Abstract
Background
Ubrogepant is a first-generation gepant-class drug approved for the oral treatment of migraine.
Objectives
This work aimed at developing an intranasal gel consisting of ubrogepant nanoparticles (NPs), with the aim of improving its therapeutic efficacy through rapid and longer action in the brain.
Materials and Methods
The ubrogepant NPs were prepared using poly(lactic-co-glycolic) acid (PLGA), polyvinyl alcohol (PVA), and polysorbate 80. These NPs were then formulated into a thermo-reversible gel by using an optimized polymer, stabilizer, and process. The gel formulation was then tested by in vitro and ex vivo assays for drug release and permeation, and the pharmacodynamics and pharmacokinetics were evaluated in Sprague–Dawley rats.
Results
The PLGA NPs were optimally formulated in a mucoadhesive gel, which caused significant improvement in permeability and drug retention in the nasal mucosa. The in vivo studies indicated higher concentrations of ubrogepant in the brain, with a rapid absorption compared to the intranasal NP or oral ubrogepant delivery. The rapid absorption and sustained levels in the brain were translated into significantly improved efficacy of the gel formulation in the nitroglycerin (NTG)-induced migraine model in rats.
Conclusion
The optimized gel formulation of the ubrogepant NPs validated the potential of intranasal use of ubrogepant for the treatment of migraine.
Introduction
Migraine, a chronic disease of neurological and episodic occurrence, is characterized by intense, unilateral, and oscillating headaches that may last from hours to days. This headache is accompanied by vomiting or nauseating symptoms, sensitivity and irritation toward light and sound, tingling sensations, and other sensory symptoms. 1 These symptoms of migraine are highly variable. 2 Migraine treatment armamentarium consists of serotonin receptor 1B/1D agonists, which are called triptans, 3 which are associated with severe vasoconstriction and are contraindicated for hypertensive patients with cardiac morbidities. 4 A new class of migraine drugs, called gepants, is devoid of the side-effect profile of triptans, with significant therapeutic efficiency. 5 Gepants are blockers of the calcitonin gene-related peptide (CGRP), a neuropeptide 6 that causes vasodilation in the cranial region of the brain and also stimulates inflammation in neurons. These effects precipitate the intense pain in migraine. CGRP is abundantly found in the trigeminovascular system, which is primarily the locus of the origin of migraine pain. 7 Small-molecule antagonists of CGRP have demonstrated compelling efficacy in migraine treatment. 5 Ubrogepant is the first drug in this class that was approved for the treatment of migraine. 8 A rapid and intense reduction in unilateral headache is the major need of migraine therapy. This need could be achieved using intranasal formulations that help in increasing the effectiveness of a drug. 9 Also, nasal delivery can be helpful in patients who are experiencing nausea and can ensure efficient brain exposure of the drug with direct targeting of the trigeminovascular system. 10 Many migraine drugs like sumatriptan and zavegepant have been successful in establishing rapid and safe pain relief in migraine patients.11, 12
Ubrogepant is a hydrophobic compound. Despite being a potent inhibitor of CGRP pathways, its pharmacokinetic availability to the brain (and the trigeminovascular system) is limited since it is a P-glycoprotein (P-gp) substrate. 13 Previously, we observed that nanoparticles (NPs) of ubrogepant improve the entry of the drug into the brain and also protect against degradation (article in press, Journal of Pharmacology and Pharmacotherapeutics). However, in humans, the plain NPs have the possibility of draining through the nasal cavity due to mucosal clearance,14, 15 which can be avoided by incorporating the drug NPs into a mucoadhesive in situ gel. 16 We believed that the formulation and delivery of ubrogepant NPs by the intranasal route would be useful in enhancing the effect of the drug. In this work, we prepared, optimized, and evaluated the intranasal gel formulation of ubrogepant using NPs and tested it for the in vivo effect.
Materials and Methods
Materials
Ubrogepant was obtained from Metrochem API Limited, Hyderabad. Poly(lactic-co-glycolic) acid or PLGA [lactide:glycolide (50:50), mol. weight 30,000–60,000] and polyvinyl alcohol (PVA), and dialysis membrane (MWCO: 12–14 kDa) were procured from Sigma (MO, USA). All other chemicals and solvents were of analytical grade. The stabilizers Poloxamer 407, Poloxamer 188, hydroxypropyl methylcellulose, and polysorbate 80 were procured from Sigma–Aldrich, USA. All other chemicals used were of analytical grade.
Ubrogepant-PLGA NPs were prepared using the w/o/w multiple emulsion solvent evaporation method. 17 The organic phase was made by dissolving ubrogepant and PLGA in dichloromethane (1 mL), which was then mixed with the aqueous phase containing 1% PVA and 1% polysorbate 80 in purified water by very slowly adding the organic phase to the aqueous phase with constant stirring at 150 rpm for 5 h. The solvent evaporated, and the nanosuspension was purified by centrifugation at 10,000 rpm for 30 min at 40°C to get the final NPs. These pellets were washed with water to remove untrapped drug, and the pellets were redispersed in water. The NPs, thus made, were lyophilized, and their size was determined using a zeta potential analyzer (Malvern), at the scattering angle of 90° at a temperature of 25°C. The size was also confirmed using scanning electron microscopy (SEM) (Zeiss, Oberkochen, Germany). The entrapped compound was measured by high-pressure liquid chromatography (HPLC) (Agilent 1100, CA, USA) using an already-reported method. 18 A solution of the drug formulation made in dichloromethane was filtered (pore size 0.22 µm, Millipore, Darmstadt, Germany) before analysis. For stability, the formulation was stored in a tightly closed vial at two different conditions: the first was at 4°C and controlled humidity of 75% relative humidity (RH), and the second at 25°C ± 2°C and controlled humidity of 75% RH. After 3 months, samples were recovered, and the stability study was assessed through EE%, particle size, and zeta potential, with visual examination for contamination, sedimentation, or color change. 18
Formulation of the Gel
The gel was prepared using the cold technique mentioned earlier.19, 20 The ubrogepant NPs (10 mg/mL) were dissolved in Polaxomer 407 (10%, w/v) and 2-hydroxypropyl-β-cyclodextrin (5%) in water and stored at 4°C. To this solution, Carbopol 934P (0.1%, w/v) solution in water was added with continuous stirring at 4°C and was stored for 24 h.
Characterization of the Gel
The gel formulation of ubrogepant NPs was assessed for phase uniformity, general morphology, gelling time, temperature for gelling, and viscosity. 21 The gelation of this formulation was evaluated using a reported method. 22 1 mL of the formulation was packed in test tubes in the water bath at 4°C. The temperature was raised in 2°C increments up to 25°C and then in 0.5°C increments for the gel-sol transition. The gelation was the temperature at which the movement was not observed after tilting by 90°. The viscosity of the gel was measured using a Brookfield viscometer (Brookfield DV III Ultra D220, Middleborough, MA, USA). 21 The drug (in the prepared gel) was mixed with dichloromethane (10 mL) in a 15 mL centrifuge tube and was vortexed to dissolve the drug inside the polymeric particles. The solution was further diluted with the same solvent, centrifuged, and filtered using a 0.2 µm filter. The drug concentration in the filtrate was measured using liquid chromatography–mass spectrometry (LC–MS). 18
The drug release from the NP of ubrogepant was tested using the dialysis method. The release of ubrogepant was performed using conventional (plain drug in gel matrix), gel, and NPs from the PLGA-NP at 37°C ± 1°C by the dialysis method. The dialysis sacs were filled with 1 mL of ubrogepant formulation and were immersed in 30 mL of pH 7.4 phosphate-buffered saline (PBS) solution. Sink conditions were maintained, and the system was continuously vortexed at 70 rpm. Samples (0.5 mL) were collected at hourly intervals and analyzed with the LC–MS method. 18
Ex vivo permeation was performed using Franz diffusion cells and a nasal mucosa obtained from slaughtered goat nasal cavities. 15 The mucosal layer (properly excised without damage) was fixed to the receptor and the donor compartment. The active surface area of the membrane was 1.256 cm2. The amount of gel equivalent to 10 mg of the drug was placed over the membrane. The temperature was kept at 37°C under continuous mixing at 100 rpm. The aliquots were taken at scheduled intervals for 24 h, and an equal amount was replenished with PBS (pH 6.4). The aliquot was analyzed to assess the drug concentration permeated through the mucosal layer. 18
In Vivo Studies
The in vivo studies were carried out in male Sprague–Dawley rats to determine the pharmacokinetics and brain uptake of ubrogepant via oral and nasal routes. Male Sprague–Dawley rats (200–225 g) were group housed (n = 3). The Institutional Animal Ethical Committee approved the animal study protocol, constituted under the Committee for Control and Supervision of Experiments on Animals (CCSEA), as per protocol no. DMIHER/IAEC/24-25/10. The animals were maintained at a controlled temperature of 25°C ± 2°C and humidity, with free access to chow, food, and water.
Pharmacokinetic Study
Rats were randomized into four groups (n = 6, with sparse sampling), which were assigned to treatments (with ubrogepant concentration of 2 mg/kg in formulation) as follows: (a) placebo control with intranasal gel vehicle, (b) ubrogepant NP suspension is administered by the oral route, (c) ubrogepant intranasal gel formulation is administered by the intranasal route, and (d) ubrogepant NP solution is administered by the intranasal route. Brain samples were weighed and homogenized in PBS at pH 7.4. The brain homogenates (200 µL) and serum plasma samples were used for the assessment of ubrogepant concentration by the LC–MS method.18, 23 The brain and blood were sampled at 0.5, 1, 2, 4, 6, 8, and 24 h. The pharmacokinetic parameters were calculated using WinNonlin software 8.4.
Pharmacodynamic Study
Nitroglycerin (NTG) (in propylene glycol, Sigma) was used to prepare the formulation for subcutaneous injection in 0.9% NaCl and administered at a dose of 10 mg/kg; the vehicle contained similar solvents, except for NTG. Ubrogepant treatment was given after 5 min of NTG treatment, as the following groups: (a) placebo control with intranasal gel vehicle, (b) ubrogepant NP suspension was administered by oral route (containing 2 mg/kg of ubrogepant), (c) ubrogepant intranasal gel formulation administered by intranasal route (containing 2 mg/kg ubrogepant), and (d) ubrogepant NP solution was administered using the intranasal route (containing 2 mg/kg of ubrogepant). These groups underwent the open-field test (10 min duration), and immediately after the open-field test, they were injected with formalin (50 µL, subcutaneously) in the upper lip to perform the orofacial formalin test (45 min duration). 24 The intranasal administration was done with a polyethylene tubing attached to an injector. 25 The volume administered was 50 mL in each nostril, and the oral formulation was made using polyethylene glycol (PEG) in water, and the NPs were dissolved in PBS for administration.
The effect of treatment on locomotion of rats was assessed by an infrared actimeter (Orchid Scientific, India). After acclimatization, the activity was measured for 5 min. In addition to the locomotor activity, the time spent in the center of the area was measured. These parameters were measured after 1 h of NTG administration. 26 For the orofacial formalin test, the rats were injected with 50 µL of formalin 1.5% (v/v) into the right upper lip. Face rubbing was evaluated by a researcher blind to treatments, counting the seconds the animal spent grooming the injected area with the ipsilateral forepaw or hind paw 0–3 min (Phase I) and 12–45 min (Phase II) after formalin injection. After that, the animals were sacrificed, and their blood, brain, and brainstem tissues were collected. The brainstem was carefully separated to dissect the trigeminal nucleus caudalis (TNC). 27 The tissue was rinsed in a sterile 0.9% NaCl cold solution, placed in cryogenic tubes, and kept at –80°C. For the gene expression processing of TNF-alpha and IL-6 gene expression, ribonucleic acid (RNA) extraction was done, and all the samples were checked for absorbance ratios (260/280 nm) for protein contamination. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used for normalization. The primers used were as previously reported. 24
Statistical Analysis
All the results were given as Mean ± Standard error of mean (SEM). All results were compared using the two-way analysis of variance (ANOVA), Student’s t-test, or a suitable comparative test as indicated in the figures. Differences are considered significant at a level of p < .05.
Results
In the present study, ubrogepant NPs were prepared by oil-in-water emulsification and solvent evaporation. The most optimized NP form had the effect of process parameters used in this emulsification solvent evaporation method on particle size, zeta potential, entrapment efficiency (EE), and polydispersity index (PDI) were investigated. Table 1 shows the details of these NPs, where the ratio of the stabilizer PVA and polysorbate 80 (both 1%) was used in an equal ratio. A particle size of NPs below 200 nm was achieved with this method. The analytical method used contained the concentration range of 1–500 µg/mL for the EE of ubrogepant.18, 23 The stability data (Table 1) indicate no degradation and change in size or other parameters in the formulation, indicating long-term stability. The negative zeta potential indicates the negative charges due to PLGA, PVA, and polysorbate 80, which may help to keep the NPs separated from each other in formulation. 28 It was also observed that though the particles tend to aggregate, probe sonication could immediately resuspend the NPs. The particle size distribution indicates spherical to oval-like nanoshapes, with a diameter lower than 200 nm (Figure 1A).
Characteristics of Nanoparticle Gel Formulation of Ubrogepant.

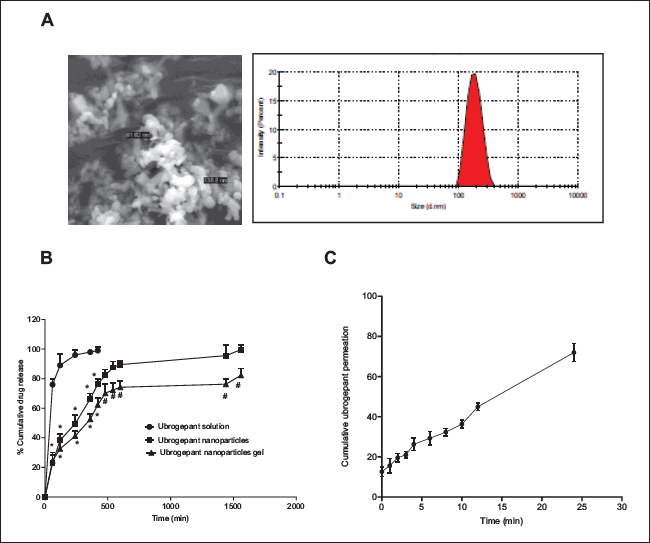
The dialysis method is used to assess the drug release from the NP formulation. 29 It is observed that the NPs are not released through the dialysis membrane, but the released drug is transported to the receiver compartment. Figure 1B shows the release pattern, indicating that the NPs release the drug in a sustained manner. Since the NPs retained the drug, the diffusion was slow, and it was slower in the gel formulation, indicating that the drug is properly maintained in the formulation.
The pharmacokinetic behavior of ubrogepant NPs and the gel formulation was assessed along with the ubrogepant drug solution using PEG, with doses that contained a similar amount (2 mg/kg) of ubrogepant active compound, and the brain and blood levels of ubrogepant were analyzed. The pharmacokinetic parameters were calculated using WinNonlin software as depicted in Table 2. As compared to the oral solution, the Cmax for the intranasal administration of the NPs was significantly higher (436.3 vs. 1,149.2 ng/mL), and the gel formulation of the NPs administered by the intranasal route showed highest Cmax (1,672.1 ng/mL), showing an improved systemic availability of the nasal NP gel over the simple NP solution by the intranasal route or the simple compound solution by the oral route. The drug elimination was relatively fast for the intranasal route for NPs, but the gel formulation of NPs showed significant improvement in retention of the drug, with the highest half-life (5.1 h for blood). The intranasal gel formulation demonstrated a faster absorption than the oral solution, which had a higher drug concentration than the oral conventional drug solution and the intranasal NP administration. The increased concentration in the blood using the gel formulation indicates that the gel formulation increased the systemic exposure of the drug, with a rapid onset of action and sustained exposure.
Pharmacokinetic Parameters After Administration of Intranasal Administration of Nanoparticles Solution or Nanoparticles Gel Formulation of Ubrogepant. The Drug was Administered at the Concentration of Absolute Ubrogepant as 2 mg/kg. Data Indicate Mean ± Standard Error of Mean (SEM), n = 6.
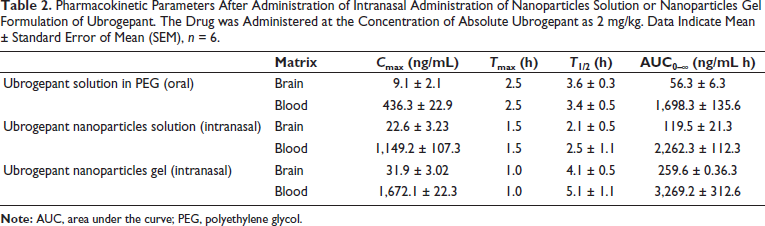
In the brain, the concentration of the drug was highest in the case of the ubrogepant NPs gel formulation, and the sustained exposure to the brain was also evident, owing to a higher Cmax (31.9 and 259.6 ng/mL) as against the conventional oral formulation and the intranasal NP solution. This higher drug concentration, higher half-life, and lower Tmax indicate faster and sustained exposure in the brain. The optimized gel formulation seems to be useful in the pharmacokinetic improvement of the ubrogepant delivery.
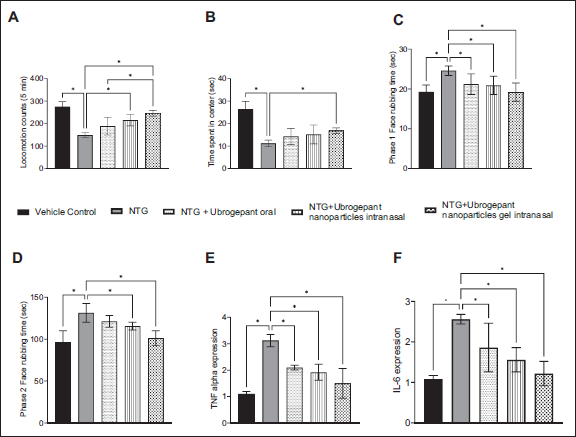
NTG administration reduced locomotion count and time spent in the central area of the open field, indicating decreased activity and potential anxiety-like behavior. This treatment also increased the inflammatory markers TNF-alpha and IL-6.
Intranasal administration of ubrogepant NPs reversed the effects of NTG, restoring normal locomotion and reducing inflammation. In the formalin-induced pain model, the gel formulation of ubrogepant NPs reduced pain behaviors in both phases, which indicates a rapid onset of action with prolonged relief from the pain response (Figure 2).
Discussion
Ubrogepant, a drug approved for the treatment of migraine by the Food and Drug Administration (FDA), is the first in its class of small-molecule oral CGRP receptor antagonists. Migraine is associated with unilateral headaches and other associated symptoms. It is generally accepted that a rapid onset of action, along with significant reach of the drug to the brain, can improve the therapeutic efficacy of anti-migraine agents. 30 The NP solution of ubrogepant was found to improve these parameters. Since a mucoadhesive gel will be useful in increasing efficacy and compliance in the treatment of migraine, we formulated a nasal gel of the ubrogepant NPs and evaluated the pharmacokinetic and pharmacodynamic profiles of the formulation in this work.
Ubrogepant works by blocking CGRP receptors, which are involved in the pain and inflammation associated with migraine. P-glycoprotein or P-gp is a protein that acts as a transporter, pumping substances out of cells, including the brain. Since ubrogepant is a P-gp substrate, it is actively transported out of the brain, reducing its effective concentration in the target areas. 5 This reduced brain availability might explain why some patients experience limited or incomplete pain relief with ubrogepant. 31 Since intranasal administration of drugs, especially their NPs, can reduce the efflux of drugs and improve brain targeting, we have used ubrogepant as the specific drug in this study. 32
The NPs were prepared using PLGA as the FDA-approved biodegradable polymer. PLGA is a biphasic polymer, which means it causes an initial release of the trapped drug molecule, with a sequential sustained release. 33 This modification in the release pattern would be useful in migraine therapy. In addition, PLGA helps to protect the drug molecule from enzymatic breakdown in the nasal milieu due to the changes in the surface properties, as observed in our work as well. 34
The multiple emulsion using the solvent evaporation method was optimized for the speed, time, and pressure of homogenization, and the optimum ratio of the organic and aqueous phases resulted in NPs of a size below 200 nm, indicating suitability for intranasal administration. 35 The zeta potential, PDI, and EE are important criteria for the successful preparation of the NPs to be used for formulations. We have found a significantly negative zeta-potential, which is desired to suspend the NPs in the gel, along with a low PDI and substantially higher EE in these NPs, which is consistent with our previous work (article in press, Journal of Pharmacology and Pharmacotherapeutics). The NPs indicate a beneficial way of formulating drugs for nasal administration. The release of the drug from the NPs, as measured during dialysis, explains that, though the NPs are shielding the drug release when compared to the solution, the release effect is sustained and enough to attain the efficacy.
As previously observed with the ubrogepant NPs, the cellular uptake of the NPs was increased and the efflux was decreased, which could be due to the drug release from the cytoplasm through changes in surface charges when the NPs with the anionic charge are retained for a longer time in the cytoplasm. 36
The release profile of ubrogepant from the conventional solution, the NPs solution, and the NPs gel formulation was tested in an in vitro system. The release pattern was found to be linear, showing a polymer-releasing approach. The conventional ubrogepant solution showed a rapid release, with almost all the drug released within 5 h. On the other hand, the NPs or the gel formulation of NPs showed an initial burst, releasing one-third of the drug in the first 5–6 h, and a slow and consistent release thereafter, which released all the drug in a total of 24 h. The permeation through the ex vivo nasal mucosa was also linear and steady, showing full permeation of the drug in 24 h. This indicated that the optimized gel formulation of the NPs can directly cause the permeation of the drug from the nasal mucosa, possibly into the brain.
The pharmacokinetics studies conducted in the Sprague–Dawley rats indicated a significant reach of the drug in the brain with all three formulations tested, although a significantly high concentration in the blood and brain was observed with the gel formulation. The conventional oral solution showed the most delay in the absorption, with the peak of the concentration in the blood reaching after 2.5 h. The Tmax, however, was decreased by an hour when the NP solution was administered by the intranasal route, and it was again decreased in the case of the intranasal administration of the gel formulation of the NPs. This early peak, however, did not translate into the early dissipation of the drug from circulation. Instead, sustained levels were seen in the blood as well as the brain in the case of intranasal gel administration. Hence, the intranasal gel formulation of the NPs showed a rapid rise in the levels in both blood and brain, with a sustained pharmacokinetic profile that resulted in high Cmax and area under the curve (AUC), with a higher half-life. Thus, the mucoadhesive formulation of the PLGA NPs has significantly improved the mucosal retention and brain penetration of the drug due to the mucoadhesive and thermo-reversible gel formulation that used Poloxamer 407, 2-hydroxypropyl-β-cyclodextrin, and Carbopol 934P, which improved stability, mucoadhesion, and resident time of the drug in the nasal mucosa, with better partitioning in the brain tissue.
The pharmacodynamic activity of the improved and optimized gel formulation of the NPs of ubrogepant was assessed by the investigation of the inflammation in the neurons of the trigeminal region, which causes pain in the cranial area, mimicking migraine in the rodent model. 37 Such migraine-like pain is induced by the NTG injection. 38 The increase in the proinflammatory cytokines, TNF-alpha and IL-6, was significantly reduced by the intranasal administration of the gel formulation of the ubrogepant NPs, in a better way than the oral formulation or plain solution of the NPs delivered by the intranasal route. Consistent with these markers, the intranasal administration of the gel formulation of ubrogepant NPs caused a significant reversal of the in vivo effects of NTG, namely, locomotion and formalin-induced pain reflexes.
The NTG model for migraine is a well-established experimental approach in animal studies to study the underlying mechanisms of migraine and investigational therapies. NTG, a nitric oxide donor, triggers migraine-like symptoms when administered, mimicking the headache, autonomic features, and pain sensitivity associated with a migraine attack. Intravenous or subcutaneous administration of NTG in healthy subjects or migraine patients also induces a delayed headache attack, like a spontaneous migraine. 39 This induced headache is generally reproducible and can be relieved with migraine medications. NTG’s mechanism of action involves the release of nitric oxide, which can cause vasodilation, nerve sensitization, and inflammatory responses, ultimately leading to migraine-like symptoms. 27 This model allows researchers to investigate the neural and vascular pathways involved in migraine, as well as the role of nitric oxide and other inflammatory mediators. The NTG model is used to evaluate the efficacy of new drugs for acute or preventive migraine treatment, as the model responds to known migraine medications. 40 Though animal studies often require relatively high doses of NTG to produce a migraine-like effect, and the effect may not fully capture the presence of aura or the genetic predisposition of patients to migraine, it is still a useful model in which the efficacy of the drugs tested can be translated to clinical effect. 41
The abundance of the CGRP/CGRP receptor system in the brain, especially in the trigeminal ganglion, is the major pathophysiological reason for the vascular headache in the case of migraine, and the CGRP antagonist ubrogepant can significantly reduce migraine symptoms by causing a reduction in trigeminal neuropathy or mechanical allodynia.7, 42 Despite its compelling efficacy, ubrogepant has some side effects that mask its ability to be a successful therapy for migraine. These side effects include liver and renal toxicity, 3 which could be decreased with lower-dose titration, as the intranasal gel formulation of the NPs shows significantly improved brain exposure and the brain-to-blood ratio of the compound.
Conclusion
The NPs of ubrogepant created using PLGA were successfully formulated in a thermoreversible gel using an optimized process that used organic to aqueous balanced phases, polymers, stabilizers, and a rationalized process of making the NPs and the gel formulation. The gel formulation not only increased the speed of absorption but also increased the mucosal availability and brain penetration of the drug with a sustained availability. The optimized gel formulation, designed for the nasal delivery of the NPs of ubrogepant, can achieve a fast and sustained action in migraine treatment, with a lower tendency to cause side effects.
Footnotes
Abbreviations
CCSEA: Control and Supervision of Experiments on Animals; CGRP: Calcitonin gene-related peptide; EE: Entrapment efficiency; FBS: Fetal bovine serum; GAPDH: Glyceraldehyde 3-phosphate dehydrogenase; HPLC: High-pressure liquid chromatography; IAEC: Institutional Animal Ethical Committee; LC–MS: Liquid chromatography–mass spectrometry; NTG: Nitroglycerin; PDI: Polydispersity index; P-gp: p-glycoprotein; PLGA: Poly(lactic-co-glycolic) acid; PVA: Polyvinyl alcohol; RH: Relative humidity; TNC: Trigeminal nucleus caudalis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal study protocol was approved by the Institutional Animal Ethical Committee constituted under the Committee for Control and Supervision of Experiments on Animals (CCSEA) as per protocol no. DMIHER/IAEC/24-25/10.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
