Abstract
Cleistanthus collinus is a toxic plant belonging to the Euphorbiaceae family, and all parts of the plant are poisonous. It is used as suicidal, homicidal, cattle and fish poison and for inducing criminal abortion. The leaves and plant parts are consumed in various forms for homicidal and suicidal and it results in symptoms such as chest pain, dyspnea, central nervous system symptoms, and vision disturbances. There is no specific antidote for the C. collinus poisoning. The percentage of mortality with C. collinus poisoning varies from 20% to 50%, and the mortality depends on the amount and mode of consumption of the leaves. The major phytoconstituents of the plant include ellagic acid, diphyllin, collinusin, cleistanthin A, and cleistanthin B. The two major compounds cleistanthin A and cleistanthin B were evaluated for pharmacological activities such as antineoplastic activity, antiviral activity, diuretic activity, neuromuscular blocking property, alpha receptor blocking property, and antioxidant effect. The current review focuses on the toxicological and pharmacological properties of this plant and its phytoconstituents.
Keywords
Introduction
The genus Cleistanthus includes around 140 species, including Cleistanthus collinus (C. collinus), C. patulus, C. schlechteri, and C. gracili. 1 C. collinus (Roxburgh Bentham [Roxb. Benth.] and Hook f.) (of the Euphorbiaceae family) is a plant known for its toxicological importance. It is known as a toxic plant and is found in Africa, India, Sri Lanka, and Malaysia. 2 In India, this plant is commonly used as a cattle poison, fish poison, abortifacient, and suicidal poison. The plant is ingested in the form of decoction and paste. 3 All parts of the plant are reported to be toxic and used as suicidal, homicidal, cattle, and fish poison and for inducing criminal abortion. 1
From 1926 to 1985, more than 1,000 cases of C. collinus poisoning were reported in various parts of Tamil Nadu. 4 The leaves of C. collinus are a kind of slow poison because they cause death after 3–5 days of consumption. 5 The percentage of mortality with C. collinus poisoning varies from 20% to 50%, and mortality depends on the amount and mode of consumption of the leaves. The mortality rate was high when the person consumed the decoction of the leaves. 6 C. collinus poisoning is a problem among rural populations, especially young women, as a method of deliberate self-harm. According to research on acute poisoning in villages, 87.8% of women consumed plant poisons, 44.5% of whom consumed C. collinus. 7 Toxicity is mainly because of its diphyllin glycosides such as cleistanthin A and cleistanthin B. 8 C. collinus leaves contain saponin, tannin, and oduvin, and the poisonous effect attributed to oduvin usually occurs after drinking the decoction of the leaves of C. collinus, leading to death. 9 The current review focuses on the toxicological and pharmacological properties of this plant and plant phytoconstituents.
Plant Profile
The C. collinus plant has its wide distribution in Southeast Asia. In India, the plant is distributed in the dry, hilly areas of the central and southern parts, such as Tamil Nadu, Karnataka, Andhra Pradesh, Puducherry, Maharashtra, Bihar, and Odisha. It is commonly found in rural and suburban areas of Puducherry, Odisha, Andhra Pradesh, Madhya Pradesh, Bihar, Karnataka, Malabar, and Tamil Nadu. 6 In Tamil Nadu, it is abundantly found in the tropical forests of the Eastern Ghats. 10 It is a shrub that has dark brown barks, oval, leathery leaves, yellowish flowers, and fruits with black spherical seeds. 11 The plant belongs to the Euphorbiaceae family, and all parts of it are poisonous. The common name of C. collinus is summarized in Table 1. 7
Common Name of C. collinus.

Clinical Features of the C. collinus Poisoning
In patients, the clinical manifestations of C. collinus poisoning include hyperthermia, hypokalemia, electrocardiography (ECG) changes (prolongation of QT interval, ST depression, and flat P wave), and the elevation of hepatic enzymatic levels such as alkaline phosphatase (ALP), serum glutamic pyruvic transaminase, and serum glutamic-oxaloacetic transaminase.2, 5 The symptoms of C. collinus poisoning include nausea, vomiting, epigastric pain, diarrhea, breathlessness, headache, drowsiness, giddiness, blurring vision, diplopia, and black clouding. 12 The patients with poisoning admitted in JIPMER Hospital, Puducherry, between September 1998 and April 2000 showed laboratory abnormalities such as hypokalemia, hyponatremia, an elevated aspartate aminotransferase/lactate dehydrogenase/creatine phosphokinase/CPK-MB, nonspecific ST-T changes and QTc prolongation on ECG, metabolic acidosis and hypoxia. 13 Clinical features of the poisoning cases include increased body temperature, early tachycardia, late bradycardia, hypotension, arrhythmia, and death.6, 13 Poisoning treated with multidose charcoal was reported to yield a better outcome in patients.14, 15
Death caused by C. collinus poisoning usually occurs around 3–5 days after its consumption, depending upon the amount and mode of consumption. The mortality rate is very high in patients who consume leaf extract. Many studies reported various causes of death, among which cardiac arrest (46%) is reported in almost all studies. In addition to cardiac causes, others like uncontrolled hypotension (33%) and respiratory failure (53%) also account for the high mortality rate in C. collinus poisoning. The case fatality rate associated with C. collinus poisoning is 30%.5, 12, 16, 17
Symptoms and Signs
The major symptoms of the poisoning include dyspnea, tachycardia, altered sensorium, muscular weakness, visual disturbances, and seizures. The plant is mainly a cardiorespiratory poison, and the clinical investigations in the patients who consumed the poison revealed ECG abnormalities, high levels of liver enzymes, hypokalemia, and high anionic gap metabolic acidosis. Major ECG changes were depressed ST segment, T-wave inversion, flat P wave, and QT prolongation.2, 5, 13, 16–19 In pediatric cases, persistent hypocalcemia was reported, and the possible mechanism proposed was hypercalciuria associated with distal renal tubular acidosis. 20
Mechanism of Toxicity
In 1997, a possible mechanism for C. collinus poisoning was proposed. C. collinus causes the depletion of thiol-containing enzymes that are responsible for the manifestation of toxicity. It was suggested that thiol compounds might be the possible antidotes for C. collinus poisoning. 21 The neuromuscular weakness caused by the poisoning resembles myasthenia gravis, which is owing to the postsynaptic acetylcholine receptor blockade. 22 The mechanism proposed for the toxicity is deoxyribonucleic acid (DNA) synthesis inhibition, increased DNA damage apoptosis, and reduced thiol-containing enzymes. 23
The other possible mechanism proposed was the oxidative stress caused by free radicals. The effect of melatonin on the heart and brain of rats administered with C. collinus was evaluated by Jayanthi et al. 24 The antioxidant effect of melatonin was demonstrated in rats administered with C. collinus. The crude extract was effective in prolonging the survival time of the rats and reducing the malondialdehyde levels in rat brains. Melatonin also decreased the inflammatory changes in the brain of C. collinus intoxicated rats. 24
The distal renal tubular acidosis in C. collinus poisoning is caused by the defect in the adenosine triphosphate (ATP) requiring the proton pump that is essential for acid secretion in the kidney. The cultured kidney cells and rat renal brush border membrane (BBM) were exposed to the decoction of the C. collinus that inhibited the vacuolar H+-ATPase activity in the renal BBM and proton pump in renal BBM vesicles. 25
Treatment of the Poisoning
There is no specific antidote for the poisoning, but n-acetyl cysteine and thiol-containing compounds are proposed to be useful as they reactivate thiol-dependent enzymes such as lactate dehydrogenase and cholinesterase. 26 The medical management of the poisoning includes gastric lavage and administration of intravenous (i.v.) fluids, close monitoring of potassium levels and replacement, cardiac pacing, and other supportive measures.
Biological Effect of the Crude Extract
The dried and powdered C. collinus leaves were extracted with chloroform, and the effect of the chloroform extract of C. collinus was tested on Swiss albino mice phrenic nerve-diaphragm preparation. The extract significantly reduced the nerve and muscle action potential amplitude and twitch tension.9, 28
The intraperitoneal (i.p.) administration of a sub-lethal (175 mg/kg; i.p.) dose of the dry powdered extract of C. collinus leaves significantly inhibited the acetylcholine action on electromyography in rats. In C. collinus poisoning cases, muscle weakness and myasthenic crisis-like syndrome were observed. The study results suggested that the C. collinus leaves block the normal function of the neuromuscular junction.22, 29
Injection of sub-lethal dose (120 mg/kg; i.p.) of the aqueous extract of C. collinus leaves causes neuromuscular impairments like myasthenia gravis in the sciatic nerve and anterior tibialis muscle of albino Wistar rats 45 minutes after administration. The lethal dose (LD) of the aqueous extract of C. collinus leaves was found to be 190 mg/kg. 30
The aqueous extract of C. collinus causes transient tachycardia, increased contractility in frog-isolated hearts, and arrhythmia, hyponatremia, renal failure, arrhythmia, and cardiac arrest in rats. 31
The C. collinus poisoning in humans caused renal failure and neuromuscular weakness without the hypokalemic milieu. Earlier reports suggested that the poisoning caused distal renal tubular acidosis and hypokalemic paralysis. Hypokalemia might be the cause of rhabdomyolysis that could be the reason for respiratory paralysis.3, 16
The antidote for C. collinus poisoning is not available to date, but a previous report suggested that cases in the state of collapse with a lung machine and oxygen tent recovered gradually after the administration of neostigmine. 32 Melatonin (15 mg/kg) increased the brain and blood malondialdehyde levels and decreased the glutathione levels in rats administered with LD50 doses of the C. collinus leaf extract. Melatonin plays an important role in C. collins-induced tissue damage, balances oxidant and antioxidant status, and ameliorates oxidative organ injury in the brain. 24
The C. collinus poisoning cases showed clinical symptoms and signs such as vomiting, subjective weakness, dyspnea, headache, giddiness, tachycardia, abdominal tenderness, tachypnea, hypoxia, crepitations, hypotension, sinus tachycardia, sinus bradycardia, ST depression, and biochemical abnormalities such as metabolic acidosis and hypokalemia. 17
The leaf fractions of C. collinus inhibited the proton pump in the renal brush border, resulting in type I distal renal tubular acidosis in Wistar rats, and the active fractions did not have a significant effect on sodium–potassium pumps. It was suggested that the active fractions did not show signs of cardiotoxicity in Wistar rats. During the study period, the rats exhibited severe respiratory acidosis, leading to central respiratory arrest and death. 18
The crude extract of the plant reduced the amplitude of norepinephrine-induced contraction of isolated tissues such as the guinea pig vas deferens and the aortic strip. The effect was dose dependent, and it indicated that the plant extract had a significant alpha-blocking property. 11 The crude extract also showed significant larvicidal activity against Culex quinquefasciatus. 33
Chemistry of C. collinus Phytoconstituents
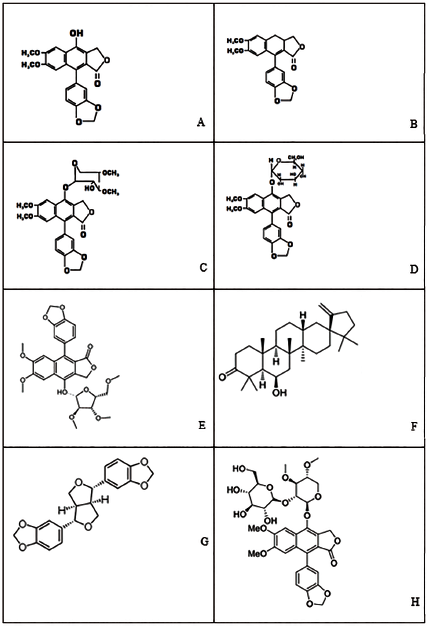
The major constituents of the genus Cleistanthus are arylnaphthalide lignans glycosides. Arylnaphthalide lignans include diphyllin, collinusin, cleistanthin, taiwanin, and other related compounds.
In 1964, two unknown compounds were isolated and purified from the fruits of C. collinus and named compound A (C37H55O2) and compound B (C31H52O2).
34
Later, Govindachari et al.
35
isolated cleistanthin, diphyllin, collunusin, and ellagic acid using the hot benzene extract of the C. collinus plant. Anjaneyulu isolated around five new compounds (wodeshiol; 3,4-dihydrotaiwanin C; cleistanthin D; Taiwanin-E 3,4-di-O-methyl-
Lakshmi et al.
38
isolated cleistanthin A and cleistanthin B from the chloroform extract of C. collinus barks, and the structures were proposed. In 1981, again, two more glycosides named cleistanthin B and cleistanthin C were isolated from the leaves of C. collinus.36, 39 The isolated diphyllin glycosides from various parts of the C. collinus have the sugar (glycone) moiety on the C-4 position of diphyllin (aglycone). Apart from cleistanthin A and cleistanthin B, taiwanin C, 3,4-dihydrotaiwanin C, taiwanin E, and 4-O-3,4-di-O methyl-β-
The furofuranoid lignans glycosides such as (+)-sesamin, 4-hydroxysesamin, paulownin, and wodeshiol were isolated from the heartwood of C. collinus. The heartwood of C. collinus also contains the dibenzylbutane lignan and dihydrocubebin.
36
Cleistanthin E: A member of the class of cleistanthins that is cleistanthin C in which the hydroxy group at position 4 of the 2,3-di-O-methyl-beta-D-xylopyranosyl moiety has been converted to the corresponding 2,3,5-tri-O-methyl-D-xylofuranoside.Chemical Structure C. collinus Phytoconstituents. (A) Diphyllin, (B) Collinusin, (C) Cleistanthin A, (D) Cleistanthin B, (E) Cleistanthin D, (F) Cleistanone, (G) (+)-sesamin and (H) Cleistanthoside A.
Pharmacological Properties of Isolated Phytoconstituents
Cytotoxic and Antitumor Effect of Cleistanthin A, Cleistanthin B, and Cleistanone
Cleistanthin A and cleistanthin B are known for their cytotoxic effects. Cleistanthin A caused the cytotoxic effect on MT2 cell lines and human phytohemagglutinin-stimulated human lymphocytes.37, 50 Cleistanthin B had less cytotoxicity against normal human cells such as bone marrow fibroblasts, peripheral blood lymphocytes, and oral fibroblasts. Cleistanthin B showed higher sensitivity in cancer cell lines than in normal cell lines. The compound cleistanthin showed chromosomal aberrations at lower doses than lethal doses. The studies concluded that the compound cleistanthin has an anticancer activity similar to other anticancer agents such as cisplatin, bleomycin, camptothecin, doxorubicin, and etoposide.23,51 Cleistanone exhibited moderate cytotoxic activity against human oral epidermoid carcinoma KB cells (IC50 = 93.6 µm), and its acetyl derivative exhibited a cytotoxic effect on MT2 cell lines.37, 47
Anti-candidal Effect of Diphyllin
In in vitro experiments, diphyllin exhibited anticandidal activity against Candida strains, such as Candida albicans, C. tropicalis, and C. glabrata with a minimal fungicidal concentration of 85–145 µg. 52
Effect of Cleistanthoside A, Cleistanthin A, and B on the Isolated Tissue Preparations
Cleistanthin A and B interacted with adrenergic and cholinergic receptors in in vitro studies on the isolated tissues. The isolated tissues used were guinea pig vas deferens and isolated heart to study the effect of the compounds on the alpha-adrenergic receptor and beta-adrenergic receptor, respectively. The other tissues used for studying the effect of cleistanthin A and cleistanthin B on nicotinic and muscarinic cholinergic receptors include rabbit vas deferens and rabbit jejunum, respectively. The study concluded that cleistanthin A and cleistanthin B have significant alpha-adrenergic receptor blocking and nicotinic cholinergic receptor blocking properties, and they do not have any effect on the beta-adrenergic and muscarinic cholinergic receptors. 53 Cleistanthin A and cleistanthin B also exerted vasodilatory action on guinea pig aorta through noncompetitive α1 adrenergic receptor inhibition. 54 Meanwhile, cleistanthoside A exhibited a direct vasoconstriction effect in isolated organ experiments. 49
Biodistribution Properties of Cleistanthin A and B
The animal models were used by Parasuraman et al. 55 to predict the biodistribution property of cleistanthin A and cleistanthin B. Normal and tumor-nude mice were intravenously injected with tagged cleistanthin A and cleistanthin B through the femoral vein and three MRI images (pre-contrast, immediate, post-contrast) were taken. It was found that cleistanthin A accumulated in the gastrointestinal tract, and cleistanthin B was distributed in the muscular tissues. 55
Effect of Cleistanthin A and Cleistanthin B on Motor Function in Mice
The patients who consumed the plant developed neuromuscular weakness and myasthenia-like crisis. The compounds of the crude extract responsible for this neuromuscular blocking property were studied by Madhav Rao et al. 56 The locomotor activity, motor function, and grip strength of mice treated with cleistanthin A and cleistanthin B were evaluated, but no significant effect of the compounds was observed. The study on the nicotinic receptor’s blocking property of the compounds using isolated mouse esophagus tissue also showed insignificant effects.
Antihypertensive Activity of Cleistanthin A and Cleistanthin B
The molecular docking and ex vivo studies showed that the compounds cleistanthin A and cleistanthin B exhibit the alpha adrenergic receptor blocking properties on the peripheral vascular system. The mean arterial pressure was measured invasively in male Wistar rats, and the effect of epinephrine, norepinephrine, acetylcholine, and histamine was recorded before and after the i.v. administration of cleistanthin A and cleistanthin B. A dose-dependent fall in the mean blood pressure in rats was demonstrated, and the effect of catecholamines was blocked after the administration of the compounds. 57
Diuretic Properties of Cleistanthin A and Cleistanthin B
Both cleistanthin A and B at doses of 12.5, 25, and 50 mg/kg also showed a significant diuretic effect when compared with the furosemide, with an increase in the urine volume in 24 hours. There was also an increase in the sodium and potassium excretion in 0–5 hours, similar to that of furosemide. There was no alteration in the pH value of the urine and chloride excretion when compared with the control group. 58 Cleistanthin A was also isolated from the plant P. toxodiifolius, a shrub commonly found in central and northeastern parts of Thailand, and is used in Thai Traditional Medicine as a diuretic. 19
Pharmacological Property of Arylnaphthalide Lignans
Antiproliferative and antileishmanial activities of diphyllin (isolated from Haplophyllum bucharicum Litv. [Rutaceae]) were reported. Diphyllin strongly inhibited the parasite internalization within macrophages. 42
Cleistanthin A caused neutrophilic granulocytosis in rodents and nonrodents and also prevented granulocytopenia induced by i.p. injection of cyclophosphamide. Cleistanthin B is also one of the constituents isolated from the leaves of C. collinus, and it is also effective against carcinogenesis. The anticancer potential of cleistanthin B was relatively lower than that of cleistanthin A. 59 The antitumor activity of cleistanthin A was evaluated using mice harboring Dalton’s ascites lymphoma and solid tumor S-180 sarcoma cells. The compound had significant antitumor activity against Dalton’s ascites lymphoma and solid tumor S-180 sarcoma cells.23, 59 Cleistanthin A caused significant chromatid aberrations in Chinese hamster ovary (CHO) cells. Exposure to a low dose of cleistanthin A did not have a significant effect on cell survival and DNA strand breaks. A high dose of cleistanthin A caused a high degree of DNA strand breaks and decreased cell viability. The CHO cells treated with a high concentration of cleistanthin A (20 µg/mL) caused apoptosis after long exposure (24 h). During the apoptosis process, DNA breaks because of enzymatic DNA cleavage were caused by cleistanthin A. 60
The genotoxic effects of cleistanthin B in CHO cells and cultured blood lymphocytes showed a decrease in cell viability and a concomitant increase in DNA strand breaks. Exposure of CHO cells for 30 minutes to cleistanthin B (1–6 µg/mL) resulted in the characteristic induction of chromatid-type aberrations. CHO cells when treated with low doses of cleistanthin B (1 µg/mL) for a short time (30 min) caused chromatid aberration but did not affect the cell viability nor DNA strand breaks significantly. The adverse effects of cleistanthin B on DNA and chromosomes and subsequent loss of viability suggested that the cell death caused by cleistanthin B was probably by apoptosis. 51 The i.v. and the i.p. administration of cleistanthin B in rats and rabbits resulted in decreased serum levels of ALP, lactate dehydrogenase, glutamate oxaloacetate transaminase, and glutamate pyruvate transaminase.
The minimum lethal dose of cleistanthoside A and cleistanthin A is 3 and 4 mg/100 g body weight, respectively. 49 The LD50 value of cleistanthin B in mice was reported to be 1.2 mg/kg body weight. 51
Conclusion
Plants were the primary sources of drugs for early drug discovery. There are various examples of drugs from plants that are utilized in treating serious diseases. They include plants such as Digitalis purpurea, Vinca rosea, and Atropa belladonna. It is also a well-known fact that all substances are poisons, and it is the dose that differentiates the drug and the poison. C. collinus and its active principles, such as leistanthins A, B, D, and E, in appropriate doses may be useful as a therapeutic option for treating many clinical conditions, and further studies are needed to evaluate it. Cleistanthins are also found in other plants that are edible or nontoxic. Arylnaphthalide lignans found in C. collinus may be an important source of new lead compounds in drug discovery research.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
