and Monitoring Studies.
Emerging Topics
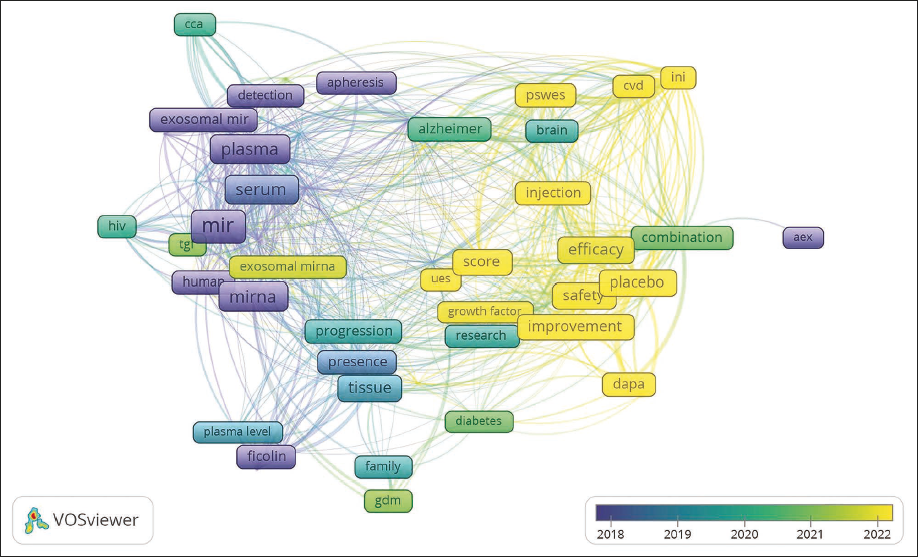
Figure 5 presents the overlay visualization of term occurrences over time, highlighting emerging topics in yellow. These recent and trending terms include efficacy, placebo, safety, improvement, combination, injection, dapa, and score, suggesting a strong recent research focus on evaluating clinical outcomes, therapeutic strategies, and treatment validation in exosome-based interventions. In contrast, earlier studies, represented in dark blue, concentrated on terms such as plasma, serum, mir, and mirna, reflecting an earlier emphasis on biomarker discovery and molecular profiling. The transition toward yellow nodes indicates a shift from foundational research to applied clinical investigations and therapeutic evaluations in the field of exosome-based clinical trials.
Overlay Visualization of Terms Extracted From Exosome-related Clinical Trial Abstracts and Titles. Terms are Color-coded Based on Their Average Publication Year, Ranging From Earlier Studies in Dark Blue (2018) to More Recent Topics in Yellow (2022). Emerging Areas Include Efficacy, Safety, Placebo, Combination, and Injection, Indicating a Growing Focus on Clinical Translation and Therapeutic Evaluation. In Contrast, Earlier Research Emphasized miR, Plasma, Serum, and miRNA, Reflecting Foundational Biomarker and Sample Analysis. This Shift Highlights the Evolution From Basic Molecular Investigations to Clinical Outcome-driven Research.
Conclusion
This systematic review, encompassing 90 clinical trials, underscores the growing clinical relevance of exosomes in diagnostics, therapeutics, and drug delivery across diverse diseases. Most studies focused on exosomal miRNAs, long non-coding RNAs (lncRNAs), and proteins as potent biomarkers for conditions such as Alzheimer’s disease, acute myeloid leukemia (AML), hepatocellular carcinoma (HCC), pancreatic cancer, and prostate cancer. Exosomes were primarily derived from plasma and serum, but also from urine, ascites, sputum, tears, breast milk, saliva, and adipose- or placenta-derived stem cells. They served as biomarkers, treatment agents, and delivery vehicles, especially in trials targeting neurodegenerative diseases, COVID-19, cancer, knee OA, spinal cord and nerve injuries, and transplant rejection. Stem cell-derived exosomes and exosome-based formulations (e.g., nebulized or injected) demonstrated promising regenerative, immunomodulatory, and anti-inflammatory effects with strong safety profiles. Notably, Alzheimer’s, COVID-19, and skin rejuvenation emerged as hot application areas, while miRNAs, placebo-controlled trials, and efficacy assessments dominated recent keywords (Figure 2). Conceptual and temporal mapping revealed evolving research from molecular profiling toward clinical efficacy, treatment combinations, and precision delivery systems (Figures 2–4). However, significant gaps remain—standardization in exosome isolation, characterization, and dosage, as well as the lack of large-scale randomized controlled trials (RCTs) and long-term outcome data, continue to limit clinical adoption. Future directions should focus on harmonized protocols, multicenter validation studies, and integration of artificial intelligence (AI) and omics tools to advance exosome-based strategies from promising research to clinical application.
Footnotes
Abbreviations
AAA: Abdominal aortic aneurysm; Akt/mTOR: Protein kinase B/mammalian target of rapamycin; ALIX: ALG-2 interacting protein X; AML: Acute myeloid leukemia; AMR: Antibody-mediated rejection; ARDS: Acute respiratory distress syndrome; BACE1-AS: Beta-site amyloid precursor protein cleaving enzyme 1-antisense; BM-MSC: Bone marrow mesenchymal stem cell; CCA: Cholangiocarcinoma; CCM: Cell-free stem cell-derived conditioned medium; CD133: Cluster of differentiation 133; CD4+: Cluster of differentiation 4 positive; CD69: Cluster of differentiation 69; CDK6: Cyclin-dependent kinase 6; COVID-19: Coronavirus disease 2019; CRP: C-reactive protein; CTL: Cytotoxic T lymphocyte; CVD: Cardiovascular disease; DC: Dendritic cell; DGF: Delayed graft function; EVs: Extracellular vesicles; exo-LncRNA TUG1: Exosomal long non-coding RNA taurine upregulated gene 1; ExoChip: Exosome microfluidic chip; GBC: Gallbladder carcinoma; GDM: Gestational diabetes mellitus; GZFL: Guizhi Fuling Decoction; HACS: Human adipose tissue stem cell-derived exosome-containing solution; HCC: Hepatocellular carcinoma; HER2/3: Human epidermal growth factor receptor 2/3; HIF-1α: Hypoxia-inducible factor 1-alpha; hPMSC-sEVs: Human placental mesenchymal stromal cell-derived small extracellular vesicles; HPE: Human platelet extract; HSP70: Heat shock protein 70; HSP90: Heat shock protein 90; HUC-MSC: Human umbilical cord mesenchymal stem cell; KS: Kaposi’s sarcoma; KSHV: Kaposi’s sarcoma-associated herpesvirus; LC3: Microtubule-associated protein 1 light chain 3; lncRNA: Long non-coding RNA; MBD: Myeloma bone disease; MCI: Mild cognitive impairment; MetS: Metabolic syndrome; MIF: Macrophage migration inhibitory factor; miRNA: MicroRNA; MSC: Mesenchymal stem cell; MVBs: Multivesicular bodies; NF-PA: Non-functioning pituitary adenoma; ONFH: Osteonecrosis of the femoral head; p53: Tumor protein 53; PaO2/FiO2: Partial pressure of oxygen/fraction of inspired oxygen; PCI: Percutaneous coronary intervention; PEL: Primary effusion lymphoma; piR-10506469: Piwi-interacting RNA-10506469; PM: Particulate matter; pNCC: Phosphorylated sodium-chloride cotransporter; PRINS: Psoriasis susceptibility-related RNA gene induced by stress; PS: Phosphatidylserine; RCT: Randomized controlled trial; RHOU: Ras homolog family member U; RIC: Remote ischemic conditioning; SDB: Sleep-disordered breathing; SOCS2-AS1: Suppressor of cytokine signaling 2-antisense 1; TDP-43: TAR DNA-binding protein 43; Tfh: Follicular helper T cell; TRAP: Traffic-related air pollution; TSG101: Tumor susceptibility gene 101; TZD: Thiazolidinedione; U-Ex Tg: Urinary exosomal thyroglobulin; UC-WJ: Umbilical cord Wharton’s jelly; VEGF-α: Vascular endothelial growth factor-alpha.
Authors’ Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation, or in all these areas, took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study does not require any ethical approval.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research article is derived from a research grant funded by the Research, Development, and Innovation Authority (RDIA), Kingdom of Saudi Arabia, with grant number (12899-jazzan-2023-JZU-R-2-1-HW). The authors also gratefully acknowledge the funding of the Deanship of Graduate Studies and Scientific Research, Jazan University, Saudi Arabia, through Project Number: (JU-20250296-DGSSR-RP-2025).
Informed Consent
The participant has consented to the submission of the article to the journal.
