Abstract
Background
Ageing is associated with progressive neuronal deficits, leading to cognitive decline and increased vulnerability to neurodegenerative diseases. Currently, there is no permanent treatment available which can cure ageing and associated neuronal deficits.
Objectives
The purpose of this investigation was to examine the neuroprotective effects of Ganoderma lucidum extract (GLE), both alone and in combination with alpha lipoic acid (ALA), on age-related neuronal deficits in old rats.
Materials and Methods
Eight groups of rats were used in this investigation. GLE and ALA were administered alone and in combination for 90 days orally. After 90 days, various physiological, behavioural, oxidative stress and histopathological parameters were assessed.
Results
Treatment with GLE and ALA significantly and dose-dependently modulates the neuronal deficits of aged rats. GLE 100 with ALA 50 showed synergistic effects on aged rats.
Conclusion
New recent data supports the benefits of GLE for cognitive deficits in aged rats.
Keywords
Introduction
As per the World Health Organisation (WHO), 80% of the population who are residing in low- and middle-income countries will be aged by 2050. 1 Ageing is generally characterised by the accumulation of a wide variety of molecular and cellular damage over time. 1 That results in function loss, biological and physiological damage, and the early stages of multiple age-related diseases. 2 The oxidative stresses and free radical deposition theories stand out among the various theories associated with ageing. 3 Two of the main consequences of neurodegenerative disorders, which include vascular dementia, Alzheimer’s disease (AD), amyotrophic lateral sclerosis, Parkinson’s disease (PD) and Huntington’s disease (HD), are progressive memory loss and an increasing inability to move. These diseases are especially prevalent in older populations.4, 5 It has been established that oxidative stress plays a part in the etiopathogenesis of memory impairment. 6 Several studies have investigated the relationship between certain levels of oxidative stress biomarkers and cognitive function as measured through Mini-Mental State Testing. According to a previous study, poor mental performance in elderly people living in institutions was linked to both higher levels of inflammatory cytokines and oxidative stress indicators, such as glutathione (GSH) peroxidase and malondialdehyde (MDA). 7 Another study found that, even though age is the primary cause of cognitive impairment, mental decline is slow in patients with elevated GSH peroxidase activity. In contrast, high GSH levels appear to accelerate cognitive impairment in elder people. This appears to be a paradoxical finding because high intracellular GSH should protect cells from free radical damage. Because GSH is a substrate for GSH peroxidase, low enzyme activity may result in an increase in reactive oxygen species (ROS) and low GSH levels. 8
Ganoderma lucidum (GL), commonly known as Lingzhi in China and Reishi in Japan, is a fungus belonging to the Ganodermataceae family. For almost 4000 years, GL has been historically identified as a medicinal mushroom, and its profound effects have been described in ancient texts. 9 This woody, glossy, black mushroom is widely distributed in China, Japan and other Asian nations. 10 GL has traditionally been consumed for its nutritional and culinary properties, as well as for its medicinal properties when utilised in folk medicine. 10 There are many pharmacologically active compounds like triterpenoids, polysaccharides, flavonoids, polyphenols, alkaloids, glycoproteins, sesquiterpenoids, meroterpenoids, steroids, benzopyran derivatives and benzoic acid derivatives. 11 GL polysaccharides (β-glucans), triterpenoids (Ganoderic acid), and peptidoglycans are the three main active components that play an important role as an effective antioxidant. 12 Many contemporary pharmacological studies have demonstrated that GL is a mushroom that has been used for both medicinal and culinary purposes. It also has biological properties that include anti-inflammatory, anti-cancer, anti-oxidative, nerve-calming, immunomodulating anti-infection, liver-detoxifying, hepatoprotective, antiviral and anti-diabetic effects.13, 14 In vitro, GL polysaccharides and triterpenoids showed antioxidant activity15, 16 and were discovered to be quickly absorbed after ingestion, increasing the subjects’ total antioxidant activity in the blood. 10
Materials and Methods
Chemicals and Equipment
Drugs and Chemicals: Quercetin, gallic acid, nitrogen blue tetrazolium (NBT) and 2,2-diphenyl-1-picrylhydrazyl (DPPH) was purchased from Himedia, India. Folin-Ciocalteu’s reagent was purchased from Central Drug House (CDH), India, and alpha-lipoic acid (ALA) was purchased from Setu Nutrition Pvt. Ltd., India. All other chemicals used in this study were of analytical grade.
Sample Collection
The GL mushroom used in this study was procured and authenticated by the ICAR Directorate of Mushroom Research, Solan, Himachal Pradesh, India [Accession number: DMRX 779]. The mushroom was cleaned with distilled water followed by air drying, ground into coarse powder and stored in an airtight container for further use.
Extraction
The extraction was done with the help of a soxhlet extraction apparatus. The hydro-ethanolic solvent was used (i.e., 50% ethanol and 50% water). 17 The weighed quantity of powdered sample was loaded into the thimble. The extraction process was run for eight hours at 60–70℃. The extract was filtered using Whatman filter paper, which was evaporated through a rota evaporator. The collected extract was then further stored in a deep freezer at -20℃.
Animals and Treatments
Before the animal research began, the protocol for the experiment was approved by the MM College of Pharmacy’s Institutional Animals Ethics Committee (Protocol no. MMCP-IAEC-110, dated 20/11/2021) and carried out in accordance with the regulations on animal experimentation issued by the Committee for Control and Supervision of Experiments on Animals (CCSEA), Government of India. Forty-eight Wistar rats, young (4–6 months old, body weight = 150–250 g) and aged rats (18–24 months, body weight = 300–350 g) were kept in typical laboratory settings with controlled humidity, temperature, and a natural light and dark cycle. (between 22 and 25°C and 50% relative humidity). Before conducting any behavioural tests, the rats were acclimatised to the laboratory environment. The eight groups listed below comprised the rats (n = 6).
Group I (Young rats): 4–6 months old rats received ad libitum diet.
Group II (Aged rats): 18–24-month-old rats received ad libitum diet.
Group III (Aged + GLE 100): Ganoderma lucidum extract (GLE) was administered (100 mg/kg per os (p.o)) daily to aged rats for three months. 44
Group IV (Aged + GLE 200): GLE (200 mg/kg/day p.o) was administered to aged rats daily for three months. 44
Group V (Aged + GLE 400): GLE (400 mg/kg/day p.o) was administered to aged rats daily for three months. 44
Group VI (Aged + ALA 50): ALA (50 mg/kg/day p.o) was administered to aged rats daily for three months.48, 18
Group VII (Aged + ALA 100): ALA (100 mg/kg/day p.o) was administered to aged rats daily for three months. 48
Group VIII (Aged + GLE 100 + ALA 50): GLE (100 mg/kg/day p.o) and ALA (50 mg/kg/day p.o) was administered in combination daily to aged rats for three months.
After three months of treatment, all the behavioural parameters (assessment of depression, anxiety, memory & pain) were performed and then blood was collected for biochemical and molecular estimations. At the end, all the rats were sacrificed and brain was isolated for biochemical, molecular and histopathological analysis).
Analysis of Behavioural Parameters
All behavioural parameters were analysed using various procedures. The Morris water maze was performed as earlier described by Vorhees and Williams and Morris19, 20 and passive avoidance test was performed as earlier described by Eagle et al. 21 to evaluate memory impairment. For measuring the anxiolytic behaviour elevated plus maze test was performed as described by Walf and Frye. 11 To measure the immobility time in unescapable environment, a forced swim test was performed as earlier described by Porsolt et al. 22 The locomotor activity was measured by using a digital actophotometer described by Bhosale et al. 23 The motor coordination was assessed using rota rod apparatus as described by Bansal and Chopra. 24 To assess the thermal nociception, tail immersion test was performed as earlier described by Courteix et al. 25 and mechanical nociception via Randal Selitto test by Santos-Nogueira et al. 26
Analysis of Biochemical and Molecular Estimations
Brain samples were washed with ice-cold saline solution containing 0.9% sodium chloride. These samples were homogenised in chilled phosphate buffer with a pH value of 7.4. The homogenised samples were used to measure various parameters, including lipid peroxidation,24, 27 superoxide dismutase (SOD),24, 28 reduced GSH,24, 29 catalase,24, 30 acetylcholinesterase activity24, 31 and tumour necrosis factor-alpha (TNF-α) 24 . The hippocampal and cortical regions of the brain were also used to measure necrosis factor kappa B 65 (nuclear factor kappa B (NF-кB)) activity. 32
Histopathological Analysis
The histopathological analysis of the hippocampal region of the rat brain sample was performed by first fixing the brain tissue in formalin, embedding it in paraffin, and slicing it into thin sections. These sections were then stained with haematoxylin and eosin (H&E), enabling the visualisation of cellular structures under a microscope and assessing neuronal morphology, tissue integrity and pathological changes in the hippocampus. Additionally, the number of densely stained, irregularly shaped pyknotic neurons was counted at a 200 µm scale bar using a 40X objective. 33
Statistical Analysis
The results were shown as means ± standard error of the mean. The behavioural, biochemical and histopathological evaluations were analysed using one-way analysis of variance (ANOVA), followed by Tukey’s post hoc test, using GraphPad Prism software version 5.01.
Results
Effect of GLE on Learning and Memory Deficits Aged Rats
Morris Water Maze Test
Figure 1 shows that on day 5, the aged group rats spent less time in the target quadrant (TSTQ) and had significantly (p < .001) greater escape latencies (EL) than the younger group rats. When compared with aged group rats, GLE therapy (200 & 400 mg/kg) and ALA (100 mg/kg) enhanced (nearly 2–2.5-fold) the TSTQ on day 5, and they considerably (p < .001) lowered EL. Furthermore, compared to aged group rats, GLE+ALA-treated rats revealed a significantly rise in TSTQ and a decline in EL. Rats treated with GLE (100 mg/kg) and ALA (50 mg/kg) showed a notable decline in EL and an increase in TSTQ quadrant; however, the impact was not statistically significant.
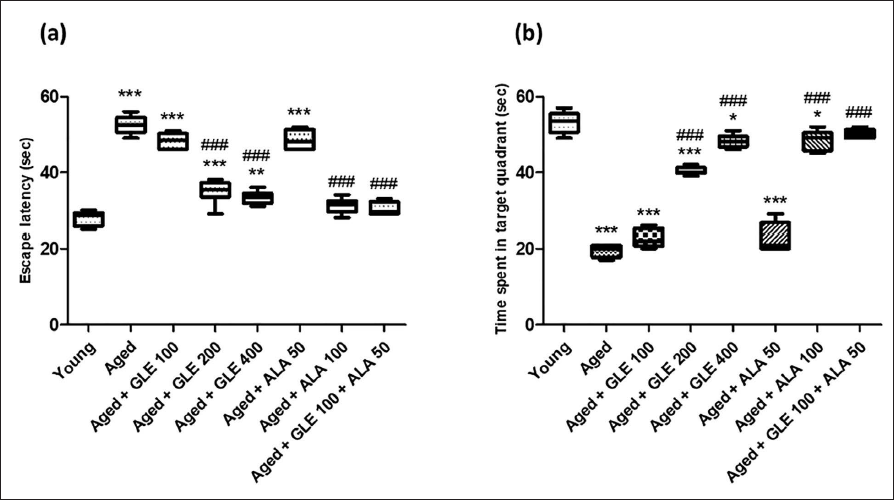
Passive Avoidance Test
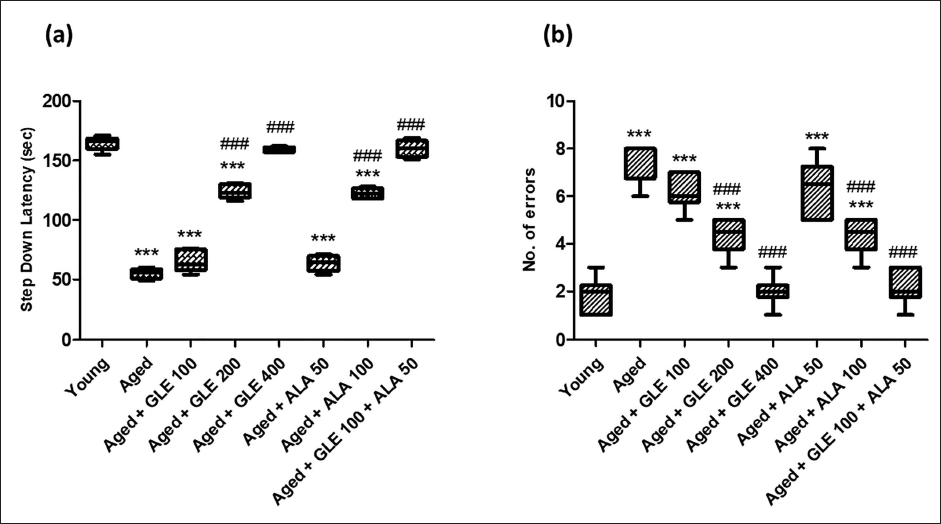
Step-down latency (SDL), a passive avoidance test, measures how long it takes an animal to step down. Increase in step down latency time is considered as parameter for learning. In comparison to the young rats, the no. of mistakes was dramatically increased (fourfold), and SDL was significantly lowered (threefold) in the older rats (Figure 2). In addition, after administering GLE (200 and 400 mg/kg) & ALA (100 mg/kg) for three months dose-dependently, aged rats’ SDL was significantly (p < .001) increased, and their number of mistakes was significantly attenuated (p < .001) in comparison to their older group rats. GLE (100 mg/kg) administered in aged rats showed a little rise in SDL and a decrease in the number of mistakes, but these effects were not statistically significant. In contrast to the aged group rats, these rats showed a substantial increase in SDL (2.9-fold) and a decrease in the number of mistakes (3.5-fold) when GLE (100 mg/kg) and ALA (50 mg/kg) were administered in combination.
Effect of GLE on Anxiolytic Behaviour of Aged Rats: Elevated Plus Maze
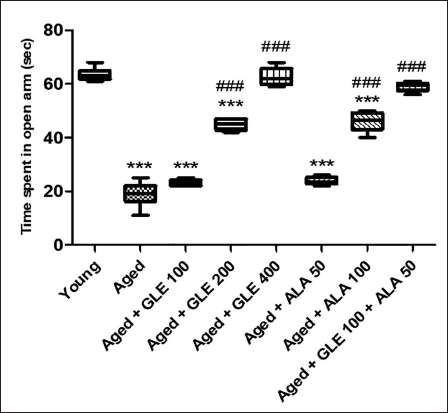
Anxiety in aged rats significantly increased (3.3-fold), as indicated by less time spent in the open arm (TSOA) compared with young group rats. GLE (200 mg/kg) and 100 mg/kg of ALA administered to aged rats increased TSOA (2.5-fold), while 400 mg/kg of GLE increased the TSOA by (3.3-fold). Rats administered with 100 mg/kg of GLE and 50 mg/kg of ALA showed a little increase in TSOA, but the effect was not significant. Additionally, these rats showed a 3.1-fold increase in TSOA with GLE (100 mg/kg) and ALA (50 mg/kg) in combination, compared with the aged rats’ group (Figure 3).
Effect of GLE on Immobility Duration (Forced Swim Test)
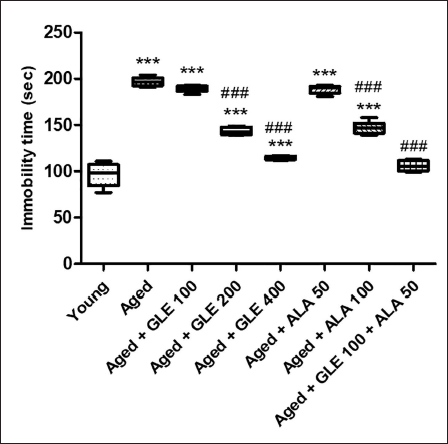
When comparing the immobility duration of older rats to that of the young group, the difference was substantial (doubled). Furthermore, compared to old group rats, the immobility period was considerably (p < .001) reduced in aged rats following treatment with GLE (200 and 400 mg/kg) and ALA (100 mg/kg) for three months, dose-dependently. Rats treated with ALA (50 mg/kg) and GLE (100 mg/kg) showed a slight reduction in immobility time, but the effect was insignificant. But when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats’ immobility period was significantly reduced – nearly 1.9-fold as compared with aged rats group (Figure 4).
Effect of GLE on immobility time. Data values are given as mean ± SD, with six animals. in each group. *Denotes p < .05 vs. young rats; **denotes p < .01 vs. young rats; ***denotes p < .001 vs. young rats; ##denotes p < .01 vs. young rats; ###denotes p < .001 vs. aged rats. GLE: Ganoderma lucidum extract, ALA: alpha lipoic acid.
GLE Effect on Locomotor Activity (Actophotometer)
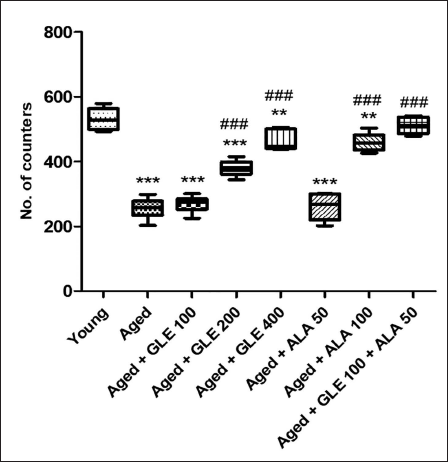
In comparison to the younger group of rats, the aged rats’ locomotor activity was considerably lower (p < .001) (Figure 5). In contrast to aged group rats, the number of counters was considerably (p < .001) higher in old rats following treatment with GLE (200 & 400 mg/kg) and ALA (100 mg/kg) for three months dose-dependently. When comparing the treatment of GLE (100 mg/kg) and ALA (50 mg/kg) to the aged rat’s group, no discernible change was seen. But when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats showed a twofold increase in the number of counters when compared with aged group of rats.
GLE Effect on Motor Coordination (Rota Rod Test)
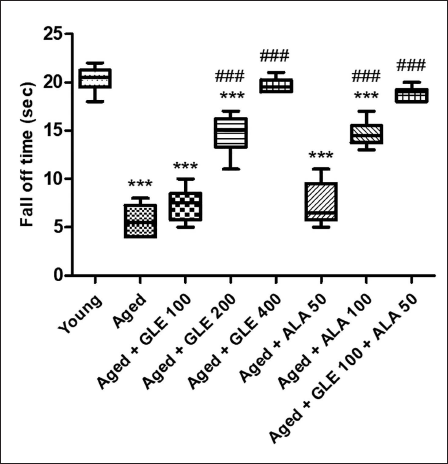
When comparing older rats to the younger group of rats, there was a substantial (3.5-fold) decline in motor coordination (Figure 6). In contrast to aged group rats, aged rats treated with GLE (200 & 400 mg/kg) and ALA (100 mg/kg) for three months dose-dependently showed a substantially longer fall-off time (p < .001) following therapy. Rats administered with GLE (100 mg/kg) and ALA (50 mg/kg) showed a noticeable increase in fall-off time; however, the effect was not statistically significant. When GLE (100 mg/kg) and ALA (50 mg/kg) were given together, however, these rats showed an increase (3.3-fold) in fall-off time when compared with aged group of rats.
Analgesic Effect of GLE
Thermal hyperalgesia (Tail immersion test).
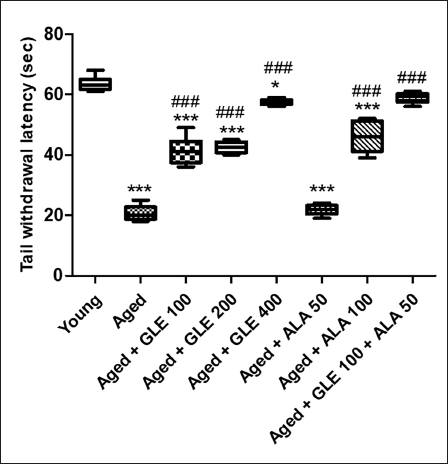
When comparing aged rats to the younger group of rats, there was a substantial threefold decrease in tail withdrawal latency (Figure 7). In contrast to aged group rats, rats treated with GLE (100, 200 & 400 mg/kg) and ALA (100 mg/kg) for three months dose-dependently showed a substantially higher tail withdrawal latency (p < .001) following therapy. Rats given 50 mg/kg of ALA did not show any discernible change. But when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats’ tail withdrawal latency was noticeably (2.8-fold) higher than that of the older group of rats.
GLE Effect on Mechanical Hyperalgesia (Randall-Selitto Test)
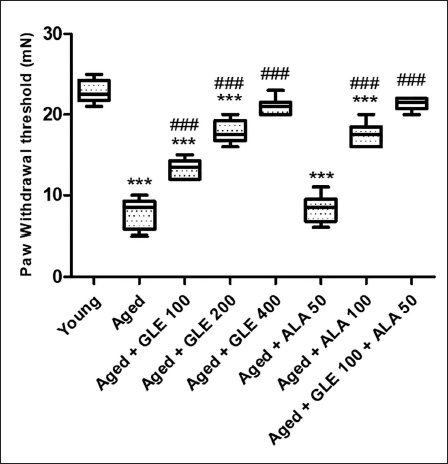
Rats in the aged group showed a considerable (2.9-fold) lower paw withdrawal threshold than those in the young group (Figure 8). The paw withdrawal threshold of elderly group rats was dramatically enhanced (2–3-fold) by treatment with GLE (200 & 400 mg/kg) and ALA (100 mg/kg). The paw withdrawal threshold of aged rats was dramatically (1.7-fold) enhanced after treatment with GLE (100 mg/kg. When comparing the administration of ALA (50 mg/kg) to the aged group rats, no discernible difference was seen. When GLE (100 mg/kg) and ALA (50 mg/kg) were given together, paw withdrawal threshold significantly increased (almost three times) in comparison to the aged group rats.
Biochemical Estimations
Effect of GLE on Lipid Peroxidation
When comparing the levels of MDA in aged rats to those in the younger group, there was a substantial (fourfold) rise (Figure 9). In contrast to aged group rats, rats treated with GLE (200 & 400 mg/kg) and ALA (100 mg/kg) for three months showed a substantial (p < .001) drop in MDA levels. Rats administered with GLE (100 mg/kg) and ALA (50 mg/kg) showed a noticeable drop in MDA levels, although this effect was not significant. But when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats’ MDA levels were decreased (3.6 times) in comparison to the aged group of rats.
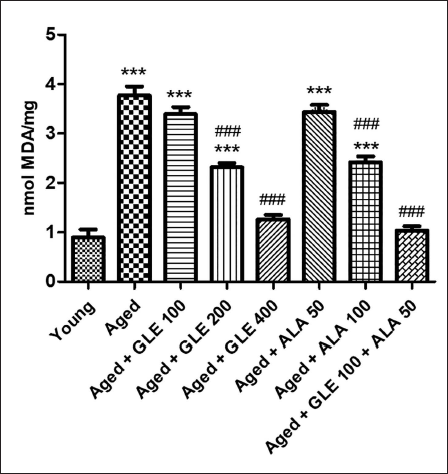
Effect of GLE on Superoxide Dismutase
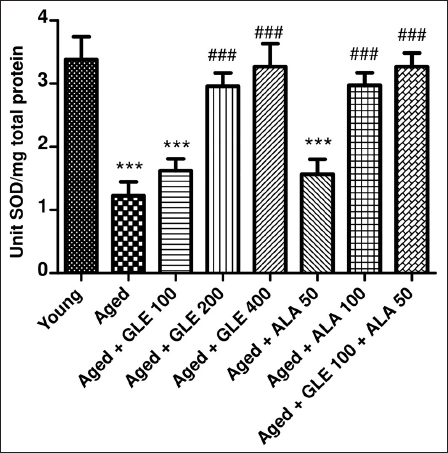
In comparison to the younger group of rats, the SOD level was considerably lower (p < .001) in the older rats (Figure 10). In contrast to the aged group rats, the rats treated with GLE (200 & 400 mg/kg) and ALA (100 mg/kg) for three months showed a substantial (p < .001) increase in SOD levels. Rats administered with GLE (100 mg/kg) and ALA (50 mg/kg) showed a noticeable rise in SOD levels, although the impact was not statistically significant. But when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats’ SOD levels significantly increased (2.6 times) in comparison to the older group of rats.
GLE effect on SOD. Data Values are Given as Mean ± SD, with Six Animals in Each Group. ***denotes p < .001 vs. young rats; ###denotes p < .001 vs. aged rats. GLE: Ganoderma lucidum extract, ALA: alpha lipoic acid.
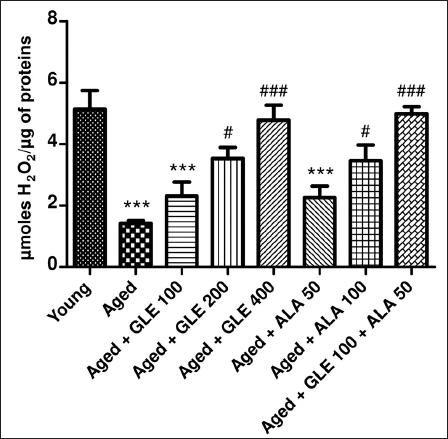
Effect of GLE on Catalase
In comparison to the younger group of rats, the amount of hydrogen peroxide (H2O2) was much lower (27.68%) in the aged group rats (Figure 11). In contrast to aged group rats, the rats treated with GLE (400 mg/kg) for three months dose-dependently showed a substantial rise in H2O2 levels (p < .001). Following treatment with GLE (200 mg/kg) and ALA (100 mg/kg), the H2O2 level rose considerably (p < .05). Rats administered GLE (100 mg/kg) and ALA (50 mg/kg) showed a notable rise in H2O2; however, the effect was insignificant. But when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats’ H2O2 levels significantly increased (3.5-fold) in comparison to the aged group of rats.
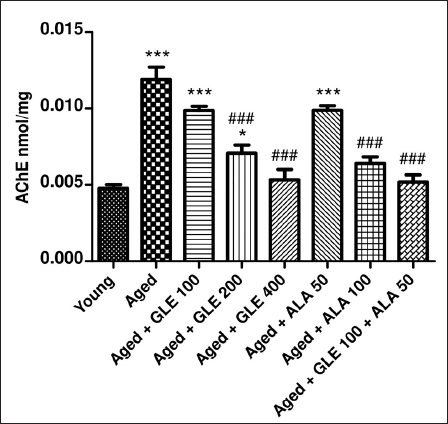
Effect of GLE on Acetylcholinesterase
The level of acetylcholinesterase (AChE) was (2.75-fold) higher in aged rats than in the young group (Figure 12). In contrast to aged group rats, the rats treated with GLE (200 & 400 mg/kg) and ALA (100 mg/kg) for three months dose-dependently showed a substantial (p < .001) reduction in AChE levels. Rats treated with GLE (100 mg/kg) and ALA (50 mg/kg) showed a noticeable drop in AChE levels, although the impact was not statistically significant. Nevertheless, when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats’ AChE levels significantly increased (2.2-fold) in comparison to the aged group rats.
Effect of GLE on Reduced Glutathione
Aged rats showed a substantial drop in GSH (p < .001) when compared with a younger group of rats (Figure 13). In contrast to aged group rats, the rats treated with GLE (400 mg/kg) for three months showed a dose-dependent rise (1.9-fold) in their GSH level. Furthermore, following treatment with GLE (200 mg/kg) and ALA (100 mg/kg), the GSH level was elevated (p < .01) in aged rats. Rats administered with GLE (100 mg/kg) showed a little rise in GSH; however, this effect was not statistically significant. When GLE (100 mg/kg) and ALA (50 mg/kg) were given together, however, these rats showed a considerable (p < .001) increase in GSH compared with aged rats.
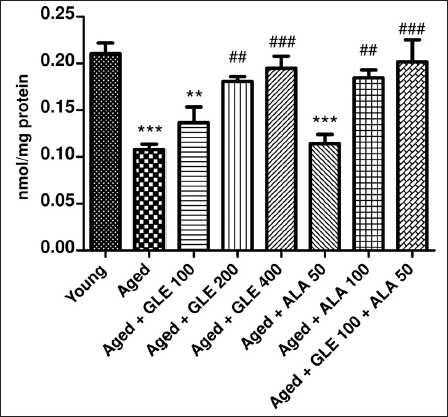
Molecular Estimation
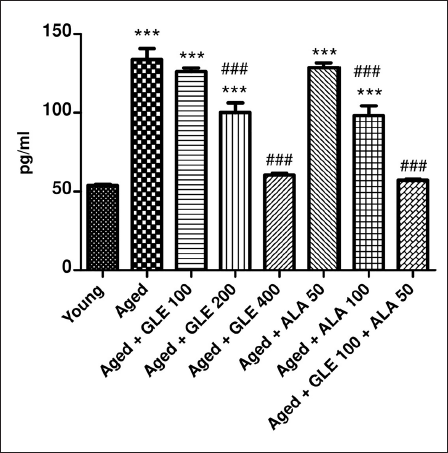
Effect of GLE on TNF-α
Compared to the younger group of rats, the aged rats exhibited a considerable (2.4-fold) rise in TNF-α (Figure 14). In contrast to aged rats, TNF-α was considerably (p < .001) reduced in aged rats following treatment with GLE (200 & 400 g/kg) and ALA (100 mg/kg) for three months in a dose-dependent manner. Rats administered with GLE (100 mg/kg) and ALA (50 mg/kg) showed a notable drop in TNF-α; however, the effect was not significant. Nevertheless, when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats showed a 2.3-fold rise in TNF-α when compared to the older group of rats.
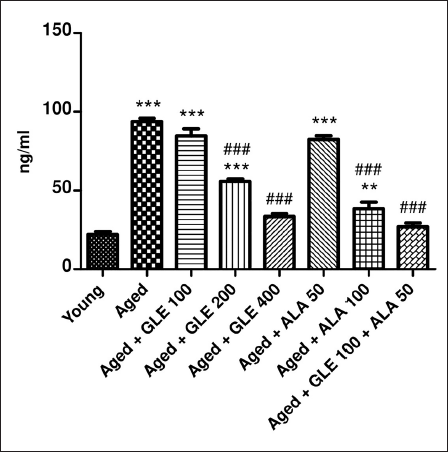
Effect of GLE on NF-кB
Aged rats showed a significant (4.2-fold) rise in NF-кB when compared with the young group of rats (Figure 15). In contrast to aged rats, NF-кB was considerably (p < .001) reduced following treatment with GLE (200 & 400 mg/kg) and ALA (100 mg/kg) for three months, depending on the dosage. Rats treated with GLE (100 mg/kg) and ALA (50 mg/kg) showed a notable reduction in NF-кB, although the effect was not significant. Nevertheless, when GLE (100 mg/kg) and ALA (50 mg/kg) were given together, these rats showed a 3.4-fold rise in NF-кB compared to the aged group rats.
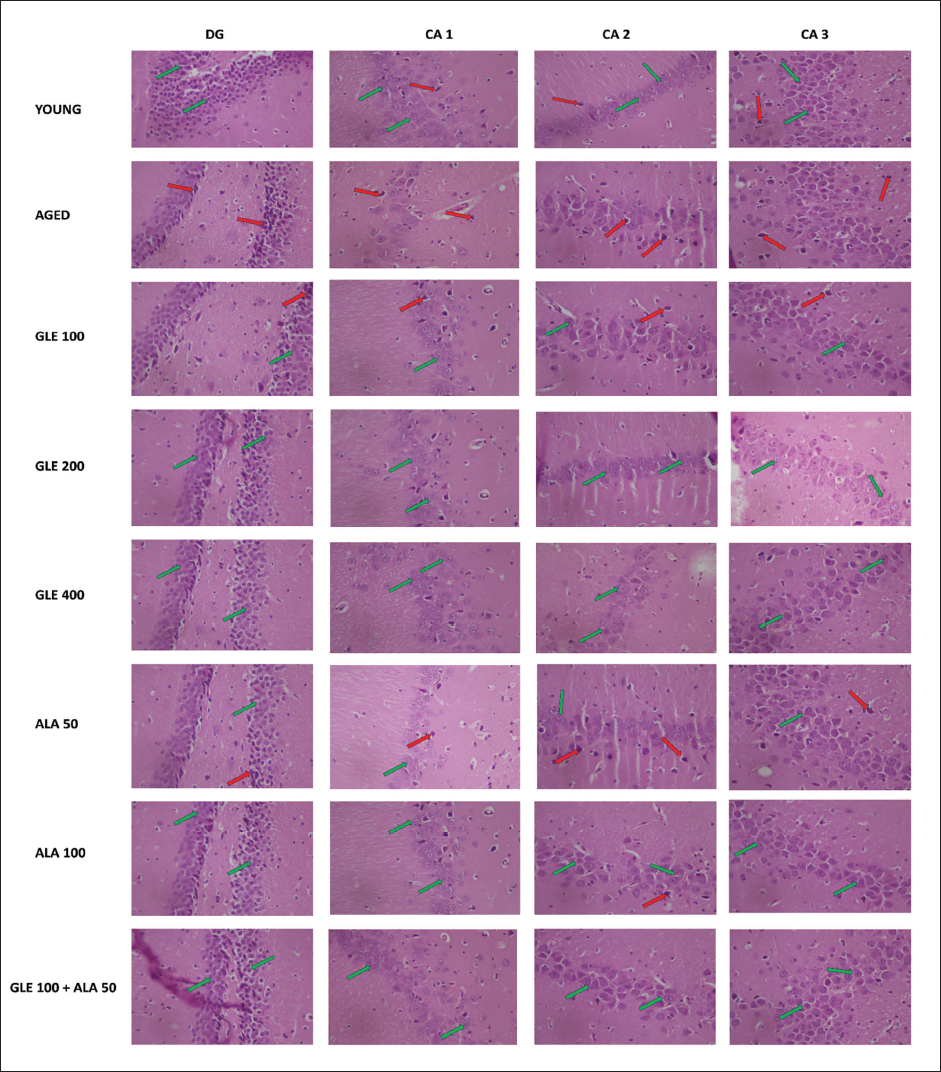
Histopathological Analysis
During the histopathological examination of rat’s brain hippocampal region, marked neuronal damage was observed in the dentate gyrus of aged rats’ brain when compared to young rat’s brain as indicated by large no of pyknotic and granule dead cells. Administration of GLE (100, 200 & 400 mg/kg) and ALA (50, 100 mg/kg) dose-dependently modulates the neuronal damage by reduction in pyknotic and granule dead cells. Further, administration of GLE (100 mg/kg) in combination with ALA (50 mg/kg) markedly reduced the neuronal damage in rat hippocampal region (Figures 16 and 17).
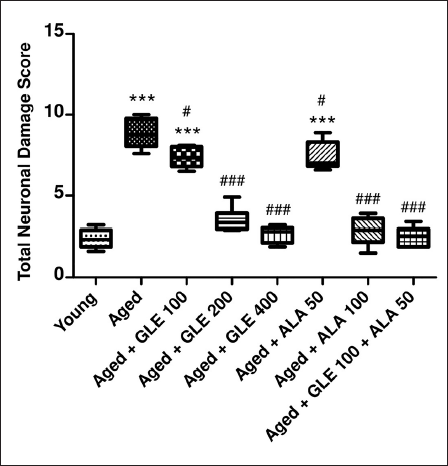
Histopathological Examination of Rat’s Brain Hippocampal Region. Red Arrow Indicates Damaged Cell and Green Arrow Indicates Healthy Cells.
Discussion
Ageing is the primary risk factor for most neuronal deficits. Free radicals are very reactive molecules or atoms with one or more unpaired electrons (s) in their outer shell that can be created when oxygen reacts with specific compounds. 34 Because these radicals are produced in cells when one electron is lost or received, they can behave as reductants or oxidants. 35 Free radicals induce cumulative oxidative damage that leads to ageing, which results in alteration in neuronal junctions in the brain that progressively worsen cognitive decline. 36 In addition, we found that aged rats had significantly higher levels of inflammation, oxidative stress indicators and impairments in memory, nociception, anxiety and depression when compared to younger rats. These findings suggest that age-related neuronal deficiencies may be the cause of neuronal deficiencies in old rats.
When compared to aged rats, the administration of GLE improves nociception, depression, anxiety, learning and memory. These outcomes were in concurrence with a previous work by Lee and Kim 36 that demonstrated how administering GL water extract to aged mice caused by D-galactose enhances their memory and learning. Further, a study by Matsuzaki et al. 37 found that a water extract from the GL mycelia culture medium reduces depression and anxiety in aged rats. Additionally, research by Li et al. 38 confirmed the effectiveness and safety of GL in rheumatoid arthritis patients. Memory, 39 depression, 40 anxiety, 41 and nociception 42 are all improved by the administration of ALA. In our research, we also found that a combination of GLE and ALA administered to aged rats dramatically improves their memory, depression, anxiety, and nociception.
Oxidative stress is a normal biological process for the development of disease. Under normal circumstances, several enzyme systems involved in in vivo redox homeostasis keep the physiologically significant intracellular amounts of ROS low. As a result, an imbalance between the body’s prooxidants and antioxidants can also be used to characterise oxidative stress. 43 MDA, SOD, GSH and catalase are examples of oxidative stress biomarkers that are crucial for determining the extent of disease and the advantages of antioxidants for general health. Oxidative stress is a crucial theory of ageing and SOD, GSH, and catalase were the first line of defence antioxidants. 44 MDA and the naturally occurring antioxidants SOD and GSH are currently thought to be the most significant oxidative stress markers among other markers. Many species’ susceptibility to oxidative stress are increased when SOD expression is reduced. 45 Decreased GSH levels in the brain are associated with neuronal loss brought on by ageing or neurodegeneration. 46 Many age-related degenerative diseases are thought to have a connection to the pathogenesis of catalase deficiency or malfunction. 47
A study by Sharma P and Tulsawani R 34 showed how MDA and GSH might improve memory deficits caused by hypobaric hypoxia by regulating neurotransmission, neuroplasticity, and preserving redox equilibrium. Further, a 2019 study by Ahmad MF et al., 48 showed that GL increases SOD activity. Additionally, a study conducted in 2024 by Hasnat MA et al.,49, 50 found that the fruiting body of GL increases catalase production. Furthermore, Ajith T et al., 51 demonstrated that GL extract inhibits AChE while also increasing the activities of antioxidant enzymes (SOD, catalase and GSH). When ALA is administered, it increases MDA, SOD, AChE, GSH and catalase activities.52–54 As a result, in comparison to aged rats, we also found that the combination of GLE and ALA considerably reduces the production of oxidative stress and increases the activities of antioxidant enzymes.
Differential expression of some genes implicated in inflammatory processes can be caused by oxidative stress-induced activation of several transcription factors. 55 For instance, research has demonstrated that the inflammatory process leading to several chronic illnesses is significantly influenced by the synthesis of TNF-α and the activation of NF-κB. 56 ROS can affect synaptic and non-synaptic neuronal transmission in brain tissues, resulting in neurodegeneration, neuroinflammation, cell death and memory loss. 57 The body can activate various immune system cells in the event of an inflammatory response. They emit a range of substances known as ‘inflammatory mediators’. The hormones bradykinin and histamine are two of them. To allow more blood to reach the injured tissue, they cause the tissue’s tiny blood vessels to grow or dilate. Reddish-pink patches that are inflamed experience heat. 58 The pathophysiology of inflammation is greatly influenced by biomarkers, including ROS, cytokines (such as TNF-α and interleukin-6 (IL-6)), chemokines, prostaglandins, C-reactive proteins (CRP), NF-κB and major immune cell types. 59 Given that ageing is a significant risk factor for cancer, systemic, sterile (non-infection-caused), age-related chronic inflammation – also known as inflamm-ageing – is considered to have an underlying etiological relationship to the illness. 60 Ageing causes an increase in CRP and other inflammatory indicators. According to Yoon et al., 61 the gut microbiota is thought to have a role in systemic chronic inflammation, changing and becoming more pro-inflammatory with age.
According to Liu et al., in lipopolysaccharide-stimulated BV2 microglial cells, GL ethanol extract suppresses the NF-κB and toll-like receptor pathway, therefore inhibiting the inflammatory response. 62 Furthermore, research by Hu et al., revealed that the down-regulation of NF-κB signalling is linked to the anti-inflammatory effects of GL triterpenoid in human Crohn’s disease. 63 Additionally, a study conducted in 2020 by Akbari et al., revealed that GL triterpenes protect against inflammatory reactions generated by lipopolysaccharides. 64 The impact of ALA supplementation on inflammatory markers in patients with metabolic syndrome and associated illnesses was documented in a study by Ying et al., 65 Furthermore, ALA suppresses NF-κB activation independently of its antioxidant action. As a result, we also found in our investigation that the combination of GLE and ALA considerably decreased inflammation caused by ageing.
Conclusion
In conclusion, the effects of GLE on neuronal impairments in old rats are supported by recent data. When compared to aged rats, GLE improves depression, anxiety, nociception, learning and memory. It also prevents oxidative stress and increases the activity of antioxidant enzymes. GLE can counteract the negative effects of oxidative stress, which can cause illnesses and premature ageing. GLE may be useful in the treatment of neurodegenerative illnesses since it inhibits the production of inflammatory mediators (TNF-α and NF-κB). In the future, GLE may prove to be effective in neurodegeneration associated with age-dependent oxidative stress. However, further research is necessary to explore its potential protective role against age-related disorders.
The current research offers strong evidence that GLE and ALA have notable neuroprotective properties against age-related neuronal impairments in rats. The results of the research highlight the capacity of GLE and ALA, both alone and when used together, to alleviate several elements of neuronal degeneration linked to the ageing process. Administration of GLE and ALA over 90 days showed a decrease in memory impairment, anxiety, depression and locomotor impairments in old rats. The reduction in these symptoms was dependent on the dosage of the administered substances. The coadministration of GLE and ALA, especially at lower dosages, exhibited synergistic benefits, augmenting cognitive and motor capabilities and promoting overall neurological well-being in elderly rats.
Age-related neuronal deficits are mostly influenced by oxidative stress and inflammation. This research demonstrates that GLE and ALA have a significant impact on reducing oxidative stress indicators, such as lipid peroxidation, while simultaneously enhancing antioxidant defences, including SOD and GSH levels. Moreover, both medications effectively reduced inflammatory markers like TNF-α and inhibited NF-κB activity, both of which are essential for neurodegenerative processes. The neuroprotective benefits of GLE and ALA are primarily attributed to their capacity to restore antioxidant equilibrium and diminish inflammatory reactions.
Behavioural tests demonstrated that treatment with GLE and ALA resulted in better cognitive functioning. This was demonstrated in the MWM test by shorter EL and longer TSTQ. In the passive avoidance test, memory recall was also improved. Furthermore, the rats that were treated showed reduced anxiety in the elevated plus maze test and reduced depression in the forced swim test. The increased motor coordination and locomotor activity in conjunction with the behavioural changes suggest that GLE and ALA have broad-spectrum advantages on neurological health.
Overall, the research provides evidence that GLE and ALA can reduce age-related neuronal impairments, suggesting their potential as therapeutic interventions to address age-related neurodegenerative issues. Moreover, additional studies to clarify the exact mechanism are warranted.
Footnotes
Abbreviations
Author Contributions
HK, SB, SS, SC and RC conceived the study. HK, RC, SC and SB were involved in protocol writing, database search, screening of articles and preparation of the final draft. SB, SG, and SC were responsible for article screening till the writing of the final draft. SG, SC, and SB were consulted for the critical revision of the manuscript to resolve any discrepancies. Finally, all the authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
The animals were approved by the MM College of Pharmacy’s Institutional Animals Ethics Committee (Protocol no. MMCP-IAEC-110, dated 20/11/2021).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
