Abstract
Background
The individual teratogenicity profiles of Lumacaftor and Ivacaftor are known, but the combined effects remain unexplored.
Purpose
To evaluate the teratogenic profile of a combination of Lumacaftor and Ivacaftor in Sprague-Dawley Rats.
Materials and Methods
This study adhered to the Committee for Control and Supervision of Experiments on Animals (CCSEA) and Organisation for Economic Co-operation and Development (OECD) guidelines. Sprague–Dawley rats were acclimated and housed individually under controlled conditions. A polygamous breeding scheme was implemented, and pregnant female rats were assigned to control, vehicle control or treatment group following OECD Test Guideline 414. Lumacaftor and Ivacaftor were administered to pregnant rats at various doses, such as MRHD, 5MRHD, 8MRHD and 10MRHD, from gestation days 6 to 17. Clinical observations, body weight and food consumption of pregnant rats were monitored. Postmortem examinations included caesarean section, reproductive performance (the number of corpora lutea, implantation sites, early and late resorptions and live foetuses) and assessment of foetal analysis. The foetal analysis incorporated external malformation evaluation, visceral examination and skeletal examination using alizarin red solution.
Results
In pregnant rats exposed to Lumacaftor and Ivacaftor, the external examination was conducted on 206, 209, 199, 184, 198 and 201 foetuses in their respective groups, each consisting of 19 litters. Within this cohort, approximately 33% of foetuses underwent visceral examination. Additionally, across six groups approximately 50% of foetuses underwent skeletal examination. Importantly, no abnormalities were detected in skeletal, visceral or histopathological assessments across the treatment groups encompassing doses of a combination of Lumacaftor and Ivacaftor at MRHD, 5MRHD, 8MRHD and 10MRHD. Additionally, no significant morbidity or mortality related to the treatment was observed.
Conclusion
In conclusion, the study indicates that the combination of Lumacaftor and Ivacaftor at doses equivalent to MRHD, 5MRHD, 8MRHD and 10MRHD did not induce significant teratogenic effects, affirming their safety during pregnancy in rats.
Introduction
Teratogenicity studies in animals are performed to assess the likelihood of a test substance inducing developmental abnormalities in the offspring of pregnant animals. 1 These abnormalities can be identified through prenatal or postnatal examination. 2 Substances may cause reproductive toxicity early in the reproductive cycle, such as during early gestation, by interfering with fertilization or implantation. Animal teratogenicity studies allow for the early identification of potential teratogenic effects of a drug, prior to human trials. This can lead to the elimination of potentially harmful drugs from further development and can prevent unnecessary exposure of pregnant women and their foetuses to potential harm. Animal teratogenicity studies can identify specific developmental stages when the foetus is most susceptible to the effects of a drug. This information can inform the timing of medication use during pregnancy and guide clinical decision-making. Animal teratogenicity studies further help identify specific organs or systems that may be affected by a drug, providing valuable information on the mechanisms of teratogenicity and guiding further research. The assessment of teratogenicity is a critical aspect of drug development and regulatory approval processes, particularly for combination drugs that may pose greater risks to foetal development. The accuracy of estimated teratogenic risk relies on well-executed experiments that involve a sufficient number of test animals, the implementation of sound animal husbandry practices and comprehensive assessments of both maternal and foetal conditions. 3 Interactions between drugs may lead to different or more pronounced toxic effects, including teratogenicity, that are not observed with individual drugs. Furthermore, it is possible that individual drugs may be safe at certain doses, but when combined, the total dose exceeds the safe limit, resulting in teratogenicity. 4 Lumacaftor and Ivacaftor are two drugs that have been approved for the individual treatment of cystic fibrosis (CF). Lumacaftor/Ivacaftor, combined with a single drug, is indicated for the treatment of CF in patients aged 2 years and older who are homozygous for the F508del mutation in the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) gene. Embryofoetal development studies in rats and rabbits were conducted with the individual components of Lumacaftor and Ivacaftor. However, there are limited data on the teratogenicity of these two drugs in combination in animal models. 5 Hence, the present study aimed to investigate the teratogenic profile of Lumacaftor/Ivacaftor in Sprague–Dawley rats. This study has the potential to contribute to clinical decision-making and ultimately improve the safety and health outcomes of pregnant women and their offspring.
Materials and Methods
Ethical Statement
The experimental protocol received approval from the Institutional Animal Ethics Committee of the Department of Pharmaceutical Sciences, Andhra University. All the experimental procedures were conducted in accordance with the Committee for Control and Supervision of Experiments on Animals (CCSEA), Organisation for Economic Co-operation and Development (OECD) guidelines.
Animals and Animal Housing
The animals used in this study were selected in accordance with the guidelines outlined in the OECD Test Guideline 414 for the conduct of teratogenicity studies. Male Sprague–Dawley rats weighing 250 ± 25 g and female Sprague–Dawley rats weighing 200 ± 20 g and aged 12–14 weeks and 10–12 weeks, 6 respectively, were obtained from M/s. VAB BioSciences, Hyderabad, Telangana (Registration No: 282/PO/RcBt/S/2000/CPCSEA), a licensed supplier who provided documentation that they were bred and housed in accordance with the CCSEA guidelines. Before the commencement of the study, a thorough health examination was conducted on all animals to ensure their suitability for inclusion. Furthermore, the rats were acclimated for 7 days to adapt to the experimental environment. Upon arrival, the rats were housed individually in clean cages lined with soft bedding. The room temperature was maintained at 22°C–25°C, the relative humidity was 40%–70% and the light/dark cycle was 12 hours. 7 All animals were provided unrestricted access to standard rodent diets and water throughout the duration of the study.
Mating Procedure
A polygamous breeding scheme was used in which one male rat was housed with two female rats. 8 The date the male rat was added to the female rats was recorded on each breeding cage card. Vaginal smears were taken at the same time each day in the morning for a minimum of 7 consecutive days using the wet smear technique. 8 The female rat was lifted at the base of her tail, and approximately 0.2 mL of saline was flushed into the vaginal cavity using a blunt-tipped disposable pipette, which was repeated twice. The saline was then expelled onto a clean microscope slide, which was covered with a coverslip. The smears were examined under a microscope at low power. Mating was confirmed by the presence of spermatozoa in the vaginal smear.9, 10 Spermatozoa presented for at least 12 hours following copulation. When pregnancy was confirmed, the female rats were separated and placed in individual cages to avoid overcrowding. The day of confirmation of pregnancy was considered day 0 of gestation.
Grouping of Animals
A total of 180 young adult Sprague–Dawley rats (60 males and 120 females) with unique identification numbers were used in the study. Pregnant females were unbiasedly assigned to the control, vehicle control or four treatment groups, each containing 20 rats. The study followed the OECD Test Guideline 414 and used four dose levels based on the Maximum Recommended Human Dose (MRHD) of Lumacaftor and Ivacaftor. Male rats were used only for breeding purposes and were not included in the study, which focused on pregnant female rats and their offspring.
Dosage and Administration of Doses
Pregnant female rats were divided into different treatment groups: the control group received 0.9% w/v normal saline solution orally, the vehicle control group received dimethyl sulphoxide (DMSO) alone and the MRHD group received the maximum recommended human dose of Lumacaftor and Ivacaftor dissolved in DMSO. The 5MRHD group received a dose five times the Maximum Recommended Human Dose (5MRHD), the 8MRHD group received a dose eight times the Maximum Recommended Huan Dose (8MRHD) and the 10MRHD group received a dose 10 times the Maximum Recommended Human Dose (10MRHD). Treatments were administered once a day, in the morning, from gestation day 6 to gestation day 17, using sterile stainless steel gavage needles (18G × 50 mm Rat Curved-end) attached to an oral gavage glass syringe. The daily treatment volume was calculated based on the animal’s most recent body weight.
Observation of the Dams
Clinical observations were made and recorded at least once a day, preferably at the same time each day, taking into consideration the peak period of anticipated effects after dosing. The condition of the animals, including mortality, morbidity, pertinent behavioural changes and all signs of overt toxicity, was recorded.
Estimation of Body Weight and Food Consumption of Pregnant Animals
During the study, all the animals were weighed on day 0, on the first day of dosing, every 3 days during the dosing period and on the day of the scheduled kill. Food consumption was recorded at 3-day intervals and coincided with the number of days of body weight determination.
Postmortem Examination
Pregnant rats were euthanized 1 day before the expected delivery. Macroscopic examination was also conducted on females who showed signs of abortion or premature delivery. Dams were examined for abnormalities during termination. Caesarean section and foetal analyses were performed. Sodium pentobarbital was used for anaesthesia. The gravid uterus was exposed through laparotomy and then detached from the placenta. Foetuses were assessed as alive or dead. After the removal of the membranes and maternal tissues, the foetuses were weighed, and the placental weight was recorded before histopathological processing.
Study Assessments
This study aimed to assess the effects of administering different doses of the combination of Lumacaftor or Ivacaftor to pregnant Sprague–Dawley rats from gestational day 6 to gestational day 17. The objectives of these studies include evaluating changes in body weight, food consumption, potential maternal morbidity/mortality and impact on reproductive performance. It was also aimed to determine the incidence of external malformations in rat foetuses and assess visceral and foetal skeletal development following exposure to combinations of various doses of Lumacaftor and Ivacaftor.
Statistical Analysis
The statistical analysis was performed by using GraphPad Prism version 8.0.1. Descriptive statistics were employed for categorical data. The Kruskal‒Wallis test and analysis of variance (ANOVA) were used for multiple comparisons of variables between and among the groups, and the chi-square test was used for the determination of the sex ratio.
Results
Change in Body Weight, Water Intake and Food Consumption of Pregnant Rats
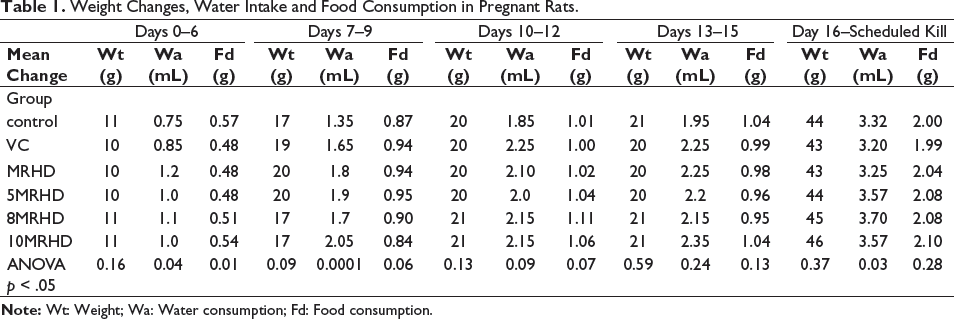
Our analysis revealed no statistically significant differences in mean weight changes among the groups at any of the examined time points. Specifically, the ANOVA p values for days 0–6 (p = .16), days 7–9 (p = .09), days 10–12 (p = .13), days 13–15 (p = .59) and day 16–scheduled kill (p = .37) all surpassed the conventional significance threshold of 0.05. These results indicate a consistent pattern in mean weight changes across all dose levels of the combination of Lumacaftor and Ivacaftor.
While all pregnant rats displayed a consistent increase in water intake throughout gestation, a fascinating interplay between time and treatment emerged. The MRHD-treated group, 5MRHD and 8MRHD groups had a statistically significant increase in water consumption during days 0–6 of gestation compared to other treatment groups (p .04). During days 7–9, pregnant rats treated with MRHD, 5MRHD and 10MRHD doses of the Lumacaftor/Ivacaftor combination exhibited a significant increase (****p < .0001) in water consumption, while the 8MRHD group did not show a significant change in water consumption during the same period compared to the control and vehicle control-treated groups. No significant change in water sconsumption was observed in all treatment groups during days 10–12 (p .09) and days 13–15 (p .24), respectively. The vehicle control groups had the lowest water consumption on day 16–scheduled kill followed by the control group. The MRHD, 5MRHD, 8MRHD and 10MRHD groups had significantly higher water consumption than the control and vehicle groups. The difference between the treatment groups and the control groups was statistically significant (days 16–18 p .02 and day 19–scheduled kill p .03). There was no difference in water consumption between the treatment groups on days 0–6. However, the control group had a significantly higher food consumption than the other groups (p .01). There was no statistically significant difference in water consumption between the control groups and the treatment groups on days 7–9 (p .06), days 10–12 (p .07) and days 13–15 (p .13). There was a statistically significant difference (p .02) in water consumption between the control groups and the treatment groups on days 16–18, where no statistical difference was identified during day 19–scheduled kill (p .28). The mean change in body weight, water consumption and food consumption in all treatment groups is depicted in Table 1.
Weight Changes, Water Intake and Food Consumption in Pregnant Rats.
Morbidity and Mortality
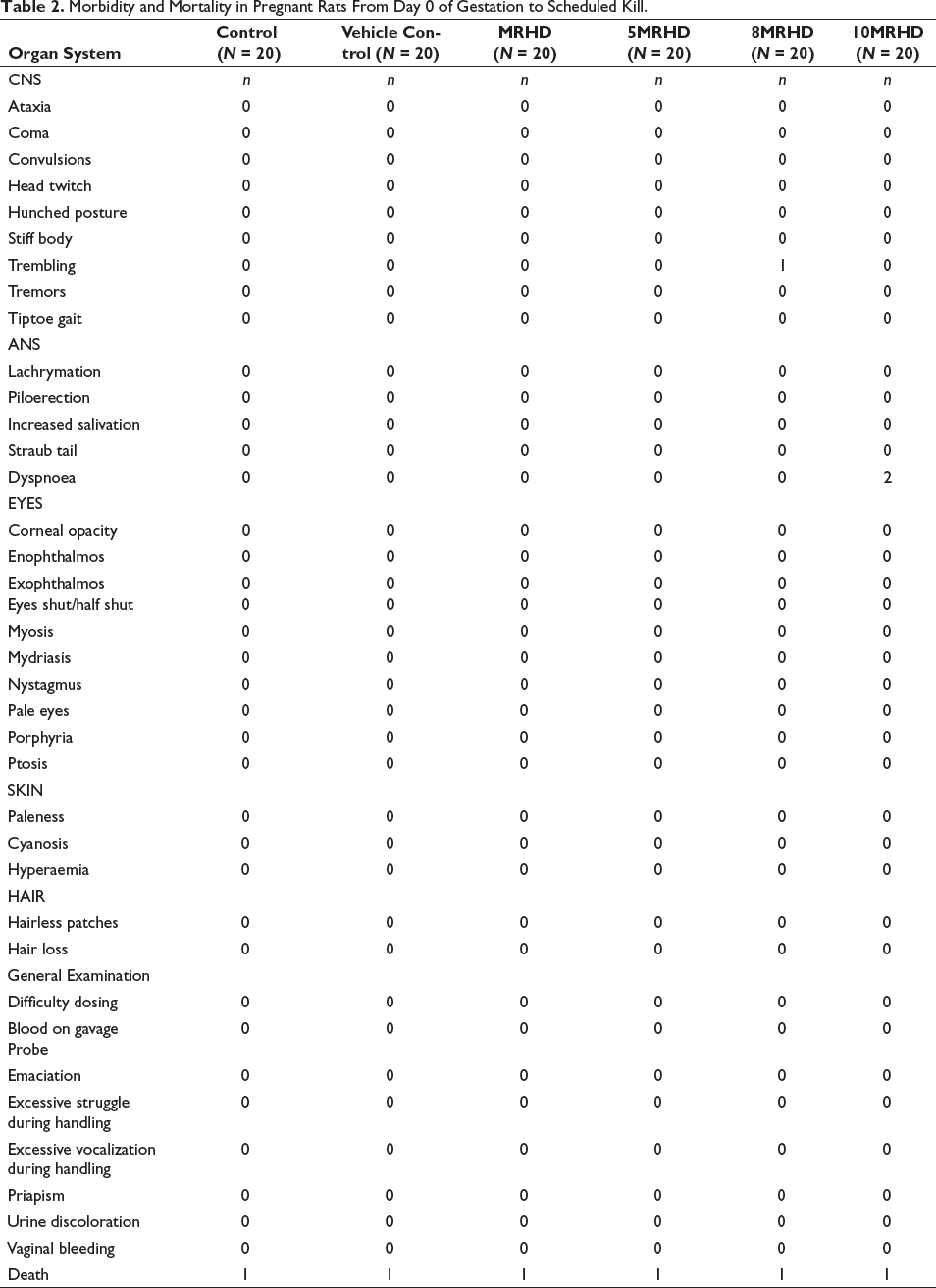
All pregnant rats were examined for signs of morbidity and mortality from day 0 of gestation to the scheduled kill. No significant illness was observed in any of the treatment groups, but two pregnant rats in the 10MRHD group had difficulty breathing, and one rat showed trembling in the 8MRHD treated group (Table 2). Six pregnant rats died prior to the scheduled kill, one in each treatment group. In the Control group, one mortality occurred during days 0–6, attributed to factors unrelated to treatment, with no further mortalities in subsequent intervals. In the Vehicle Control group, one mortality on days 7–9, shortly after treatment initiation, may not directly link to treatment, and no additional mortalities were observed. The MRHD group had one mortality on days 10–12, 5MRHD on days 13–15, 8MRHD on days 13–15 and 10MRHD on days 7–9, indicating isolated events during different intervals.
Morbidity and Mortality in Pregnant Rats From Day 0 of Gestation to Scheduled Kill.
Reproductive Performance
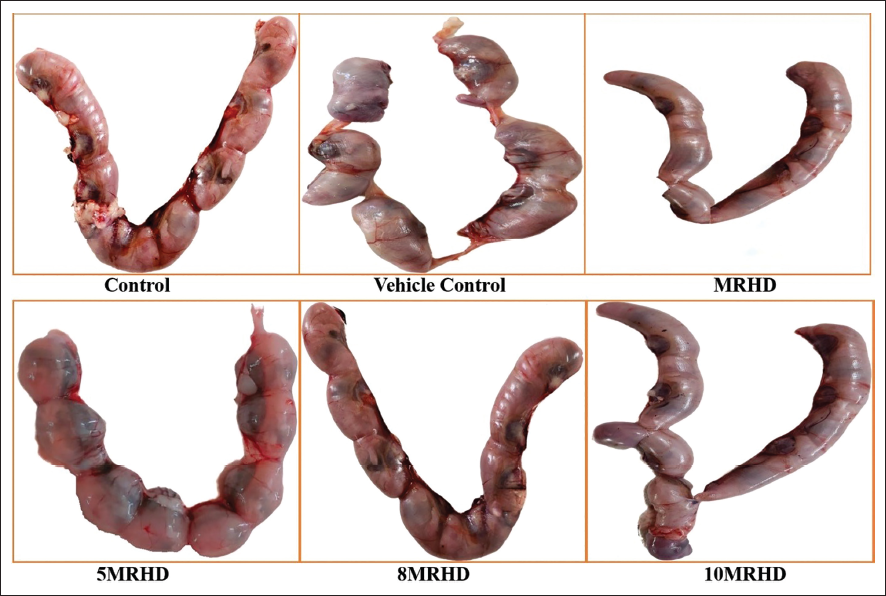
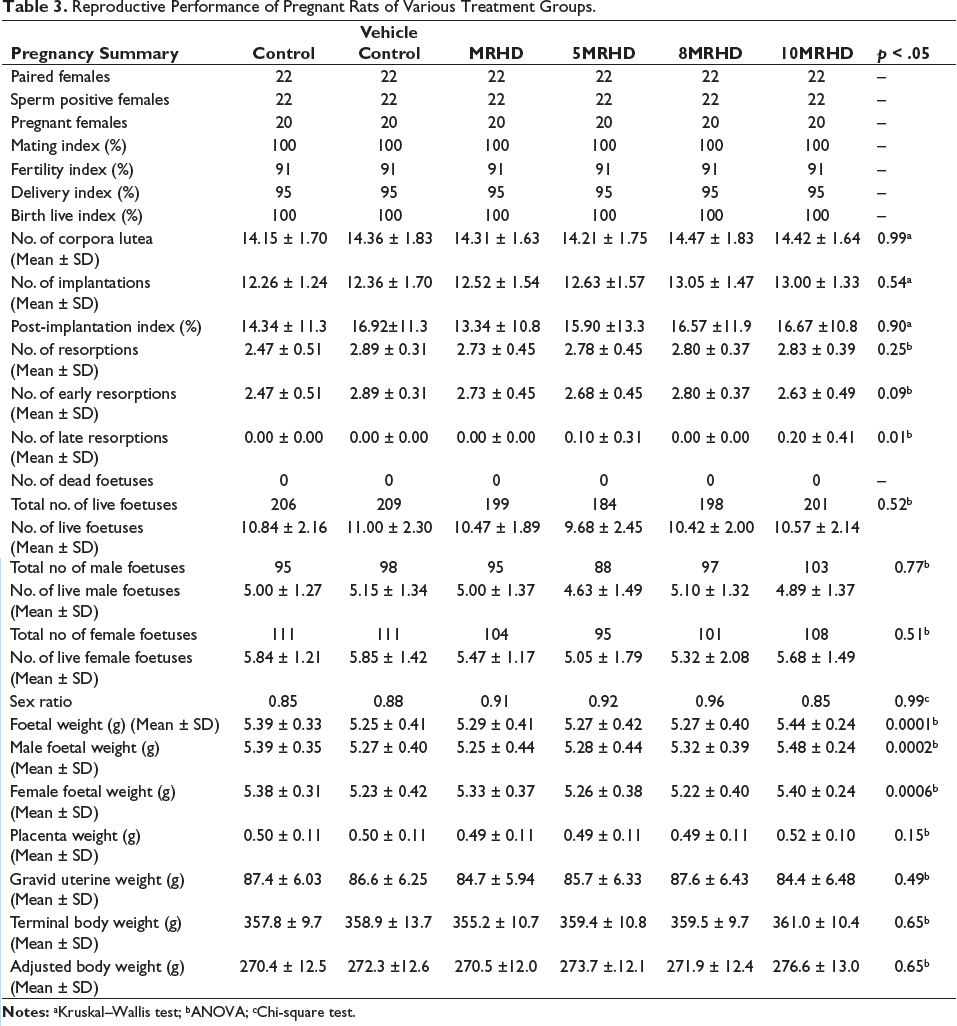
On the day of the scheduled kill, animals were administered with sodium pentobarbital to anaesthetize the dams. Following the collection of the uterus (Figure 1) and ovaries, the gravid uterus was weighed, and various parameters were quantified, including the number of corpora lutea, implantation sites, early and late resorptions, live foetuses and their sex. The weights of the foetal and placental tissues were also determined. The following reproductive indices were calculated 11 : Mating Index, Fertility Index, Delivery Index, Birth Live Index and Post-Implantation Loss Index (Table 3).
Reproductive Performance of Pregnant Rats of Various Treatment Groups.
External, Visceral and Skeletal Examination
Morphological abnormalities of the foetuses were assessed through an external examination, which involved careful evaluation of the eyes, ears, tail, head, mouth and limbs. No visible signs of birth defects, such as cleft lip, club foot or other external malformations, were detected in any of the foetuses (Figure 2).
Foetuses Removed From Uterus by Caesarean Section.
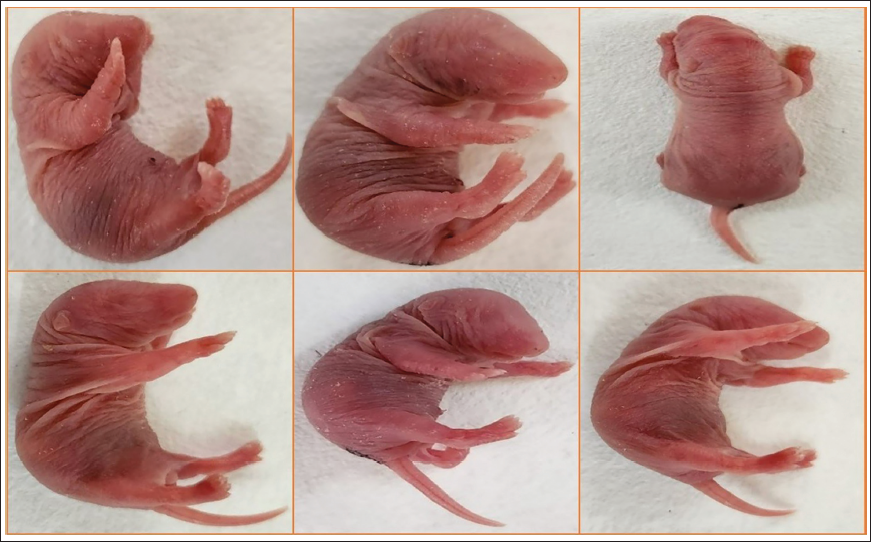
After removal from the uterus, the foetuses were euthanized, and one-third of the foetuses from each litter were promptly fixed in Bouin’s solution for 14 days to allow proper fixation. 12 The structures of each section were examined in cranial-caudal order, and the size, form, shape, symmetry and position of the internal structure were noted. 13
The examination of foetal parameters across different treatment groups, including Control, Vehicle Control, MRHD, 5MRHD, 8MRHD and 10MRHD, revealed no statistically significant differences, as indicated by the p value of .52. A total of 206, 209, 199, 184, 198 and 201 foetuses were examined in their respective groups, with 19 litters examined in each group. The absence of small foetuses (runt) and the low occurrence of large foetuses (>6 g) across all groups further demonstrate the uniformity in foetal size distribution. Notably, the weight of the large foetuses in the Control group was 6.2 g, while other groups exhibited comparable weights. Additionally, no general abnormalities were observed in any of the treatment groups, including the absence of abnormalities in foetal eyes, ears, nose, mouth and jaw, trunk, limbs, paws and digits and tail. These findings collectively suggest a lack of discernible treatment-related effects on foetal morphology and development, reinforcing the safety profile of the combination of Lumacaftor and Ivacaftor at doses multiple times of MRHD.
A total of 206, 209, 199, 184, 198 and 201 foetuses were examined across the respective groups, with 19 litters examined in each group. Within this cohort, 69, 70, 66, 61, 66 and 67 foetuses were subjected to visceral examination, constituting approximately 33% of the total foetal population in each group. Notably, no abnormalities were detected in coronal sections, lungs and trachea, heart, liver, stomach or kidneys across any of the treatment groups (Figure 3). These findings collectively suggest a consistent lack of treatment-related effects on visceral foetal parameters, reaffirming the overall safety profile of the administered doses of a combination of Lumacaftor and Ivacaftor.
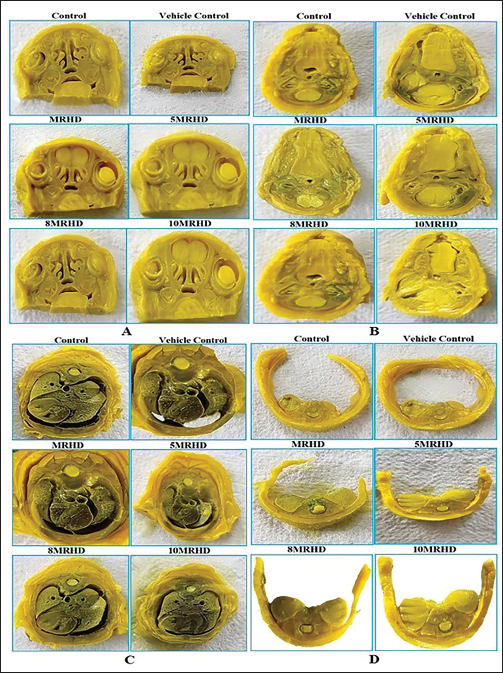
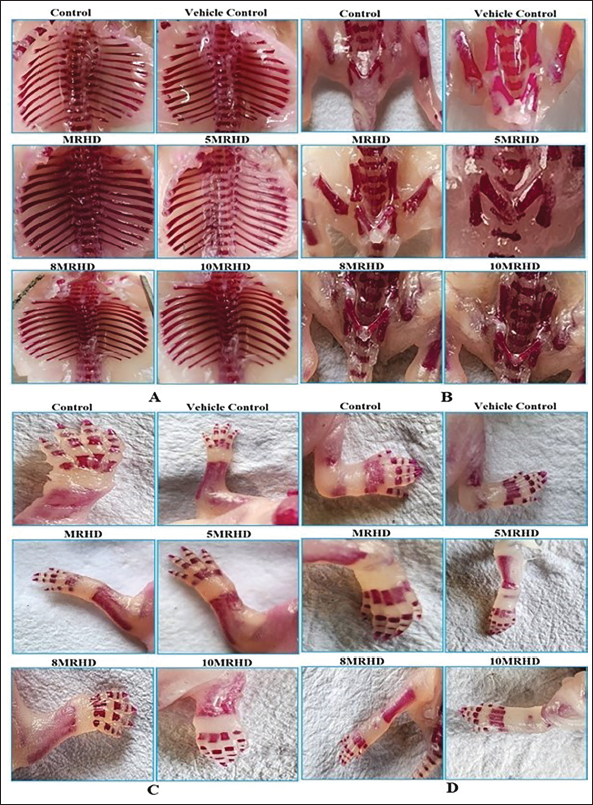
In a rigorous examination of 1,197 foetuses across six groups (19 litters per group), approximately 103, 105, 100, 92, 99 and 101 foetuses, constituting 50%, were subjected to skeletal examination. This assessment revealed no treatment-related abnormalities in cervical, thoracic, lumbar or caudal vertebrae, ribs, sternebrae, thoracic and pelvic girdles, forelimbs/forepaws or hind limbs/hind paws (Table 4). This consistent absence of malformations across all treatment groups (Figures 4 and 5) indicates a safety profile for foetal skeletal development with these administered doses.
External, Visceral and Skeletal Examination of Foetuses.
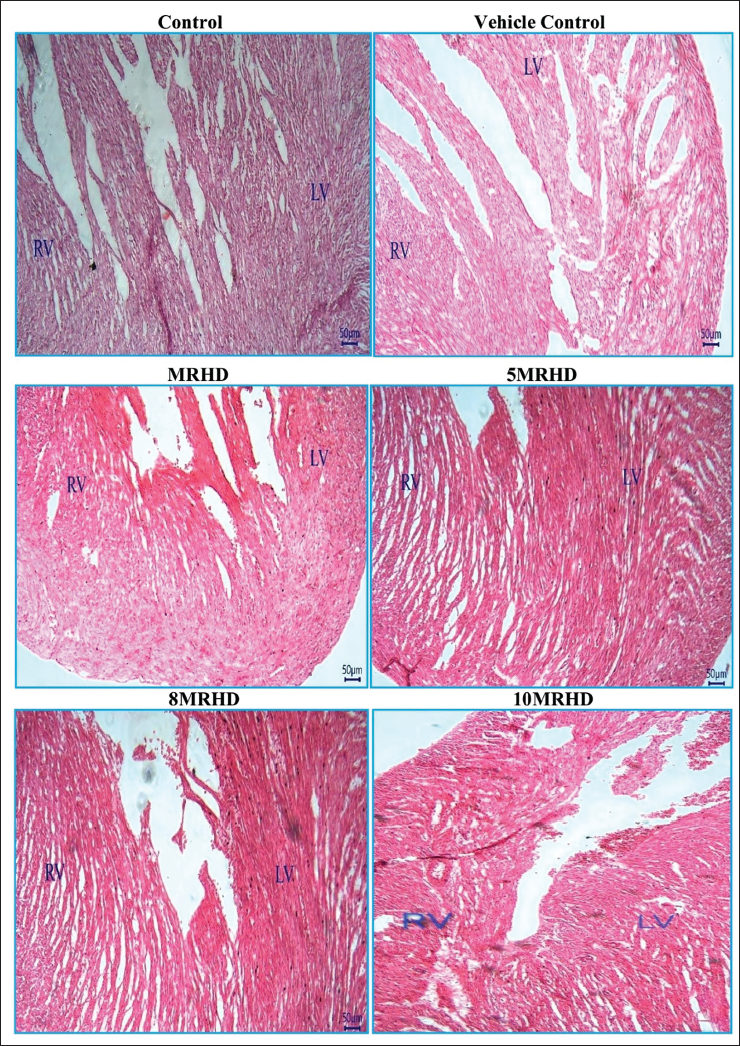
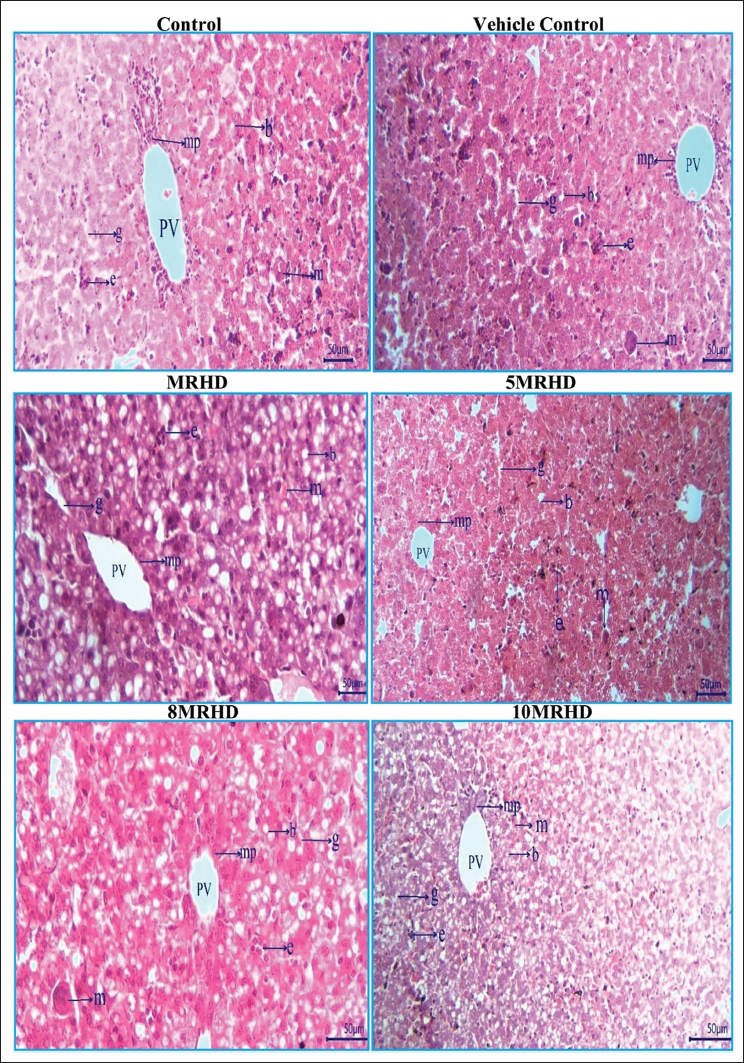
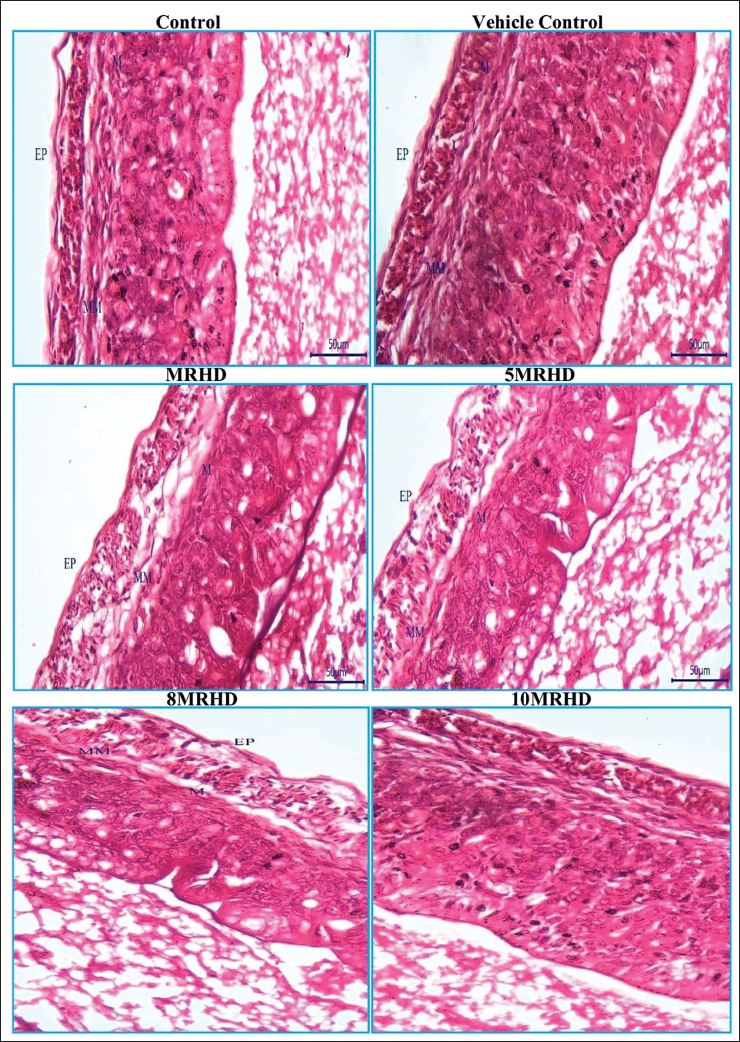
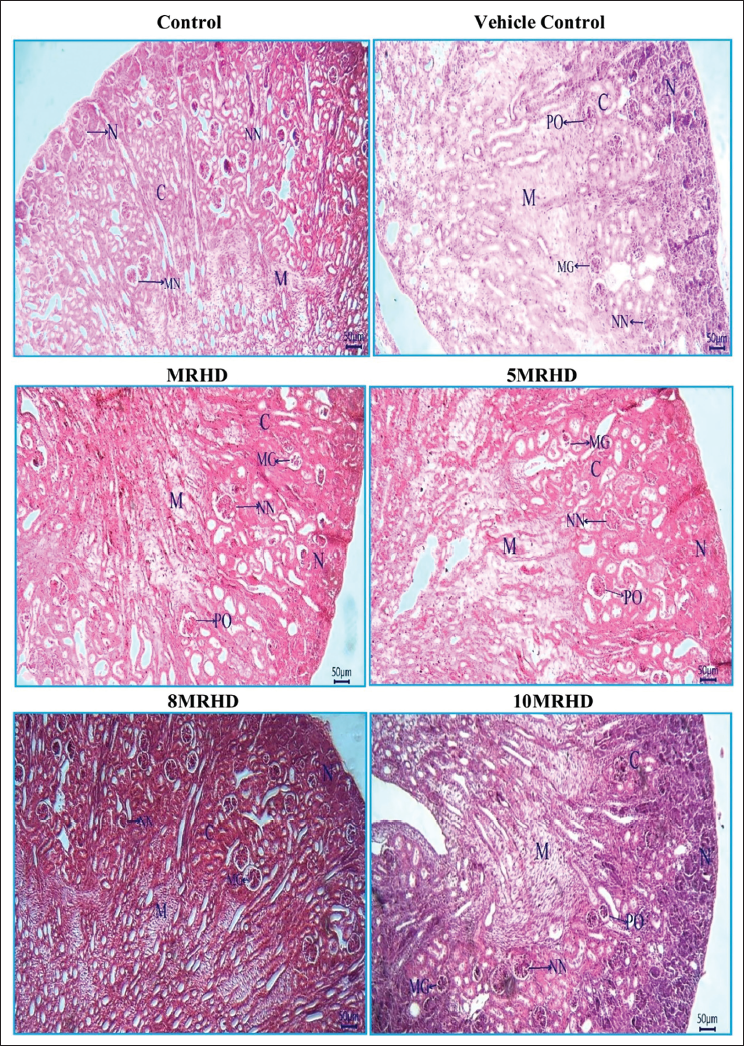
The histopathological examination of rat foetal tissues following maternal exposure to Lumacaftor and Ivacaftor combination at various doses revealed reassuring findings across multiple organs. Lung development showed no adverse effects, with normal bronchioles and alveoli observed in all dosage groups. Cardiovascular examination demonstrated structurally normal right and left ventricles without septal defects, affirming positive cardiovascular profiles (Figure 6). Liver examination indicated a consistent and reassuring pattern of normal development, with no apparent teratogenic effects on hepatocyte cords, glycogen storage, hematopoietic activity and biliary structures (Figure 7). In the stomach, the presence of a well-developed, keratinized epithelium in the non-glandular region suggested maintained structural integrity, supported by the prominence of the muscularis mucosa and deep eosinophilia in the lamina propria (Figure 8). Kidney examination showed typical structural and developmental processes, with no observable teratogenic effects on nephrogenic zones and nephrons at tested dosage levels, that is MRHD, 5MRHD, 8MRHD and 10MRHD (Figure 9). These comprehensive findings collectively emphasize the safety of the Lumacaftor and Ivacaftor combination during foetal development, providing valuable insights into teratogenic profile across vital organ systems.
Discussion
Teratogenicity studies in animals are performed to assess the likelihood of a test substance to induce developmental abnormalities in the offspring of pregnant animals. Teratogens can cause birth defects or abnormalities in developing foetuses by disrupting various stages of pregnancy through several mechanisms such as altering DNA and cell division, altering cell signalling and gene expression, inducing apoptosis, disrupting cell differentiation and migration and disrupting cell differentiation and migration. 14 Several factors contribute to the teratogenic profile of a substance which include a higher dose of a chemical substance, and timing of exposure, as vulnerability to chemical exposure is greater during the early stages of embryonic development than during the foetal development period. Maternal metabolism towards a specific drug can influence the metabolites that ultimately reach the foetus. Additionally, differences in placental membranes, transport rate, formation and biotransformation can also impact foetal exposure to the teratogenic agent. 15 Furthermore, the principles of toxicokinetics and pharmacokinetics of different classes of compounds influence the degree of penetration of the placental membrane. Generally, the greater the lipid solubility of an agent, the more likely the potential for foetal exposure through maternal–foetal circulation. 16
Combination therapy can increase the efficacy of treatment by targeting multiple pathways or mechanisms of disease. The use of multiple drugs with different mechanisms of action can reduce the likelihood of the development of drug resistance. Further, combination therapy can allow for lower doses of each drug to be used, which can reduce the risk of adverse effects while maintaining efficacy. The potential for teratogenicity with drug combinations is not always predictable based on the known teratogenicity of the individual drugs. 17 The combination of Lumacaftor and Ivacaftor was first approved by the US Food and Drug Administration (US FDA) for the treatment of CF in patients aged 12 years and older who have two copies of the F508del mutation in the CFTR gene (FDA, 2015). Existing literature suggests that Lumacaftor and Ivacaftor exhibited no adverse effects on fertility and reproductive performance in rats, up to doses eight times MRHD for Lumacaftor and 10 times the MRHD for Ivacaftor. Dose selection in this study, including MRHD, 5MRHD, 8MRHD and 10MRHD, was based on exposures extrapolated from a prior 6-month repeat-dose toxicity study in rats, as indicated by existing literature. The pregnant rats were treated with MRHD, 5MRHD, 8MRHD and 10MRHD throughout the organogenesis.
Our study findings reveal that there were no significant effects on reproductive performance and foetal abnormalities in any of the treated groups. External examination of foetuses revealed no runt (foetus < 3 g), no anasarca, conjoined twins and cutis aplasia. Further, an external examination of the foetal cranium showed no signs of acephaly, anencephaly, cranioschisis, domed head, exencephaly, macrocephaly or microcephaly in any of the litters treated with any of the treatment group. Similarly, ablepharia, exophthalmos, anophthalmia, macrophthalmia and microphthalmia of the eyes, anotia, macrotia, microtia and deformed pinna of ears, arrhinia, deficit naris of the nose, cleft lip, cleft palate of the mouth, spina bifida, scoliosis of the trunk and adactyly, brachydactyly, oligodactyly, polydactyly, syndactyly of limbs, paws and digits were not observed, confirming any effect on the formation of external malformation. During visceral examination, the coronal section of the head is normal without hydrocephaly and asymmetric pattern. Sections of the lungs and trachea were normochromic and showed no fused, enlarged, supernumerary lobes. The sections of the heart revealed no septal defects, cardiomegaly or valvular alteration. The liver is normal, without fused or supernumerary lobes. No signs of hepatomegaly were observed in litters of all treatment groups. Kidneys are normally sized without hydronephrosis. For skeletal examination of the foetuses, one-half of the foetuses from each litter were stained using alizarin red S solution. 18 No skull bone detected with reduced ossification. Cervical, thoracic, lumbar and caudal bones are adequately formed without any incomplete ossification. Similarly, ribs (13 pairs) and sternebrae (six pairs) were adequate. The clavicle, scapula bone and the regions of the pelvic girdle such as ilium, ischium and pubis were symmetric with adequate ossification. The distal phalanges, proximal phalanges, metacarpals of forelimbs and fore paw, distal phalanges, proximal phalanges, metatarsals of hind limbs and hind paw are adequate in number without any deformities in all the examined foetuses of all treatment groups, thus suggesting no significant external malformations, skeletal abnormalities even after administration of Lumacaftor and Ivacaftor combination at a dose multiple times higher than the MRHD. The maternal mortality is also similar in all treatment groups and none of the maternal deaths were treatment-related.
No adequate and well-controlled studies of Lumacaftor and Ivacaftor have been conducted in pregnant women. The extent of exposure to Lumacaftor and Ivacaftor in pregnant women during clinical trials was very limited.
Teratogenicity studies in animals do not necessarily confirm teratogenicity in humans for various reasons. Recognition or identification of teratogens in humans is challenging due to therapeutic dosages being significantly lower than those administered to animals for inducing malformations. Pregnant women typically do not undergo prolonged drug therapy or receive numerous drugs. Establishing causation or association in humans demands extensive analysis and a large number of controlled cases. Moreover, chemicals displaying teratogenic properties in animals may lack normal exposure routes in humans, leaving their potential effects untested in the human population. 19
Limitations of this study include the focus solely on rodent models, specifically pregnant rats. While rodents are a common choice for teratogenicity studies, it may limit the generalizability of the findings to other species, including humans. A parallel study using non-rodent models, such as rabbits, would provide a more comprehensive assessment of the teratogenic potential across different species. Furthermore, this study primarily concentrated on evaluating teratogenic effects during gestation. However, to obtain a more holistic understanding of the safety profile, a separate investigation should be conducted to assess the impact of the combination of Lumacaftor and Ivacaftor on the offspring during the lactation period. This is crucial as potential adverse effects may manifest postnatally.
Conclusion
The results of this study suggest the pregnant rats treated with a combination of Lumacaftor and Ivacaftor at a dose of MRHD, 5MRHD, 8MRHD and 10MRHD did not exhibit any significant teratogenic effects on foetuses, suggesting that the Lumacaftor and Ivacaftor combination is safe during pregnancy. Conducting a parallel investigation using non-rodent models, such as rabbits, is essential for a more comprehensive assessment across diverse species. Furthermore, the need for further studies during lactation is essential to comprehensively assess the safety profile of the combination of Lumacaftor/Ivacaftor. This additional investigation is crucial for evaluating potential postnatal impacts and ensuring a thorough understanding of the overall safety during this critical period.
Abbreviations
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for financial support under the Research Fellowship scheme by the University Grants Commission, a statutory body of the Government of India through an Act of Parliament for the coordination, determination and maintenance of standards of university education in India.
Statement of Ethical Approval and Informed Consent
The experimental protocol received approval from the Institutional Animal Ethics Committee of the Department of Pharmaceutical Sciences, Andhra University. All experimental procedures were conducted in accordance with the Committee for Control and Supervision of Experiments on Animals (CCSEA), Organisation for Economic Co-operation and Development (OECD) guidelines.
