Abstract
Background
Helminthiasis is a major concern in the livestock sector due to the immense afflictions in the production and economy of the farmer. Chemical anthelmintics are used to combat them and parasites are acquiring resistance against these drugs. Hence, the development of new drugs or alternatives is the need of the hour for combating such infestations. This study was undertaken to determine the effect of methanolic leaf extract of Tectona grandis on strongyles.
Materials and Methods
The dried leaves of Tectona grandis were subjected to Soxhlet extraction using methanol, and the extract was tested for its anthelmintic activity in vitro using egg hatch assay and larval motility assay. A dose of 250, 125, 62.5, 31.25, 15.625, and 7.8125 mg/ mL was used for the study. Fresh strongyle ova were exposed to different concentrations of the extract and observed after 48 hours for the hatch. L3 larvae obtained from coproculture were subjected to treatment with extract at different concentrations, and mortality per unit time was calculated. Fourier transform infrared (FTIR) spectroscopic analysis and Gas chromatography-mass spectrometry (GC/MS) were performed to identify the chemical nature of the extract.
Results
There was a dose-dependent inhibition of hatch and larval mortality with a maximum inhibition of hatch at 250 mg/mL. There was a cent percent hatch in control wells and no loss of progressive motility in the larval motility test. On exposure to the extract, the larvae progressively lost their motility, and finally, there were caesurae of movement which indicated their death. The extract at 250 mg/mL killed all the larvae by 30 min, whereas, at 31.25 mg/mL, the mortality was 66.6% after 2 hr.
Conclusion
The results suggest that methanolic extract from Tectona grandis leaves has a promising anthelmintic property and further studies are required for the isolation of active molecules.
Introduction
One of the most substantial socioeconomic agricultural enterprises in India is the raising of livestock. The most common constraint faced by the farming community is the frequent occurrence of parasitic infections among the livestock herds. Successful and economically sustainable production of livestock is adversely undermined by the invasion of parasitic nematodes into the gastrointestinal (GI) tract of animals. Among the GI nematodes, Strongyle sp. can cause a nutritional imbalance with malnourishment as the nutrients are redirected from the absorption site towards the body sites for the revitalization of tissues that are injured by the parasites. 1 Affected animal shows deteriorative signs like a decline in body weight, impaired growth, disrupted state of reproduction, blood loss with anemia, and diarrhea, which can finally lead to fatality as its grave outcome. 2 In addition to its effect on compromising the health of animals, helminthiasis has deleterious consequences on the economic progress of a nation and worldwide. To confront these parasites, anthelmintics are routinely provided in livestock farms on a deliberate and frequent basis; meanwhile, risks with the administration of chemotherapeutics and the acquisition of anthelmintic resistance by the parasitic species toward different groups of anthelmintics are currently turning to be more obvious with significant exacerbation.
Anthelmintic resistance is an inheritable suppression of susceptibility to an anthelmintic in the parasitic community that was formerly sensitive to the same anthelmintic. The surge in anthelminthic resistance is mostly due to the under dosage, inappropriate and extensive use of anthelmintics as a therapeutic agent against various sorts of parasitic infections. 3 This ultimately promoted a serious threat to the warfare against parasitism as there is a perceivable drop in the exploration of novel anthelmintics by the pharmaceutical industry. Hence in this instance, phytotherapy has earned immense recognition as a complement to classical chemical anthelmintics. 4 Compared to synthetic chemicals, these herbal-based remedies are less burdensome, more reliable, efficacious, and have minimal detrimental effects. 5 Hence, this investigation used the methanolic leaf extract of Tectona grandis to examine its ovicidal and larvicidal properties against strongyles.
Tectona grandis, a multi-faceted and remarkably eminent medicinal tree, is a distinctive reservoir of various bioactive compounds, and its dimensional stability is noted. In Bangladesh, Thailand, China, India, and Pakistan, T. grandis, often known as teak, is widely dispersed. It is a type of tropical hardwood tree included in the Lamiaceae family of flowering plants. 6 Teak has legitimate credibility as a superior timber worldwide due to its outstanding physical and mechanical attributes, such as flexibility, durability, rigidity, and resilience to deterioration. 7 The primary components of plants have a wide range of pharmacological properties, including anti-bacterial, antioxidant, anti-fungal, anti-inflammatory, anti-pyretic, analgesic, anti-diuretic, and hypoglycemic effects. Teak leaves have hemostatic characteristics, making them suitable for applying on the skin cuts to halt bleeding. 8 Topical application and oral administration of leaf extract showed significant improvement in collagenation, wound contraction, and breaking strength. 9
The growing interest in medicinal plants as an alternative source of bioactive compounds that are biodegradable into non-toxic products and potentially useful for the control of parasites has been sparked by factors such as the emergence of resistant populations, high cost, risk of environmental pollution, and reduction in animal production due to low effectiveness. In this context, this study was undertaken to establish the anthelmintic activity of alcoholic leaf extract of Tectona grandis.
Materials and Methods
Plant Material
The leaves of Tectona grandis were collected from different parts of the district of Thrissur and authenticated by a Botanist at St. Thomas College, Thrissur. The herbarium was prepared and deposited in the repository at the College of Veterinary and Animal Sciences, Mannuthy. It was dried under shade and pulverized in a mechanical pulverizer with temperature control. They were methanol-extracted in a Soxhlet extraction system at 60°C till the solvent in the extraction chamber was colorless, dried in a rotary vacuum evaporator keeping a vapor temperature less than 40°C, and kept chilled until use.
Phytochemical Screening
The methanolic extract of T. grandis leaves was tested for the presence of various active chemical constituents, namely steroids, alkaloids, tannins, phenolic compounds, flavonoids, glycosides, and saponins by qualitative tests. 10
Fourier Transform Infrared Spectroscopy (FTIR) Analysis
A Perkin-Elmer spectrum two FTIR spectrometer with attenuated total reflectance was used for FTIR analysis of the extract obtained. An overhead ATR attachment was installed at the sample station. A small quantity of the sample was carefully placed on the diamond crystal surface to cover the ATR diamond window and focus the laser beam. Each spectrum’s absorbance was recorded under the 60 N value. 11
Gas Chromatography - Mass Spectrometry (GC-MS) Analysis of Potent Extract/Fraction
Gas chromatography (Trace 1300) was used to separate the components of the extract, and the separated contents were then analyzed using TSQ 8000 MS/MS. On a TSQ-2MS capillary column (30 m 0.25 mm; ID 0.25 mm film), the chemicals were separated. The sample was filtered using a 0.22 m syringe filter after being dissolved in methanol and used for additional analysis. The temperature of the column oven was programmed to rise from an initial temperature of 80°C for two minutes, then rose at a rate of 15°C/min to 150°C for one minute, and then rose at a rate of 10°C/min to 250°C for five minutes. The oven ran for 20 minutes. The ion source temperature was 230°C, whereas the injection temperature was 290°C. One mL min-1 of helium was employed as the carrier gas in this experiment. It had a 70-eV ionizing energy. By gathering the full-scan mass spectra within the scan range of 40–350 amu, all data were acquired. The National Institute of Standards and Technology (NIST) MS Search 2.0 library was used to identify the compounds. 12
Assessment of the Anthelmintic Activity
Egg Hatch Assay
Fresh Strongyle ova were collected from fecal samples of goats, concentrated by centrifugation, washed with distilled water, and used. The extract was diluted to concentrations of 500, 250, 125, 62.5, 31.25, and 15.625 mg/mL in a total volume of 1 mL. Tyrode’s solution served as a negative control. About 500 eggs/mL of Tyrode’s solution was counted, and 500 µL of this was seeded in each well of a marked 6-well tissue culture plate and then added with equal volume of the dilutions of the extract as described earlier. The effective concentration of the drug in each well was thus reduced to 250, 125, 62.5, 31.25, 15.625, and 7.8125 mg/mL. The positive control was treated with albendazole at concentrations from 0.01 to 0.05 mg/mL, whereas the negative control was treated with the diluent. The culture plates were incubated at 27°C for 48 hours of incubation, and then a drop of Lugols Iodine was added to each well to stop the hatching of eggs Under a dissecting microscope, hatched larvae (either dead or alive) and unhatched eggs were counted (magnification 40×) in all the triplicate wells. 13 The percentage of unhatched ova was then calculated.
Larval Mortality Test
Five grams of Strongyle sp. infected goat dung was cultured for ten days at room temperature with appropriate humidity in the dark to produce L3 larvae. Tyrode’s solution was used to rinse the larvae into Petri plates. Some adjustments were made to the technique of Rahman et al., 2011 14 for assessing the larvicidal activity. A hundred motile larvae were gathered in 100 µL of water, and the same amount of extract diluted in distilled water was then added. Extracts were made according to the egg hatch assay. The loss of motility of the larvae was checked every 30 minutes, and the percentage of larvae found non-motile/ dead was calculated. The presence of immobile and rod-shaped larvae was looked into as the primary criterion for death, while those larvae that showed some type of movement or curvature in their body were considered to be alive. The efficacy was expressed as the percentage of larvicidal activity using the following formula:
% Larvicidal activity = (% viability inhibition per well/% viability inhibition in control well) × 100.
Statistical Analysis
Calculation of IC50
In this study, the half-maximal inhibitory concentration or the effective concentrations that were able to inhibit 50% of exposed ova or larvae (IC50) was calculated using GraphPad Prism version 5.00 (GraphPad Software, San Diego, California, USA). The concentrations of the extracts that were used for the assay were log-transformed, and the percentage of hatch or motility obtained for each treatment based on the three replicates per concentration was analyzed by non-linear (least squares) regression using the model log (inhibitor) vs. response - variable slope.
Results
Phytochemical Screening
The herbarium prepared was deposited in the repository of the Department of Veterinary Pharmacology and Toxicology, College of Veterinary and Animal Sciences Mannuthy with accession number HERB/CVA/24/2023. The result of the phytochemical screening of the methanolic extract of T. grandis (MTG) is summarized in Table 1. Phytochemical screening revealed that MTG contains a mixture of active chemical constituents such as steroids, phenolic compounds, tannins, flavonoids, and saponins.
Phytochemical Screening of Methanolic Leaf Extract of Tectona grandis Leaf.
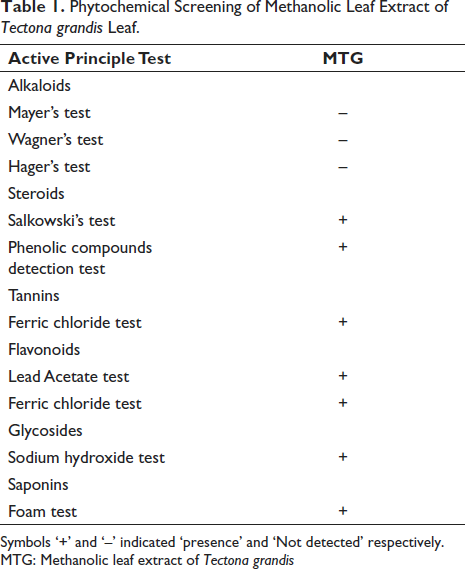
Symbols ‘+’ and ‘–’ indicated ‘presence’ and ‘Not detected’ respectively.
MTG: Methanolic leaf extract of Tectona grandis
Fourier Transform Infrared Spectroscopy (FTIR) Analysis
The compounds obtained by comparing the spectra of the extract using FTIR and FLUKA library are listed in Table 2, and the spectrum of the methanolic extract of T. grandis leaves is given in Figure 1.
List of Structurally Similar Compounds of Methanolic Extract of T. grandis Leaf.
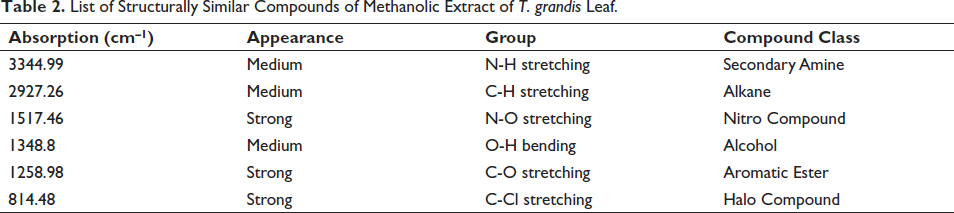
FTIR Spectrum of Methanolic Extract of T. grandis leaf.
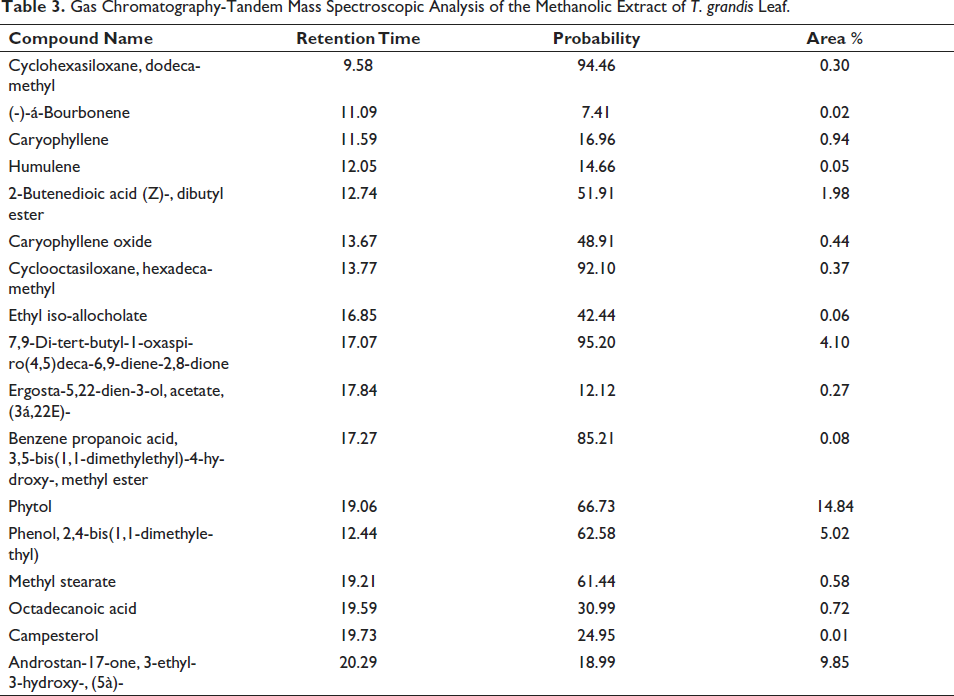
Gas Chromatography-Tandem Mass Spectrometry (GC-MS/MS) Analysis
Phytochemicals identified by GC-MS and the spectrum obtained in Figure 2 for methanolic T. grandis are given below with their probability and respective retention time in Table 3.
GCMS Spectrum for the Methanolic Extract of Tectona grandis.
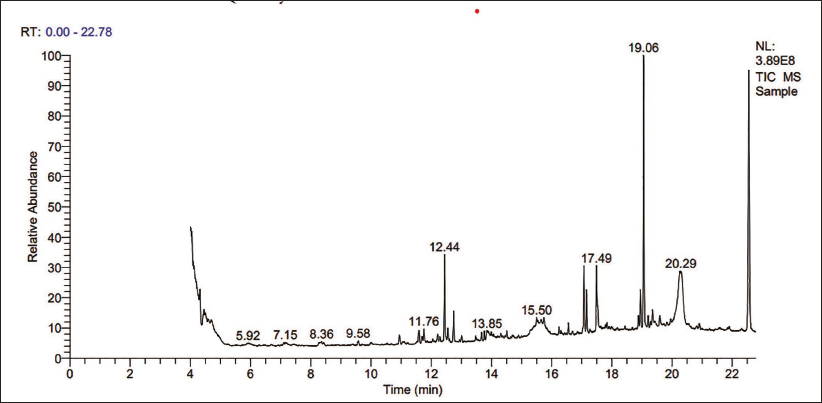
Gas Chromatography-Tandem Mass Spectroscopic Analysis of the Methanolic Extract of T. grandis Leaf.
Assessment of Anthelmintic Activity
Egg Hatch Assay
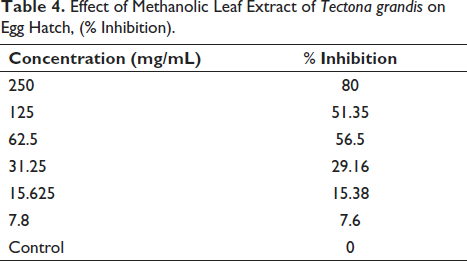
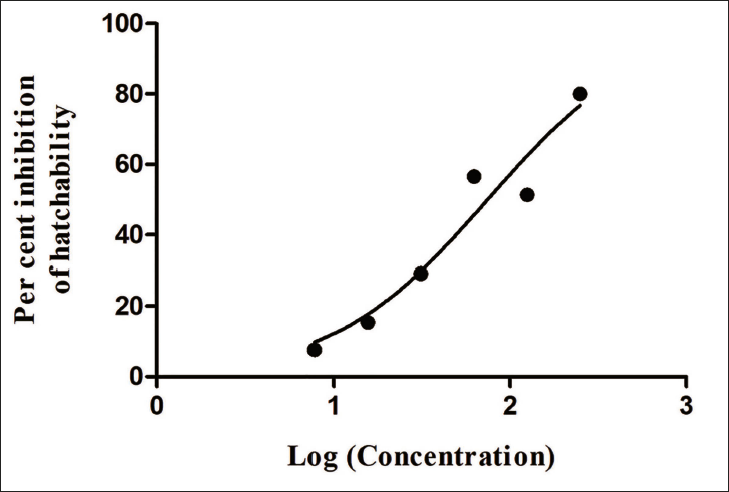
The ova were collected from goats that were maintained on a local farm and were not dewormed in the past six months with any chemical anthelmintic. The fecal samples were collected from the rectum of six animals maintained on the farm and were pooled before they were concentrated for the study. From the results represented in Table 4, it is seen that the methanolic extract of T. grandis showed strong and dose-dependent ovicidal activity against the ova of strongyles from goats. There was a maximum inhibition of hatch at 250 mg/ mL with a very early onset of the ovicidal activity as evidenced by shrinkage of ova, whereas a cent percent hatch was noticed in control wells. The half-maximal inhibitory concentration (IC50) calculated was 74.541.194 mg/ mL. Figure 3 shows the dose-response curve of percentage inhibition on egg hatchability against various log concentrations of the extract. The ova that were treated with the standard drug showed no hatching even at the lower dose and hence may be considered to be more potent compared to the test substance.
Effect of Methanolic Leaf Extract of Tectona grandis on Egg Hatch, (% Inhibition).
Larval Mortality Test
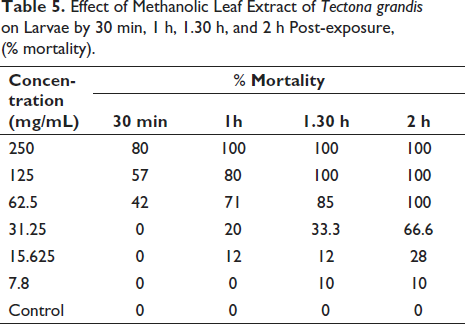
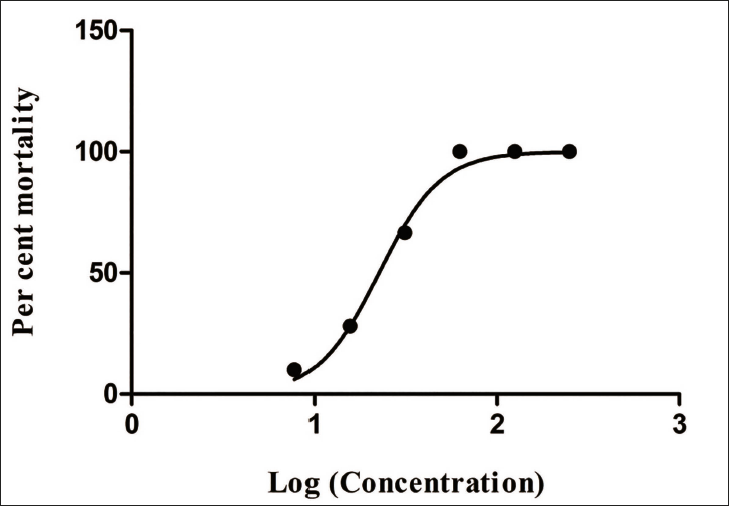
The results (Table 5) indicate that there was a dose-dependent inhibition of larval mortality when they were exposed to different concentrations of the extract and the positive control. In negative control, no loss of progressive motility in larval motility was observed even after five hours of exposure. On exposure to the extract, the larvae progressively lost their motility, and finally, there was a complete cessation of movement which indicated their death. The death of larvae that were exposed to the test drugs was dose and time-dependent. The extract at 250 mg/mL killed all the larvae by 30 min, whereas, at 31.25 mg/mL, the mortality was 66.6% after 2 hr. The half-maximal inhibitory concentration (IC50) after 2 h was calculated to be 22.571.0560634 mg/mL. By the end of the 4th hour, there was loss of motility even at the lower concentrations. Since the positive control drug caused loss of motility of larvae at all concentrations by the second hour, that time was used to calculate the IC50 of the test substance. Figure 4 shows the dose-response curve of per-cent mortality of larvae after 2 h against various log concentrations of MTG.
Effect of Methanolic Leaf Extract of Tectona grandis on Larvae by 30 min, 1 h, 1.30 h, and 2 h Post-exposure, (% mortality).
Dose–Response Curve of Percent Inhibition on Egg Hatchability Against Various Log Concentrations of MTG. *Albendazole at all doses produced 100% inhibition.
Dose–Response Curve of Percent Mortality of Larvae After 2 h Against Various Log Concentrations of MTG.
Discussion
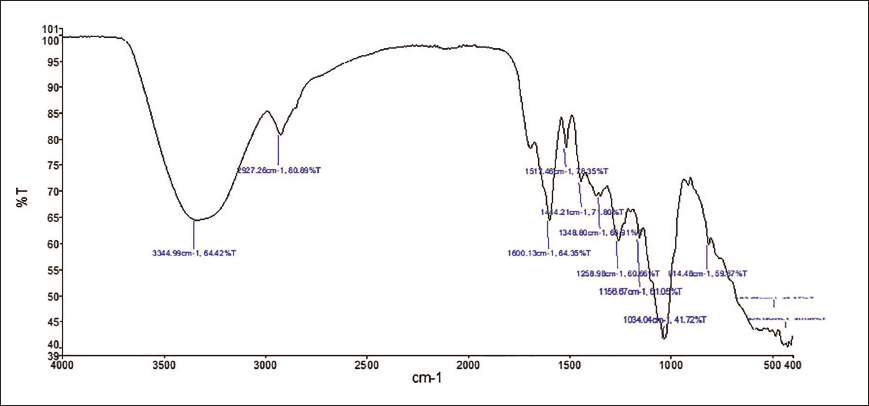
Synthetic chemical anthelmintics have been the pillar for the treatment and control of helminthiasis for decades, and their indiscriminate use has led to the development of anthelmintic resistance. 15 Much of the latest research focuses on the use of plants in the development of treatment or alternatives and adjuncts in the therapy of infections, inflammation, and neoplasia. 16 In this study, we investigated the anthelmintic activity of the methanolic extract of Tectona grandis leaves by using egg hatch assay and larval mortality test. The preliminary phytochemical screening by qualitative color reactions and GCMS revealed the presence of steroids, phenolic compounds, tannins flavonoids, and saponins. The GCMS analysis revealed the presence of caryophyllene, caryophyllene oxide, humulene, Campesterol cyclohexasiloxane, dodecamethyl, benzene propanoic acid, 3,5-bis(1,1-dimethylethyl)-4-hydroxy-, methyl ester, phytol, phenol, 2,4-bis (1,1-dimethylethyl, methyl stearate while FTIR analysis confirmed the presence of secondary amine, alkane, nitro compound, alcohol, aromatic ester, and halo compounds. Leaf extract of Tectona grandis contains various phytoconstituents like steroids, tannins, glycosides, reducing sugar, terpenoids, flavonoids, saponins, etc., which are reasonable for diverse pharmacological effects like anti-inflammatory, anti-malarial, anthelmintic, laxative, anticancer, antipyretic properties. 17 Also, the alcoholic leaves extract of Tectona grandis was found to be efficacious against parasitic infections in humans, and studies have shown that the alcoholic extract of T. grandis leaves showed anthelmintic activity against earthworms, 18 but has not been evaluated against gastrointestinal nematodes or their immature forms, ova, etc. They attributed this activity to the presence of various steroids, tannins, glycosides, reduced sugar, terpenoids, flavonoids, and saponins in the extract. The presence of these phytochemicals was implicated in the antimicrobial activity against Staphylococi where they caused the destruction of cell membranes and thus inhibited cellular functions. 19
The effect of the extract on the larvae is measured by in vitro tests, including larval motility (mortality) assays, larval development assays, larval migration inhibition assays, etc.20, 21 The adulticidal activity of plant extract against adult nematode parasites is assessed by its impact on changes in motility, paralysis, and death of the worms. 22 Screening at all three stages will reveal the exact mechanism of action of the plant extract as the anthelmintic activity of a wide spectrum of conventional drugs can be on any of the three stages, like eggs, larvae, adult, or combined. There are different reports that prove the anthelmintic activity of phytochemicals by evaluating their effect on the ova of parasites, larvae, and adults. Phytoconstituents like saponins and tannins in leaf extracts of Parkia biglobosa impeded the developmental cycle and growth progression of the egg of a bovine nematode parasite. 13 Various research conducted in our lab has shown that the phenolic, tannin, and flavonoid content of the extracts of Mallotus phillipensis, Azadirachta indica, Murraya koenigii, Allophyllus cobbe, Vitex negundo, and Ocimum sanctum contributed to their anthelmintic action.11, 23–26
The Gas Chromatography mass spectroscopy revealed the presence of around 20 major compounds with bioactivity. Caryophyllene and caryophyllene oxide that were detected in the extract are bicyclic sesquiterpenes which are active components in the essential oils derived from spices that have been proven to induce apoptosis and anticancer effect. 27 Another metabolite of caryophyllene, humulene, was also detected in the extract, which also has a proven antiproliferative effect. 28 There are reports of anthelmintic activity of essential oils that contain β-caryophyllene or its oxide including those from Hyptis dilatata Benth, Mesosphaerum suaveolens Kuntze, 29 flowers of Bidens sulphurea, 30 Tanacetum vulgare, 31 Croton zehntneri and Lippia sidoides,32, 33 Coriandrum sativum, Tagetes minuta, Alpinia zerumbet and Lantana camara. 34 Another bioactive compound detected was campesterol, with a hydroxyl group in position C-3 of the steroid skeleton and saturated bonds throughout the sterol structure, with the exception of the 5-6 double bond in the B ring. It has antimicrobial and anti-inflammatory properties,35, 36 whereas plants that contain these sterols like Imperata cylindrical, 37 and Urochloa distachya 38 showed potent anthelmintic activity.
From the results of the study, it is evident that the methanolic leaf extract of Tectona grandis showed significant ovicidal and larvicidal activity by its lethal effect on both stages. Higher doses of leaf extract notably restricted the hatching of ova. It was observed that the larva was formed inside the ova but could not hatch out. After 30 minutes of incubation with the extract, larvae exposed to the extract were discovered to be dying slowly and were progressively dead in a dose-dependent manner. Based on the research findings obtained in the study it can be interpreted that the leaf extract of T. grandis could be affecting the process of energy generation and its utilization of the larvae.
Conclusion
From the results of the study, it could be concluded that the methanolic extract of leaves of T. grandis possessed good anthelmintic activity as evidenced by the results of egg hatch assay and larval motility assay in Strongyle sp. A lead for creating an innovative and secure anthelmintic can come from the further separation of the active chemicals.
Abbreviations
Footnotes
Acknowledgments
The authors acknowledge Kerala Veterinary and Animal Sciences University, Kerala, India, for the facilities provided.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
