Abstract
Background
Obesity is associated with a higher prevalence of endometrial hyperplasia, thereby increasing the risk of endometrial and ovarian cancers. The precise mechanisms linking obesity to endometrial hyperplasia remain unclear, but dysregulation of adipose tissue homeostasis is known to play a significant role. Hypertrophied adipocytes in obese individuals secrete various bioactive substances, including cytokines, growth factors, hormones, and metabolites. Additionally, hyperplastic adipocytes exhibit enhanced aromatase activity, leading to increased estrogen synthesis, which further promotes the development of endometrial hyperplasia.
Purpose
The purpose of this study is to explore the anti-inflammatory and anti-proliferative activities of the poly unsaturated fatty acids.
Methodology
An extensive literature survey has been performed to identify the role of adipokines and elevated endogenous estrogen levels in activating cell survival signaling pathways, such as PI3K/Akt/mTOR, MEK/ERK1, and JAK–STAT in endometrial cells and their possible role in Endometrial Hyperplasia. Further, the possible beneficial anti-inflammatory and anti-proliferative effects of polyunsaturated fatty acids (PUFAs) such as eicosapentaenoic acid (EPA), docosahexaenoic acid (DHA), and arachidonic acid (AA) were explored.
Results
Numerous studies suggest the beneficial role of dietary fats, such as EPA, DHA, and AA in modulating the growth of endometrium in obesity-induced endometrial hyperplasia. PUFAs can activate adenosine monophosphate-activated protein kinase (AMPK), which inhibits gluconeogenesis and lipogenesis. It also phosphorylates acetyl-CoA, leading to a decrease in malonyl-CoA, which inhibits mitochondrial CPT1. Additionally, AMPK activation promotes β-oxidation, and PPAR-γ mechanisms by down regulating the NF-kB pathway involved in endometrial hyperplasia.
Conclusion
This review sheds light on the potential of PUFAs in mitigating estrogen synthesis, adipokine secretion, and endogenous aromatase activity in obesity induced endometrial hyperplasia. Furthermore, it critically evaluates the role and mechanisms of PUFAs in attenuating obesity-associated endometrial hyperplasia and reducing the risk of ovarian cancer.
Introduction
Obesity is a chronic condition that has been escalating worldwide, affecting both adults and children, and is associated with a range of health issues, including type 2 diabetes, hypertension, sleep apnea, fatty liver, atherosclerosis, degenerative diseases, metabolic syndromes, and cancers.1, 2 Recent research has revealed a concerning link between obesity and gynecological cancers, particularly endometrial and ovarian cancers, in women, leading to increased morbidity and mortality.3–5 This situation poses a significant burden on healthcare and the economy, especially in India, where the prevalence of endometrial cancer is projected to surge by over 50% by 2030, posing a global healthcare threat. 6 The current standard treatments for ovarian and endometrial cancers involve surgery, radiation, chemotherapy, and combination therapies. However, in obese women, surgical removal of the uterus, tubes, and ovaries for early-stage cancers can be challenging. 7 Additionally, obese individuals may face greater difficulties in coping with radiation and chemotherapy compared to lean cancer patients. 8 These limitations have prompted scientists to explore alternative treatment and prevention strategies for obese ovarian and endometrial cancer patients.
In recent years, researchers have focused on investigating the potential of dietary molecules in combating endometrial and ovarian cancers. Adipocytes play a crucial role in regulating energy storage and homeostasis, producing hormones, growth factors, and adipokines. 9 Excessive fat accumulation in white adipose tissue (WAT) can lead to obesity, and it is worth noting that the uterus is surrounded by visceral adipose tissue in humans. 10 Endometriosis, a chronic inflammatory disease characterized by the presence of endometrial tissue outside the uterus, is progesterone-resistant and estrogen-dependent, leading to dysmenorrhea, infertility, pelvic pain, heavy menstrual bleeding, and other gynecological problems. 11 This disease can also affect adjacent organs such as the fallopian tubes, bladder, and rectosigmoid colon, impacting 5–15% of women of child-bearing age and 30–50% of infertile women. 12 Among the 20 most common tumor types, endometrial cancer exhibits the strongest association with obesity, resulting in 200,000 new cases annually and 270,000 deaths. 13 Furthermore, ovarian cancer ranks as the 11th most prevalent cancer among women, with over 22,000 new cases estimated to be diagnosed in the coming year, primarily affecting women in the age range of 55–64 years, different from endometriosis14, 15 Numerous studies have shown a strong positive correlation between body mass index (BMI) and the incidence of endometrial and ovarian cancer,16, 17 highlighting the significant attention drawn to the association between obesity and these cancers.
To shed light on the emergence of endometrial and ovarian malignancies in obese individuals, many scientists and researchers have focused their efforts on investigating adipose-derived variables and their significance.18–20 In this regard, peroxisome proliferator-activated receptor-gamma (PPARγ), a nuclear hormone receptor, plays a central role in regulating adipocyte differentiation, 21 lipid metabolism, 22 and insulin sensitivity. 23 Its activation enhances adipocyte function and lipid storage, contributing to the development of WAT and obesity.24, 25 Additionally, dysregulation of PPARγ signaling in adipocytes and other tissues may contribute to the development of hyperplasia in the endometrium and ovaries, potentially promoting cancer development. 26 In the context of obesity and its implications in endometrial and ovarian cancers, dietary molecules such as PUFAs have garnered attention for their ability to modulate PPARγ activity. 27 Omega-3 fatty acids, a type of PUFA, have been shown to activate PPARγ and exhibit anti-inflammatory and anti-cancer properties. 18 Discussing the signaling and metabolism of PPARγ lays the foundation for the subsequent sections of this review. These sections delve into the potential implications of these molecular processes in endometrial and ovarian cancers, particularly in the context of obesity. By integrating information about PPARγ, its role in adipose tissue function, and its modulation by polyunsaturated fatty acids (PUFAs), this review provides a more comprehensive understanding of the link between obesity, adipose tissue, and the development of gynecological cancers. This approach allows readers to gain deeper insights into the rationale behind exploring adipokines, PUFAs, and their potential therapeutic implications in the context of endometrial and ovarian cancers, offering a more insightful perspective for the review article.
Fat Deposited Adipocytes and their Secretions
Adipocyte hypertrophy refers to the enlargement of adipocytes, which are the specialized cells responsible for storing and releasing fat in the body. 28 This process leads to alterations in adipocyte function and the release of various secretory products, including adipokines, free fatty acids, and inflammatory molecules. 29 These secretions contribute to the complex interplay between adipose tissue, metabolism, inflammation, and the development of obesity-related disorders. 30 Understanding the mechanisms underlying adipocyte hypertrophy and its secretions is important for unraveling the pathophysiology of obesity and developing therapeutic strategies to mitigate its adverse effects. A substantial part of the human body, adipose tissue accounts for 20–40% of the body’s total volume and is essential for controlling inflammation, lipid and glucose metabolism, and energy homeostasis. 31 Adipose tissue pertains in three distinct forms: WAT, which stores energy as fat; brown adipose tissue (BAT), which is specialized for producing heat and is involved in thermogenesis; and beige adipose tissue, which combines the traits of both WAT and BAT and has the potential to be thermogenic under specific circumstances. Additionally, adipose tissue acts as active sites for sex steroid metabolism and produces adipokines, which possess both systemic (endocrine) and local (autocrine or paracrine) functions. 32 Preadipocytes mature into fully functional adipocytes by accumulating excess lipids and undergoing abnormal expansion. Consequently, hypertrophic adipocytes experience fibrosis, cellular stress, and local hypoxia, leading to inflammation and adipocyte dysfunction. 33 These hypertrophic adipocytes alter proinflammatory factors, including monocyte chemoattractant protein-1 (MCP-1), which causes local invasion of immune cells and recruitment of macrophages. 34 Further, they stimulate immune cells such as T cells and recruit macrophages, resulting in complement activation and the production of inflammatory cytokines. 35 In response to adipocyte inflammation and through their paracrine interactions, macrophages become activated, releasing inflammatory substances. This process leads to the formation of crown-like structures, which serve as microscopic indicators of macrophage-adipocyte inflammation. 36 Additionally, macrophages secrete various proinflammatory mediators in adipose tissue, including prostaglandin (PGE2), 37 interleukin (IL)-1β, 38 IL-6, tumor necrosis factor-α (TNF-α), 39 transforming growth factor-beta 1 (TGF-β1), 40 leptin, 41 angiopoietin-1, 42 hepatocyte growth factors such as vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), 43 growth hormones, 44 and other bioactive compounds. 45 Table 1 presents information on the levels of adipokines, inflammatory markers, and growth factors in normal adipocytes and hypertrophic adipocytes, and Figure 1 shows their paracrine actions. However, the excessive and inappropriate accumulation of mature white adipocytes is a defining characteristic of obesity. Despite this understanding, the precise mechanisms through which mature adipocytes influence the initiation and advancement of cancer cells in the endometrium and ovaries are still not fully understood or clear.
Impact of Ovarian Cancer Progression and Metastasis.

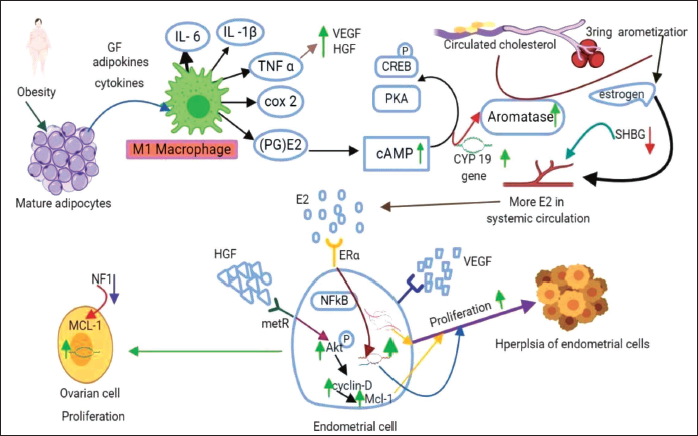
Adipocyte secretion and hyperplasia refer to the processes by which adipocytes, or fat cells, secrete various molecules and grow in number, respectively. These processes are crucial for the functioning of adipose tissue, which serves as the body’s primary energy storage site in the form of fat. 46 Adipokines are a class of bioactive molecules secreted by adipocytes, and they play essential roles in numerous physiological processes, including inflammation, 47 metabolism, 48 and insulin sensitivity. 49 Adipokines can be classified as cytokines or hormones because of their ability to act as signaling molecules and regulate cellular responses. They are involved in various physiological functions, such as regulating energy homeostasis, appetite, and glucose metabolism. Leptin, for instance, is an adipokine that plays a crucial role in appetite regulation and energy balance. It acts on the hypothalamus in the brain, suppressing appetite and increasing energy expenditure.
Adiponectin, on the other hand, enhances insulin sensitivity and has anti-inflammatory properties, making it beneficial for metabolic health. While adipokines have been extensively studied in the context of metabolic diseases, their specific involvement in cancer development, particularly endometrial and ovarian cancers, is an area of ongoing research. Some adipokines have been linked to cancer development and progression due to their effects on inflammation and insulin sensitivity.
PUFAs are essential fatty acids that cannot be synthesized by the body and must be obtained from dietary sources. Common types of PUFAs include omega-3 fatty acids and omega-6 fatty acids. PUFAs have been studied extensively for their potential health benefits, including their anti-inflammatory properties and potential role in cancer prevention. Emerging research suggests that PUFAs may modulate adipokine secretion. For instance, omega-3 fatty acids have been associated with increased adiponectin levels, which may contribute to improved insulin sensitivity and reduced inflammation. On the other hand, omega-6 fatty acids may influence the production of other adipokines, potentially affecting the inflammatory state and metabolic function. The relationship between adipocyte secretion, adipokines, PUFAs, and cancer development is complex and not yet fully understood. Adipose tissue produces a wide array of bioactive molecules, and only a subset of these molecules has been identified as being critically linked to endometrial hyperplasia and other cancers. Some adipokines may promote a proinflammatory environment or contribute to insulin resistance, both of which are factors associated with cancer development. However, other adipokines, like adiponectin, have shown potential anti-cancer properties due to their anti-inflammatory and insulin-sensitizing effects. 50 Figure 2 represents a simplified overview of the interactions between adipokines, PUFAs, and cancer development. To fully comprehend these complex relationships, further research is required to elucidate the specific roles of individual adipokines, the influence of PUFAs on their secretion, and their collective impact on cancer development. 21
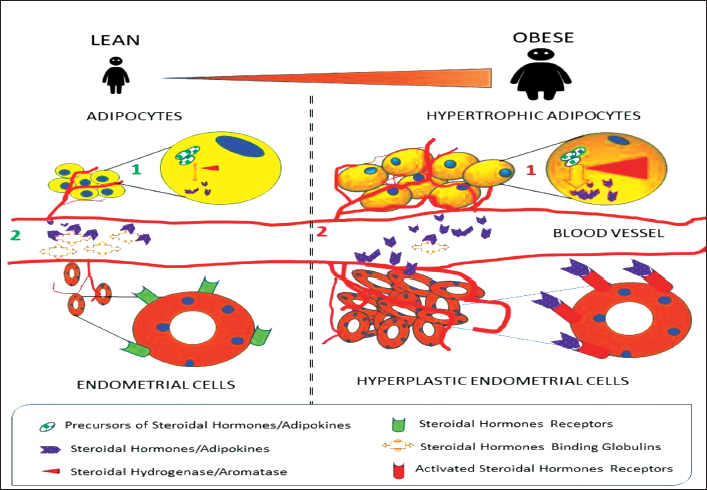
Adipokines can exert their effects locally within the adipose tissue, influencing the behavior of neighboring cells and the overall adipose microenvironment. However, many adipokines can also enter the bloodstream and act as circulating hormones, affecting distant organs and tissues throughout the body. The systemic effects of adipokines can have broad implications for overall health, as these molecules may influence metabolic regulation, immune responses, and other physiological processes in various organs. Consequently, the dysregulation of adipokine secretion and hyperplasia may contribute to the development of various diseases, including cancer.
Leptin and Adiponectin
Leptin is a 16-kilodalton (16 kDa) protein that is secreted by mature adipocytes and plays a crucial role in regulating energy balance and body weight. 51 It acts on the hypothalamus in the brain to suppress appetite and increase energy expenditure. 52 However, leptin is not solely involved in metabolism; it also has other physiological effects, including potential implications in cancer development and progression. 53 Regarding endometrial and ovarian cancers, there is evidence suggesting that leptin may contribute to the development of these diseases. 54 Leptin receptors are present in the endometrium, and leptin signaling can influence various processes involved in endometrial cancer progression. Mammalian cells have two major isoforms (obesity receptor): Ob-Ra short form and Ob-Rb long form. 55 High levels of leptin have been associated with an increased risk of endometrial cancer. Obese receptors Ob-Rb and Ob-Ra are highly active in inflammation. When the mature adipocytes secrete more leptin, it binds to the OB-receptor and activates the Janus kinase–signal transducer of activation (JAK–STAT) signaling pathway. Activated STAT subsequently causes the upregulation of apoptosis inhibitor encoding genes, cell cycle regulators such as B-cell lymphoma-2 (BCL-XL), myeloid cell leukemia (MCl-1), 56 and cyclin-D, which increase the cell proliferation, thereby leading to oncogenesis. 57 Leptin can promote cell proliferation and inhibit apoptosis in endometrial cancer cells through activation of signaling pathways involved in cell growth and survival. 58 Additionally, leptin can stimulate angiogenesis, which is the formation of new blood vessels, thus promoting tumor growth and metastasis. 59
Leptin and its receptors are also found in ovarian tissues, and studies have suggested that leptin may contribute to the development and progression of ovarian cancer. 60 Rapid JAK/STAT3 pathway activation by leptin results in the activation of two critical cell-growth signaling pathways: Extracellular signal-regulated kinases (ERK) and protein kinase B (AKT). Leptin-induced activation of nuclear factor kappa B (NF-κB) inhibits apoptosis in cancer cells via the nuclear factor kappa B inducing kinase (NIK)/nuclear factor kappa B inducing kinase (IKK) signaling pathway. Adiponectin directly lowered the survival of normal human endometrial stromal cells without changing the levels of AdipoR1 and AdipoR2. Adiponectin has an anti-proliferative effect on endometrial cancer via stimulating the activation of the activated protein kinase (AMPK) pathway and decreasing the activation of the phosphatidylinositol (PI3K)/AKT, mitogen-activated protein kinase (MAPK)/ERK1/2, and IGF pathways. 61 Serum IL-6 concentrations were significantly greater in healthy females during the luteal phase of the menstrual cycle. Furthermore, decreased blood levels of dehydroepiandrosterone were associated with increased IL-6 concentrations, and vice versa. 61 Leptin can promote cell proliferation, inhibit apoptosis, and enhance the invasive potential of ovarian cancer cells. It can also stimulate the production of other growth factors and cytokines involved in cancer progression. 62 Furthermore, patients with ovarian cancer have reported having worse outcomes when their leptin levels are high. While there is evidence connecting leptin to endometrial and ovarian cancers, it is important to recall that the precise pathways through which leptin contributes to these cancers are not entirely understood. The relationship between leptin and cancer is complex and likely involves interactions with other factors in the tumor microenvironment. 63 It is important to mention that obesity is a known risk factor for endometrial and ovarian cancers. Obesity is characterized by excess adipose tissue and high leptin levels. 64 Adipose tissue produces more leptin when a person is obese, which may explain why certain malignancies are more common and aggressive in obese people. Adiponectin stimulates the connection between phosphotyrosine interacting with PH domain and leucine zipper 1 (APPL-1) and AMPK, which in turn promotes glucose uptake through glucose transporter 4 (GLUT4), hence insulin-sensitizing skeletal muscle and adipose tissue. Given that insulin resistance is linked to an increased risk for a number of cancers, low levels of adiponectin may therefore raise the risk for oncogenesis. Adiponectin reduces the migration of endothelial cells induced by VEGF and inhibits the proliferation of endothelial cells driven by fibroblast growth factor-2 (FGF-2). Recombinant mouse adiponectin was injected intralesionally into hypervascularized murine fibrocarcinomas, resulting in a 60% reduction in tumor volumes and weights. This was followed by an increase in tumor apoptosis, which was mediated by activating caspase-3. These results imply that adiponectin may function as a strong angiogenesis inhibitor, activating apoptosis, and thereby preventing tumor growth. 65
The structure of adiponectin is the only factor that determines its particular biological action. The high molecular weight (HMW) form of adiponectin, whose active form makes up nearly 70% of circulating adiponectin in healthy subjects, has the most noticeable role in protecting against diabetes and improving insulin sensitivity, leading to different metabolic diseases in the case of impaired multimerization sensitivity. Non-HMW adiponectin, or complexes with lower molecular weight, exhibit stronger anti-inflammatory effects. Adiponectin has an anti-atherosclerotic action in the cardiovascular system. Furthermore, it has been demonstrated that this adipokine, with its pro- and anti-inflammatory qualities, regulates immune system cell function and plays a significant role in human reproduction. 66
Growth Factors
One group of adipokines that has gained significant attention is growth factors, which are proteins that regulate cell growth, proliferation, and differentiation. 67 Growth factors play crucial roles in normal physiological processes like tissue development, wound healing, and immune response. 68 However, dysregulation of growth factors can contribute to the development and progression of various diseases, including cancer. Adipokines secreted by adipose tissue can affect the development and function of the endometrium. Some growth factors released by adipocytes, such as insulin-like growth factor 1 (IGF-1), PI3-k/Akt, and Ras/MAPK pathway, are both activated by IGF-1, leading to increased glucose metabolism, increased protein synthesis, and increased cell proliferation. PI3-kinase/Akt suppresses apoptosis by blocking Bcl-2-associated agonist of cell death (BAD) and forkhead protein (FKHR) and activates nuclear factor-κB for cell survival. Through direct binding in the extracellular space, IGF-1 binding proteins regulate the bioavailability of IGF-1. Through direct interactions with cell membrane-bound proteins like integrins, insulin-like growth factor-binding proteins also have a number of IGF-independent effects. 69 Insulin resistance-related tumors include breast cancer, prostate cancer, ovarian cancer, and endometrial cancer. Appropriate receptor expression has been found in a number of these tumors. Furthermore, via activating through AdipoRs, which were expressed in both tissue samples and cell lines, adiponectin inhibits the proliferation of endometrial cancer. 70 VEGF and adiponectin have been implicated in the pathogenesis of endometrial cancer. 71 Increased levels of these growth factors can promote cell proliferation, angiogenesis, and inflammation, which are all associated with tumor growth and progression. 72 Growth factors can influence the microenvironment of the ovaries, potentially contributing to ovarian cancer development. 73 Growth factors such as TGF-β and insulin-like growth factor binding protein 1 (IGFBP-1) have been associated with ovarian cancer progression and poor prognosis. 74 Obesity is characterized by an increase of mature white adipocytes, the major adipocyte subtype in adult humans, together with excess whole-body AT. The hypertrophied adipocytes in obese individuals secrete higher levels of VEGF, EGF, leptin, and angiopoietin-1 (ANGPT1). These chemicals are necessary for AT to be able to connect with many types of cells. Angiogenesis and endothelial cell proliferation both depend on VEGF, a major endothelial cell growth factor. VEGF signals are transduced via the transmembrane tyrosine kinase receptors VEGFR-1 and VEGFR-2, which are expressed on the vascular epithelium. VEGFR-2 is the primary functional receptor that transduces angiogenic and VEGF-mediated signals, whereas VEGFR-1 may act as a decoy receptor. After ligand binding, VEGFR-2 undergoes autophosphorylation of its tyrosine kinase domain. Tyr1175 phosphorylation forms a docking site where the adaptor protein Shb and the PI3 kinase p85 subunit can attach. PI3K substrates on the cell membrane, which is translocated by the p85 subunit, regulate the PI3K/AKT/mTOR pathway. 75 Forty-eight percent of ovarian cancers express the EGF-receptor (EGFR), while 10–35% of ovarian tumors have activated EGFR. There are several EGFR ligands in ascites. Ascites components also induce EGFR transactivation in addition to ligand activation. Through transcriptional activation, EGFR activation controls the production of matrix metalloproteinase (MMP). PEA-3, a transcription factor belonging to the ETS family, is associated with the advancement of disease and is implicated in the control of MMP that is dependent on EGF. Transmembrane (MT1-MMP) or membrane-associated (MMP-9) proteinases’ pericellular location can be modulated by non-transcriptional methods of MMP regulation. Intracellular adaptor proteins, which convey signals through cascades like the RAS/RAF/MEK/MAPK and PI3K/AKT cascades, are responsible for the transduction of EGFR signals. In these signaling cascades, the downstream proteins have the ability to go from the cytoplasm to the nucleus, where they bind to transcription factors and the complexes they form, including MYC, ETS like-1 (ELK), and activator protein-1 (FOS/JUN). Numerous cellular responses, including proliferation, differentiation, motility, survival, and carcinogenesis, are regulated by signal transduction via the EGFR family to downstream pathways and cascades. 76
The precise mechanisms through which these variables contribute to cancer development are currently being investigated, as the relationship between adipocyte secretions, growth factors, and cancer is intricate and multifaceted. However, by understanding the interactions between growth factors, adipose tissue secretions, and gynecological tumors, it may become possible to identify potential therapeutic targets and develop preventive and treatment strategies. PPARγ also plays such a prominent role in illness, many medication research efforts are focused on finding PPAR agonists. Fatty acids and their derivatives in large doses have the ability to activate PPAR. Long-chain PUFAs, such as leukotriene B4 and 8-S-hydroxyeicosatetraenoic acid, always have more effects than other PPAR activators. Additionally, a number of prostanoids, including 15-deoxy-12, 14-prostaglandin J2, and 15-hydroxyeicosatetraenoic acid, can activate PPAR. It is well known that 15d-PGJ2 has an impact. Thiazolidinediones (TZDs), which are artificial PPAR ligands, are widely known for their exceptional effectiveness in controlling insulin sensitivity and blood glucose levels. 129
Estrogen
Estrogen, primarily produced by the ovaries in premenopausal women, is a group of hormones that play a crucial role in the development and functioning of the female reproductive system. However, adipose tissue, especially visceral fat, can also produce estrogen through the conversion of androgens into estrogens by the enzyme aromatase. 77 This becomes particularly relevant after menopause when ovarian estrogen production decreases significantly, and adipose tissue becomes a more significant source of estrogen. Estrogen can influence the development of endometrial cancer and ovarian cancer as it is known to stimulate the growth of the endometrium, which is the lining of the uterus. 78 Prolonged exposure to high levels of estrogen without the counterbalancing effects of progesterone can result in abnormal thickening of the endometrium, thereby increasing the risk of endometrial cancer. 79 Obesity, which is associated with greater amounts of adipose tissue and increased estrogen production, has been identified as a risk factor for endometrial cancer. The relationship between estrogen and ovarian cancer is complex, and some studies suggest that heightened exposure to estrogen may be linked to an increased risk of developing specific types of ovarian cancer, particularly endometrioid and clear cell ovarian cancers.80, 81 However, the precise mechanisms behind this association are not fully understood. It is important to note that other factors, including genetics and hormonal imbalances, also play a role in the development of ovarian cancer.
Aromatase
Aromatase is the enzyme responsible for converting androgens into estrogens. Although it is true that aromatase is expressed in various tissues, including adipose tissue, the direct involvement of adipocyte-secreted aromatase in the development of endometrial and ovarian cancers is not well-established. In endometrial cancer, the predominant risk factor is prolonged exposure to estrogen without the counterbalancing effects of progesterone. 82 While adipose tissue can produce estrogen through aromatase activity, the main source of estrogen in premenopausal women is the ovaries. 83 After menopause, when ovarian estrogen production declines, adipose tissue becomes a more significant source of estrogen. However, the majority of estrogen production in postmenopausal women occurs in peripheral tissues, including adipose tissue, rather than ovarian tissue. Therefore, while adipose tissue-derived estrogen may contribute to endometrial cancer risk in postmenopausal women, it is not solely mediated by adipocyte-secreted aromatase. 84
The relationship between aromatase and ovarian cancer is not as well-established as it is in breast cancer. 33 Aromatase expression and activity in ovarian cancer cells have been reported in some studies, suggesting a potential role in the local production of estrogens within the ovarian tumor microenvironment.85, 86 However, the precise contribution of adipocyte-secreted aromatase to the development of ovarian cancer is not fully understood. Ovarian cancer has various subtypes with different underlying molecular mechanisms, and the involvement of estrogen in the development of different subtypes can vary. 87 Development of endometrial and ovarian cancers is influenced by a combination of genetic, hormonal, and environmental factors. While estrogen and aromatase are known to play a role in these cancers, their exact contributions are still under investigation.
Excess Fat Deposition
Hyperplasia is known to play a significant role in both obesity and the development of endometrial and ovarian cancers. Obesity, often caused by an imbalance between calorie intake and energy expenditure, can result in hyperplasia, leading to an increase in the number of adipocytes. In obesity, the enlarged adipose tissue releases various factors, such as adipokines, inflammatory cytokines, and hormones like estrogen. These factors can disrupt the body’s metabolic balance and contribute to the development of obesity-related health issues like insulin resistance, 88 type 2 diabetes, 89 cardiovascular diseases, 90 and certain types of cancer.91, 92 Estrogen stimulates the proliferation of endometrial cells, which can lead to hyperplasia. If left untreated, endometrial hyperplasia can progress to endometrial cancer. 93 The risk of progression depends on various factors, including the type of hyperplasia and the presence of cellular atypia. In the context of ovarian cancer, hyperplasia can be a precursor to the development of certain types of ovarian tumors (ovarian epithelial tumors). 94 Ovarian epithelial hyperplasia may progress to ovarian epithelial carcinoma, which is the most common and aggressive form of ovarian cancer. However, not all cases of hyperplasia lead to cancer, and other factors, including genetic mutations and environmental influences, play a role in tumor development.
The treatment strategies for endometrial and ovarian cancers typically involve a combination of surgery, radiation therapy, chemotherapy, and targeted therapy. The specific treatment plan depends on the stage of the cancer, the individual’s overall health, and other factors, and it is determined by a healthcare team. While diet alone cannot cure endometrial and ovarian cancers, it can play a supportive role in controlling and managing these cancers. Diets rich in PUFAs, which refer to fatty acids that have the potential to lower blood cholesterol levels, may have beneficial effects. 95 Certain types of PUFAs, such as omega-3 fatty acids, have been shown to have cholesterol-lowering effects and are often recommended in the context of endometrial and ovarian cancers.96, 97 Omega-3 fatty acids can impact the production and release of adipokines, which are molecules that influence various physiological processes. 98 In the case of endometrial hyperplasia, the influence of PUFAs on adipokines may have implications for the development and progression of the condition. 99
Crosstalk Between Adiponectin and Leptin Insulin and Estrogen
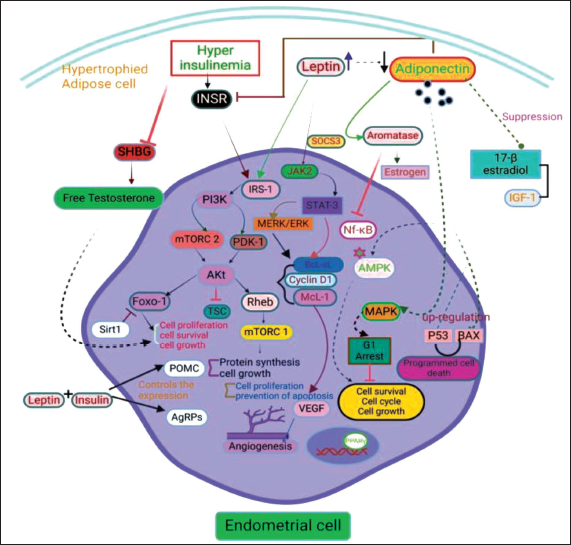
When leptin binds to the leptin receptor (LepRb), three tyrosine residues (Y985, Y1077, and Y1138) are phosphorylated, activating the receptor-associated kinase JAK2 through transphosphorylation. SOCS3 (suppressors of cytokine signaling) inhibits the mRNA expression of JAK–STAT that is stimulated by leptin. Through Foxo1 (forkhead box O1) and the signal transducer and STAT3, insulin and leptin control the expression of AgRP (agouti-related protein) and POMC (proopiomelanocortin). The orexigenic neuropeptide AgRP’s Foxo1-dependent expression is suppressed by Sirt1. 100 Insulin binding to either hybrid or homodimeric INSR receptors causes certain residues in the receptor subunit’s cytoplasmic tail to be autophosphorylated. This, in turn, activates effector proteins, including INSR substrate (IRS) proteins, through the phosphotyrosine-binding (PTB) and Src homology 2 (SH2) domains. Following the recruitment of PI3K to the plasma membrane and its activation into phosphatidylinositol (3,4,5)-trisphosphate (PIP3), insulin signaling is then enhanced. Phosphoinositide-dependent protein kinase-1 (PDK1) then phosphorylates Akt/PKB at the Thr308 residue. An essential step for the best activation of Akt/PKB’s kinase activity is Ser 473 phosphorylation, which is achieved in parallel by insulin’s activation of the mTORC2. After being triggered, Akt/PKB phosphorylates the tuberous sclerosis complex (TSC), preventing its guanosine triphosphate (GTPase) activating protein action in the direction of the small GTPase Ras homolog enriched in brain (Rheb), so permitting the stimulation of mTORC1 downstream. IR is encouraged by sustained mTORC1 activity, which may be brought on by an excess of nutrients. Angiogenesis, proliferation, metabolism, survival, and differentiation are just a few of the numerous biological processes that are involved in the development of tumors that are regulated by the PI3K/Akt/mTOR axis. 101 Both granulose and epithelial ovarian cancer cells produce adiponectin receptors, and their expression is linked to a more advanced stage of the disease as well as a shorter overall and progression-free survival. Additionally, adiponectin suppresses the proliferation-promoting effects of 17β-estradiol and IGF-1 on OVCAR-3 and SKOV-3 ovarian cancer cells by downregulating the expression of their receptors. In OVCAR3, OVCAR4, and A2780 ovarian cancer cells, stimulation of adiponectin receptors has also been demonstrated to cause G1 cell cycle arrest and encourage apoptosis. 65 Mainly acting through the liver kinase B1/AMP-activated protein kinase (LKB1/AMPK) pathway, adiponectin inhibits signaling pathways that are involved in the initiation of the cell cycle, cell growth, and survival. These pathways include c-Jun N-terminal kinase (cJNK), 1/2 (ERK1/2), PI3K/protein kinase B (Akt), and STAT3. Adiponectin also controls the expression of several other proteins involved in the cell cycle and programmed cell death, including p53 and Bax, which are upregulated, and c-myc, cyclin D1, and Bcl-2, which are downregulated. Furthermore, by inhibiting the phosphorylation of NF-κB, a transcription factor involved in controlling the activity of several proinflammatory cytokines, adiponectin has anti-inflammatory effects. 65 Figure 3 shows schematic representation.
PUFAs: Structural Integrity of Dietary Fatty Acids (DFAs): Impact on Metabolic Health
A plethora of both epidemiological and interventional studies suggest that DFAs distinctly influence metabolic health based on their structural integrity. 102 PUFAs are subclassified into two types, that is, n-3 (omega-3) and n-6 (omega-6) fatty acids. These PUFAs are obtained through dietary sources including fatty fish and seed/vegetable oils, and they are considered to be pivotal in several physiologic processes, including inflammation. The metabolites of PUFA, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), were shown to be beneficial in combating metabolic health. 103 By showing the different effects on lipid metabolism, conversely, the plasma levels of palmitate, a free fatty acid derived from saturated fats, were found to be elevated and attributed to the lipotoxicity in obese individuals, suggesting the distinct roles of fatty acids depending on their structural integrity. 104
PUFAs are involved in cascades including inflammation, 105 oxidative stress, 106 estrogen metabolism, 107 cell survival and proliferation, 108 extracellular matrix remodeling, 109 and remodeling of whole-body lipid metabolism. 110 Essential PUFAs can be classified as ω-3 or ω-6 fatty acids, and they have been the subject of research regarding their impact on serum cholesterol levels and their potential role in cancer treatment, including ovarian cancer. Omega-3 fatty acids, in particular, have been investigated for their anti-cancer characteristics and their potential benefits in managing ovarian cancer. 111
PUFAs and Aromatase Enzyme
As previously reported, the enzyme aromatase turns androgens into estrogens, and this conversion mainly takes place in peripheral tissues such as adipose tissue, the liver, and the brain. Estrogens play crucial roles in various physiological processes, such as sexual development, reproductive function, and bone health. Research suggests that PUFAs, particularly long-chain omega-3 fatty acids, can modulate the activity of the aromatase enzyme.112, 113 Long-chain omega-3 fatty acids, such as EPA and DHA, are abundant in fatty fish and certain plant sources like flaxseed and walnuts. Several studies have demonstrated that omega-3 fatty acids can inhibit the expression and activity of aromatase, potentially leading to reduced estrogen synthesis.114, 115 According to these studies, the main mechanism associated with obesity and fatty acids is the interaction between genes and fatty acids. This interaction influences the composition of membrane fatty acids, transcriptional regulation, and post-transcriptional processes, ultimately modifying gene expression. 116 Therefore, PUFAs are considered DFAs since they are essential for the human body and must be obtained from the diet. Unlike saturated fats and monounsaturated fats, which can be produced by the body or obtained from dietary sources, polyunsaturated fats cannot be synthesized by the body and therefore must be consumed through food. These can modulate the expression levels of genes and transcription factors involved in cellular responsiveness to metabolic signals, lipid metabolism, and energy regulation. 117 The impact of DFAs on obesity depends on their chain length and degree of desaturation.
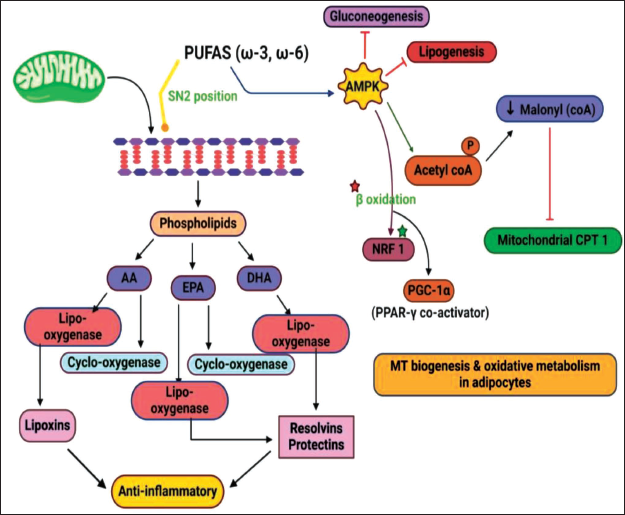
Two major transcription factors that interact with DFAs are sterol regulatory element-binding protein-1 (SREBP-1) and PPAR. Both of these factors play key roles in gene regulation, cellular differentiation, development, and the metabolism of lipids, proteins, and carbohydrates.118, 119 The impact of PUFAs on aromatase levels and inflammatory markers has been studied, but the exact mechanisms and optimal dosages are still being investigated. Additionally, other possible mechanisms may also influence the synthesis of higher estrogen levels in hypertrophied adipocytes. PUFAs have been shown to reduce the risk of certain types of cancers, although many of the molecular mechanisms behind this effect remain unknown. Some mechanisms have been elucidated and are depicted in Figure 4, but there is still much to be discovered regarding the molecular mechanisms by which DFAs reduce cancer risk.
Anti-inflammatory and Anti-cancer Properties of PUFAs
PUFAs, such as EPA and DHA, have been extensively studied and shown to possess beneficial properties.11, 120 They have potent anti-inflammatory effects, inhibiting the production of inflammatory molecules and alleviating symptoms of chronic inflammatory conditions. 121 PUFAs also have potential as anti-cancer agents, interfering with cancer cell growth and survival, and reducing the risk and progression of various cancers.96–99, 102 They can enhance the effectiveness of chemotherapy and radiation therapy while minimizing side effects.122, 123 These effects are attributed to PUFAs’ ability to modulate cellular processes, including gene expression, signal transduction pathways, and cell membrane composition, impacting cell signaling and receptor function.97–99 PUFAs have an endogenous proresolution activity that helps regulate the innate inflammatory response by interfering with the conversion of arachidonic acid (AA). 124 EPA and DHA can reduce C-reactive protein (CRP) levels and counteract the increase in proinflammatory cytokines caused by a high-fat diet. 125 PUFAs activate NF-κB signaling and upregulate PPARγ, while decreasing PGE2 levels in serum. 126 They also inhibit superoxide production, phagocytosis, MHC II expression, and the release of leukocyte antigens, TNF-γ, IL-1β, and IL-6. 127
Regular long-term consumption of EPA and DHA through diet has shown an inverse association with the risk of endometrial cancer. 128 This suggests that PUFAs can reduce the risk and progression of cancer. Women with high levels of circulating and tissue n-3 PUFAs also have a lower risk of endometrial cancer, highlighting the potential of these fatty acids in combating cancer pathogenesis.129, 130 Epidemiological studies support the inverse association between dietary consumption of n-3 PUFAs and the risk of endometrial cancer. 131 Mechanistic pathways have been proposed to explain how dietary PUFAs exert their beneficial effects on metabolic health and endometrial cancer. 132 However, the precise mechanisms by which n-3/n-6 PUFAs affect adipocyte-derived growth factors and their subsequent effects on endometriosis and ovarian cancer pathogenesis remain poorly understood.
Explaining the possible distinctions between DHA and EPA requires an understanding of the molecular mechanism by which omega-3 PUFA controls adiponectin expression. The transcription of the adiponectin gene has been identified as being critically regulated by nuclear PPARγ. It has been shown that DHA and EPA activate PPARγ in vitro to enhance adiponectin expression; however, this finding is debatable. So, it may suggest that PUFAs potentially reduce the different types of cancers, including ovarian cancer. 133 n-3 PUFAs inhibit tumorigenesis at different sites, while n-6 PUFAs, particularly AA, promote tumor growth and inflammation. DHA acts as an endogenous ligand of retinoid X receptor (RXR-α), impacting lipid metabolism.132, 134 Signaling pathways are the major therapeutic target for n-3 PUFAs. mTOR plays a role in cell growth, energy, proliferation, metabolism, and survival. mTOR consists of two complexes, mTORC1 and mTORC2, with distinct functions. Upregulation of the mTOR signaling pathway is a drug target in endometrial cancer and contributes to carcinogenesis and progression. 135 Alpha-linolenic acid (ALA) and its metabolite 13(S)-HODE have been associated with anticarcinogenic properties. The metabolites of 20-carbon PUFAs, such as leukotrienes and prostaglandins, are linked to cancer through processes like angiogenesis, cell proliferation, metastasis, and apoptosis. 136
EPA and DHA, PUFA’s, modulate gene expression through transcription factors, including PPARγ, which regulates fat cell differentiation and lipid metabolism. 137 PPAR, a major transcription factor, mediates the metabolic effects of PUFAs, including lipid catabolism. AMPK, a metabolic sensor, controls intracellular metabolic fluxes like lipid oxidation and lipogenesis. AMPK inhibits lipogenesis by phosphorylating acetyl-Co-A carboxylase, leading to decreased malonyl-CoA levels and potentially inhibiting mitochondrial carnitine palmitoyltransferase 1 (CPT-1). 138 AMPK also inhibits gluconeogenesis and induces mitochondrial biogenesis through NRF-1 and PPARγ coactivator PGC-1α (Figure 4). NF-κB, activated by toll-like receptor (TLR4), induces proinflammatory gene expression. 18 n-3 PUFAs can activate β-arrestin/transforming growth factor-beta-activated protein kinase 1 binding protein 1 beta (TAB-1) signaling, inhibiting cytochrome 450 family 19 subfamily (CYP-19) gene transcription and blocking NF-κB-mediated induction of proinflammatory factors, as observed in breast cancer cells, which may also apply to endometrial cells. 139
Conclusion
Estrogen synthesis is upregulated in obesity, along with fluctuating levels of cytokines and growth factors, which will further bind to the cell surface estrogen receptors (ERα and ERβ), leading to endometrial hyperplasia. This estrogen-induced hyperplasia of the tumor was found to be reduced by inhibiting the gene transcription of CYP19 and inflammatory markers through PUFAS, by reducing endogenous aromatase, which plays a role in the biosynthesis of estrogen (E2). The anti-inflammatory and antitumorigenic properties of PUFAs make it more interesting. Hence, obesity-induced endometriosis and ovarian cancer are treated by oral delivery of PUFAs. Recommending patients with obesity-induced endometriosis and ovarian cancer to have foods containing PUFAs also may serve the purpose.
Abbreviations
Footnotes
Acknowledgments
The authors acknowledge the financial support by the Indian Council of Medical Research.
Author Contributions
Athuru Supriya: Investigation, validation, and writing—original draft.
Ammu V. V. V. Ravi Kiran: Revised draft and proofread.
Thaggikuppe Krishnamurthy Praveen: Writing—revised draft, proofread, and supervision.
Declaration of Competing Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Indian Council of Medical Research (No. 3/1/2(10) Obs/2021-NCD-II).
