Abstract
Background
Diabetes mellitus is a chronic metabolic disorder and a fast-growing global problem. Studies have shown that Linum usitatissimum has anti-inflammatory and antioxidant properties that decrease blood sugar and cholesterol levels, and Moringa oleifera possesses hepatoprotective, anti-inflammatory and hypoglycaemic effects. Hence, this study investigated both extracts’ hypolipidemic and hepatoprotective effects on diabetes.
Objectives
To study the improvement in glucose level, lipid levels (total cholesterol, high-density lipoprotein, low-density lipoprotein) and hepatic parameters after giving L. usitatissimum and M. oleifera seed extract combination in diabetic rats.
Materials and Methods
A total of 36 healthy male Wistar rats were taken and divided into six groups (groups I–VI). Diabetes was induced by administering a single intraperitoneal injection of streptozotocin in overnight fasted rats. The mixture of aqueous extract of L. usitatissimum and M. oleifera seeds was prepared and given to rats of groups IV–VI daily for 4 weeks. After 28 days, the blood sample was collected from all the rats, and biochemical parameters were determined.
Results
All the values are expressed as mean ± standard deviation (SD). Significant decreases in lipid levels, urea, creatinine and liver function tests were seen at the end of 1 month in all the three groups receiving the extract.
Conclusion
From our study, it was found that the administration of L. usitatissimum and M. oleifera seeds extract possesses excellent hypolipidemic and hepatoprotective properties, making them effective in preventing long-term complications of diabetes.
Keywords
Introduction
Diabetes represents a complex systemic ailment characterised by deficient insulin secretion and disruptions in fat and protein metabolism. In India, a substantial 11.8% of the population grapples with this condition, with a higher prevalence observed in urban locales than in rural areas. 1 Current therapies for diabetes encompass oral hypoglycaemic agents such as glipizide, gemigliptin and glibenclamide, as well as injectable insulin, they are not without their limitations and drawbacks. Thus, there arises a critical need to explore alternative treatments that are effective, cost-efficient and exhibit fewer side effects, particularly in the realm of herbal hypoglycaemic agents. Recent research highlights the potential of various plant extracts to lower blood glucose levels in experimental animal models, with perceived lower toxicity compared to synthetic counterparts. 2
In the context of dietary interventions for managing diabetes, flaxseed (Linum usitatissimum) emerges as a noteworthy contender. Renowned for its rich lignan content, including secoisolariciresinol diglucoside (SDG), and essential fatty acids like α-linolenic acid (ALA). Flaxseed also boasts a notable amino acid profile featuring arginine, glutamic acid and aspartic acid. Its medicinal properties, ranging from anti-inflammatory to antioxidant effects, contribute to reducing blood sugar and cholesterol levels, enhancing insulin sensitivity and mitigating the risk of obesity.3, 4
Similarly, tropical endemic Moringa oleifera has many uses in traditional medicine. It has been suggested that this plant’s leaf has antioxidant and therapeutic qualities that could aid in the management and treatment of diabetes and its consequences. M. oleifera, with its protein-rich composition, abundant nutrients and diverse array of antioxidants such as phenolic acids, tannins, flavonoids and glucosinolates, has garnered attention for its pharmacological potential. Studies indicate its hepatoprotective, anti-inflammatory and anti-diabetic properties.5–7
Thus, this study endeavours to assess the combined impact of Linum and Moringa extracts on lipid levels and liver function tests (LFT) in diabetic-induced rats. By targeting these aspects, the aim is to mitigate the long-term consequences of diabetes and possibly provide novel avenues for disease management.
Materials and Methods
The study was conducted in the animal house, located in the Department of Pharmacology after getting approval from the Institutional Animal Ethics Committee (Ref. No KIIT/KIMS/IAEC/08/21) at Kalinga Institute of Medical Sciences (KIMS), Bhubaneswar, Odisha and protocols were followed according to the guidelines of the committee for the purpose of control and supervision of experiments on animals (CPCSEA). The study period was 1 month (30 days). Seeds of L. usitatissimum (flax seeds—Figure 1) and M. oleifera (Moringa seeds—Figure 2) were purchased from a local market. Streptozotocin was purchased from Hi-Media Laboratory Pvt., Ltd., India. Glucometer, Glimepiride, Metformin and Mupirocin ointment were procured from KIMS Pharmacy.
Flax Seed.

Moringa Seeds.

Preparation of Seed Extract
L. usitatissimum and M. oleifera seeds were taken and shade dried. Their outer shells were removed manually. Seeds were then grounded in mortar and pestle and then soaked in distilled water separately. For this purpose, about 100 g of the powdered material was soaked in 2 L of distilled water and they were shaken thrice daily. After that the solution was sieved using a sieve of a particular mesh size or through a Mushin cloth. Then, the extract was concentrated by using a rotary evaporator under 50–60°C. The aqueous extract obtained was collected in an airtight glass container in a refrigerator at 2–8°C for further use in the experiment. Respective doses for the study were prepared by reconstituting the extract in distilled water. After the extracts were prepared, L. usitatissimum and M. oleifera extracts were taken in 4:1 ratio and mixed thoroughly. 4 Then, the sample was stored in an airtight glass container in the refrigerator at 2–8°C for further use in the experiment.
Grouping of Animals
For the experiment to be conducted, 36 rats were taken and were divided into six groups.
Group I—Vehicle control: rats will receive a regular diet and saline.
Group II—Diabetic control (rats are diabetic and will receive a normal diet).
Group III—Treatment control, diabetic rats treated with glimepiride (1 mg/kg) and metformin (500 mg) combination.
Group IV—Diabetic rats were treated with 1 mL extract (mixture of L. usitatissimum and M. oleifera seed extract at ratio 4:1) (100 mg/kg).
Group V—Diabetic rats were treated with 2 mL extract (mixture of L. usitatissimum and M. oleifera seed extract at ratio 4:1) (200 mg/kg).
Group VI—Diabetic rats were treated with 4 mL extract (mixture of L. usitatissimum and M. oleifera seed extract at ratio 4:1) (400 mg/kg).
Induction of Experimental Diabetes
Experimentally, diabetes was induced in rats by administering a single intraperitoneal (i.p.) injection of streptozotocin at 40 mg/kg in overnight fasted rats in groups II–VI. 5 All animals received 5% glucose solution orally in order to prevent drug-induced hypoglycaemia in the early phase. Blood glucose level was measured through tail tipping using a glucometer at 24, 48, 96 and 168 h after streptozotocin administration, and blood glucose levels above 13.8 mmol/L (250 mg/dL) were considered diabetic and included in the study.
Experimental Design
The study was conducted on 36 male albino rats weighing between 190 and 200 g. Those rats were familiarised with the experimental conditions for 6 months and were grouped and housed in standard rat cages during the experiment. They were maintained at 25°C and provided standard laboratory animal feed and water throughout the experimental period of 1 month.
After induction of diabetes, the rats (groups IV–VI) were treated with the extract (1, 2 and 4 mL mixture of L. usitatissimum and M. oleifera seed extract at a ratio of 4:1, respectively) for 4 weeks. The blood sample was collected on day 0 (3 days after induction of diabetes) and at the end of the 4th week through retro-orbital plexus puncture for the biochemical parameter estimation like total cholesterol (TC), high-density lipoprotein (HDL), low-density lipoprotein (LDL), alanine transaminase (ALT), alkaline phosphatase (ALP) and aspartate aminotransferase (AST).
Estimation of Biochemical Parameters
During the experiment, the random blood glucose levels were estimated every week (0, 7, 14, 21 and 28 days) to assess the change in glycaemic parameters via glucometer.
Also, lipid levels (TC, HDL, LDL) and liver functioning test (AST, ALT, ALP) were measured on days 0 and 28. The biochemical parameters were carried out in the KIMS laboratory in Bhubaneswar. Blood sugar level was estimated by glucose O-toluidine method. 6 The lipid profile was estimated by the CHOD POD method. 7 Triglycerides and VLDL were estimated by enzymatic colorimetry. 8 HDL was estimated by the dextran sulphate method. 9 LDL was measured by estimating the cholesterol content. 10
Statistical Analysis
All the values are expressed as a Mean ± Standard deviation (SD). The data were analysed by one-way analysis of variance (ANOVA) and post hoc methods using socscistatistics.com. A level of p < 0.05 was considered statistically significant. A level of significance was noted and interpreted accordingly.
Results
This study was carried out in the animal laboratory of the Pharmacology Department in KIMS, Bhubaneswar. The study was initiated only after getting approval from the Institutional Animal Ethics Committee (IAEC). The seeds of L. usitatissimum and M. oleifera were bought from the local market and were authenticated. After that, their extract was (as mentioned above) and graded doses were taken based on the toxicity studies. A total of 36 male Albino Wistar rats weighing 190–200 g were selected for the analysis and divided into six groups containing six rats each. Diabetes was induced in rats by using streptozotocin. Group I was the vehicle control, group II was diabetic control and group III was treatment control. Groups IV–VI were test treatment control groups receiving varying doses of extract of the seeds. Random blood glucose levels were noted on days 0, 7, 14, 21 and 28. The hypolipidemic effect was observed by measuring the lipid parameters (TC, LDL, HDL) while the hepatoprotective effect was observed by measuring the hepatic parameters (AST, ALT, ALP) at the end of the 28th day.
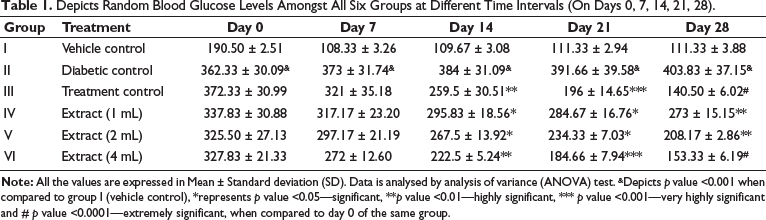
Table 1 shows a significant reduction in blood glucose parameters, showing the glycaemic effect of the drug combination in diabetic rats after the extract was given at different doses.
Depicts Random Blood Glucose Levels Amongst All Six Groups at Different Time Intervals (On Days 0, 7, 14, 21, 28).
From Table 2, it is observed that there is a significant reduction in LDL and TC level and increased HDL level, showing the hypolipidemic effect of drug after giving the extract at different doses.
Depicts the Evaluation of Lipid Parameters (TC, LDL and HDL) Amongst All Six Groups, at the End, that is, 28th Day.

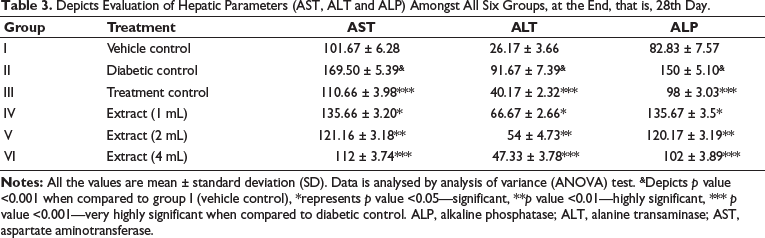
From Table 3, it is observed that there is a significant reduction in AST, ALT, ALP levels, showing the hepatoprotective effect of the drug after giving the extract at different doses.
Depicts Evaluation of Hepatic Parameters (AST, ALT and ALP) Amongst All Six Groups, at the End, that is, 28th Day.
Discussion
Diabetes mellitus encompasses various disorders characterised by glucose intolerance and elevated blood sugar levels due to insulin dysfunction. 11 Prolonged hyperglycaemia can lead to severe damage to the nerves and blood vessels, resulting in conditions like heart attacks, blindness, stroke, kidney failure and limb amputation. 12
According to the International Diabetes Federation (IDF), in 2010, diabetes affected 6.4% of the global population, that is, about 285 million people, with projections estimating an increase to 7.7%, that is, about 439 million people. Diabetes contributed to 1.5 million fatalities in 2019, of which 48% occurring were under the age of 70. However, the adverse effects of diabetes can be mitigated through medication, diet, physical activity, treatment and regular screenings for complications. 13
Various drugs have been developed to regulate blood sugar levels in individuals with type II diabetes, including insulin, oral anti-diabetic agents (OAAs) and incretin-based drugs. Among OAAs, sulfonylurea-type drugs like glimepiride and glibenclamide stimulate pancreatic cells to release insulin, lowering blood glucose levels. Insulin sensitizers such as TZDs (pioglitazone and rosiglitazone) and metformin directly reduce blood glucose levels and insulin resistance. 14
However, current anti-diabetic medications often lack efficacy and can cause undesirable side effects. For example, insulin secretagogues are associated with hypoglycaemia, weight gain and β cell death. Moreover, existing drugs are primarily designed to manage diabetes rather than cure it. Metformin, derived from the French lilac plant, has become a leading treatment for type II diabetes. It is essential to explore effective, low-cost and safer herbal hypoglycaemic agents as alternatives. 15
Among indigenous remedies, L. usitatissimum (flaxseed) and M. oleifera seeds stand out as particularly effective in addressing the damage caused by diabetes. Belonging to the Linaceae family, flaxseed, also known as linseed or common flax, is a flowering plant. It is rich in dietary fibre, several B vitamins, protein and minerals. Flaxseed contains both insoluble and soluble fibre, with 10 g providing 3 g of insoluble fibre and 1 g of soluble fibre, which aids in lowering blood cholesterol levels. Flaxseeds are abundant in omega-6 fatty acids (linoleic acid), omega-3 fatty acids (primarily ALA) and omega-9 fatty acids (oleic acid), along with saturated fat and palmitic acid. 16
Lignans present in flaxseeds act as potent antioxidants, enhancing insulin sensitivity and slowing down the progression of diabetes. It also has been found to inhibit the phosphoenolpyruvate carboxykinase gene expression, which codes for a crucial enzyme responsible for liver glucose synthesis. They help reduce blood glucose and insulin levels. The soluble mucilaginous dietary fibres, omega-3 fatty acids and lignans in flaxseed contribute to stabilizing blood sugar levels. The insoluble fibre, rich in lignans, takes longer to digest, resulting in a gradual release of sugar into the bloodstream. Antioxidant properties of lignans further support insulin sensitivity and hinder the advancement of diabetes. 17 Additionally, it helps reduce fasting blood glucose, glycated haemoglobin and overall glycaemia, potentially lowering the risk of heart disease. This could be attributed to its high dietary fibre content and low glycaemic carbohydrate composition. 18 According to research by Kelley et al., blood sugar levels in rats fed 0.5% conjugated linoleic acid and 0.5% flax oil for 4 weeks decreased by 20%. 19
In the treatment and management of diabetes, M. oleifera seeds have shown promise as beneficial agents. Studies have revealed their hypolipidemic, hypoglycaemic, anticancer, antioxidant and hepato-protective effects, along with anti-inflammatory activity. Moringa seeds have been shown to lower fasting blood glucose levels and enhance serum antioxidant enzymes making it a useful treatment for Types 1 and 2 diabetes. Flavonoids present in Moringa scavenge reactive oxygen species from mitochondria, protecting beta cells and helping to control hyperglycaemia in diabetic patients. 20
In this study, rats were given a single intraperitoneal injection of streptozotocin following overnight fasting to induce diabetes. Rats in groups II–VI received streptozotocin at 40 mg/kg dose. Following diabetes induction, rats in groups IV–VI were treated with a mixture of L. usitatissimum and M. oleifera seed extract at varying doses (1, 2 and 4 mL, respectively) for 4 weeks. Blood samples were collected via retro-orbital plexus puncture at day 0 and the end of the 4th week.
The study revealed progressive changes in biochemical parameters such as ALT, TC, LDL, HDL, AST and ALP. The diabetic control group experienced a considerable increase in blood glucose levels, but the vehicle control group showed negligible variation in these values. When the mixed extract was administered, blood glucose levels gradually decreased. The highest dose of the extract demonstrated a more significant reduction, comparable to the standard group treated with a combination of glimepiride and metformin (Table 1).
The extract appears to contain biologically active substances with hypoglycaemic properties, such as flavonoids and tannins, based on the observed decrease in glycaemic indices. This may be explained by increased insulin synthesis, higher utilization of glucose in the periphery, inhibition of the body’s synthesis of glucose, or inhibition of intestinal absorption of glucose. 21 Similar findings were reported in studies by Kapuriya et al. and Woldekidan et al.22, 23
Compared to non-diabetic rats, diabetic rats had higher serum levels of lipids such as triglycerides, LDL and TC. This suggests that insulin insufficiency has caused dysregulation in metabolic pathways. Administration of the combined extract daily significantly reduced levels of these lipids, including TC, HDL, LDL and triglycerides, compared to diabetic control rats. The highest dose of the extract showed a more pronounced reduction in TC and LDL levels, comparable to the standard (glimepiride and metformin combination). This protective effect on membrane-bound lipoprotein lipase, which is involved in the hydrolysis of triglycerides and is decreased in diabetes, is thought to be the cause of this effect, which inhibits the oxidation of lipids. These findings suggest the extract’s potential in preventing diabetic complications and improving lipid metabolism in diabetics. Similar results on hypolipidemic parameters were reported in studies by Vargas-Sánchez et al. and Saleh et al.24, 25
High serum aminotransferase levels, which are frequently seen in diabetics, are linked to liver problems and diabetic sequelae, including neuropathy, retinopathy and restricted joint movement. Despite metabolic regulation and body mass index, these enzyme activities continue to change. 26
Higher levels of tissue toxicity biomarker enzymes, such as AST and ALT, were seen in the diabetic control group of rats in a 28-day study comparing rats with and without diabetes, suggesting hepatocellular injury. However, daily administration of combined seed extract resulted in a significant reduction in AST, ALT and ALP levels, particularly at higher doses, comparable to standard medication (glimepiride and metformin combination). This reduction may be attributed to decreased gluconeogenesis and ketogenesis, possibly due to improved insulin sensitivity.
Similar findings on AST, ALT and ALP parameters have been reported in previous studies, reinforcing the potential of seed extract in mitigating liver damage associated with diabetes. 27
The combination of L. usitatissimum and M. oleifera seed extract at a ratio of 4:1 demonstrates anti-glycaemic, hypolipidemic and hepatoprotective effects, as evidenced by reductions in blood glucose, lipid levels (TC and LDL) and liver enzymes (ALT, AST and ALP) levels.
Conclusion
From our study, it can be concluded that there was a significant reduction in glycaemic parameters as well as the plant extracts possess excellent hypolipidemic and hepatoprotective properties due to their content of antioxidant compounds such as phenols, glucomoringin and flavonoids. Therefore, it can be said that this extract is efficient in reducing the long-term complications of diabetes like dyslipidaemia. The presented results show that L. usitatissimum and M. oleifera seed extract can serve as a promising additional therapy approach in managing diabetes. The present study creates further opportunities to separate the active constituents from flaxseed and M. oleifera seeds that are accountable for anti-diabetic activity and to clarify their mechanism of action. More studies are required that drive primary preclinical and clinical outcomes together with standardized product invention development to optimize the dose of polyphenols and lignans within an edible product that could be administered along with classic therapy. Therefore, it can be said that this extract is effective in reducing the long-term difficulties of diabetes like nephropathy, dyslipidaemia and so on.
Footnotes
Abbreviations
Acknowledgments
The authors thank the staff and technicians of the Department of Pharmacology, KIMS, Bhubaneswar, Odisha, India.
Authors Contribution
Nitu Sahoo: Concept, design, definition of intellectual content, literature search, clinical studies, experimental studies, data acquisition, data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review.
Dilip Kumar Bishi: Concept, design, definition of intellectual content, literature search, clinical studies, experimental studies, data acquisition, data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review.
Vartika Srivastava: Concept, design, definition of intellectual content, literature search, clinical studies, experimental studies, data acquisition, data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review.
Chaitali Pattanayak: Concept, design, definition of intellectual content, literature search, clinical studies, experimental studies, data acquisition, data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review.
Sougata Sarkar: Data analysis, statistical analysis, manuscript preparation, manuscript editing and manuscript review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
This study has been approved by the IEAC, KIMS, Bhubaneswar.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
