Abstract
Background
Pulmonary arterial hypertension is controlled by the dual endothelin receptor antagonist bosentan monohydrate. Recent advances in microneedle technology offer controlled, prolonged drug release.
Objectives
This study formulated and evaluated a bosentan monohydrate-loaded liposome-based dissolved microneedles (MNs) array patch.
Materials and Methods
Bosentan monohydrate liposomes were prepared using the ethanol injection method and incorporated into microneedle patches. The dissolving microneedles (DMNs) were prepared by using a two-step casting method. The first 3D computer-aided design (CAD)-designed master mold for the MNs was fabricated using SLA 3D printing and replicated using polydimethylsiloxane (PDMS). The mold was filled with a mixture of polyvinyl alcohol (PVA), polyvinylpyrrolidone (PVP-K30), PEG-400, and bosentan-loaded liposomes, then centrifuged and dried. Optimization using response surface methodology (RSM) focused on particle size, zeta potential (ZP), and entrapment efficiency (EE).
Results
The optimized liposomes had an average size of 238.06 nm, a ZP of −34.33 mV, and an EE of 75.3%. Morphological analyses via microscopy and scanning electron microscopy (SEM) confirmed spherical liposomes with smooth surfaces. Drug release studies indicated a controlled release over time, reaching approximately 90% cumulative drug release (CDR) at 600 min. Kinetic modeling showed a zero-order release pattern. Mechanical testing of the MNs demonstrated a fracture point at 0.42 N, ensuring adequate strength for skin penetration. Ex vivo tests on rat skin confirmed nearly 100% penetration of the 15 × 15 microneedle array.
Conclusion
The study concludes that the optimized formulation of liposomal-loaded DMNs is easy to administer, non-invasive, and biodegradable, offering a promising approach for controlled drug release in managing pulmonary hypertension (PH).
Introduction
The chronic, progressive illness known as pulmonary hypertension (PH) has a variety of origins, typically involving harmful changes in the small arteries of the lungs or a congestive vasculopathy brought on by elevated pulmonary vein pressure, which may result in right heart failure and if not treated, eventually death.1, 2 The annual incidence of pulmonary arterial hypertension (PAH) is around 2.4 cases per million persons, with a prevalence that ranges from 15 to 50 cases per million. Additionally, the high mortality rate of up to 30% within 3 years underscores the serious and often grim prognosis associated with this condition. 3 When precapillary PH, which is defined by a mean pulmonary artery pressure (mPAP) of more than 25 mmHg and a pulmonary artery wedge pressure (PCWP) of less than 15 mmHg, is discovered during a right-heart catheterization (RHC), PAH is identified.4, 5 One may categorize PH into five distinct clinical subgroups: (a) PAH; (b) PH as a consequence of left ventricular dysfunction; (c) PH linked to respiratory disorders, hypoxia, or hypoventilatory syndromes; (d) PH resulting from pulmonary arterial blockages, such as chronic thromboembolic pulmonary hypertension (CTEPH); and (e) a variety of PH etiologies with different underlying mechanisms, such as sarcoidosis and sickle-cell disease. 1 As a result, the efficient management of PAH continues to be a crucial therapeutic concern.
One of the biggest clinical challenges in treating PAH effectively is still present. Prostate cyclin analogs, prostacyclin receptor agonists (PRAs), soluble guanylate cyclase stimulators, and phosphodiesterase-5 inhibitors (PDE5i) are the three main routes that have been the focus of PAH therapy development during the past 20 years. 6 Several targeted therapeutic agents have been formulated for the treatment of PAH, including prostanoid, phosphodiesterase type 5 inhibitors, and endothelin receptor antagonists (such as ambrisentan and bosentan). 2
Bosentan is a dual endothelin receptor antagonist that is derived from sulfonamide and pyrimidine, and it is mainly used to treat PAH. This type of hypertension affects the blood vessels in the lungs and the right portion of the heart. The neurohormone endothelin-1 (ET-1) works by attaching itself to endothelin type A (ETA) and endothelin type B (ETB) receptors that are present in vascular smooth muscle and endothelium. Elevated levels of ET-1 in the plasma and lung tissue of PAH patients suggest its involvement in the disease’s pathogenesis. Bosentan is an antagonist of the endothelin receptor types ETA and ETB that is both competitive and selective, with ETA receptor affinity being somewhat greater. 7
Bosentan monohydrate was chosen for dissolving microneedles (DMNs) due to its efficacy in reducing pulmonary vasoconstriction and vascular remodeling in PAH. Its solubility and stability make it suitable for transdermal delivery, improving bioavailability and patient compliance, unlike other PAH treatments.
A corresponding number of aqueous compartments are encased in one or more lipid bilayers to form the vesicular colloidal structures known as liposomes. 8 They have demonstrated considerable improvements in the administration and medicinal effectiveness of different kinds of active pharmacological substances. Enhanced liposome delivery optimizes the pharmacodynamics and pharmacokinetics of various medications. 9
Compared to conventional methods, the transdermal route offers several potential advantages, such as avoiding first-pass metabolism, extending and predicting the duration of action, minimizing undesired side effects, utility of drugs with short half-lives, improvement of physiological and pharmacological response, preventing fluctuations in medication levels, differences between patients, and—above all—patient convenience. Various tactics have recently been applied to improve the transdermal distribution of bioactive compounds.10, 11
The microneedle technology has received attention recently as a method for the controlled and prolonged release of medications. Microneedles (MNs) array patches are a non-invasive, user-friendly microinjection tool made up of tiny needles (ranging from 25 to 1,500 µm) that, when applied to the skin, puncture the outer layer (stratum corneum) and form temporary microchannels. This process allows the direct delivery of therapeutic agents into the deeper layers of the skin. 12 They minimize the total usage load while improving patient comfort and compliance by avoiding close contact with the blood vessels and nerves. 13 Various types of MNs have been documented in the literature. However, recent years have seen a significant increase in interest toward polymer-based DMNs. 12 DMNs are composed of water-soluble, biodegradable, or biocompatible polymers that dissolve in tissue fluid to deliver the nano-drug in a precise and regulated manner. Their biocompatibility eliminates the necessity for post-treatment removal and lowers the risk of irritation or infection. 14
DMNs offer significant advantages over traditional transdermal systems or injectable formulations for bosentan monohydrate by providing a painless, minimally invasive delivery method. This improves patient compliance by eliminating the need for frequent injections or messy patches, offering a convenient, self-administered, and discreet treatment option with sustained drug release, and avoiding microbial infection due to frequent administration.
DMNs are particularly beneficial for the PAH because many PAH treatments require continuous or frequent administration. Microneedle patches are easy to self-administer and less intimidating than traditional needles, leading to better compliance. For PAH, drugs that act systemically, such as endothelin receptor antagonists, can be delivered effectively through this route, and also microneedles can be designed to release drugs at a controlled rate, improving therapeutic outcomes by maintaining steady drug levels, which is crucial in managing PAH.
This study focuses on creating and analyzing bosentan monohydrate-loaded liposome microneedle array patches. DMNs were chosen to decrease the frequency of dosing and to minimize side effects commonly associated with more frequent drug administration methods. To achieve this objective, bosentan monohydrate-loaded liposome dispersions were formulated and optimized using design of experiments (DoE) techniques and then evaluated for their liposome characteristics in terms of different parameters, such as vesicle size (VS), zeta potential, microscopy study, and scanning electron microscopy (SEM). In the second step, for the final formulation, a liposome dispersion filled with bosentan monohydrate was freeze–dried and loaded into DMNs (bosentan monohydrate-loaded liposome-DMNs). The bosentan monohydrate-loaded liposome-DMNs were assessed for their SEM, in vitro drug release, skin penetration, and mechanical strength of the dissolving microneedle patches.
Materials and Methods
Materials
Bosentan monohydrate (purity ≥99%) was obtained from the Yarrow Chem Products; lecithin soya 30% was procured from HiMedia Laboratories; polyvinylpyrrolidone-K30 was procured from LOBA Chemie Pvt. Ltd.; potassium dihydrogen orthophosphate and di-sodium hydrogen orthophosphate were procured from Thermo Fisher Scientific India, Mumbai; cholesterol and polydimethylsiloxane (PDMS) were procured from the Yarrow Chem Products; oleic acid, polyvinyl alcohol (PVA), polyethylene glycol 400, and sodium chloride were procured from SD Fine-Chem Limited. Each and every solvent that was utilized was of analytical grade and obtained from Hayman Group Ltd.
Methods
Compatibility Studies of Drug and Excipients
The pure drug and excipients were examined using Fourier transform infrared spectroscopy (FTIR) spectroscopy (Shimadzu digital IR spectrometer, Japan) within the 400–4,000 cm⁻1 range to identify significant interactions.
Powder X-ray Diffraction Studies
X-ray powder drug diffraction patterns, granulated lipid-based formulation, were recorded using a D/teX Ultra2 detector diffractometer (Rigaku Miniflex 600 6G), standard runs using a 40 kV voltage, a 15 mA current, and a scanning speed of 2.00°/min over an axis of 2θ and range of 3°–90° were used.
Preparation of Optimized Bosentan Monohydrate-loaded Liposomes
Ethanol injection was used to create bosentan monohydrate liposomes, as described in the literature. 15 A mixture of soya phosphatidylcholine, cholesterol, oleic acid, and the drug was dissolved in ethanol. Simultaneously, a measured amount of phosphate buffer saline (PBS, pH 7.4) was stirred on a magnetic stirrer. Next, a 20-gauge needle (0.9 × 25 mm) was used to progressively inject the ethanolic lipid solution into the phosphate buffer solution (pH 7.4) to form vesicles, and the mixture was stirred for 1 h at 500 rpm. The liposome formulation was subsequently sonicated at a speed of 30 and a temperature of 25°C for 5 min using probe sonication.
Formulation of Dissolving Microneedle Patches Containing Bosentan Monohydrate-loaded Liposomes
The master mold’s initial 3D computer-aided design (CAD) model was designed using software (Figure 1), then sliced and exported in STL format. The master mold for the microneedle arrays was fabricated using SLA 3D printing technology (Creality Halot Mage, China) with standard resin. The mold had a diameter of 16 mm, with needle dimensions of 1,000 µm in height, 300 µm in base diameter, and a pitch of 250 µm for a 15 × 15 array. To create a working mold with needle cavities, PDMS compound (a 10:1 mixture of elastomer and curing agent, manually mixed and degassed) was poured onto the master mold and cured at room temperature for 48 h. After curing, the compound was extracted from the master mold and prepared to create microneedle array patches. PVA, polyvinylpyrrolidone (PVP-K30), PEG-400, and bosentan monohydrate-loaded liposomes were mixed together and added to the PDMS molds in the prescribed amounts. Following centrifugation, the molds were allowed to dry for 48 h at ambient temperature and 45% relative humidity in an isolation chamber. The MNs were meticulously removed from the mold after drying, and then they were described. 16
PVA, PVP-K30, and PEG-400 were chosen for the microneedle base due to their complementary properties, ensuring optimal drug delivery and structural integrity. PVA provides mechanical strength, PVP-K30 enhances solubility, and PEG-400 improves flexibility, ensuring efficient drug release and strong, yet effective delivery.
(a) Computer-aided Design of Master Mold, (b) 3D Printed Master Mold, (c) Dissolving Microneedle Patch.

Formulation Optimization
The liposome formulations were statistically optimized using response surface methodology (RSM), which allowed for the determination of the optimal process conditions and the identification of important variables and how they interact with fewer trial runs. The independent variables selected for this study included the concentration of soya phosphatidylcholine (X1), cholesterol (X2), and oleic acid (X3), each varied at three levels: low (−1), medium (0), and high (+1). The optimization focused on particle size (VS-Y1), zeta potential (ZP-Y2), and entrapment efficiency (EE-Y3) (Table 1). A Box–Behnken design was employed via Design Expert 12 software to generate a total of 17 experimental runs, as detailed in Table 3, which presents the work plan in terms of both coded and real values for the parameters and the constraints on the dependent variables.
The Entire Experimental Plan Interns of Coded and Actual Values of Selected Variables and Constraints of Dependent Variables for Box–Behnken Design.
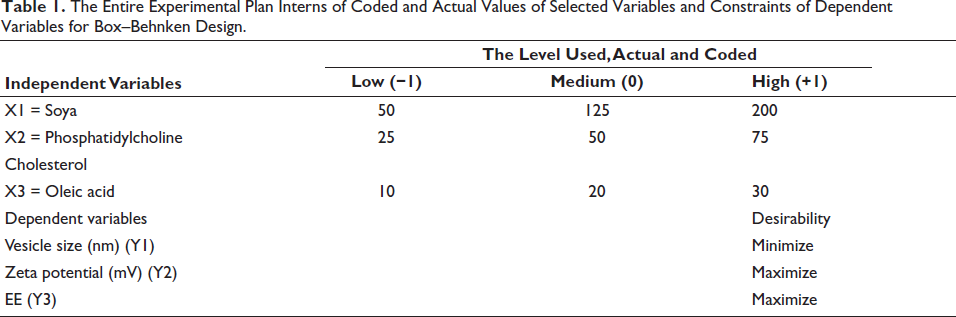
In optimizing the liposomal formulation using RSM, key criteria included lipid concentration, lipid-to-drug ratio, and surfactant type. The optimization process helped identify the best formulation conditions, resulting in improved particle size, enhanced ZP for stability, and higher EE, ensuring effective and stable drug delivery.
Analysis of variance (ANOVA) was used to statistically validate the produced polynomial equations. Various statistical models, including the 2FI and quadratic models, were applied to the experimental runs. A comparison of many statistical indicators, including relative standard deviations, coefficient of variation (CV), many correlations coefficient (R2), and both predicted and adjusted R2 values, was used to determine which model suited the data the best. Quantifying the response for every experimental run was done using quadratic regression, followed by a thorough investigation.
Characterization of Optimized Bosentan Monohydrate-loaded Liposomes
Determination of VS and ZP
To measure VS and ZP, 1 mL of the liposomal colloidal dispersion was diluted with distilled water to a total volume of 10 mL, and 3 mL of this diluted sample was then used for analysis. Liposome size and ZP were assessed using a Litesizer 500 (Anton Paar, Austria) with 658 nm dynamic light scattering (DLS) at 25°C and a 90° scattering angle. The liposome size is reported as the average diameter, determined by the Litesizer Nano software through cumulant analysis of signal intensity. Each sample was measured in triplicate, and the results are presented as the average value. 17
Determination of EE
The liposomal colloidal dispersions were subjected to centrifugation using a refrigerated centrifuge (Eppendorf, Germany) at 7,500 rpm and 15°C for 1 h. After centrifugation, the concentrated liposomes were retained at the top of the filter, while the filtrate, which contained the unentrapped drug, was collected at the bottom of the tubes. The free drug concentration of this filtrate was then determined using a UV spectrophotometer (Shimadzu 1800, Japan) set to dilute it with phosphate buffer (pH 7.4) at a wavelength of 267 nm. Next, using the given formula, the percentage of encapsulation efficiency (%EE) was ascertained.
18
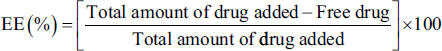
Photo-microscopic Analysis and Scanning Electron Microscopic Study
To capture the images of an optimized freeze–dried liposomes, the samples were positioned on a clean glass slide, and a coverslip was then placed over the top of the liposome samples, and the slide was placed on the microscopic stage, and the focus was adjusted until liposome images were clearly visible by using upright Motic BA310E optical microscope at 40× magnification with BioVis software.
To analyze the surface structure of the optimized liposome formulation, SEM from Zeiss was employed. The samples were mounted on stubs, coated with aluminum via sputtering, and then examined using the SEM.
Characterization of Bosentan Monohydrate-loaded Liposome-based Microneedle Array Patches
Photo-microscopic and SEM of Dissolving MNs
In order to take pictures of a microneedle that dissolves, the samples were positioned on a clean glass slide, and the slide was placed on the microscopic stage, and the focus was adjusted until MNs images were clearly visible by using upright Motic BA310E optical microscope at 40× magnification with BioVis software.
In Vitro Drug Release and Drug Release Kinetic Data
Bosentan monohydrate drug-loaded liposome microneedle array patches were subjected to an in vitro release investigation utilizing a USP type II dissolving device (Electrolab, TDT-08L, Navi Mumbai, India). The microneedle array patch was kept at 37°C and agitated at 100 rpm while submerged in 900 mL of PBS at pH 7.4 (release medium). At various time points (0, 30, 60, 120, 180, and up to 600 min), to verify sink conditions, 5 mL samples were removed and swapped out for a corresponding volume of release medium. Drug release was quantified using a UV–visible spectrophotometer. 15
Different mathematical models can be used to analyze drug release kinetics, assessing how drug levels change over time from 0 to 600 min. The effectiveness of these models in describing dissolution profiles was evaluated using various plots: the zero-order model, with the cumulative percentage of drug release plotted against time; the First-order model, with the cumulative percentage of drug remaining plotted against time; the Higuchi model, with the cumulative percentage of drug release plotted against the square root of time; and the Korsmeyer–Peppas model, with the cumulative percentage of drug release plotted against the logarithm of time. 19
Mechanical Strength Study
A universal testing machine (UTM, Deesha Implex, India) was utilized to evaluate the liposome microneedle array patch filled with bosentan monohydrate in terms of mechanical strength. To separate and test a single microneedle, the patch was sectioned. At first, there was a 3 mm space between the top cylindrical rod and the microneedle array patch. The upper rod then descended at a rate of 1 mm/min until the microneedle broke, using a 100 N load cell to measure the force and displacement. The machine recorded these values to generate a force-displacement curve, which was used to determine the failure force of the single microneedle. 20
Insertion Study of MNs
To assess whether MNs penetrate the skin, bosentan monohydrate-loaded liposome-based microneedle array patches arranged in a 15 × 15 configuration were inserted into rat skin with IAEC approval. The patches were attached with 1 cm long screws under light stress to a wooden plate. The MNs were pressed into the skin by applying approximately 0.4 N of force with a thumb for 2 min, after which the patches were removed. 21
Results
Compatibility Studies of Drug and Excipients
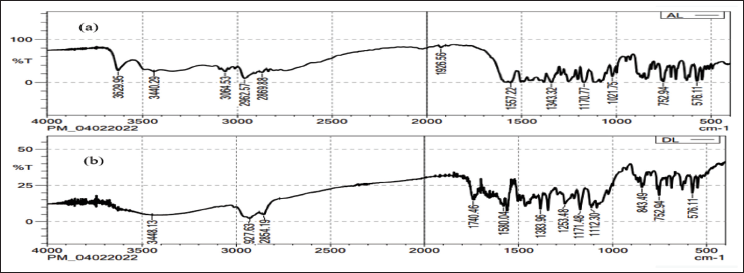
The IR spectroscopy method was employed to investigate drug-polymer interactions. About 5 mg of the sample was combined with dry KBr, and the spectra were recorded across a wavelength range of 4,000–400 cm⁻ 1 . By comparing the spectra of the pure drug with those of the physical mixture containing bosentan monohydrate, soya lecithin, cholesterol, PVA, and PVP, all drug peaks were observed in the physical mixture. This analysis indicated compatibility between the drug and all the excipients used in the formulation, as shown in Table 2 and Figure 2.
Interpretation of Fourier Transform Infrared Spectroscopy (FTIR) Spectra of Pure Bosentan Monohydrate and Physical Mixture.
Powder X-ray Diffraction Studies
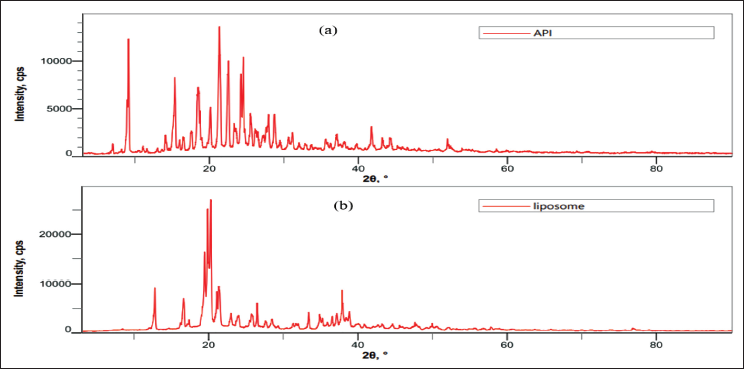
The X-ray powder diffraction (XRD) examination of pure drug and optimized liposome formulation reveals significant insights into the physical state of bosentan monohydrate. The diffractograms indicate the broader as well as decrease in a number of peak intensities characteristic of bosentan monohydrate within the liposomal formulations compared to pure drugs, as shown in Figure 3. This broader and decrease peak intensities suggest that bosentan monohydrate exists in an amorphous or disordered crystalline phase within the lipid matrix compared to a pure drug. This transformation from a crystalline to an amorphous state is crucial as it may enhance the drug’s solubility and bioavailability, highlighting the efficacy of the liposomal formulation.
Fourier Transform Infrared Spectroscopy (FTIR) Spectra (a) Pure Drug (Bosentan Monohydrate), (b) Physical Mixture of Final Formulation.
X-ray Powder Diffraction (XRD) Spectra of (a) Pure Bosentan Monohydrate, (b) Optimized Liposome Formulation.
Formulation Optimization
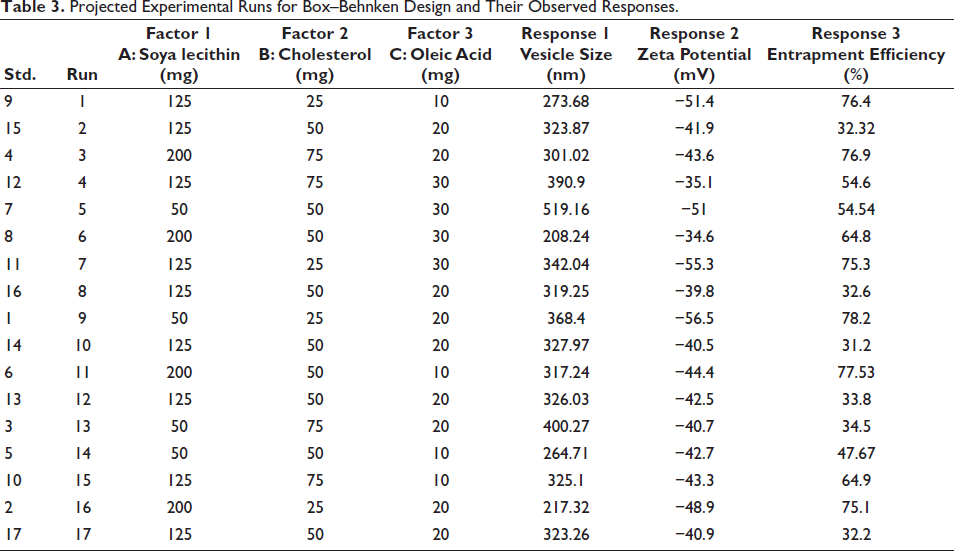
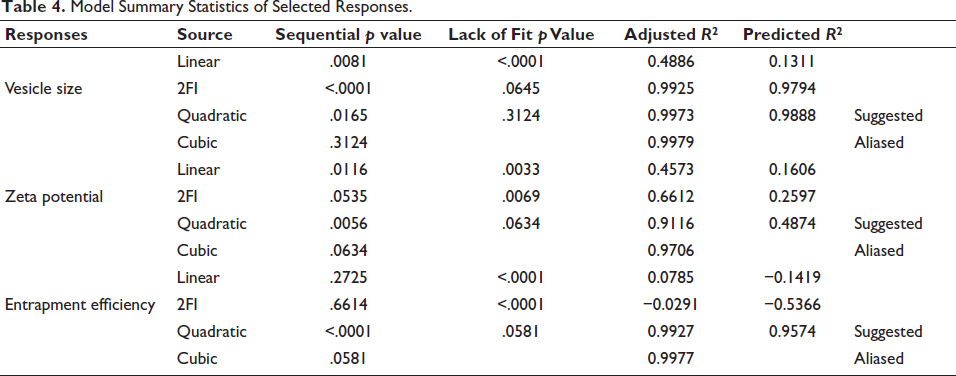
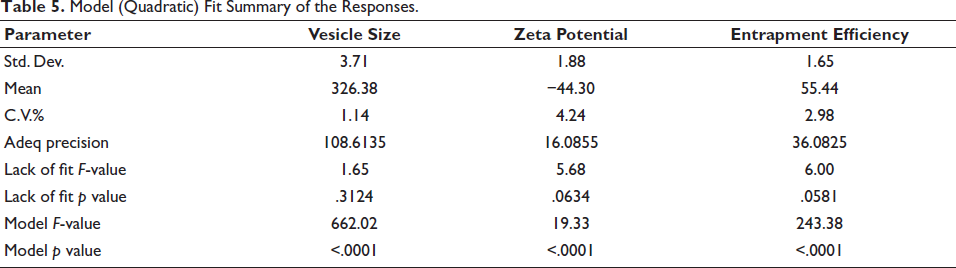
The Box–Behnken design with three factors and three levels was used in the RSM to identify the optimal concentrations and interactions of factors affecting particle size, ZP, and entrapment efficacy. Seventeen experimental runs were conducted, with results shown in Table 3. The particle size (VS) varied from 208.24 to 519.16 nm, the ZP ranged from −34.6 to −56.5, and the EE ranged from 31.2% to 78.2%. These results were analyzed for individual responses and parameter effects using statistical models and ANOVA. The quadratic model was chosen for all responses based on model summary statistics, Type-I sequential sum of squares, and fit summary.
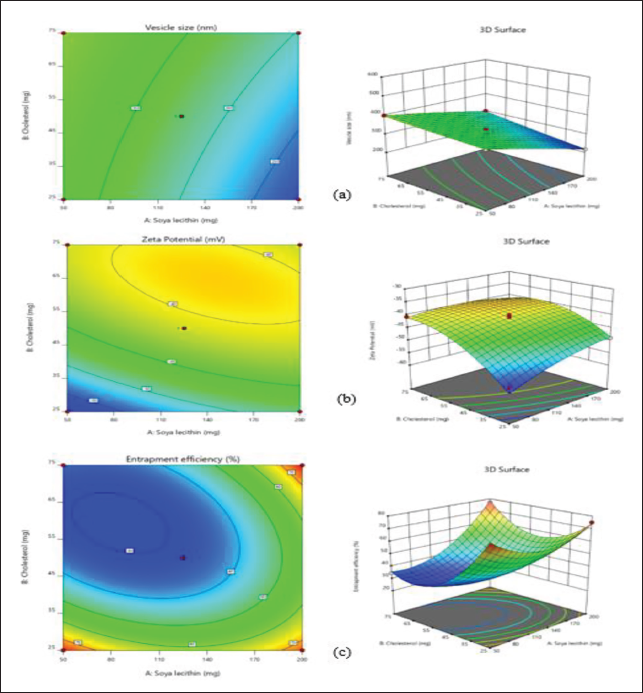
Three-dimensional plots are useful for illustrating how interactions between chosen factors affect the selected responses. The quadratic model was chosen for all results, as indicated by the Type-I sequential sum of squares and the fit summary, due to its superior F-value, p value, and R2 value. Notably, the quadratic model had the smallest p value (.0001) among all polynomial orders (Table 4 and Figure 4). The ANOVA coefficients provided can help identify which factors are significant and assess their impact. To illustrate the primary effects of these factors and their interactions, contour plots and 3D response surface graphs (RSGs) are required (Figure 5). Using the desired function [D], various models produced from an experiment can be optimized by setting minimum PS, maximum ZP, and maximum EE as constraints for each result, among other parameters, to create an overlay graph (Table 5). At the optimal levels of the independent variables, the combined desirability plots achieved the highest D value of 0.962, and the most critical responses were clustered together in the overlay plot (Figure 6). Using these predicted ideal ratios of 200 mg of soya lecithin, 71.06 mg of cholesterol, and 29.99 mg of oleic acid, an optimized liposome formulation was developed and tested. The results of this experimental formulation were then compared to theoretical predictions to validate the setup and assess its effectiveness.
Projected Experimental Runs for Box–Behnken Design and Their Observed Responses.
Model Summary Statistics of Selected Responses.
Model (Quadratic) Fit Summary of the Responses.
The experimental and predicted values showed a strong correlation with the chosen responses. The probability distribution confirms that the residuals are evenly distributed, indicating a linear pattern. Standard statistical methods may not be suitable, but examining the visible plot is effective. Additionally, a general residual plot (external studentized residuals vs. standard probability percent) was used to assess and validate the accuracy of the modified model (Figure 4).
Normal Probability and Model Residuals Versus Test Orders for (a) Vesicle Size (VS), (b) Zeta Potential (ZP), and (c) Entrapment Efficiency (EE).
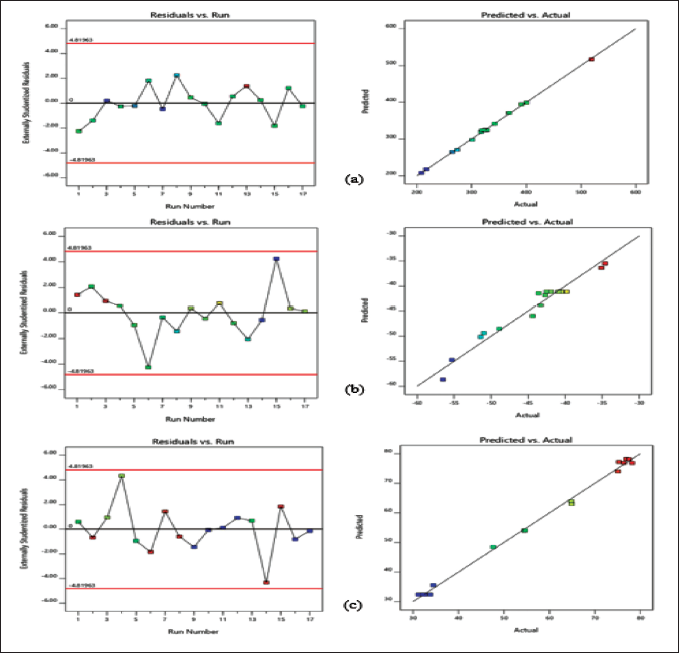
To evaluate the impact of the measurable effects of the factor treatments, an ANOVA was run. Multiple regression analysis was used to create polynomial equations from the data. The formulas produced by the optimum model’s outcomes are listed below:
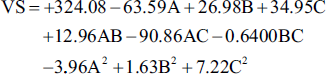
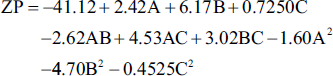
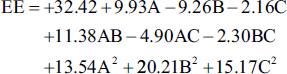
Table 6 presents the p values and ANOVA coefficients for each of the two replies. The model coefficients’ relevance was evaluated using these findings. Furthermore, RSM was used to analyze and understand the effects of each parameter on the responses, as illustrated in Figure 5.
Contour Plots and 3D Response Surface Plots for (a) Vesicle Size (VS), (b) Zeta Potential (ZP), and (c) Entrapment Efficiency (EE).
Overlay Plot.
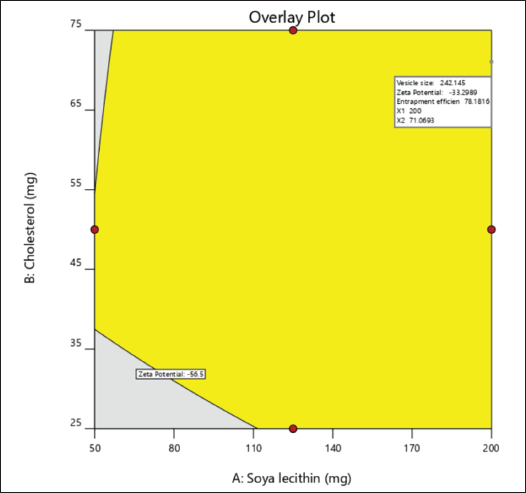
The model order obtained from statistical analysis was standardized using the desirability function (D). The ideal levels of the variables that were independent in the desirability curve plot correlated to the highest D value of 0.962 for both replies. Subsequently, this configuration achieved a VS of 242.117 nm, a ZP of −33.2988, and an EE of 78.1999. The relative error, obtained by comparing predicted and experimental values, is presented in Table 6.
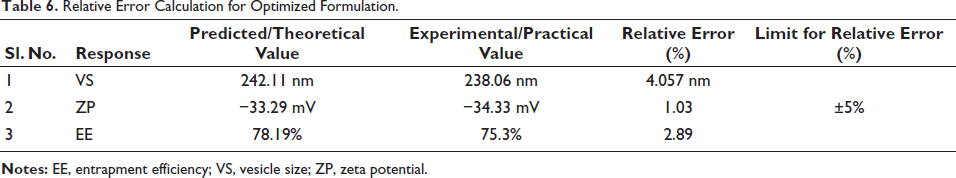
Relative Error Calculation for Optimized Formulation.
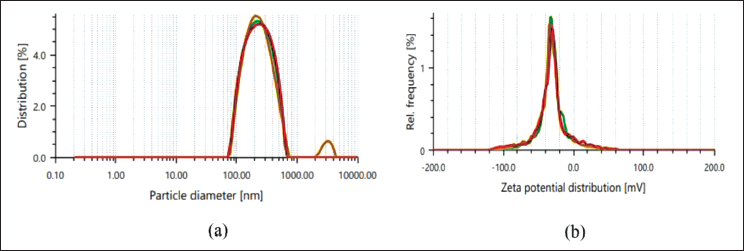
VS and ZP
The optimized formulation was characterized to assess its VS and ZP using DLS and ZP analysis techniques, respectively, both of which are critical parameters influencing the stability and biological performance of liposomal formulations. The average VS of the prepared liposomes was found to be 238.06 nm as portrayed in Figure 7a, as determined by DLS. The narrow size distribution, with a polydispersity index (PDI) of 0.252, suggests a fairly uniform and homogenous population of liposomes.
The ZP values were measured to assess the external charge and stability of the optimized liposome formulation. The formulated liposomes exhibited a ZP of −34.33 mV as portrayed in Figure 7b, suggesting high stability. This greater surface charge is indicative of more electrostatic repulsion between liposome vesicles, which helps prevent aggregation and maintains high stability.
(a) Vesicle Size (VS) Distribution, (b) Zeta Potential (ZP) of the Optimized Formulation of Liposomes.
EE
The EE of the enhanced liposomal composition was assessed to determine the capacity of the liposomes to encapsulate the active therapeutic agent. The EE values were determined by using quantitative analysis through UV-spectroscopy. The results demonstrated a significant EE of 75.3% for the optimized liposomal formulation.
Photo-microscopic and Scanning Electron Microscopic Study of Optimized Liposomes
The morphological characteristics of the optimized liposomes were meticulously analyzed using photo microscopy and SEM. Photo-microscopic analysis revealed that the liposomes exhibited a predominantly spherical shape, confirming the successful formation of the vesicular structures (Figure 8a). Further structural elucidation using SEM provided detailed insights into the surface morphology of liposomes. The SEM images highlighted the smooth and intact surface of the liposomes, indicating the absence of any aggregation or fusion phenomena (Figure 8b), supporting their suitability for transdermal applications. Additionally, the high-resolution SEM analysis revealed the precise structural integrity of the liposomes, showcasing their potential for enhanced stability and encapsulation efficiency. Additionally, the high-resolution SEM analysis revealed the precise structural integrity of the liposomes, showcasing their potential for enhanced stability and encapsulation efficiency. The combined findings from the photo-microscopy and SEM studies ensure their efficacy in the transdermal therapeutic delivery system.
(a) Photo-microscopic Image, (b) Scanning Electron Microscopy (SEM) Image of Liposomes.
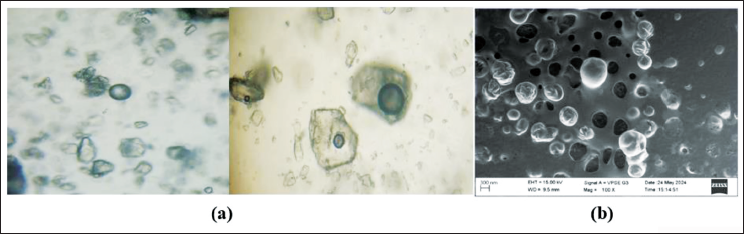
Photo-microscopic and SEM Study of Dissolving MNs
Microscopic analyses of DMNs revealed their structural integrity and uniformity (Figure 9). High-resolution images showed sharp tips essential for effective skin penetration, with MNs uniformly coated in the therapeutic agent.
Image of Microscopic Analyses of Dissolving Microneedles (DMNs).

In Vitro Drug Release and Drug Release Kinetic Data of Dissolving MNs
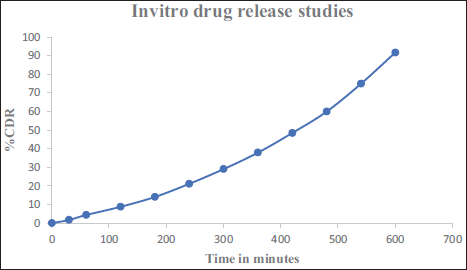
Figure 10 illustrates an in vitro study of drug release from a dissolving microneedle patch containing bosentan monohydrate-loaded liposomes, showing the cumulative drug release (CDR) percentage over time, measured in minutes. Before being released, bosentan monohydrate must migrate from the lipid bilayer to the outer surface of the liposomes. The water-soluble PVP, PVA, and PEG400 dissolve in the buffer solution to facilitate this process. The medication is released as soon as bosentan monohydrate comes into contact with the dissolving media. The CDR increases steadily over time, indicating a gradual release of the drug. The trend suggests a controlled release over time, with the drug release reaching close to 90% CDR at around 600 min.
In Vitro Drug Release Study of Optimized Liposome Loaded Dissolving Microneedle Array Patches.
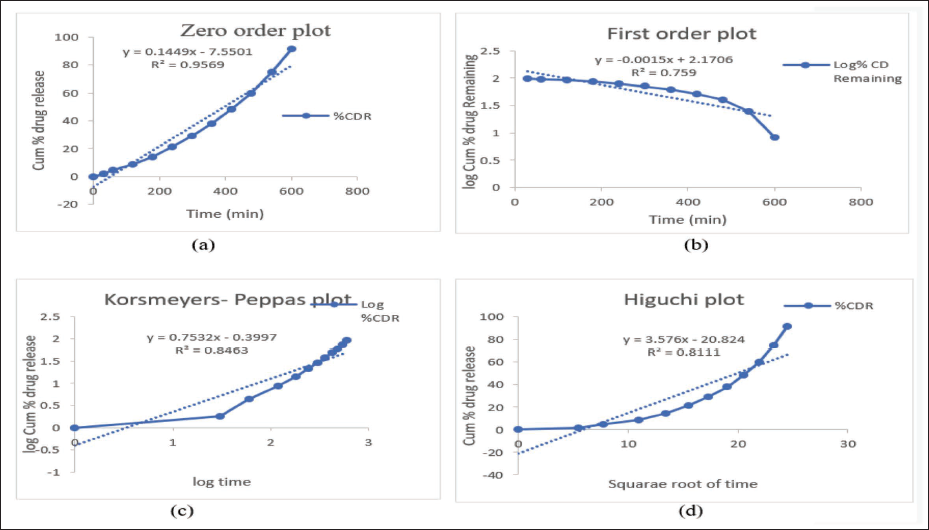
In the drug release kinetic studies, the release profiles of the optimized liposomes loaded DMNs patch were evaluated to understand the release mechanism and rate. The data, plotted as CDR over time, exhibited distinct kinetic patterns. The release kinetics were analyzed using various models (Figure 11a-d), such as zero-order, First-order, Korsmeyer–Peppas, and Higuchi models, to determine the best fit for the observed release data. The medication release followed zero-order, according to the data, suggesting regulated release.
Zero-order drug release is preferred for treating PH because it ensures consistent drug levels, avoiding peaks and troughs that can cause side effects or reduced efficacy. This steady release enhances therapeutic outcomes, aligns with the chronic nature of the disease, improves patient compliance, and minimizes dosing frequency and systemic side effects.
(a) Zero-order, (b) First-order, (c) Korsmeyer–Peppas, (d) Higuchi Plot of Drug Release Kinetic Studies of Optimized Liposome Loaded Microneedles (MNs) Array Patch.
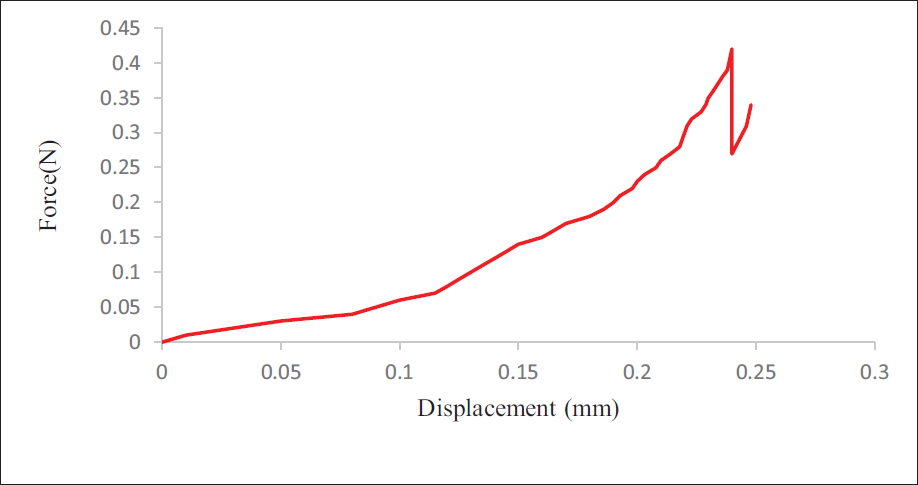
Analysis of the DMNs Mechanical Strength
In the study of the mechanical strength of DMNs, the fracture point was recorded at 0.42 N strength (Figure 12). This indicates the MNs capability to withstand the stress before breaking, which is crucial for ensuring effective and reliable drug delivery in the transdermal therapeutic system.
Mechanical Property of Microneedle Array Under a Compression Force.
Insertion Study of DMNs
Successful transdermal drug delivery depends on MNs’ capability to penetrate the skin with sufficient mechanical force. In this study, a 15 × 15 array of 225 MNs was gently applied to the skin. While rat skin is inherently elastic, it rapidly loses its elasticity with increased force. As seen in Figure 13, almost all of the MNs effectively penetrated the skin.
Microneedle Array Patch Skin Penetration Investigation Following Rat Skin Implantation.

Discussion
The chronic, progressive illness known as PH has a variety of origins, typically involving harmful changes in the small arteries of the lungs or a congestive vasculopathy brought on by elevated pulmonary vein pressure, which may result in right heart failure and, if not treated, eventually death. Bosentan is a dual endothelin receptor antagonist that is derived from sulfonamide and pyrimidine and is mainly used to treat PAH. The microneedle technology has received attention recently as a method for the controlled and prolonged release of medications. MNs array patches are a non-invasive, user-friendly microinjection tool made up of tiny needles (ranging from 25 to 1,500 µm) that, when applied to the skin, puncture the outer layer (stratum corneum) and form temporary micro-channels. The IR spectroscopy method was employed to investigate drug-polymer interactions. About 5 mg of the sample was combined with dry KBr, and the spectra were recorded across a wavelength range of 4,000-400 cm⁻ 1 . By comparing the spectra of the pure drug with those of the physical mixture containing bosentan monohydrate, soya lecithin, cholesterol, PVA, and PVP, all drug peaks were observed in the physical mixture. The XRD analysis of the pure drug and the optimized liposome formulation provides valuable insights into the physical state of bosentan monohydrate. The diffractograms indicate a broader as well as decrease in the number of peak intensities characteristic of bosentan monohydrate within the liposomal formulations compared to the pure drug. This broader and decrease in peak intensities suggests that bosentan monohydrate exists in an amorphous or disordered crystalline phase within the lipid matrix compared to the pure drug. This transformation from a crystalline to an amorphous state is crucial as it may enhance the drug’s solubility and bioavailability, highlighting the efficacy of the liposomal formulation in stabilizing and delivering bosentan monohydrate in a more readily absorbable form. The Box–Behnken design consists of three factors, and three levels were used in the RSM to identify the optimal concentrations and interactions of factors affecting particle size, ZP, and entrapment efficacy. Seventeen experimental runs were conducted, with results shown in Table 3. The particle size (VS) varied from 208.24 to 519.16 nm, the ZP ranged from −34.6 to −56.5, and the EE ranged between 31.2% and 78.2%. The optimized formulation was characterized to assess its VS and ZP using DLS and ZP analysis techniques, respectively, both of which are critical parameters influencing the stability and biological performance of liposomal formulations. The average VS of the prepared liposomes was found to be 238.06 nm, as portrayed in Figure 7a, as determined by DLS. The narrow size distribution, with a PDI of 0.252, suggests a fairly uniform and homogenous population of liposomes, which is crucial for their application in transdermal patches to ensure consistent drug delivery.
The ZP values were measured to assess the external charge and stability of the optimized liposome formulation. The formulated liposomes exhibited a ZP of −34.33 mV, suggesting high stability. This greater surface charge is indicative of more electrostatic repulsion between liposome vesicles, which helps in preventing aggregation. The EE of the enhanced liposomal composition was assessed to determine the capacity of the liposomes to encapsulate the active therapeutic agent. The EE values were determined by using quantitative analysis through UV-spectroscopy. The results demonstrated a significant EE of 75.3% for the optimized liposomal formulation. The morphological characteristics of the optimized liposomes were meticulously analyzed using photo-microscopy and SEM. Photo-microscopic analysis revealed that the liposomes exhibited a predominantly spherical shape, confirming the successful formation of the vesicular structures. Further structural elucidation using SEM provided detailed insights into the surface morphology of liposomes. The SEM images highlighted the smooth and intact surface of the liposomes, indicating the absence of any aggregation or fusion phenomena, supporting their suitability for transdermal applications. Microscopic analyses of DMNs revealed their structural integrity, and high-resolution images showed sharp tips essential for effective skin penetration, with MNs uniformly coated in the therapeutic agent. In vitro study of drug release from a dissolving microneedle patch containing bosentan monohydrate-loaded liposomes, showing the CDR percentage over time, measured in minutes. To be released, the water-soluble PVP, PVA, and PEG400 dissolved in the buffer solution. Then, before being released, bosentan monohydrate needs to go from the bilayer of lipids to the liposomes’ outer surface. The medication is released as soon as bosentan monohydrate comes into contact with the dissolving media. The CDR increases steadily over time, indicating a gradual release of the drug. The trend suggests a controlled release over time, with the drug release reaching close to 90% CDR in 5 h. The mechanical strength of DMNs, the fracture point was recorded at 0.42 N strength. This indicates the MNs capability to withstand the stress before breaking, which is crucial for ensuring effective and reliable drug delivery in the transdermal therapeutic system. Optimal transdermal drug delivery depends on MNs’ capability to penetrate the skin with sufficient mechanical force. In this study, a 15 × 15 array of 225 MNs was gently applied to the skin. While rat skin is inherently elastic, it rapidly loses its elasticity with increased force. As seen in Figure 13, almost all of the MNs successfully pierced the skin.
Conclusion
In this research, a 3-factor, 3-level Box–Behnken design was utilized to optimize liposome formulations for transdermal drug delivery, focusing on VS, ZP, and EE. The study involved 17 experimental runs, resulting in liposomes with VSs ranging from 208.24 to 519.16 nm, ZPs between −34.6 and −56.5 mV, and entrapment efficiencies between 31.2% and 78.2%. The optimized liposomes exhibited an average VS of 238.06 nm and a ZP of −34.33 mV, indicating a stable formulation with a PDI of 0.252, which is favorable for consistent drug delivery. The EE was notably high at 75.3%, demonstrating the liposomes’ effective encapsulation capacity. Morphological analysis through photo-microscopy and SEM confirmed that the liposomes were predominantly spherical with a smooth surface, free from aggregation. Additionally, the DMNs, loaded with the optimized liposomes, showed structural integrity and uniformity, with sharp tips essential for skin penetration. In vitro drug release studies indicated a controlled release of bosentan monohydrate, with a CDR of close to 90% over 600 min. Kinetic analysis revealed that the release followed zero-order kinetics, suggesting a controlled release mechanism. The mechanical strength of the MNs was satisfactory, with a fracture point of 0.42 N, ensuring their effectiveness in drug delivery. The study concludes that the optimized formulation of liposomal-loaded DMNs is easy to apply, non-invasive, biodegradable, and effective for transdermal drug delivery, offering a promising approach for controlled drug release with high encapsulation efficiency and stability for therapeutic applications.
Footnotes
Abbreviations
ANOVA: Analysis of variance; CAD: Computer-aided design; CDR: Cumulative drug release; CV: Coefficient of variation; DLS: Dynamic light scattering; DMNs: Dissolving microneedles; DoE: Design of experiments; EE: Entrapment efficiency; ET: Endothelin; FTIR: Fourier transform infrared spectroscopy; MNs: Microneedles; PAH: Pulmonary arterial hypertension; PBS: Phosphate buffer saline; PCWP: Pulmonary artery wedge pressure; PDE5i: Phosphodiesterase-5 inhibitors; PDI: Polydispersity index; PDMS: Polydimethylsiloxane; PH: Pulmonary hypertension; PRA: Prostacyclin receptor antagonist; PVA: Polyvinyl alcohol; PVP-K30: Polyvinylpyrrolidone; RHC: Right heart catheterization; RSM: Response surface methodology; SEM: Scanning electron microscopy; SLA: Stereolithography; VS: Vesicle size; XRD: X-ray powder diffraction; ZP: Zeta potential.
Acknowledgments
The authors acknowledge Sri Adichunchanagiri College of Pharmacy, King Faisal University and Vidya Siri College of Pharmacy, Bangalore for providing support to carry out the present work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Grant No. KFU242975].
