Abstract
Background
Er-Miao-San (EMS) is a classic prescription in Traditional Chinese Medicine (TCM) for the treatment of colorectal cancer (CRC) and has shown promising therapeutic effects in clinical practice. However, the specific components and molecular mechanisms of EMS remain unclear.
Purpose
The aim of this study was to analyze the effective components and molecular mechanisms of EMS in treating CRC through network pharmacology techniques and experimental validation.
Materials and Methods
The Traditional Chinese Medicine Systems Pharmacology database was used to screen the main active chemical components and targets of the EMS formula. The compound structures were verified using the PubChem database, which is an organic small-molecule bioactivity database. GeneCards and OMIM databases were utilized to predict target genes related to CRC. The Cytoscape 3.8 software was used to construct a “Drug-Active Ingredient-Target-Disease” intersection network. The STRING database was employed to analyze the core target protein–protein interaction network shared by EMS and CRC. The core targets were further subjected to Gene Ontology enrichment analysis and Kyoto Encyclopedia of Genes and Genomes pathway enrichment analysis using the R language. Molecular docking between the core targets and the major active chemical components of EMS was performed using AutoDock software. The impact of the core targets on the prognosis of CRC patients was analyzed using the R language. Subsequently, we validated the potential mechanisms predicted by network pharmacology for the inhibition of CRC cell proliferation by the key proteins in the relevant pathways through CCK8 cell proliferation assays and Western blot experiments.
Results
Molecular docking results showed good docking affinity between the key active components, such as quercetin and baicalein, of EMS and the core targets. Kaplan–Meier survival analysis demonstrated a close correlation between the core targets and the survival prognosis of CRC patients. Cellular experiments showed that EMS significantly inhibited the proliferation of CRC cells and may promote apoptosis and autophagy of CRC cells by suppressing the expression of key proteins in the PI3K/AKT pathway.
Conclusion
The TCM formula EMS exerts anti-CRC effects through multiple pathways and targets, improving the prognosis and extending the survival period of CRC patients. This study provides preliminary insights into the effective components and molecular mechanisms of EMS in the treatment of CRC, which were preliminarily validated through molecular docking and experimental approaches.
Keywords
Introduction
Colorectal cancer (CRC) is one of the most common malignant tumors in the gastrointestinal tract. According to the latest global cancer burden data released by the International Agency for Research on Cancer (IARC) of the World Health Organization in 2020, there were approximately 1.93 million new cases of CRC and 940,000 deaths globally in 2020, making it the third most common cancer and the second leading cause of cancer death, accounting for 10% and 9.4% of the total number of cancer cases and deaths, respectively. The data released by IARC show that there were approximately 550,000 new cases of CRC and nearly 300,000 deaths in China in 2020. 1 It is a major public health problem that poses a threat to human health.
The main treatments for CRC are surgery, postoperative chemotherapy, targeted therapy, and immunotherapy, but the serious side effects of drugs and drug resistance severely limit the clinical treatment of CRC. 2 In Asian countries, traditional Chinese medicine (TCM) has been widely accepted as an alternative therapy for malignant tumors. Clinical and pharmacological studies have shown that many natural products of Chinese herbal medicine have biological functions, such as multitarget inhibition of proliferation, promotion of cell apoptosis, inhibition of tumor invasion and metastasis, antiangiogenesis, and regulation of autophagy, reversing chemotherapy resistance in vivo and playing an antitumor role.3, 4 Er-Miao-San (EMS) is a classic prescription used in TCM for the treatment of gastrointestinal tumors. It is composed of two medicines, Atractylodes Rhizoma and Phellodendri chinensis Cortex, and was first recorded in the book “Shi Yi De Xiao Fang,” which was used by ancient Chinese doctors to treat arthritis. In clinical practice, we found that EMS can effectively prolong the survival time of patients with malignant tumors and improve their quality of life. Pharmacological studies of the herbs in the EMS formula also suggest that it has pharmacological effects such as heat-clearing and detoxifying, promoting bile flow, stimulating the gastrointestinal tract, and antitumor activities. 5
As a traditional treatment method in China, Chinese herbal medicine plays a unique role in the adjuvant treatment of CRC. 6 However, although EMS has certain therapeutic effects in the treatment of CRC, its specific mechanism of action has not been clarified. Thanks to the rapid development of modern Chinese medicine databases and computer network topology analysis and bioinformatics, this study used network pharmacology to investigate the potential interactions between the herbs, active compounds, and potential targets in EMS, explore the binding modes and affinity between active compounds and potential targets through molecular docking technology, and to obtain a clearer understanding of the mechanism of action. We also performed experimental verification of the potential mechanism of inhibiting colon cancer of critical proteins in relevant pathways predicted by network pharmacology to provide reference and research ideas for its clinical application and further mechanism research.
Materials and Methods
Screening of Active Ingredients and Potential Targets of EMS
The identification of effective active ingredients within EMS was conducted utilizing the Traditional Chinese Medicine Systems Pharmacology Database (TCMSP) (
Acquisition of CRC Target Genes
CRC-related targets were retrieved and screened through the GeneCards (
Construction and Analysis of the “Active Ingredient-Target-Disease”
Relationship Network Overlapping targets between active ingredients of EMS and CRC were identified using the Venny online platform (
Construction of Protein–Protein Interaction (PPI) Networks and Screening of Core-acting Proteins
To elucidate potential targets of EMS in CRC, a PPI analysis was executed using the STRING database. Cytoscape 3.8.0 software, in conjunction with the cytoHubba plugin, was utilized for network analysis based on a topology network algorithm, aiding in the identification of core functional targets.
GO (Gene Ontology) Analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) Pathway Enrichment Analysis
The GO database (
Molecular Docking
Download the molecular structure of the drug from the PubChem database (
Survival Analysis of Core Target Genes
Survival analysis of core target genes employed the survminer and survival R packages on the TCGA-COAD dataset. Colon cancer patients were stratified into high-expression and low-expression subgroups based on the optimal cutoff point. Overall survival (OS) indicators, risk ratio (HR) with 95% confidence intervals, and log-rank p values were estimated, with statistical significance set at p < .05.
Experimental Validation
Cell Line
The human colon cancer cell lines HCT116 and LoVo were obtained from the American Type Culture Collection (ATCC, Manassas, USA). HCT116 and LoVo cells were grown in McCoy’s 5a modified medium with 10% fetal bovine serum (FBS), which was prepared by ATCC (#30-2007). The cells were cultured at 37°C in a 5% CO2 environment.
Preparation of EMS Extract
EMS recipe was obtained from Chinese Medicine Pharmacy of the Shanghai Traditional Chinese Medicine Hospital. EMS was composed of 15 g of P. chinensis Cortex Chinensis and 15 g of Atractylodes lancea. Place EMS (15 g of P. chinensis Cortex Chinensis and 15 g of A. lancea) in a round-bottom flask according to the proportions. Add 10 times the volume of 95% ethanol and reflux extract for 1.5 hours using an electric heating mantle. Filter out the residue, reduce the pressure, and concentrate the filtrate to dryness, weigh it, and calculate the extraction yield. Dry the residual herbal materials in a fume hood to remove the ethanol solvent. Extract the dried residues with 10 times the volume of distilled water in reflux for 2 rounds, each lasting 1.5 hours, starting the timing from the boiling point. Combine the extracts, reduce the pressure, and concentrate to a density of 1.05 g/mL (60°C). Tilt and pour out the concentrated extract, let it cool to room temperature (RT) and extract twice with an equal volume of ethyl acetate. Reduce the pressure and concentrate the ethyl acetate layer and water layer separately to dryness, then weigh and calculate the yield. Perform quality control by using high-performance liquid chromatography to determine the content of major components such as astragaloside IV in the EMS formula. Prior to the experiment, weigh an appropriate amount of the extracted powder, and dissolve it thoroughly in RPMI 1640 medium using methods such as vortex, ultrasound, and water bath. Filter the solution using a 0.22 µm filter and prepare it to the desired concentration. Store it at 4°C for later use.
CCK-8 Assay
The HCT116 and LoVo cells in the logarithmic growth phase were seeded at a concentration of 8,000 cells per well in a 96-well plate. After the cells adhered, the serum-free culture medium was replaced, and the cells were starved overnight. Different concentrations of EMS extract, specifically 25 µg/mL, 50 µg/mL, 100 µg/mL, 150 µg/mL, 200 µg/mL, and 250 µg/mL were prepared to establish the control groups. After 48 hours, a 10% CCK-8 solution was added, and the cells were cultured at 37°C for 1 hour. The absorbance of the HCT116 cells was measured at 450 nm using an enzyme-linked immunosorbent assay analyzer to calculate cell viability.
Western Blot Analysis
Western blot analysis was performed using primary antibodies from Abcam, including pan-AKT (ab8805), pan-AKT (phospho T308) (ab8933), interleukin (IL)-6 (ab233706), tumor necrosis factor-alpha (TNF-α) (ab183218), vascular endothelial growth factor A (VEGFA) (ab46154), and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (ab59164). All primary antibodies were diluted 1:1,000, and the secondary antibody (ab6721, Abcam) was diluted 1:10,000. RIPA lysis buffer (Beyotime) containing 1% protein inhibitor and 1% phosphatase inhibitor (Beyotime) was used at a ratio of 100 µL RIPA per 1 × 10 6 cells or 200 µL RIPA per 20 mg tissue. The protein concentration was quantified using the Enhanced BCA Protein Assay Kit (Beyotime) according to the manufacturer’s instructions. After electrophoresis separation on 10% SDS-PAGE (Solarbio), equal amounts of protein (25 µg) were transferred onto the PVDF membrane (Millipore, MA, USA) by blotting. After blocking with 5% skim milk at RT for 1.5 hours, the membrane was incubated with primary antibody at 4℃ overnight. Afterward, the membrane was washed with 1 × TBST (Solarbio) three times and incubated with the secondary antibody at RT for 2 hours, followed by washing three times with 1 × TBST. All proteins were visualized using Immobilon Western Chemiluminescent HRP Substrate (Millipore). In brief, the reaction mixture was added uniformly onto the membrane (protein surface), and the membrane (protein surface) was then gently put into a colorimeter. The film was scanned or photographed, and the band intensity was evaluated using Image Lab 3.0 software (Bio-Rad Laboratories).
Results
Screening of Active Ingredients in EMS and Prediction of Potential Targets
After screening and deduplication based on the OB and DL values in the TCMSP database, a total of 46 potentially active ingredients in EMS were obtained. Among them, 37 active ingredients were from P. chinensis Cortex and 9 active ingredients were from Atractylodes Rhizoma (Table 1). The compound structures of these active ingredients were verified using the PubChem database. By further screening in the TCMSP database, 455 potential target proteins were identified for the active ingredients of P. chinensis Cortex, and 67 potential target proteins were identified for the active ingredients of Atractylodes Rhizoma, resulting in a total of 522 target proteins. After deduplication, 197 potential target proteins for EMS were obtained. This indicates a high similarity in the target proteins between the two medicinal ingredients of EMS, suggesting a synergistic effect. Subsequently, the relationships between EMS, active ingredients, and target proteins were imported into Cytoscape 3.8.0 software for data visualization. In the visualization, the active ingredients are represented by yellow nodes, the potential predicted target proteins are represented by green nodes, and the connections between active ingredients and target proteins indicate their interaction (Figure 1).
The Main Effective Components and Relevant Parameters of Er-Miao-San (EMS) in Treating Colorectal Cancer (CRC).
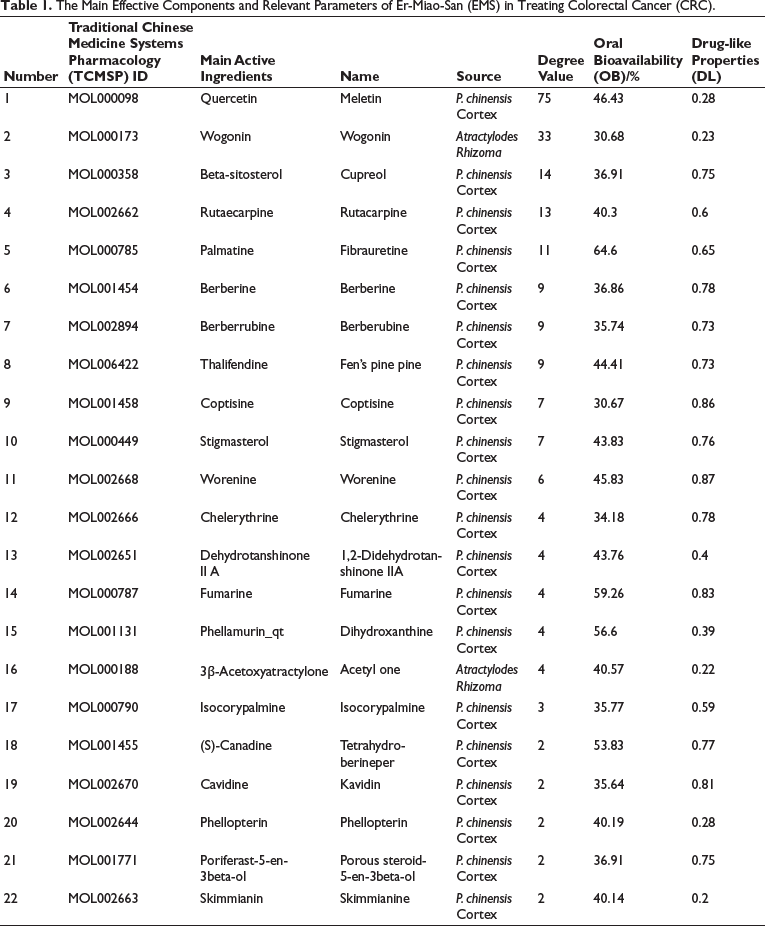
Screening for CRC Disease-related Targets and Targets Involved in the Treatment of CRC by EMS
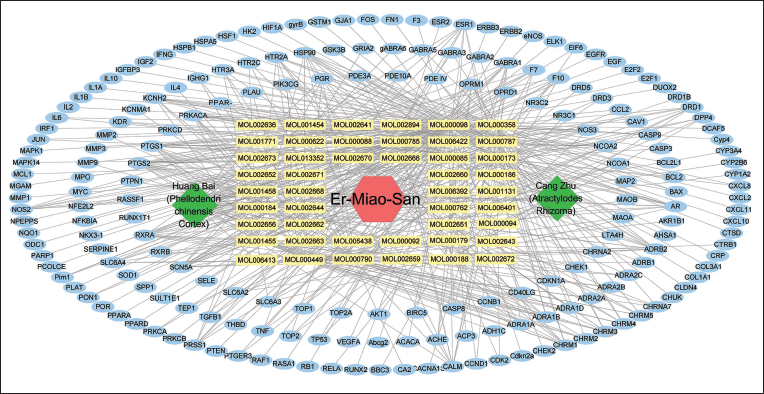
In the quest for disease-related targets associated with CRC, a comprehensive search using the keyword “Colorectal cancer” in the GeneCards disease database yielded a substantial 10,746 CRC-related targets. Refinement with a stringent criterion of “Relevance Score > 10” resulted in the identification of 1,490 CRC disease-related targets. Simultaneously, the DisGeNET database contributed an additional 166 CRC-related targets, while the MalaCards database yielded 1,109 CRC-related targets, with 1,074 targets meeting the criteria of “Score > 25.” After the meticulous elimination of duplicates, a consolidated list of 1,748 CRC disease-related targets was achieved (Figure 2A).
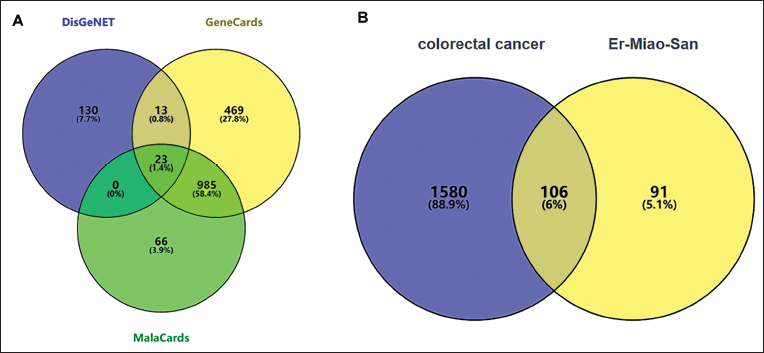
After the intersection with the potential targets of the active components, 106 possible targets for CRC were obtained (Figure 2B).
Screening for Key Effective Ingredients of EMS in the Treatment of CRC
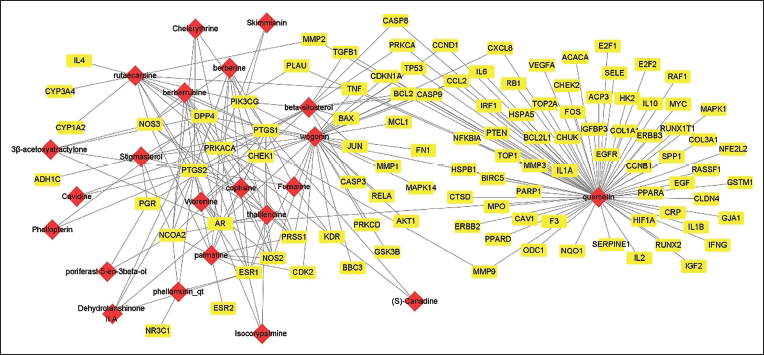
A comprehensive “Active Ingredient-Target-Disease” relationship network diagram was meticulously constructed utilizing Cytoscape 3.8.0 software. This network effectively illustrates the 106 potential target proteins associated with EMS in the treatment of CRC, along with their corresponding 22 active ingredients (Figure 3). In this graphical representation, red nodes signify active ingredients, yellow nodes denote CRC treatment target proteins, and the edges symbolize the interactions between active ingredients and CRC treatment target proteins. Employing the Network Analyzer tool within Cytoscape 3.8.0 software, topological analysis was conducted on the network. The outcome of this analysis highlighted quercetin, wogonin, beta-sitosterol, rutaecarpine, and palmatine as key pharmacological molecules, potentially holding central regulatory roles in the treatment of CRC with EMS (Table 1).
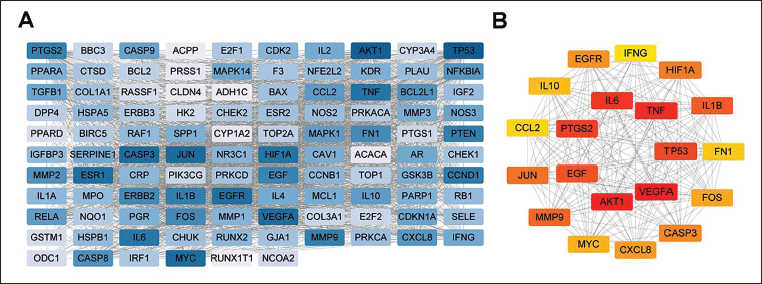
PPI Network Construction and Core Protein Screening
We submitted the 106 potential CRC treatment targets to STRING, a PPI database, for protein interaction analysis. Utilizing the cytoHubba plugin with the MCC algorithm in Cytoscape 3.8.0 software, we conducted a topological network analysis, identifying and filtering core proteins (see Figure 4A). The color intensity in the visualization reflects the assigned values to each target. The darker coloration signifies a higher assigned value, indicating the likely significance of these proteins in the regulatory network. Through the MCC algorithm, several protein nodes, including VEGFA, AKT1, TNF, IL6, and PTGS2, exhibited higher parameters, suggesting their potential core role in the treatment of CRC with EMS (Figure 4B).
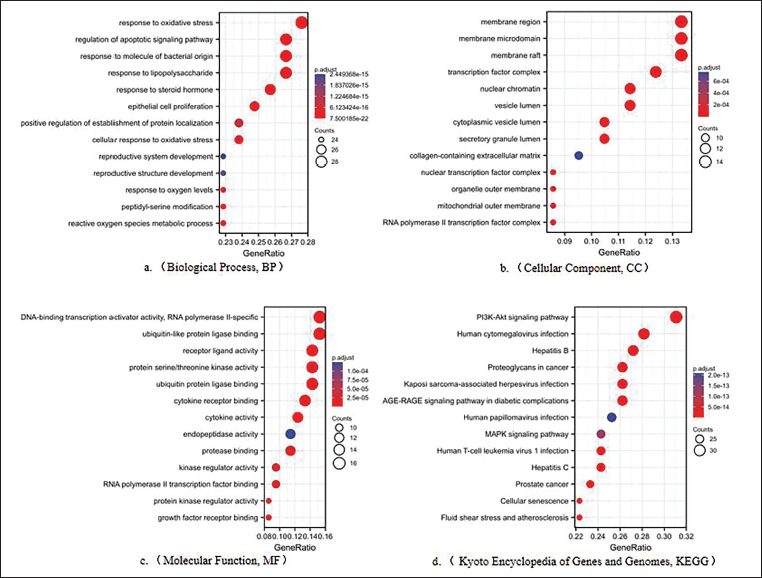
Biological Function Enrichment Analysis
To delve deeper into the biological functions of EMS in treating CRC, we conducted a comprehensive biological function enrichment analysis of the 106 target proteins. Employing the ClusterProfiler and ggplot2 packages in R 3.5.0 software, we set a filtering criterion of p < 0.05 to analyze and visualize potential biological functions (Figure 5). The GO enrichment analysis, focusing on BP, illuminated that EMS may exert its anti-CRC effects through various BP. These include oxidative stress, lipopolysaccharide response, bacterial response, apoptosis, steroid hormone response, and epithelial cell proliferation. In the CC analysis, target proteins implicated in the anti-CRC effects of EMS were predominantly enriched in cellular membrane components (membrane rafts, membrane microdomain, membrane region), transcription factor complexes, RNA polymerase II transcription factor complexes, luminal structures, mitochondrial outer membranes, and nuclear chromatin. The MF analysis suggested that EMS is also involved in molecular processes such as ubiquitin-protein ligase activity, DNA-binding transcription activator, RNA polymerase II, serine/threonine kinase activity, cytokine (ligand/receptor) activity, and peptidase activity. Furthermore, KEGG pathway enrichment analysis unveiled associations between EMS therapeutic effects and several signaling pathways, including cancer, immune system, endocrine metabolism, and apoptosis. Notable pathways encompassed the PI3K-AKT signaling pathway, protein-polysaccharide in cancer, mitogen-activated protein kinase (MAPK) signaling pathway, cell senescence, IL17 signaling pathway, endocrine resistance, and apoptosis pathways.
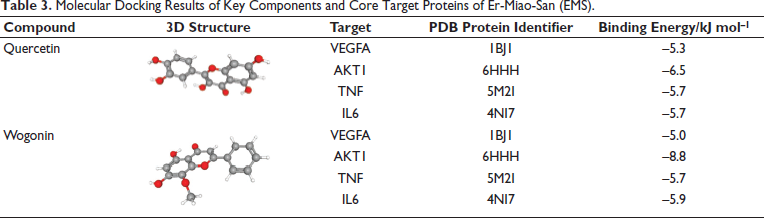
Docking Analysis of Key Components in EMS and Core Target Proteins
To establish the connection between the key components of EMS and the core targets, we obtained the protein crystal structures corresponding to the targets from the PDB database. Additionally, we consulted relevant literature to understand the protein active sites for VEGFA, 12 AKT1, 13 TNF, 14 and IL6. 15 Based on this information, we determined the protein pockets (Table 2). We conducted computer-simulated molecular docking verification between the key components, quercetin, and wogonin of two Chinese medicines, P. chinensis Cortex and Atractylodes Rhizoma, and the top four core targets VEGFA, AKT1, TNF, and IL6 in topological network analysis. 12 The results of the docking simulations demonstrated that both quercetin and wogonin exhibited commendable binding abilities with VEGFA, AKT1, TNF, and IL6 (Table 3 and Figure 6). Affinity values are indicative of the binding performance between the ligand and receptor proteins. A lower affinity value suggests a more favorable interaction between the molecule and protein. An affinity value less than 0 indicates spontaneous binding of the ligand molecule to the receptor protein, while a value smaller than –5.0 kJ mol⁻¹ signifies excellent binding between the molecule and the protein.
Target Gene Protein Pocket Coordinates and Grid Box Sizes.

Molecular Docking Results of Key Components and Core Target Proteins of Er-Miao-San (EMS).
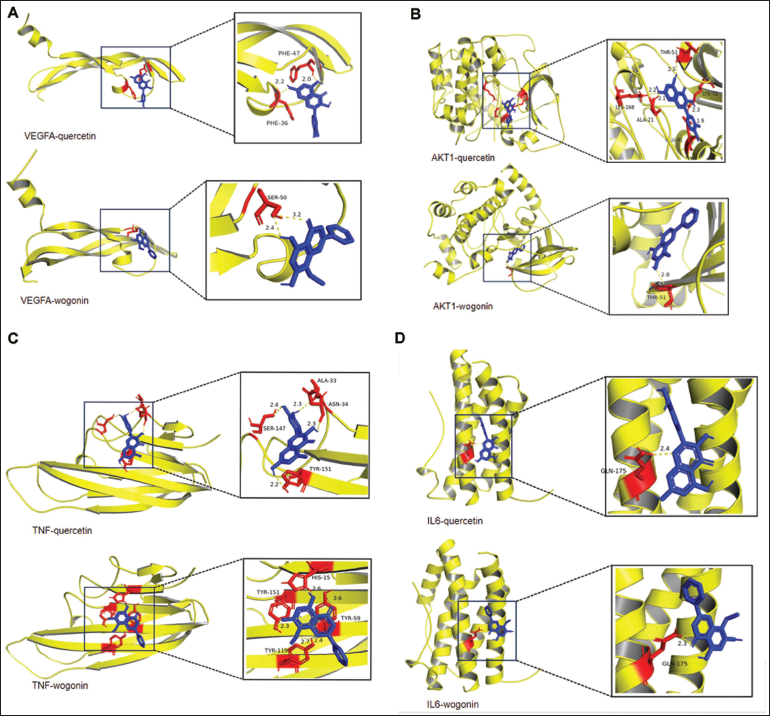
In Figure 6, the blue part represents the drug molecule, the yellow part represents the target protein structure, the left figure represents the overall binding pattern between the two, and the right represents the enlarged picture of molecule and protein binding.
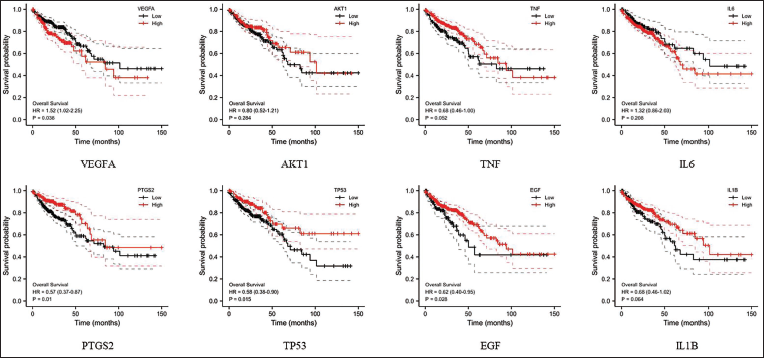
Survival Analysis of Core Target Genes
To further analyze the association of EMS anti-CRC core targets with CRC disease prognosis. Utilizing R 3.5.0 software along with the survminer and survival R packages, we examined the molecular expression patterns in CRC clinical patients sourced from the TCGA-COAD dataset. Employing the optimal cutoff point, CRC patients were stratified into high and low expression subgroups for the core target genes, with a significance threshold of p < 0.05. Subsequently, the survival prognostic significance of these core target genes in CRC was analyzed based on the OS indicator. The results unveiled that among the top eight core target genes VEGFA, TNF, PTGS2, TP53, EGF, and other targets exhibited noteworthy prognostic significance in CRC (Figure 7), indicating that EMS may exert a therapeutic role in prolonging the survival of CRC patients by targeting relevant targets.
Validation of the Colorectal Cancer Effect of EMS in vitro
To further investigate the inhibitory effect of EMS on CRC cell proliferation, we performed in vitro experiments. HCT116 and LoVo cells were treated with increasing concentrations of EMS for 24 hours, and cell viability was measured. As shown in Figure 8, EMS significantly inhibited the growth of HCT116 (Figure 8A) and LoVo cells (Figure 8B), with IC50 values of 122.47 ± 3.432 µg/mL and 98.432 ± 5.891 µg/mL, respectively, indicating a good inhibitory effect of EMS on the proliferation of CRC cells.
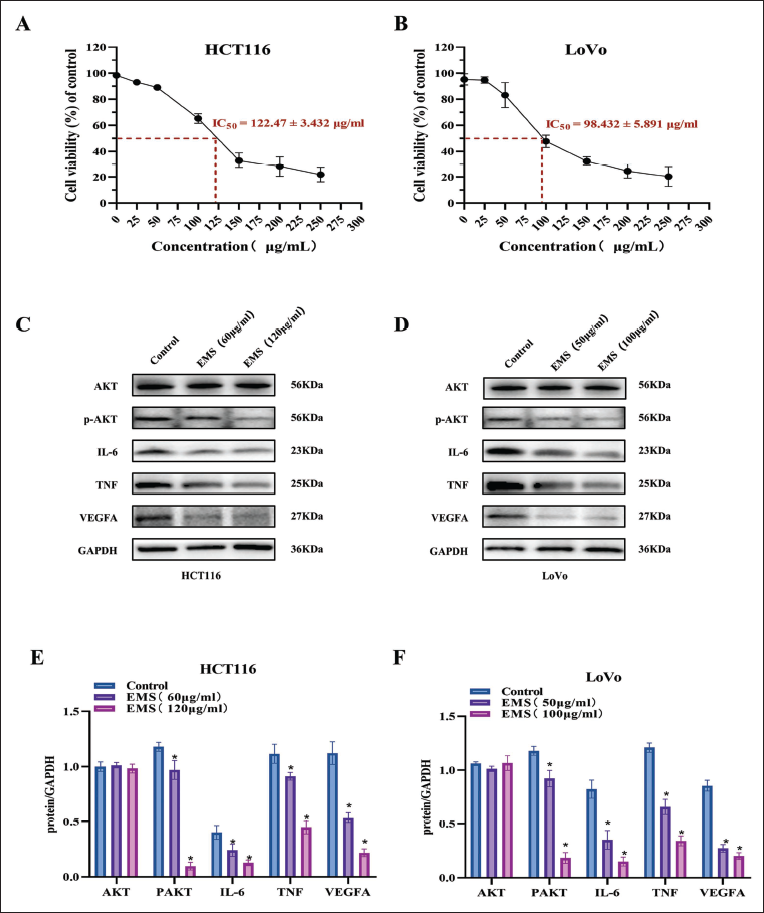
The network pharmacology and molecular docking results suggested that the PI3K/AKT signaling pathway might play an important regulatory role in the process of EMS inhibiting CRC cell proliferation, we further conducted immunoblot analysis. The results showed that after EMS intervention, the phosphorylation levels of the key molecules of the PI3K/AKT signaling pathway were significantly inhibited in HCT116 and LoVo cells, along with downregulation of core molecules such as VEGFA, TNF, and IL6 (Figure 8C-F). These findings indicate that EMS promotes apoptosis and autophagy levels in HCT116 and LoVo cells through the PI3K/AKT signaling pathway.
Discussion
CRC is one of the malignant tumors that seriously damage human life. In recent decades, its incidence and mortality have been on the rise. TCM, as a unique treatment method in China, has shown great advantages in prolonging the survival time and improving the quality of life of patients with CRC. Modern pharmacological studies have found that two Chinese herbal medicines, P. chinensis Cortex and Atractylodes Rhizoma, in EMS have pharmacological effects such as clearing heat and detoxification, promoting bile flow, stimulating the gastrointestinal tract, and antitumor, but their specific mechanisms of action are still unclear.
This study conducted computational analysis through pharmacology, disease databases based on network pharmacology, bioinformatics, and topology algorithms to discover that 22 key pharmacological molecules in EMS may exert anti-CRC therapeutic effects through 106 targets. The BP involved include oxidative stress, cell apoptosis, cell proliferation, and tumor metabolism, and the treatment effects are exerted through pathways such as regulating the PI3K-AKT signaling pathway, the cancer protein-polysaccharide signaling pathway, and the MAPK signaling pathway. Among the 22 key pharmacological molecules, quercetin, wogonin, beta-sitosterol, rutaecarpine, and palmatine may be the key pharmacological molecules in the treatment of CRC with EMS. Specifically, quercetin has been shown to have multiple anti-tumor effects, including inhibiting tumor cell proliferation, reversing tumor cell multidrug resistance, and enhancing antitumor effects when combined with other agents. 16 Wogonin is a natural flavonoid compound that can promote tumor cell apoptosis, block the cell cycle of tumor cells, inhibit tumor angiogenesis, increase the infiltration of immune cells, and enhance immune function.17, 18 Beta-sitosterol is a steroid-like plant oil with various pharmacological activities. It can inhibit tumor occurrence and development, induce tumor cell differentiation, stimulate tumor cell apoptosis, play an antioxidant role, and inhibit tumor cell proliferation by activating the sphingomyelin cycle. 19 Rutaecarpine is a naturally occurring indole alkaloid that can inhibit the proliferation of a variety of tumor cells, including cervical cancer, colon cancer, lung cancer, melanoma, T lymphocytic leukemia, prostate cancer, and breast cancer. 20 Palmatine is a bioactive alkaloid that can enhance immune function by regulating cytokines and immune cells. 21
Based on our predictions of active ingredient targets and “active ingredient-potential target-disease” relationship network construction as well as PPI analysis, VEGFA, AKT1, TNF, IL6, PTGS2, TP53, EGF, IL1B, and other core targets may play critical regulatory roles in the anti-CRC effects of EMS. Tumor proliferation and metastasis rely on the blood vessels generated to provide nutrients. Therefore, blocking tumor angiogenesis is currently a hot topic in tumor therapy. VEGFA, as a vascular endothelial growth factor, increases tumor vascular permeability, promotes the proliferation of endothelial cells, and induces tumor angiogenesis when highly expressed. 22 Clinical studies have confirmed that anti-VEGFA drugs have good therapeutic effects in CRC treatment. EGF, as a growth factor, can promote the growth and proliferation of epithelial cells and is an important messenger for signal communication between tumor cells and matrix cells. TNF, IL6, and IL1B, as inflammatory cytokines, play different roles in inflammation, cell differentiation, lipid metabolism, and cell apoptosis. PTGS2 has been found to be highly expressed in many benign cancer precursors and malignant tumors can promote tumor angiogenesis, enhance tumor cell invasion and metastasis, and inhibit cell apoptosis.23, 24 AKT1 is a key molecule in the PI3K-PTEN-AKT-mTOR signaling pathway and plays an important regulatory role in tumor cell growth, apoptosis, invasion, and metabolism. 25 TP53 is a tumor suppressor gene. Mutations of TP53 are widely present in many types of cancer cells. When this gene is mutated, p53 becomes inactive, leading to the formation of tumors. 26
Through GO function enrichment and KEGG pathway enrichment analysis, we found that EMS may exert various biological functions through signaling pathways such as PI3K-Akt signaling pathway, cancer protein-polysaccharide signaling pathway, MAPK signaling pathway, cell senescence pathway, IL17 signaling pathway, endocrine resistance pathway, and apoptosis pathway in cancer treatment, immune regulation, endocrine metabolism, tumor cell apoptosis, etc. Combined with our molecular docking verification, the key active ingredients in EMS, quercetin, and wogonin, have good binding ability against the core targets screened from the PPI network. Moreover, most of the targets in the anti-CRC core targets of EMS are also meaningful in CRC prognosis, indicating that EMS may exert a therapeutic effect on CRC patients’ survival by targeting relevant targets. Especially, the in vitro experimental results show that EMS can significantly inhibit the phosphorylation of key molecules in the PI3K/AKT signaling pathway and downregulate core molecules such as VEGFA, TNF, and IL6.
In summary, based on our network pharmacology and molecular docking studies, the key bioactive ingredients of EMS, quercetin, wogonin, etc., through signaling pathways such as PI3K-Akt, cancer protein-polysaccharide, and MAPK, regulate core targets such as VEGFA, AKT1, TNF, and IL6, to exert therapeutic effects for cancer treatment, immune regulation, endocrine metabolism, and tumor cell apoptosis. This provides research direction and reference for future clarification of the specific effective ingredients and mechanism of action of EMS in the treatment of CRC.
Conclusion
EMS, a classical Chinese medicine formula, emerges as a promising therapeutic approach in the fight against CRC. Its effectiveness is evidenced by its ability to influence diverse pathways and targets, ultimately enhancing the prognosis, and extending the survival of individuals grappling with CRC. This study provides bioinformatic insights that could pave the way for a deeper understanding of the mechanisms of action of the EMS.
This comprehensive analysis not only affirms EMS efficacy but also lays the groundwork for further exploration into the intricate workings of this classical Chinese medicine. By elucidating the complex interplay between EMS and the biological pathways associated with CRC, this study not only validates its therapeutic potential but also offers a roadmap for future research endeavors.
Footnotes
Abbreviations
Authors’ contributions
Jianlin Ren and Jing Lin conceived and designed the study. Chenyue Yuan, Yong Zhao, and Yuting Yan performed the data analysis. Lin Zhou and Jinyan Chu wrote the paper. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Multiple research projects funded by National Natural Science Foundation of China (82174452, 81873279), Shanghai Municipal Science and Technology Commission (19401935200, 22S21901000), Shanghai Shenkang Hospital Clinical Three-Year Action Plan (HDC2020CR2047B), Shanghai Medical Innovation Development Foundation, and Shanghai University of Traditional Chinese Medicine (WLJH2021ZY-MZY026).
