Abstract
Omega-3 polyunsaturated fatty acids (OPFA) have beneficial effects on inflammatory reactions and production of cytokines. They decrease the release of 5HT by platelets and possess vasorelaxant activity. This led them to be tried in the prophylactic treatment of migraine. After 4 weeks of a single-blind placebo run-in period, patients were randomized and treated in double-blind condition by placebo or OPFA 6 g a day for 16 weeks, followed by a 4-week placebo run-out period. The intention to treat population included 196 patients. Those who received all four treatment periods included 96 patients taking OPFA and 87 taking placebo. The primary efficacy analysis was the number of migraine attacks during the last 4 weeks of treatment. During this period, the mean number of attacks was 1.20 ± 1.40 in the OPFA group and 1.26 ± 1.11 in the placebo group (NS). The total number of attacks during the 4-month period of the study was significantly different between groups: 7.05 in the placebo group, 5.95 in the OPFA group (P = 0.036). Mean intensity, mean duration of the attacks and rescue medication use, were not significantly different between the two groups. Except for a significant difference against OPFA for eructations, the tolerance was satisfying. Despite a run-in placebo period of 1 month, a very strong placebo effect was observed in this trial: 45% reduction of the attacks between run-in and 4-month treatment period (55% in the OPFA group, P = 0.058). Finally, this large study did not confirm two previous studies based on a small number of patients.
Introduction
Non-pharmacological approaches and several pharmacological treatments are available for migraine prophylaxis but many patients complaint of side-effects of these drugs (1, 2). Thus new drugs are expected in this field; migraine prophylactic therapy requires a medical approach tailored to each patient.
In 1985 and 1986, two double-blind cross-over studies (3, 4), performed on a small number (5/15) of patients, reported a decrease in migraine attack frequency with the use of a product rich in ω-3 polyunsaturated fatty acids (OPFA, Maxepa, Pierre Fabre Medicamed, Boulogne, France). This product, used in the present study, is an oil from fish flesh containing 30% ω-3 PUFA (eicosapentanoic acid, EPA, 18%; and docosahexaenoic acid, DHA, 12%). It reduces serum triglycerides, has a platelet anti-aggregating effect and presents a vasorelaxant activity (5–7).
Polyunsaturated fatty acids (PUFA), which represent approximately 30% of intracellular fatty acids, are essential dietary components, not synthesized by humans. The fatty acid composition of lipid tissues can be modified by the composition of PUFA in the diet. DHA is the most abundant ω-3 PUFA in tissues, especially at the level of phospholipid membranes of the neurones. Several studies have shown that ω-3 acids are required for the optimal functioning of the nervous system (8, 9). Additionally, ω-3 PUFA have beneficial effects on both specific and non-specific inflammatory reactions and the production of cytokines; this led them to be tried therapeutically in several immunological and inflammatory processes. Some experimental and immunological studies have involved platelets, cytokines and inflammation, at least in part, in the pathophysiology of migraine (10), and the release of serotonin by the platelets is decreased after treatment by Maxepa (3). These physiopathological elements and the previous therapeutic studies led us to conduct a large multicentre study, vs. placebo, of this product as a potential prophylactic treatment for migraine.
Methods
Two hundred and fifty patients, between 18 and 65 years old, consulting general practitioners (39 participating centres), having given their written informed consent and fulfilling the IHS criteria for migraine with or without aura (11), and having between two and six attacks a month, were selected for this study. The protocol was accepted by an independent ethics committee. The flowchart is shown in Fig. 1.

Design of the study. Duration: 24 weeks with 16 weeks of double-blind study.
Patients were excluded from the study if they fulfilled any of the following criteria: arterial hypertension, an ongoing neurological or psychiatric illness, a prophylactic anti-migraine treatment ongoing or stopped less than 1 month before the study, a hormonal treatment for less than 3 months, or an anti-inflammatory treatment of long duration.
After a single blind placebo run-in period (D28 to D0) and confirmation of the inclusion criteria, eligible patients were included in the study.
The sample size calculation was based on a decrease of the number of attacks by 5% in the placebo group and 25% in the OPFA group; with an alpha-risk at 5% and a power of 90% the number of patients needed was estimated at 65 by group.
The patients were randomized into two groups and treated in double-blind conditions with placebo (olive oil and lactose) or Maxepa (omega-3 polyunsaturated fatty acids, OPFA), six capsules of 1 g each per 24 h (three in the morning, three in the evening) for 16 weeks (D0 to D112). This double-blind phase was followed by a 4-week period of single-blind placebo run-out (D112 to D140). Each patient noted in a diary the frequency of migraine attacks and their intensity, assessed on a four-point scale: O=pain absent, 1=mild pain, 2=moderate pain, 3=severe pain.
Patients were defined as responders when the number of attacks at the end of the comparative period (month 4) decreased by 50% or more compared with the number of attacks during the run-in period.
The duration of the attacks, the acute treatment used and the degree of functional impairment were also assessed from 0 (able to work) to 3 (requirement of bed rest). The number of attacks was calculated for each 28-day period from the patient diary cards.
The primary efficacy criterion was the mean number of migraine attacks during the last period of treatment (the last 4 weeks of treatment D85–D112).
The secondary efficacy parameters were the duration, the intensity of the attacks, the use of acute treatment and the number of responders.
Compliance was assessed by calculating the ratio of real : theoretical capsule consumption according to the returned capsule count. Statistical analysis was performed for efficacy on the intention to treat and per protocol population, and for tolerance on the intention to treat for the whole population, using the Student's t-test for continuous variables, the Wilcoxon test for ordinal variables and the χ2 or Fisher's test for non-ordinal variables. Baseline between-group comparability with respect to demographic variables and migraine parameters was also assessed. All the statistical tests have been interpreted at the 5% significance level.
Results
At the end of the placebo run-in period, 22 patients did not fulfil the inclusion criteria (seven for non-compliance, four lost to follow-up, 11 having less than two or more than six migraine crises during this period). Thirty-two recruited patients presented recorded observations of doubtful authenticity. Following an audit by the clinical Quality Assurance Department, these patients were excluded from the intent-to-treat analysis. The intention to treat population included 196 patients (100 patients were treated by OPFA and 96 by placebo). The characteristics of the two groups at inclusion are shown in Table 1. After consideration of all protocol deviations, the per protocol population consisted of 167 patients, 85 patients taking OPFA and 82 taking placebo.
Characteristics of the two patient groups at inclusion (D0)
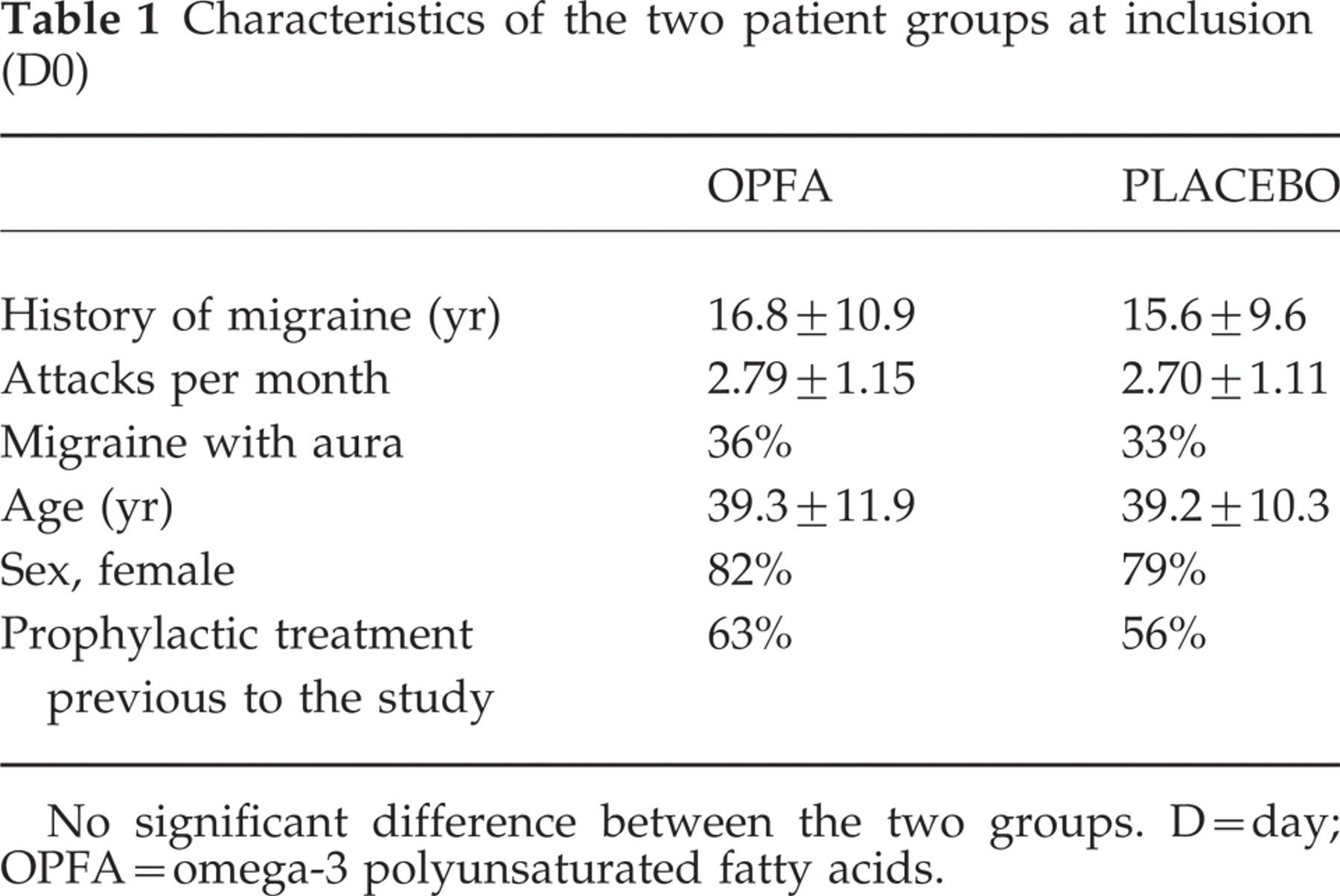
No significant difference between the two groups. D = day; OPFA = omega-3 polyunsaturated fatty acids.
The population of completers (intent-to-treat population), having received all four treatment periods, was composed of 183 patients, 96 taking OPFA and 87 taking placebo.
During the 16-week comparative treatment period, 13 patients withdrew from the study: six for lack of efficacy, six due to the patient's decision, and one was lost to follow-up.
The compliance analysis performed on the completers population shows average compliance of 94.5% and 93.7% in the OPFA and placebo groups, respectively (a non-significant difference, P = 0.87).
Primary efficacy criterion
The mean number of migraine attacks (intent-to-treat study) is shown in Table 2. At the end of the inclusion period, the mean number of migraine attacks in the OPFA group was 2.79 ± 1.15 vs. 2.70 ± 1.11 for the placebo group. The analysis of the 4-week periods from D0 to D112 does not show a significant difference between the OPFA and the placebo groups regarding the number of migraine attacks. During the last 4-week period (D85 to D112) the main efficacy criterion, i.e. the mean number of migraine attacks, was 1.20 ± 1.40 in the OPFA group and 1.26 ± 1.11 in the placebo group (non-significant, NS).
Mean number of migraine attacks (intent to treat analysis)

OPFA = omega-3 polyunsaturated fatty acids.
However, if one compares the mean number of attacks during the totality of the comparative treatment period (D0 to D112) the number of migraine attacks was 5.95 ± 4.01 for the OPFA group compared with 7.05 ± 4.12 for the placebo group (P = 0.03). In addition, the per-protocol analysis of the mean frequency of migraine attacks from D0 to D112 shows a significant difference in favour of OPFA (OPFA group 5.99 ± 3.90 attacks, placebo group 7.19 ± 4.19) (P = 0.03).
Even though this difference is significant, it is doubtful that this would be translated into significant clinical efficacy as this would represent six attacks on average in 16 weeks with OPFA, seven attacks with placebo.
Secondary criteria analysis
During the double-blind comparative period (D0–D112) the mean intensity and the mean duration of the attacks were not significantly different between the two groups (intensity: 2.21 ± 0.67 and 2.28 ± 0.64 for the OPFA and placebo groups, respectively; duration: 15.5 ± 13.2 h and 18.2 ± 16.9 h for the OPFA and placebo groups, respectively). In contrast a significant difference was found between the two groups in favour of OPFA for the number of responders (OPFA group 74 responders, 26 non-responders; placebo group 53 responders, 43 non-responders – P = 0.006).
To evaluate the possible remanent effect of the drugs, the patients were followed-up in single-blind conditions (taking only the placebo) from D112 to D140. The cumulative number of attacks during this period was 71 in the OPFA group, 95 in the placebo group (non-significant difference). The mean number of attacks during this period was 1.11 ± 1.43 in the OPFA group, 1.51 ± 1.22 in the placebo group (NS).
Despite a run-in placebo period of 1 month, a very strong placebo effect was observed in this trial: 45% reduction of the attacks between run-in and 4-month treatment period (55% in the OPFA group, P = 0.058).
Tolerance
This analysis was conducted on an intent-to-treat population of 196 patients, 100 having received OPFA and 96 placebo. 148 adverse events were recorded. The main reported adverse events (AEs) concerned:
The nervous system, central and peripheral: AEs noted in 5% of cases with OPFA and 8% with placebo; principally non-migrainous headaches (NS).
The digestive tract (32% effects of this type with OPFA against 18.8% with placebo; main AEs concern nausea, 5.1%, and eructations, 4.6%). A significant difference against OPFA is noted for the eructations (8% in the OPFA group, 1% in the placebo group).
Three serious adverse events, without relation to the study treatment, were declared. All patients concerned were taking placebo.
Five patients withdrew from the study due to non-serious adverse events (nausea, diarrhoea). All five patients were taking placebo.
Discussion
The first attempt to introduce omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) in therapeutic diets was derived from the Dyerberg and Bang epidemiological study (12), describing a low incidence of coronary artery disease in Greenland Eskimos despite their high intake of fat. The dietary fat of this population came from sea food and contained large quantities of EPA and DHA. For example, a 100-g diet of fatty fish (salmon, herring, eel, mackerel) contains 1–2 g of EPA and DHA (13).
Dietary ω-3 fatty acids from fish and fish oil have hypolipaemic effects in normal subjects and in hypertriglyceridaemic patients with combined types IV and V hyperlipaemia, decreasing plasma triglycerides and to a lesser extent plasma total cholesterol (9). This results from an important reduction of triglycerides and VLDL synthesis in the liver by ω-3 fatty acids (7, 14, 15).
After their intake, omega-3 fatty acids are incorporated into the membranes and modulate their reactivity: in vitro they inhibit the platelet aggregation, and modify some haemostasis parameters such as the production and structure of thromboxanes, and the concentration and activity of different coagulation factors, leading for example to thromboxane A3 and PG I3 at the platelet level. These changes reduce platelet aggregation and favour a vasodilating effect (16, 17).
Omega-3 fatty acids are very important components of cerebral phospholipids and have an important impact on the development of the central nervous system and of the retina. This was demonstrated recently in premature infants (8, 9). In the rat an experimental deficiency in omega-3 fatty acids is responsible for Na+−K+−ATPase pumps inducing a cerebral dysfunction.
A variety of non-pharmacological and pharmacological (dihydroergotamine, serotonin antagonists, tricyclic antidepressants, calcium channel blockers, alpha-agonists, sodium valproate, monoamine oxidase inhibitors, NSAIDs) treatments are available for migraine prophylaxy and have been tested in double-blind studies vs. placebo (1, 2). However, the results are not entirely satisfactory and side-effects such as weight gain (pizotifen, flunarizine, amitryptiline, for example), sedation (oxetorone, amitryptiline) or discomfort (betablockers) are a major problem with prophylactic drug treatment of migraine and new drugs are needed.
A parallel design was chosen for this trial and the corresponding number of patients needed to treat was calculated. Dose–response curves for the efficacy of OPFA have not been studied but the dose of 6 g a day was chosen according to previous studies with this product. As proposed by Tfelt-Hansen (18), we used migraine attack frequency as the main parameter and we excluded other parameters such as changes in headache indices. Attacks were reported by the patients in their diary and at each visit the study coordinator assured that patient data were well documented.
During this 6-month study we looked for a time-effect, which is the decrease of attack frequency after entering the trial, regardless of what treatment is given, and we observed a very strong placebo effect. This strong placebo effect could be related to the selection of our migraineurs in a general practice, as attested by a low mean number of attacks in contrast to the majority of publications originating from headache clinics, or to a close relationship between the GP and the patient.
Our study fails to show, on the main criterion (the number of attacks in the last 4 weeks of the comparative treatment period), a significant difference compared with placebo and we did not find the positive results demonstrated on a small number of patients by the two previous studies (5, 6).
