Abstract
Drugs which directly counteract nitric oxide (NO), such as endothelial receptor blockers, NO-synthase inhibitors, and NO-scavengers, may be effective in the acute treatment of migraine, but are also likely to be effective in migraine prophylaxis. In the underlying pilot study the prophylactic effect of the NO scavenger hydroxocobalamin after intranasal administration in migraine was evaluated. Twenty patients, with a history of migraine of <1 year and with two to eight migraine attacks per month, were included in an open trial. A baseline period was followed by an active treatment period of 3 months with 1 mg intranasal hydroxocobalamin daily. Patients were instructed to complete a diary in which details of each attack were described. A reduction in migraine attack frequency of ≥50% was seen in 10 of 19 patients, which corresponds to 53% of the patients (responders). A reduction of ≥30% was noted in 63% of the patients. The mean attack frequency in the total study population showed a reduction from 4.7 ± 1.7 attacks per month to 2.7 ± 1.6 (P< 0.001). For the responders the migraine attack frequency was reduced from 5.2 ± 1.9 (baseline) to 1.9 ± 1.3 attacks per month (P < 0.005), while for those who did not respond a non-significant reduction was found: 4.1 ± 1.4 to 3.7 ± 1.5 (P > 0.1). A reduction was also observed for the total duration of the migraine attacks per month, the total number of migraine days per month and the number of medication doses for acute treatment used per month. This is the first prospective, open study indicating that intranasal hydroxocobalamin may have a prophylactic effect in migraine. As a percentage of responders in prophylactic trials of > 35-40% is unlikely to be a placebo effect, a double-blind study is warranted.
Introduction
Shortly after the invention of nitroglycerin, it was noticed that this substance is capable of inducing a violent headache (1). Nitroglycerin, administered as a short continuous intravenous infusion, has been used to induce headache in healthy subjects and in migraine patients (2). In these studies nitroglycerin induced an immediate headache response during the infusion. Only in migraine patients was this non-migraine headache followed by a genuine migraine attack with a time delay of several hours after the infusion (2). Nitroglycerin may be regarded as an exogenous source of nitric oxide (NO) (3). These and other observations have urged Olesen to propose endogenous NO as a candidate for the causative molecule in migraine (4).
As migraine attacks develop several hours after a NO challenge, it has been suggested that NO may be involved in the early phase of spontaneous migraine attacks. Therefore, drugs which directly counteract NO, such as endothelial receptor blockers, NO-synthase inhibitors and NO-scavengers, may be effective in the acute treatment of migraine, but are also likely to be effective in migraine prophylaxis (4).
The efficacy of a nitric oxide synthase inhibitor has been demonstrated as an acute treatment of migraine attacks (5) as well as of tension-type headache (6). However, to our knowledge no NO-scavenger has been tested in migraine as an acute treatment, or as a prophylactic drug. Hydroxocobalamin (OHB12) appears to be such a NO-scavenger (7) and is a likely candidate for further investigation.
Oral formulations of OHB12 do not lead to elevated plasma concentrations, as its bioavailability is very low (8). Alternative routes are the intranasal and intramuscular administration of OHB12. Both Bruins Slot et al. (9) and Van Asselt et al. (10) have demonstrated a good absorption of OHB12 after intranasal administration in cobalamin-deficient patients. Moreover, it has been demonstrated that intranasal administration of 5 mg OHB12 to healthy volunteers in each nostril gives serum concentrations of OHB12 comparable to intramuscular administration of 1 mg OHB12 (11).
Therefore, we performed an open study to evaluate the prophylactic effect of the NO scavenger hydroxocobalamin after intranasal administration in migraine.
Methods
Patients
Patients with a history of migraine for >1 year were diagnosed according to the International Headache Society (IHS) criteria (12). They suffered from at least two and at most eight migraine attacks per month. Exclusion criteria were the use of any prophylactic drug in the 3 months prior to inclusion, tension-type headache >5 days per month, age <16 years, (wish for) pregnancy or lactation, anaemia (haemoglobin cut-off point for women 7.5 mmol/l and for men 8.5 mmol/l), use of vitamin B12-containing supplements, acute or chronic rhinitis, abuse of alcohol or other substances and abnormal findings of the liver or kidney function tests.
The protocol was approved by the local Ethics Committee.
Design and treatment
After written informed consent to participate in the study and medical approval, patients started with a baseline period of 1 month (month 0) in which no prophylactic medication was used. This baseline period was followed by an active treatment period of 3 months. The patients used 1 mg intranasal OHB12 daily by administering 50 µl of a 2% hydroxocobalamin aqueous solution (prepared according US patent 5801 161 and EU patent 0.735.859). The multidose spray flacons were a gift from Pfeiffer (Radolfzell, Germany).
During the 4-month period patients were instructed to complete a diary in which details of each attack, fulfilling the IHS criteria, were described. Patients were allowed to treat their migraine attacks with their habitual methods.
Parameters and statistics
The primary endpoint was the number of patients with a reduction of ≥50% in migraine attack frequency in month 3 compared with baseline (responders). Secondary efficacy parameters were number of attacks per month, total duration of migraine in hours per month, number of days with migraine, mean headache severity of each attack per month and amount of rescue medication used per month. The headache severity was expressed as: 0, no headache; 1, mild headache; 2, moderate headache; or 3, severe headache. At regular times the following parameters were determined to document the safety of the treatment: γGT, ALAT, ASAT, LDH, creatinine, number of reticulocytes, erythrocytes, Hb and cobalamin serum concentration.
Pre- and post-active treatment, rhinoscopia anterior followed by a nasal endoscopy was performed by an ENT specialist to assess any detrimental effects on the nasal mucosa.
Patients were instructed to describe every Adverse Event (AE) they noted. At the end of the study patients were asked to give their opinion of the efficacy of the new drug for several parameters.
Patient compliance was controlled by weighing the nasal flacons at the end of the study.
Statistical comparisons for the migraine parameters between baseline (month 0) and month 3 were performed using the non-parametric two-tailed Wilcoxon signed ranks test. The results are expressed as mean ± SD. Only descriptive statistics were performed for the clinical chemistry parameters.
Results
Twenty patients, both with and without aura, were included in the study, five males and 15 females with a mean age of 41±14 years (range 20–70 years). One patient (female) withdrew from the study within 2 weeks after the start of the active medication, because of acne. As her participation in the study had been of too short duration, she was excluded from the analysis. The amount of drug used varied from 84% to 120%, which we regarded as a reasonably good patient compliance for a daily therapy of 3 months.
Efficacy
The results are summarized in Table 1. The mean number of attacks per month was 4.7±1.7 during baseline, ranging from three to eight attacks per month. After 3 months of therapy the mean number of attacks had decreased to 2.7±1.6 attacks per month (P<0.001), ranging from 0 to six attacks per month. A reduction in migraine attack frequency of ≥50% was seen in 10 of 19 patients, which corresponds to 53% of the patients (responders). A reduction of ≥30% was noted in 63% of the patients. For the responders the migraine attack frequency was reduced from 5.2±1.9 (baseline) to 1.9±1.3 attacks per month (P<0.005), while for those who did not respond a non-significant reduction was found: 4.1±1.4 to 3.7±1.5 (P>0.1, see Fig. 1).
Migraine parameters before and after 3 months therapy with 1 mg intranasal hydroxocobalamin once daily in 19 patients
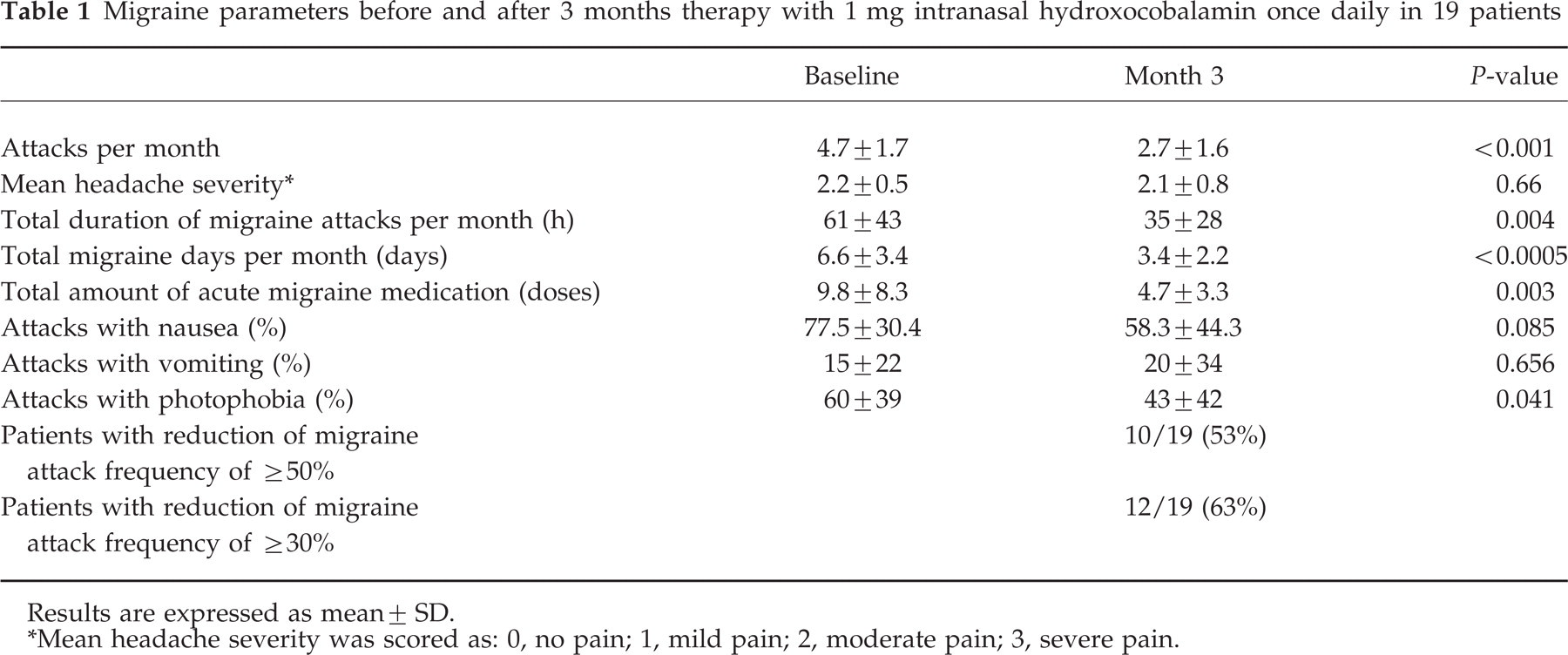
Results are expressed as mean± SD.
∗Mean headache severity was scored as: 0, no pain; 1, mild pain; 2, moderate pain; 3, severe pain.

Migraine attack frequency per month for the total population and for the two subgroups of responders and non-responders. ∗P<0.005; compared with baseline (month 0).
The reduction in migraine attack frequency for responders was more pronounced and statistically significant for the number of moderate (month 0: 2.2; month 3: 0.7; P=0.014) and severe (month 0: 2.2; month 3: 0.7; P=0.039) headaches per month than for the number of mild headaches (month 0: 0.8; month 3: 0.5; P=0.46; see Fig. 2). For the non-responders there was no significant reduction in any of the headache scales. There appeared to be no statistical difference in the mean severity of all migraine attacks per month for the total study population, nor for one of the subgroups. A reduction was also observed in the total duration of migraine attacks per month, the total number of days with migraine per month and the number of medication doses for acute treatment used per month (see Table 1). No statistically significant change was seen in the mean duration of the single attacks. The type of rescue medication used by the patients were paracetamol (acetaminophen) eight patients; acetylsalicylic acid seven patients; NSAIDs 11 patients; triptans 10 patients; ergotamine three patients. Some patients used more than one type of medication.

Migraine attack frequency per month for mild, moderate and severe headaches for the subgroup of responders (n=10). ∗P<0.05; compared with baseline (month 0).
Associated symptoms
In the whole study population there was no significant change in the percentage of attacks in which nausea or vomiting occurred (Table 1). However, for the subpopulation responders a statistically significant reduction was observed in the percentage of attacks in which nausea occurred: month 0, 85±21%; month 3, 45±44% (P=0.017). In the total population a reduction was seen in the percentage of attacks with photophobia: month 0, 60±39%; month 3, 43±42% (P=0.041).
Adverse events
No serious AEs were encountered. As mentioned above, one patient withdrew, as she developed acne on the hairy scalp. Three weeks after discontinuation of the OHB12, acne was not resolved completely.
Adverse events appeared mainly at the start and disappeared after a few days. Most of the AEs were restricted to the nose, such as decongestion (n=5), local irritation (n=6) and wheezing (n=3). One patient mentioned that her leg cramps has disappeared when she started using OHB12. At the end of the study, these cramps appeared again. Another patient mentioned the disappearance of her pain in the neck. One patient, familiar with constipation, noticed that diarrhoea occurred after starting with OHB12. This was, however, self-limiting. Except for the acne, all patients rated their AE as mild and no patient stopped therapy with this study drug because of an AE.
Three of the five men mentioned independently that ‘several times a feeling in the head occurred as if they were going to develop a severe migraine attack, but the migraine did not break through’. Rhinoscopia revealed no anomalies and no detrimental effects on the mucosa were observed.
Patient impressions
At the end of the study, 13 of 19 patients rated the reduction in migraine attack frequency as ‘obvious’, while four considered the effect as small. For the other parameters the choices of the patients were less clear, which corresponds to the results obtained from the diary. There was no clear improvement in the subjective feeling of the patients apart from the migraine. They did not feel less tired.
Clinical and haematological parameters
A summary of the values is given in Table 2. Only one patient had at baseline a cobalamin concentration below normal values (150–600 pmol/l). The cobalamin concentration in the total study population increased from 257±78 pmol/l to 1953±2451, 3246±2608, 5708±3976 and 7475±4378 pmol/l at, respectively, the end of week 1, and months 1, 2 and 3. Despite the increase in cobalamin in serum, no increase was observed in haemoglobin, erythrocytes or reticulocytes, suggesting that the patients had no deficiency.
Clinical and haematological parameters before and after 3 months therapy with 1 mg hydroxocobalamin intranasally once daily in 19 patients

Results are expressed as mean± SD.
As expected, the parameters for kidney and liver function stayed normal (see Table 2). None of the patients exceeded the normal values pre- or post-treatment.
Discussion
Design of the study
NO is hypothesized to play an important role as the causative molecule in migraine headache (4). After the administration of a NO-donor (nitroglycerin) a considerable delay of several hours was seen before a migraine attack developed (13). This suggests that NO may be involved in the early phase of migraine attacks (4). Therefore, a NO-scavenger, administered to a patient as an acute migraine therapy, may be less effective because NO may already have triggered a cascade of reactions leading to the migraine symptoms. The presence of high (blood) concentrations of a NO-scavenger in the early phase of a migraine attack may be more effective to prevent the development of a full-blown migraine attack. Therefore, we have chosen for an open label prophylactic design of this pilot study on a limited number of patients treated with daily OHB12 as a nasal spray.
Efficacy
This is the first study to demonstrate a reduction in migraine attack frequency by daily intranasal hydroxocobalamin. Overall, a mean reduction was seen of two migraine attacks per month per patient. A reduction in migraine attack frequency of ≥50% was seen in 53% of the patients.
For the responders the effect of fewer migraine attacks per month was strengthened as the decline was more pronounced and statistically significant in the moderate or severe attacks. From the patients' perspective this might be seen as a double gain.
The 50% used in the definition of a responder (defined by the IHS (12)) is arbitrarily chosen. Patients may even be satisfied with a reduction of 30% or even less. In the present study a reduction in migraine attack frequency of ≥30% was seen in 63% of the patients.
For the interpretation of the result of a prophylactic open study, more knowledge of the placebo-response is necessary. In a meta-analysis the percentage of responders has been quantified in 23 double-blind placebo-controlled clinical studies (14). In the placebo group, a responders' percentage was found of 23.5±8%, while for the active group this was 45.5±15.5%. Therefore, a percentage of responders of >35–40% in prophylactic trials is unlikely to be a placebo response. The results of double-blind studies, however, cannot be extrapolated to the present study, which has an open label design. Nevertheless, the results of our study, with 53% of the patients showing a reduction in attack frequency of ≥50%, can be characterized as promising and encouraging to perform further double-blind studies. In the present study, an increase in migraine attack frequency was observed in two patients, no change in another two patients, while 15 patients reported a decline in their migraine attack frequency in month 3 compared with month 0.
Efficacy (defined as a decline of ≥50% of the migraine attacks) was seen only in a part of the study population (53%), a phenomenon also seen in other prophylactic migraine therapies. The observation that not all patients responded to OHB12 may be explained by a different pathophysiology of the migraine in the non-responders. Another explanation might be that the plasma concentrations were too low for an adequate NO-scavenging effect in all patients. If the mild adverse event profile is taken into account, higher dosages may be possible. However, with the limited data available at present no clear relation could be detected between cobalamin serum concentrations and efficacy.
Safety and tolerance
Oral administration of OHB12 has an extremely low bioavailability, which makes this route inadequate (8). Intramuscular administration gives cobalamin levels which are high enough, but daily injections are not preferred if other routes are available. The once-daily nasal administration of 1 mg OHB12 gives cobalamin serum levels in the range of 5000–10 000 pmol/l.
The cobalamin concentration in blood in this study was high compared with normal values (150–600 pmol/l). It should be noted that after injection of 1 mg OHB12 serum levels of 20 000–30 000 pmol/l are reached. For the treatment of cyanide intoxication a single intravenous dose of 5 g OHB12 has been administered, which is 5000 times the therapeutic dose to treat cobalamin deficiency (15). Intravenous administration of 5 g OHB12 gives maximal cobalamin levels of >200 µmol/l without serious adverse events. It is clear that the levels after daily nasal administration (mean 7400 pmol/l after 3 months) offer no safety risk.
One patient withdrew early as she developed acne on the hairy scalp. No relation could be established between the administration of OHB12 and the acne. In none of the other clinical trials with intranasal OHB12 was such an adverse event observed (8–11). Characteristic of the vitamin B12-induced acne, only reported for cyanocobalamin, is the spontaneous disappearance after discontinuation of the vitamin B12 therapy within 8–10 days (16). In this case the acne did not disappear for more than 3 weeks after discontinuation of the intranasal OHB12.
The intranasal administration of OHB12 has been studied in healthy volunteers and in vitamin B12-deficient patients (9, 10). No serious toxicity or adverse events were seen in these studies, nor in this study. Therefore, we consider the intranasal treatment with OHB12 in this dosage range as safe.
Mode of action
NO, a known environmental pollutant, has come to be considered an important physiological mediator (17). It not only maintains the vasodilatory tone (formerly known as endothelium derived releasing factor (EDRF)), but also acts as a neuronal messenger. The role of NO in the pathology of migraine may be a direct dilatation of cerebral and extracerebral blood vessels, or via a direct effect on perivascular sensory nerves. This may be important to know if selective NO-synthase inhibitors or NO-scavengers are tested. OHB12, in contrast with some other NO-scavengers (18), is not selective, nor is the NO-synthase inhibitor L-NMMA (19), which is the only NO-synthase inhibitor tested in humans in migraine.
The efficacy of drugs of NO-synthase inhibitors has been demonstrated for the acute treatment of migraine and chronic tension-type headache (5, 6). This is the first report in which the efficacy of a NO-scavenger has been demonstrated for the prophylactic therapy of migraine. This strengthens the hypothesis of Olesen that NO plays an important role in the pathophysiology of migraine (4).
Vitamin B12 in migraine
Vitamin B12 or cobalamin exists in the human body as methylcobalamin, adenosylcobalamin, hydroxocobalamin and cyanocobalamin. Only the latter two are used as drugs for the treatment of patients. Hydroxocobalamin contains a hydroxy-group in the cobalt co-ordination complex instead of the cyano-group characteristic of cyanocobalamin.
Cyanocobalamin has been used extensively for the initial and maintenance treatment of pernicious anaemia and other forms of cobalamin deficiency, but hydroxocobalamin is the drug of choice (20).
Almost 50 years ago Urdahl-Aasen observed that the migraine attacks of a patient with anaemia perniciosa resolved after treatment with vitamin B12 (21). This observation led to the use of vitamin B12 in some people. Urdahl-Aasen noticed that intramuscular administration of cyanocobalamin 15 µg a week might diminish or even abolish migraine attacks in at least a part of his population (22). Due to the encouraging results a therapeutic investigation was carried out in 14 patients with migraine and six cases of psychogenic headache (22). The patients were treated with two to three injections of 15 µg cyanocobalamin in the first week, followed by the same dose weekly during 2–4 months. Only 2/14 migraine patients observed a good result, but a favourable effect was seen in 4/6 patients with psychogenic headache. All six patients with psychogenic headache reported, without being asked, a relation between the injections and a general stimulating action. Therefore the effect on the headache may be due to an increase in general well-being.
The studies by Urdahl-Aasen and Dalsgaard-Nielsen have stimulated Dalsgaard-Nielsen and Trautmann to perform a double-blind placebo-controlled study on 29 patients more than 18 years after the first observations (23). During 2 months every 2 weeks 2 mg cyanocobalamin were administered intramuscularly. In the group treated with cyanocobalamin (n=15), four patients reported a good result and two a considerable improvement. For the placebo group (n=14), a good result was obtained for two patients and a considerable improvement for five patients. No correlation between the results and the presence of psychic factors was demonstrated in this study. The authors concluded that no therapeutic effect attributable to cyanocobalamin was demonstrated.
The effect seen in the present study is not attributable to the treatment of a (subclinical) deficiency, as such an improvement would have been observed in haematological parameters. It is also unlikely that the administration of OHB12 caused an increase of well-being, as this was not noticed by the patients.
In the studies described above cyanocobalamin was used as a vitamin B12 preparation. We hypothesize that OHB12 might be effective in migraine because of its NO-scavenging activity. Since cyanocobalamin has no NO-scavenging activity, in contrast to OHB12, it is not surprising that in the earlier trials no effect was seen in migraine patients. It should also be noted that the dose administered in the early trials was much lower than that used in the present study.
It is concluded that this is the first prospective, open study indicating that intranasal OHB12, as a NO-scavenger, may have a prophylactic effect in migraine. As a percentage of responders in prophylactic trials of >35–40% is unlikely to be a placebo effect, the results of these investigations warrant a double-blind protocol.
Footnotes
Acknowledgements
The contribution of Dr P. G. Verduyn (Maasland Hospital, Sittard) for the nasal endoscopy is gratefully acknowledged. We thank W. J. T. A. Hermans (Department of Clinical Chemistry, Sittard) for the analysis of OHB12.
