Abstract
A 2-week study in rats identified target organs of oxfendazole toxicity to be bone marrow, epididymis, liver, spleen, testis, and thymus. Female rats had greater oxfendazole exposure and exhibited toxicities at lower doses than did males. Decreased white blood cell levels, a class effect of benzimidazole anthelmintics, returned to normal during the recovery period. The no observed adverse effect level was determined to be >5 but <25 mg/kg/d and the maximum tolerated dose 100 mg/kg/d. The highest dose, 200 mg/kg/d, resulted in significant toxicity and mortality, leading to euthanization of the main study animals in this group after 7 days. Oxfendazole did not exhibit genetic toxicology signals in standard Ames bacterial, mouse lymphoma, or rat micronucleus assays nor did it provoke safety concerns when evaluated for behavioral effects in rats or cardiovascular safety effects in dogs. These results support the transition of oxfendazole to First in Human safety studies preliminary to its evaluation in human helminth diseases.
Introduction
The majority of benzimidazole (BZ) carbamate anthelmintics in veterinary and human clinical use were approved over 30 years ago. Despite their extensive use for soil-transmitted helminths (STH) and other parasitic diseases, the BZs albendazole (ALB) and mebendazole (MEB) are far from perfect. Their efficacy against some human helminth species is clearly suboptimal 1,2 and if submitted today would probably not obtain approval for some of these indications. A very limited number of treatments are available for some zoonotic helminth infections, such as neurocysticercosis and echinococcosis, and all available treatments for these diseases are less than ideal in terms of efficacy or safety and not appropriate for use in control programs. 3
In addition, widespread use of BZs in animal health practice has led to drug resistance in helminths of domestic animals (sheep, cattle, horses, goats, etc). 4 Although there has been little evidence of resistance in helminth species that affect humans (due in part to the differing approach to treatment of human helminths, with, until recently, single annual dose treatment being the norm), attempts at control of a number of human helminth species such as lymphatic filariasis and STH have resulted in a massive increase in drug use, with whole communities being treated. As a response to the potential threat of drug resistance, 5 as well as to increase the drug armamentarium available for most parasitic diseases (particularly tissue-dwelling larval helminths), there is a pressing clinical need for alternative drugs to be developed.
A number of BZ anthelmintics used in veterinary medicine have been considered for transition to human use, including oxibendazole and oxfendazole (OXF). In addition, flubendazole, already approved for use as an intestinal anthelmintic, is also being developed as a treatment against adult filarial worms causing river blindness and elephantiasis 6 in humans. Oxfendazole has long been under preclinical investigation as a potential new treatment for neurocysticercosis and echinococcosis. The initial placebo-controlled study of Taenia solium cysticercosis in pigs (the normal host for larval forms of the cestode) showed that a single high dose of OXF (30 mg/kg) but not praziquantel (PZQ; 50 mg/kg PZQ) could eliminate the encysted larvae 7 as observed in necropsy 10 to 12 weeks after drug administration. A follow-up study demonstrated the dose-dependent cysticidal effects of OXF in several tissues, with no viable cysts being observed at the highest dose of OXF tested, 30 mg/kg. 8 A third study confirmed that cysts do not die immediately, the decrease in viable cysts becoming more pronounced across the 12-week time frame of the study 9 ; 12 weeks after the administration of a single dose of OXF, viable cysts had been fully eliminated from muscle, heart, and tongue and fully cleared from the brains of 3 of 4 pigs studied. Importantly, the pharmacokinetic study of OXF disposition in pigs revealed that the metabolic half-life (t1/2) of OXF is long, 21.6 hours, 10 a value comparable to its t1/2 in ruminants.
Although there is considerable experience with BZ anthelmintics, both individually and as a class, many of the available data come from experience postregistration rather than from extensive preclinical testing. Since the original work on most BZ anthelmintics was conducted some 30 to 40 years ago, the toxicological data used for the original approvals are limited, often unpublished, and do not comply with current regulatory standards. The lack of a requirement to update basic toxicological data or to conduct new studies once a compound is approved means that there has been little incentive to undertake new studies and even less to place any data gathered into the public domain. Thus, in order to transition any BZ from animal to human use, it is necessary to fill the current data gaps and to bring the dated toxicology package up to current regulatory standards. Typically, the areas lacking in these earlier toxicological evaluations are genetic toxicology, pharmacokinetics and toxicokinetics (TKs), safety pharmacology, and the species and numbers of animals studied. Using the available published data on OXF, 11,12 the data from the original toxicology package, 13 and knowledge of BZ class toxicology, 14 a series of OXF studies in rat and dog were devised to provide missing data and to bring the toxicology package up to date in support of an Investigational New Drug application for a First in Human (FIH) study. Toxicokinetic evaluation is also a key component of the present studies, as TK data for OXF have not been available in the public domain.
Methods
Animal Care
Animal care and housing were in accordance with the National Research Council Guide for the Care and Use of Laboratory Animals (1996) and the Animal Welfare Standards incorporated in 9 CFR Part 3, 1991. All use of animals was approved by the Institutional Animal Care and Use Committee and conducted at SRI International (Menlo Park, California) in a facility accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International.
Rat Pharmacokinetics
Three groups of Sprague-Dawley rats, each consisting of 3 males and 3 females, were administered OXF, 1 group at each of the following pharmacologic doses: 5 mg/kg intravenously (iv), 15 mg/kg orally, or 60 mg/kg po. Oral dose administration was by gavage. Blood samples were drawn into EDTA-containing tubes for determination of OXF concentration predose and at 5, 15, and 30 minutes, 1, 2, 4, 8, 12, 24, 32, and 48 hours (iv group) or 0.5, 1, 2, 4, 6, 8, 12, 24, 32, and 48 hours (oralgroup).
Plasma was separated from the blood samples by centrifugation within 30 minutes of collection. Rat plasma (80 μL samples) was treated with 350 µL of acetonitrile (containing 25 ng/mL MEB as an internal standard) to precipitate plasma proteins. These mixtures were then vortexed for 15 minutes and suspensions were then clarified by centrifugation (18 000g, 15 minutes); 300 µL of the resulting supernatants were transferred to a polypropylene container for solvent evaporation at 40°C. The dried samples were reconstituted with 150 µL 25% (v/v) acetonitrile in Milli-Q water. The samples were sonicated for 5 minutes and then vortexed for an additional 10 minutes. Samples were analyzed by liquid chromatography tandem mass spectrometry analysis using a Waters 2795 Alliance Integrated System (Waters Corp.; Milford, MA) with a Phenomenex Gemini C6-Phenyl column (3 µm, 50 × 2.0 mm 110A) (Torrance, CA). Mobile phase was A= 0.1% (v/v) acetic acid in water and B = 0.1% (v/v) acetic acid in acetonitrile. Study samples were quantified using a set of calibration standards prepared in blank matrix that were processed in parallel. This analytical method had an lower limit of quantitation of 5 ng/mL. Pharmacokinetic parameters were calculated from the resulting data with WinNonlin Professional (Pharsight Corp., Mountain View, CA).
Rat Toxicokinetics
Groups of Sprague-Dawley rats (3 male and 3 female per dose) were orally administered 5, 25, 100, or 200 mg/kg of OXF for 14 days, and blood samples were drawn for determination of plasma OXF levels on days 1 and 14 at 0.25, 1, 3, 6, 12, and 24 hours following OXF administration. Samples were processed and analyzed as described earlier for the rat PK study.
Rat Functional Observational Battery
The functional observational battery (FOB) was conducted on the toxicology study rats (5 male and 5 female per group) at 3 to 5 hours following oral administration of 0, 5, 25, 100, or 200 mg/kg of OXF on day 1 of dosing. The FOB components, designed to fulfill the requirements of the Food and Drug Administration (FDA) Guidance, 15 included home-cage observations, handling observations, open-field observations, and reflexes and physiological measurements. Each component had several end points that were evaluated to yield approximately 30 end points in total. Parametric data included rearing, defecation, urination, landing foot splay, fore- and hind-limb grip strength, body weight, and body temperature. Positive control groups of rats were administered amphetamine (5 mg/kg, males) or carbaryl (50 mg/kg, females).
Two-Week Toxicology Study in Rats
Groups of Sprague-Dawley rats (10 male and 10 female per dose) were orally administered 5, 25, 100, or 200 mg/kg of OXF for 14 days, doses spanning those studied in previously conducted rat toxicology studies. 13 Half the animals (5 males and 5 females) were sacrificed 24 hours after the last dose administered, and the other half was kept on study for an additional 2 weeks without dosing for evaluation of recovery from any changes resulting from OXF administration. Clinical observations were recorded once daily (approximately 2-4 hour postdose) on days of treatment, or more often as clinical signs warranted, and on the day of sacrifice. Animals were examined for any altered clinical signs, including gross motor and behavioral activity, and observable changes in appearance. Body weights were determined before dosing and weekly thereafter. Food consumption was recorded at 24-hour intervals once a week throughout the study. Blood samples were taken 24 hours after administration of the last dose (day 15 or earlier when animals in the high-dose group were euthanized) and on day 28 for the recovery group. Measurements included hematology and clinical chemistry measurements. Urine for urinalysis (protein, glucose, bilirubin, urobilinogen, and cell content) was collected during the week prior to each scheduled sacrifice. At necropsy, major organ systems and tissues were collected and weighed and then examined for gross lesions as well as for microscopic changes.
Dog Cardiovascular Safety and Tolerability Study
Three male and three female beagle dogs were orally administered vehicle followed by rising doses of OXF (5, 25, or 100 mg/kg), with a 2-week interval between doses. Postdose evaluations included clinical observations (4-8 hour postdose when possible, or more often as clinical signs warranted), body weight, body temperature, clinical pathology (hematology, serum chemistry, and coagulation), and cardiology parameters (blood pressure, heart rate, and electrocardiogram).
Genetic Toxicology Evaluation
Ames test
The potential mutagenic activity of OXF was examined in the Salmonella typhimurium–Escherichia coli/microsome plate incorporation assay. 16 –18 A range-finding experiment was conducted with strain TA100 over doses ranging from 125 to 4000 μg/plate in the presence and absence of a metabolic activation (MA) system containing 5% Aroclor 1254-induced rat-liver microsomes (S9). The maximum test concentration was based on the limit of OXF solubility. Mutagenicity was then studied in 2 experiments with 5 tester strains TA1535, TA1537, TA98, and TA100 and E coli strain WP2 (uvrA) at doses ranging from 125 to 4000 μg/plate, in both presence and absence of MA containing 5% S9 (first experiment) or 10% S9 (second experiment).
Mouse lymphoma gene mutation assay
The ability of OXF to induce mutations at the thymidine kinase locus (tk) in the L5178Y mouse lymphoma cell tk +/− → tk −/− gene mutation assay was examined using a standard procedure. 19,20 Cells were exposed to OXF for 4 and 24 hours in the absence of MA and for 4 hours in the presence of MA. Additional details on cell growth, positive control treatments, data collection, and statistical analysis were published earlier. 21 The end points evaluated were cell growth during expression periods, relative suspension growth (RSG), relative total growth (RTG), relative cloning efficiency, mutation frequency (MF), and numbers of small (≤ 0.6 mm in diameter) colonies from trifluorothymidine-resistant cells.
Micronucleus evaluation
Bone marrow smears were prepared from the left femur of each rat (5/sex/group) taken on day 15 in the main group of rats in the 2-week toxicology study. All the main group slides and positive control (cyclophosphamide) slides, the latter prepared under a separate Good Laboratory Practice regulations study and randomized in with the study slides, were stained, coded, and evaluated for the presence of micronuclei using standard procedures. 22 –24
Results
Rat Pharmacokinetics
Rats administered OXF by the iv or the oral route exhibited plasma levels of the drug (Table 1), with higher exposures seen at the 60 mg/kg than at the 15 mg/kg po dose. The t1/2 for OXF was approximately 2.5 hours. The PK parameters obtained following iv administration indicated that OXF distributed to extravascular locations and was eliminated rather quickly (only 10% remained at about 8 hours postdose). There was a possible trend for higher systemic exposure, extravascular distribution, and longer elimination time in females compared to males, and these sex-dependent differences were more apparent at the higher oral doses.
Calculated Parameters From Rat PK Study.
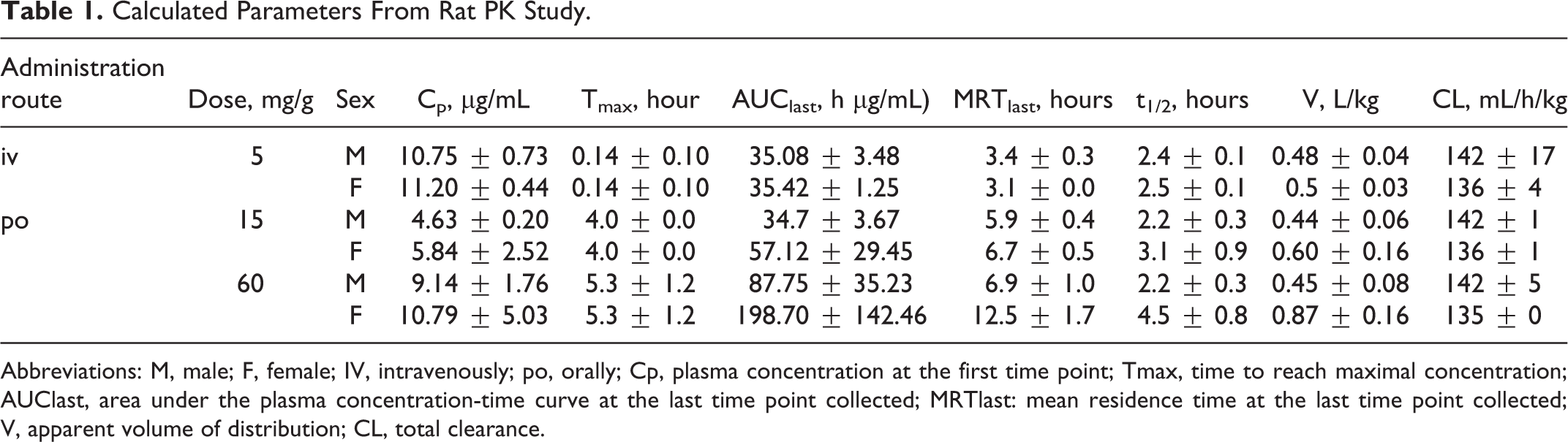
Abbreviations: M, male; F, female; IV, intravenously; po, orally; Cp, plasma concentration at the first time point; Tmax, time to reach maximal concentration; AUClast, area under the plasma concentration-time curve at the last time point collected; MRTlast: mean residence time at the last time point collected; V, apparent volume of distribution; CL, total clearance.
Rat Toxicokinetics
Rats orally administered OXF exhibited dose related, although not fully dose proportional, exposure to OXF, as expressed in the Cmax and area under the curve (AUC; Tables 1 and 2; Figure 1). Exposures were higher in females than in males, findings paralleled by higher volumes of distribution and t1/2 values. Note, for example, that females administered a 100-mg/kg oral dose of OXF had Cmax and AUC values similar to the values in males receiving 200 mg/kg (Table 2 and Figure 1).
Toxicokinetic Parameters of Oxfendazole on Day 1 and Day 14.
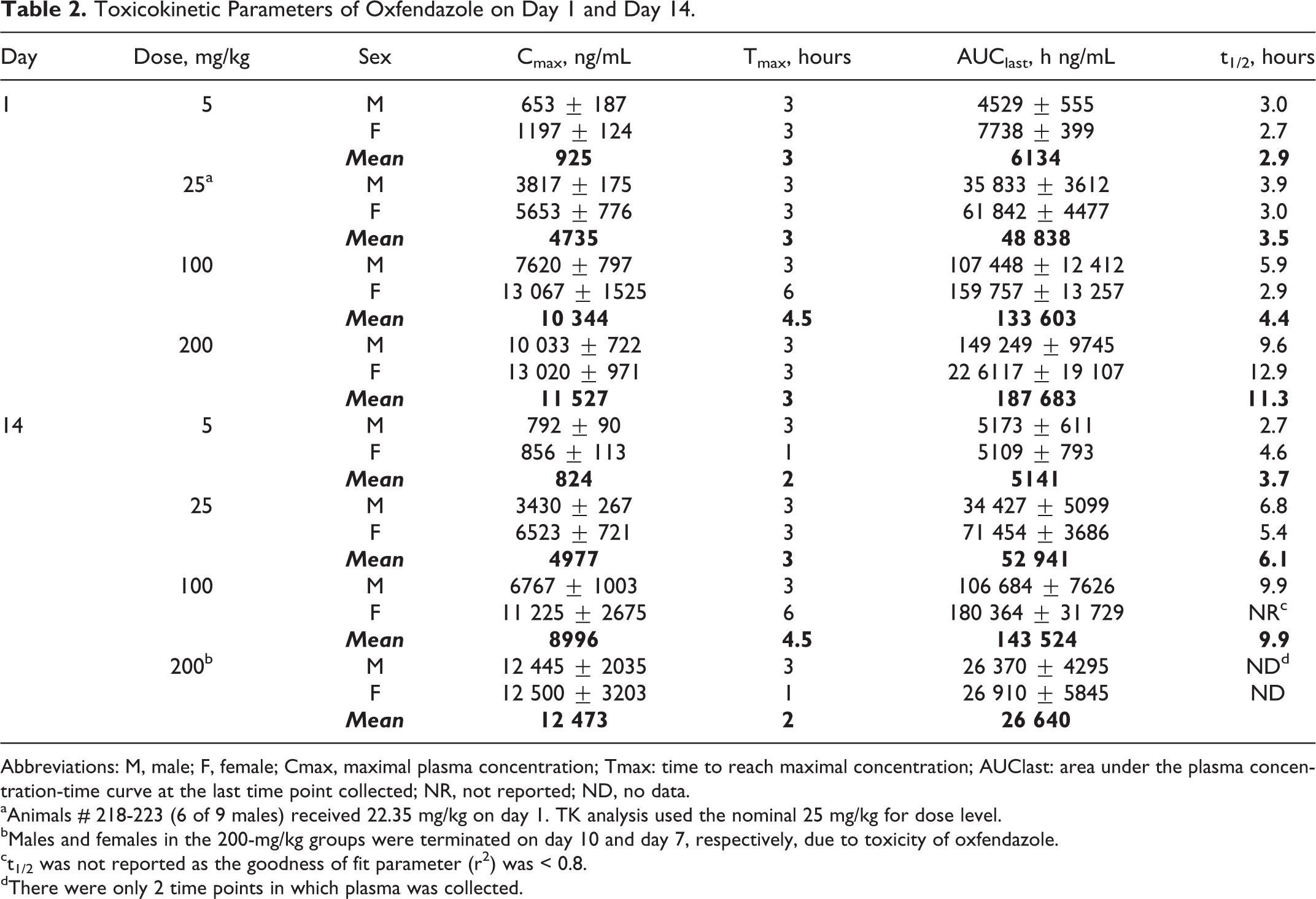
Abbreviations: M, male; F, female; Cmax, maximal plasma concentration; Tmax: time to reach maximal concentration; AUClast: area under the plasma concentration-time curve at the last time point collected; NR, not reported; ND, no data.
aAnimals # 218-223 (6 of 9 males) received 22.35 mg/kg on day 1. TK analysis used the nominal 25 mg/kg for dose level.
bMales and females in the 200-mg/kg groups were terminated on day 10 and day 7, respectively, due to toxicity of oxfendazole.
ct1/2 was not reported as the goodness of fit parameter (r 2 ) was < 0.8.
dThere were only 2 time points in which plasma was collected.
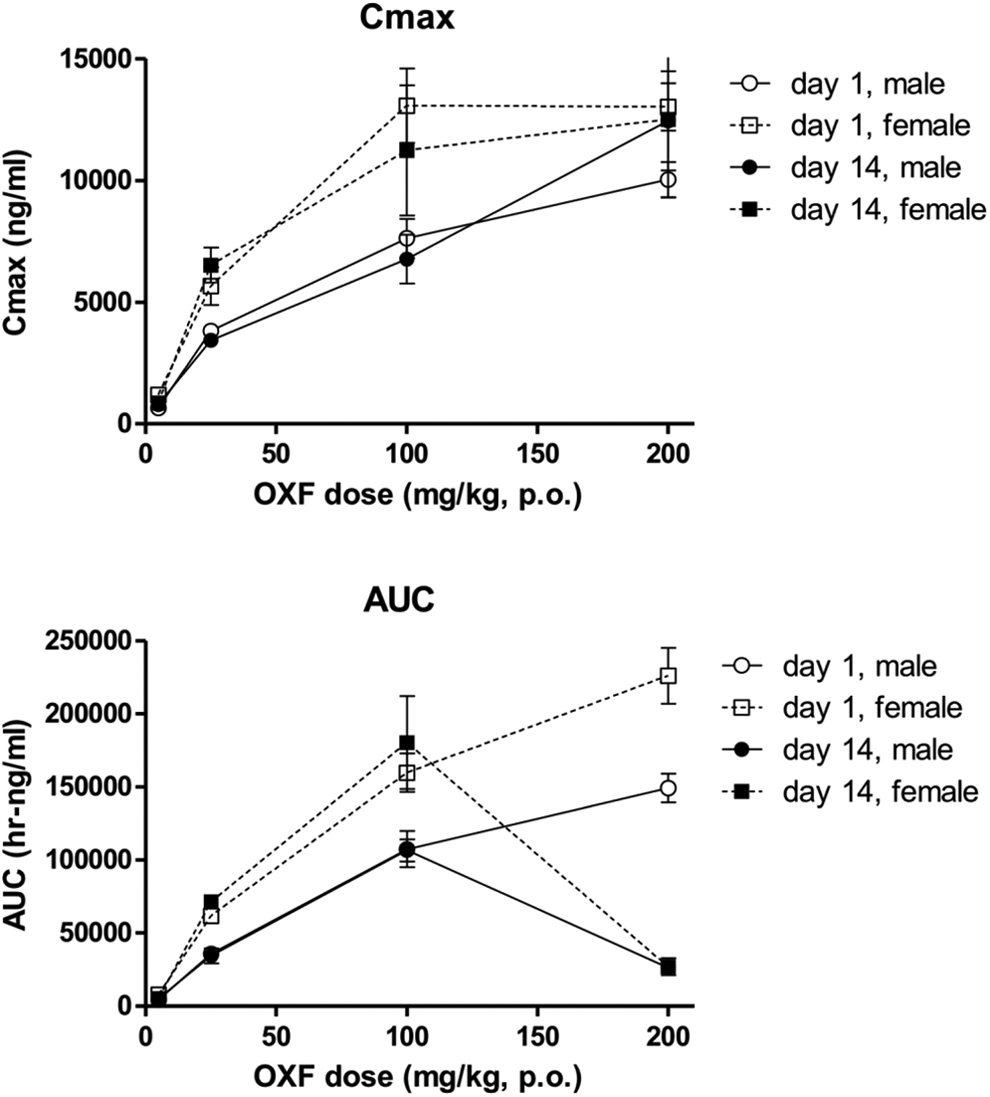
Exposure to oxfendazole in rat toxicity study. Shown are the Cmax (upper panel) and area under the curve (AUC; lower panel) values obtained in the rat 2-week toxicity study. The symbols represent the calculated oxfendazole values (±SEM) determined in the plasma of rats administered the stated oxfendazole doses. Data were obtained from 3 rats per gender per dose. Note that males and females in the 200-mg/kg group were terminated on days 10 and 7, respectively, because of toxicity. SEM indicates standard error of the mean.
Rat Functional Observational Battery
No changes at any FOB end point (home-cage, handling, open field, reflexes, and physiology components) were observed in male rats treated with OXF compared with rats administered vehicle. Female rats treated with OXF exhibited a statistically significant reduction in landing foot splay, but in the absence of a dose-dependent relationship, similar observations in males, or significant findings for related end points, this finding was not considered to be toxicologically relevant. In contrast, treatment of rats with the positive control substances, amphetamine or carbaryl (data not shown), resulted in significant changes in behavior and physiology.
Two-Week Toxicology Study in Rats
Following repeated oral administration of OXF (5, 25, 100, or 200 mg/kg/d), clinical changes were seen at only the 2 highest dose levels, 100 and 200 mg/kg. Observations included hunched posture; swelling (soft or hard) in the chin, muzzle, throat, and genitalia; paraphimosis; red discharge in the nose, mouth, and head; ruffled fur; hypoactivity; and yellow discolored stool and/or diarrhea. Mortality was seen in some females treated with 200 mg/kg OXF after 6 to 8 days of repeated dose administration. Of the 19 females on test, 6 were found dead between days 7 and 9, and 6 were sacrificed in moribund condition on day 7. This high-dose group was terminated before the scheduled sacrifice, and the recovery group was allowed to continue, without treatment, after 7 days of dosing. In general, the OXF-related treatment mortality and clinical findings were more severe in females than in males at the same dose levels.
A significant decrease in body weight gain in the high-dose group (compared with the control group) was observed in the first week of treatment and in the recovery phase after the last dose administration. On day 8, the high-dose group was 5% and 10% lower than control body weights in males and females, respectively. Mean weight gain in the high-dose group during the day 1 to 8 interval was 28 g versus 44.3 g in controls in males, and 0 g versus 16.5 g in females. Although sporadic changes in food consumption, both increases and decreases, were observed throughout the study, there was no clear association between food consumption and body weight changes in any of the treatment groups.
A dose-dependent decrease in white blood cell (WBC) count was seen at day 15 in females, with corresponding decreases in WBC subtypes (lymphocytes, neutrophils, monocytes, eosinophils, and basophils). The effect on WBC was severe in females, with decreases relative to controls of 8%, 13%, 51%, and 75% at 5, 25, 100 (on day 15), and 200 mg/kg (on day 7), respectively. Levels returned to normal historical ranges by the end of the recovery period (day 22 or 28). Effects seen in males were much less severe, with decreases in WBC seen in only the 200 mg/kg group (36% decrease in day 8 males vs day 15 control male level). White blood cells recovered in surviving animals once OXF administration was suspended. Minor effects on red blood cell (RBC) count were seen, but these were less significant than the WBC effects. None of the clinical chemistry changes were considered toxicologically relevant.
The primary target organs of toxicity identified microscopically were bone marrow, epididymis, liver, spleen, testis, and thymus. Bone marrow cell line (myeloid megakaryocytes and neutrophils) depletion was present on day 8 in male rats given 200 mg/kg OXF and on days 15 and 8 in female rats administered 100 and 200 mg/kg, respectively. This finding is consistent with the pronounced effects on WBC seen in these animals. The presence of epididymal multinucleated sperm precursors in rats given 25, 100, and 200 mg/kg OXF was noted on study day 15 (25 and 100 mg/kg) and day 8 (200 mg/kg), respectively. This finding was still present in the recovery groups in male rats treated with OXF at 100 mg/kg (day 28) and 200 mg/kg (day 22). Bilateral tubular atrophy was present on day 8 in the testes of 2 rats given 200 mg/kg OXF, and it was present unilaterally on day 28 in the testes of 1 rat given 5 and 1 given 100 mg/kg OXF. These effects, consistent with the decreased testis weight noted at necropsy, were not seen on day 22 in the recovery animals from the 200-mg/kg/d treatment group, indicating that these effects are reversible following cessation of treatment.
Hepatic midzonal fatty change was present in male and female rats given 25 and 100 mg/kg OXF (day 15) and those given 200 mg/kg OXF (day 8). After a 14-day recovery, for females in the 25, 100, and 200 mg/kg groups and males in the 100 and 200 mg/kg groups, the changes were still present. This effect is consistent with mottled, pale, dark, or discolored livers seen at necropsy. However, no effects on liver enzymes were seen in the clinical chemistry results, suggesting that the fatty change did not result in overt hepatotoxicity or compromised hepatic function; therefore, the toxicologic significance of this observation is unclear. Splenic and thymic lymphoid depletion was present on day 8 in male and female rats given 200 mg/kg OXF and on day 15 in female rats given 100 mg/kg OXF. On recovery day 28, splenic lymphoid depletion was still present in 1 female animal in the 100 mg/kg group, and thymic lymphoid depletion was still seen in 3 females in the 100 mg/kg group and 1 female in the 200 mg/kg group.
Thus, repeat daily oral administration of the highest dose of OXF, 200 mg/kg/d, resulted in significant toxicity and mortality in Sprague-Dawley rats. The main study animals in this group were euthanized after 7 consecutive daily oral dose administrations. Hematology, necropsy, organ weight, and histopathological results identified bone marrow, epididymis, liver, spleen, testis, and thymus as potential target organs of toxicity. On the basis of the toxicology end points evaluated in this study, the maximum tolerated dose (MTD) of OXF is approximately 100 mg/kg/d for a 14-day oral dose administration in rats. The no observed adverse effect level (NOAEL) is considered to be greater than 5 mg/kg/day but less than 25 mg/kg/day for a 14-day oral dose administration in rats.
Dog Cardiovascular Safety and Tolerability Study
Following the administration of single oral OXF doses of 5, 50 and 100 mg/kg to 3 conscious dogs of each sex, no changes were observed in the cardiovascular (CV) parameters measured (blood pressure, heart rate or electrocardiogram). Neither were there any measured changes in clinical observations, body weight, postdose body temperature, or clinical pathology (hematology, serum chemistry, and coagulation). Thus, based on the parameters evaluated in this study, the MTD was not reached but appears to be greater than 100 mg/kg for administration of a single oral dose of OXF to dog. The NOAEL is at least 100 mg/kg for a single oral dose administration of OXF in beagle dogs.
Genetic Toxicology Evaluation
Ames test
No cytotoxicity was seen at any dose level in the range-finding experiment conducted with strain TA100. In the follow-up experiments, precipitate was seen on the plates at OXF doses of 2000 μg/plate, but no statistically significant increase in the number of revertant colonies was seen under any test condition. Control values were consistent with historical values obtained in this test and laboratory. Oxfendazole was judged to be nonmutagenic under the test conditions used in this study; therefore, OXF was determined to be negative in the bacterial reverse mutation assay.
Mouse lymphoma gene mutation assay
In the gene mutation assay using L5178Y mouse lymphoma cells, cytotoxicity (< 20% RSG) was observed following exposure to OXF levels ≥200 μg/mL for 4 hours in the presence of MA. In an initial mutagenicity experiment conducted at dose levels of 20, 40, 80, 160, and 320 μg/mL in the presence of MA for 4 hours, clones were obtained at all but the highest dose; inadequate cell growth precluded cloning at this dose. Neither statistically significant increases in MF compared with controls nor statistically significant dose-related increases in MF were found in the clones obtained from the treated cultures. In the follow-up experiment at OXF dose levels of 10, 20, 40, 80, and 160 μg/mL for 4 hours in the presence of MA, data from the highest dose level were not included in the statistical analysis, since its RTG was less than 10%. Neither statistically significant increases in MF compared with controls nor statistically significant dose-related increases in MF were observed in the treated cultures.
In the absence of MA, cytotoxicity was observed at 400 μg/mL for 4-hour exposure and at ≥25 μg/mL for 24-hour exposure. In the initial mutagenicity experiment conducted at dose levels of 10, 20, 40, 80, 160, and 320 μg/mL in the absence of MA for 4-hour exposure, clones were obtained from cells treated with all doses. Because inadequate RTG was observed at 320 μg/mL, these data were not included in the statistical analysis. Neither statistically significant increases in MF compared with controls nor statistically significant dose-related increases in MF were observed in the treated cultures. In the follow-up experiment, conducted at dose levels of 1.25, 2.5, 5, 10, and 20 μg/mL for 24 hours, clones were obtained from cells treated at all dose levels. Because inadequate RTG was seen at the 10 and 20 μg/mL dose levels, these results were not included in the statistical analysis. Neither statistically significant increases in MF compared with controls nor statistically significant dose-related increases in MF were found in the treated cultures. Under the test conditions, OXF met the criteria for a negative response as defined for this study.
Micronucleus evaluation
No significant suppression of polychromatic erythrocytes (PCEs) occurred among RBCs, and no statistically significant increase in the frequency of micronucleated PCE were seen in rats treated with OXF for 2 weeks. Thus, OXF was considered negative for the induction of micronuclei under the test conditions used in this mammalian bone marrow micronucleus assay.
Discussion
Pharmacokinetics and Toxicokinetics
The metabolic t1/2 observed here for OXF in rat is shorter than that found in any other monogastric (dog, 6 pig 10 , and horse 25 ) or multi-gastric (cattle 25 and sheep 25 ) species previously reported. In fact, the 2-hour t1/2 observed for rat is one-tenth of that observed in most other species studied; of the other species studied, only dog exhibited a t1/2 of less than 20 hours, namely 5.5 hours.
In the present study, exposure to OXF in female rats exceeded that measured in males. The gender difference increased with OXF dosage through the 100 mg/kg level and was reflected in higher Cmax (ng/mL) and AUC (h-ng/mL) values in females than in males.
Although measurement of OXF metabolites was not part of the present study, a previously conducted rat PK study 25 found that OXF sulfone was the predominant plasma species across the 24 hours following OXF administration, accounting for 71% of the OXF-related moieties quantified. In contrast, in most other species studied, including cattle, 25 sheep, 25 dog, 6 and pig, 10 OXF itself was the predominant 0 to 24-hour plasma analyte. In horse, as in rat, an OXF metabolite predominated, but in horse that metabolite was fenbendazole 25 rather than oxfendazole sulfone as found in rat. 25
In contrast, following oral administration of the anthelmintic BZ ALB, the parent compound is barely detectable in the plasma. Rather, the analytes present are ALB sufoxide and a secondary metabolite, ALB sulfone, 26 the former considered generally responsible for the drug’s therapeutic and toxic effects. Another BZ anthelmintic, MEB, is itself considered the active moiety; its metabolism proceeds via inactivation pathways, reduction, and conjugation. 26
Toxicology
Oxfendazole
In the present rat studies, significant decreases in body weight at the higher OXF doses were accompanied by lowered WBC levels and a corresponding decrease in WBC types, including neutrophils. The observed target organs of toxicity, bone marrow, epididymis, liver, spleen, testis, and thymus corroborate those stated in the Review Summary of the New Animal Drug Application (NADA) approval. 13 In 2-week toxicology studies in rat, OXF reduced neutrophil levels at the higher dose levels, 33 mg/kg in females and 100 mg/kg in both females and males. In those NADA-supporting studies, doses of 33 and 100 mg/kg were also associated with reduced activity in lymphoid, testicular, and bone marrow tissues. Importantly, in both the NADA supporting study 13 and in the present investigation, WBC levels returned to normal during the study recovery phase.
The gender difference in toxicity observed in the present study is probably due to differences in OXF exposure, being higher in female than in male rats administered the same oral doses. It is not known whether female rats were also exposed to higher levels of the metabolite OXF sulfone or whether female rats biotransformed OXF more slowly to its sulfone, leaving higher levels of the parent compound. Indeed, in the present PK study, the t1/2 for OXF is longer in female than in male rats, especially at the higher oral dosage. Females also had a higher OXF volume of distribution following the drug’s oral administration. In any case, the greater toxicity in female rats is probably due to greater exposure to OXF, to an OXF metabolite, or both.
The target organs of toxicity in dog 13 appear to be similar to those observed here and previously in rat: In 2-week toxicology studies in dog, 13 some animals in all OXF treated groups (11, 33, or 100 mg/kg) exhibited reduced myeloid maturation in bone marrow, and most OXF-treated dogs exhibited splenic tissue and thymic atrophy. These supportive findings are particularly interesting, given the difference in the predominant OXF-related analyte in the 2 species, OXF in dog and OXF sulfone in rat, suggesting that both OXF and OXF sulfone have similar toxicologic profiles.
Because the toxicities observed in the present rat studies may have been induced by moieties other than OXF itself, the predictive value of the present toxicology findings to those that will be observed in human is presently unknown, as the metabolic profile of OXF in humans has not yet been determined. However, rat and dog are the 2 most widely used animal species for assessment of human risk of small molecules. Differences in metabolism and pharmacokinetics aside, these species are reasonable surrogates for the estimation of potential human risk, and data from these species will ultimately be used to set conservative, safe starting doses for initial human clinical trials.
Other BZs
Only limited data from mouse, rat, and dog toxicology studies on other BZs exist in the public domain; summaries of these data are available in published reviews from the regulatory agencies, many of which have been complied by Dayan. 26 From these data, the toxicology profiles of ALB and MEB appear similar to that of OXF. For example, repeated administration of high doses of ALB to rats led to leukopenia, enlargement of the liver, and testicular hyperplasia. 26 Similarly, administration of MEB in the diet to rats for 13 weeks led to an inhibition of body weight gain, anemia, hepatocyte vacuolation, liver weight gain, and testicular damage. 26 Most probably, the testicular effects of ALB derive from its disruption of microtubules, 27 a mechanism shared by other BZs, including OXF and MEB.
Despite limited publically available nonclinical metabolism and toxicology data, the BZ, ALB, and MEB are widely used as anthelmintic agents in both veterinary practice and human treatment. 26,28 Undoubtedly, ALB’s safety profile and ease of administration has contributed to its use in the treatment of whole communities. 28 The similar preclinical toxicologic profile of OXF to those of ALB and MEB hold promise for a suitable clinical safety profile for OXF.
Genetic Toxicology
Oxfendazole was not mutagenic in a 5 tester strain Ames assay, with or without MA, a finding similar to that obtained for ALB. 26 Further, OXF did not induce mutations in a mouse lymphoma cell line, with or without MA. Oxfendazole was also negative for the induction of micronuclei in a mammalian (rat) micronucleus assay, whereas ALB gave a positive mouse lymphoma cell response. 26 Indeed, ALB as well as its metabolite ALB sulfoxide induced nondisjunction in cultured human lymphocytes 29 at doses lower than those at which micronuclei were observed. And importantly, the lymphocytes of patients with hepatic hydatid disease showed increased sister chromatid exchange and micronuclei following ALB treatment. 30 Thus, with a cleaner preclinical genetic toxicology profile, OXF may have a more favorable clinical profile in this regard.
Although the current study demonstrated lack of genotoxicity of OXF in the 3 most widely used genotoxicity assays, previously published reports have presented somewhat conflicting results. El-Makawy et al 31 reported OXF to be both genotoxic and teratogenic in mice, whereas Holden et al 32 reported no chromosomal effects of OXF in human lymphocytes. Based on the current data, conducted in full compliance with current FDA testing requirements, genotoxicity does not appear to be a significant risk factor for OXF, giving it a more favorable profile than other BZs.
Toward First in Human Studies
Earlier work on OXF includes chronic toxicity studies in rat and dog. 13 In those 1-year studies, rat was more sensitive to the adverse effects of OXF than was dog, the no effect dose for rat being 0.7 mg/kg/d and for dog being 13.5 mg/kg/d. Extrapolating (using a body surface area calculation) from the results of those chronic OXF toxicology studies, the most conservative estimate of a no-effect chronic dose for humans (based on an NOEL derived from a 1-year study) is 0.13 mg/kg. A conservative dose of 0.3 mg/kg has been proposed as the starting point for a single dose Phase I OXF study. Furthermore, since the principal toxicology target organs appear to be the bone marrow and liver, safety monitoring for the proposed Phase I study includes clinical chemistry indicators of bone marrow and liver function. Coupled with OXF’s lack of induction of behavioral effects in rat, CV effects in dog or genetic toxicology signals in standard Ames bacterial, mouse lymphoma, or rat micronucleus assays, the results of the present rat pharmacokinetic, TK, toxicology, and safety studies support the investigation of OXF as a potential anthelmintic medication against human helminth species.
Footnotes
Author Contributions
Codd contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Ng contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. McFarlane contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Riccio contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Doppalapudi contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Mirsalis contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy Horton contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; an d agreed to be accountable for all aspects of work ensuring integrity and accuracy Gonzalez contributed to conception or design; contributed to acquisition, analysis, or interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Garcia contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy. Gilman contributed to conception or design; contributed to acquisition, analysis, or interpretation; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded in whole or in part with Federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under Contract No. HHSN266200600011C / N01-AI-60011. HHG is supported by a Wellcome Trust Senior International Research Fellowship in Public Health and Tropical Medicine.
