Abstract
Cystic fibrosis (CF) is a common life-limiting genetic condition caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. The CFTR protein is present in many epithelial cells in the body, and CF is characterised by suppurative lung disease, exocrine pancreatic insufficiency, and elevated sweat chloride. Traditionally, treatment of CF has involved managing the complications of defective CFTR function, such as airway clearance techniques for impaired lung mucociliary clearance and pancreatic enzyme replacement for pancreatic insufficiency. More recently, treatments have been developed which improve CFTR function. The aim of this article is to summarise the evidence surrounding treatments that improve CFTR function and their implications for clinical practice.
Keywords
Cystic fibrosis (CF) is the most common life-limiting autosomal recessive condition in whites and is caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene. 1 The CFTR protein functions principally as a chloride channel expressed on the epithelial surface of the lungs and other epithelial lined surfaces. The CFTR also regulates the epithelial sodium channel. 2 Absent or reduced CFTR function in the lungs of patients with CF results in airway surface liquid with altered pH and increased viscosity, causing a cycle of repeated infection and inflammation, resulting in progressive lung damage. 3 Impaired CFTR function also affects other organs with complications, including pancreatic exocrine insufficiency, meconium ileus, absence of vas deferens in men, CF-related liver disease, CF-related diabetes, and elevated sweat electrolytes. 1
Until recent times, CF treatments have all been aimed at managing the complications of defective CFTR, such as enzyme replacement therapy for pancreatic exocrine insufficiency, antibiotics for lung infection, and airway clearance and mucolytics for thick airway secretions. 4 Over time, therapeutics have been developed specifically for CF, such as inhaled recombinant human DNase (a mucolytic) and specific nebulised antibiotics such as TOBI (nebulised tobramycin). These, however, have not addressed the problem of altered CFTR function. This has led to slowing of the inevitable decline in lung function in patients with CF undertaking a large number of treatments, with patients often struggling to adhere to their treatment regimen. 5 Treatments aimed at restoring CFTR function have the potential to correct the underlying defect, arrest decline in lung function, and reduce patient treatment burden.
The aim of this article is to examine the evidence regarding medications that improve CFTR function and to discuss the implications of these treatments for the future of CF management.
CFTR Mutations
More than 2000 different mutations have been identified in the CFTR gene, although it is not clear that they all cause disease. 6 The disease-causing mutations are classified into 6 functional classes based on their effect on the CFTR protein. 7 The most common mutations are listed in Table 1. Class I mutations are usually due to a nonsense mutation causing a premature stop codon and result in no functional CFTR being made. These mutations may be treated by medications which promote read-through of the premature stop codon. Class II mutations are caused by mutations which cause production of a misfolded CFTR protein that is destroyed prior to reaching the cell membrane. These mutations are addressed by a CFTR corrector, which is a class of medications which improves intracellular transport of CFTR to the cell membrane. 8 Class III mutations are often referred to as gating mutations and result from missense mutations that lead to a CFTR protein which reaches the cell membrane; however, it has reduced open probability with reduced or absent function. CFTR potentiators are medications that improve CFTR function at the cell membrane and hence address class III mutations. 9 Class IV mutations are also missense mutations causing amino acid substitutions that lead to a CFTR that when open has reduced conductivity. Potentiators may also improve channel conductivity and hence help class IV mutations. Class V mutations are missense mutations causing a splicing defect that leads to a reduced overall production of CFTR. Class VI mutations are missense mutations which result in decreased CFTR stability at the membrane leading to decreased CFTR function.
Most common cystic fibrosis transmembrane conductance regulator mutations by mutation classes. 10
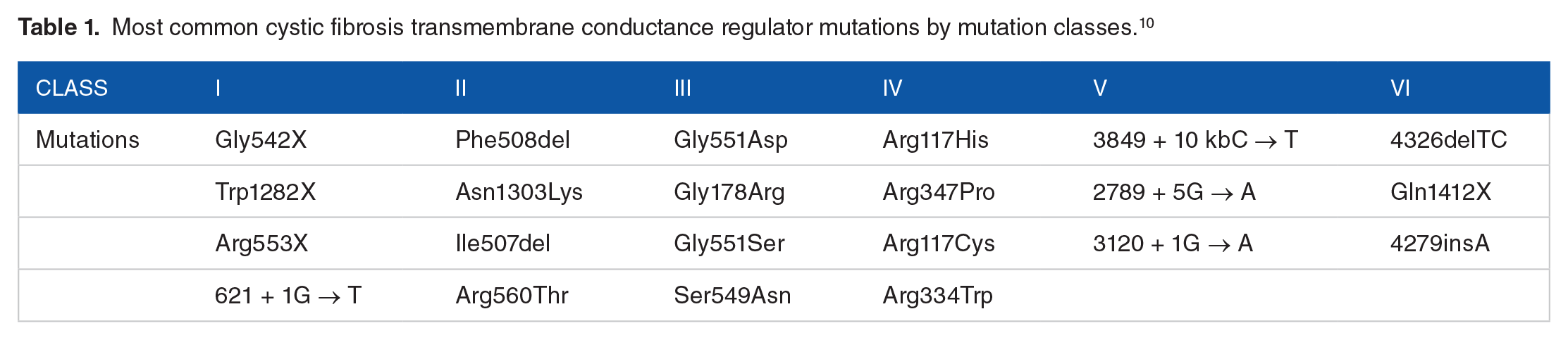
It is possible for 1 mutation to cause multiple different functional deficits in the CFTR. For instance, Phe508del on chromosome 7, the most common mutation causing CF, is a 3–base pair deletion that causes a single amino acid deletion from the CFTR. It leads to CFTR protein misfolding (class II), reduced channel opening time (class III), conductivity (class IV), and reduced protein stability (class VI). 8
Treatments Aimed at improving CFTR Function
The evidence from the major trials of medications that improve CFTR function are summarised in Table 2.
Summary of clinical trials of medications improving CFTR function.
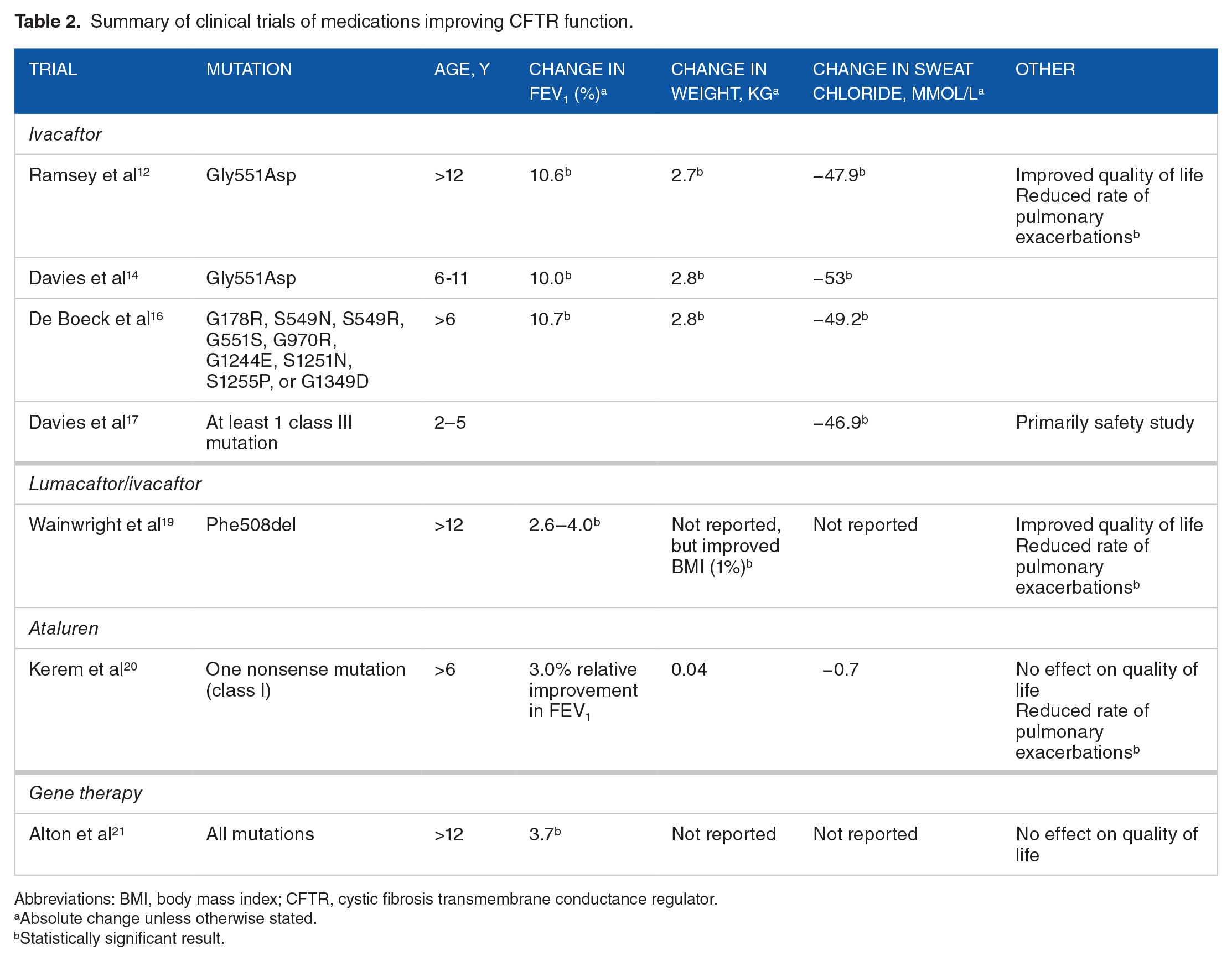
Abbreviations: BMI, body mass index; CFTR, cystic fibrosis transmembrane conductance regulator.
Absolute change unless otherwise stated.
Statistically significant result.
Ivacaftor
Ivacaftor was identified via high-throughput screening, a process where more than 200 000 compounds were tested for their suitability as potentiators. 9 Ivacaftor was demonstrated to improve the time the CFTR channel remained open and increased cell surface fluid in an in vitro trial of respiratory epithelial cells affected by the Gly551Asp mutation (the most common class III mutation). 9 It was estimated to improve chloride secretion by Gly551Asp CFTR to 50% of normal values. In a phase 2 trial of 39 adults, who had at least 1 Gly551Asp mutation, ivacaftor was shown to improve CFTR function (measured by nasal potential difference and sweat chloride levels) and lung function. 11 These findings were confirmed in a phase 3 randomised control trial of 167 patients 12 years or older who received ivacaftor or placebo. 12 At baseline, the mean age was 25.5 years with a mean forced expiration volume in the first second of aspiration (FEV1) of 63.6%. The trial showed that after 24 weeks of treatment, the change in FEV1 predicted from baseline was 10.6% (P < .001) greater in the treatment group, although it is unclear why change in FEV1 at 24 weeks was the primary end point when the trial lasted 48 weeks. Those in the treatment group gained more weight (2.7 kg, P < .001), had a lower sweat chloride (−47.9 mmol/L, P < .001), were less likely to have a pulmonary exacerbation (55% reduction in risk of exacerbation; hazard ratio: 0.455; P < .001), and reported a better quality of life (CF questionnaire–revised respiratory domain). The reduction in sweat chloride to the normal range suggests that ivacaftor is acting on CFTR and that sweat chloride is a useful biomarker of CFTR function. 13 The next phase 3 randomised controlled trial of ivacaftor was conducted in patients aged 6 to 11 years, and over 48 weeks, it was shown to improve lung function as measured by FEV1 (10.0%, P < .001) and weight (2.8 kg, P < .001) and reduce sweat chloride (−53 mmol/L, P < .001). 14
Using US CF registry data, these benefits in lung function and nutritional status have been shown to be maintained over a 3-year period. 15 Based on this evidence, ivacaftor (marketed as Kalydeco; Vertex Pharmaceuticals, Boston, MA, USA) has been approved for use in many countries, including the United States, European Union (although individual countries within the union decide whether to commission it or not), Canada, Australia, and New Zealand. 6
Ivacaftor has also been trialled for other gating (class III) mutations. In a double-blind crossover trial involving 39 patients with at least 1 non-Gly551Asp gating mutation (G178R, S549N, S549R, G551S, G970R, G1244E, S1251N, S1255P, or G1349D), ivacaftor was shown to improve FEV1, body mass index (BMI), sweat chloride, and quality of life. 16 As a result, it has been approved for use in patients with these mutations in Europe, United States, Canada, and Australia. 6
More recently, in the KIWI trial, ivacaftor has been trialled in the 2- to 5-year-old age group. The safety, pharmacokinetics, and pharmacodynamics of ivacaftor were assessed in this age group in an open-label trial lasting 24 weeks, involving 43 patients who had 1 class III mutation. 17 The study showed that the only adverse effect was deranged liver function tests and that pharmacokinetic and pharmacodynamic characteristics were similar to adult patients. Although this study was not powered to assess efficacy of treatment, ivacaftor did improve sweat chloride (−46.9 mmol/L). Based on this trial, ivacaftor has been approved for use in the 2- to 5-year-old age group in the United States, Europe, and Canada. 6
The KONDUCT trial examined the effect of ivacaftor in patients with at least 1 Arg117His CFTR mutation, which causes impaired channel conductance and gating (classes IV and III). 18 In this trial, ivacaftor did not significantly improve FEV1, although it did show benefits of reduced sweat chloride and improved quality of life. A subgroup analysis showed an improvement in lung function in patients aged 18 years and older who had more significant disease at enrolment, but in 6- to 11-year-olds, FEV1 appeared to be worse in the ivacaftor group. Intron 8 polythymidine length, which modifies CF disease severity in Arg117His mutation, 22 did not affect response to ivacaftor. Currently, ivacaftor is approved for use in the United States in children and adult patients, but only for those aged more than 18 years in Europe and Canada. 6
In a phase 2 randomised placebo-controlled trial of patients homozygous for Phe508del, ivacaftor did not improve lung function, sweat chloride, quality of life, or weight. 23 These results showed that although the Phe508del mutation is associated with a gating defect, to have a clinically significant improvement, the primary CFTR misfolding issue must be addressed.
The mechanism by which ivacaftor improves CFTR gating function is still unclear. In vitro studies demonstrate improvement in CFTR function as measured by chloride transport, although how it does this is unknown. 9 In vivo studies have shown improved mucociliary clearance (as measured by scintigraphy), although there have been no direct studies on the effect of ivacaftor on airway surface liquid. 24 In vivo studies also show improved gastrointestinal pH offering an explanation for improved weight gain. 24
To date, no significant adverse effects of ivacaftor have been identified, although surveillance is ongoing. 6 Some concerns have been raised about elevated liver transaminases and cataract development, and so the monitoring of liver function and regular ophthalmological examination is recommended. 6 A potential for iatrogenic harm is drug-drug interactions. Ivacaftor is a CYP3A substrate, so dose reduction is recommended if a strong CYP3A inhibitor (ie, clarithromycin, azole antifungals) is being used. 25
Lumacaftor
Lumacaftor was the first corrector to undergo extensive trials. During in vitro trials involving human bronchial epithelium (HBE) derived from a patient homozygous for Phe508del (Phe508del HBE), lumacaftor was shown to improve CFTR processing in the endoplasmic reticulum. 8 Lumacaftor was also shown to improve CFTR function as measured by patch clamp recordings showing rescued Phe508del-CFTR chloride current. 8 Lumacaftor’s effect was improved when it was used in conjunction with ivacaftor, an expected result as Phe508del CFTR is also gating mutation. However, the improvements in chloride transport seen with lumacaftor/ivacaftor (25% of normal) were modest when compared with those seen with ivacaftor in Gly551Asp (50% of normal).8,9
In a phase 2 trial of patients aged 18 years or older, who were either Phe508del homozygote (160 of 188) or Phe508del compound heterozygote (28/188), lumacaftor monotherapy had no effect on sweat chloride; however, when combined with ivacaftor, it reduced sweat chloride (−9.1 mmol/L). 26 This reduction was modest when compared with those seen in studies of ivacaftor in patients with the Gly551Asp mutation (−47.0 mmol/L). 12
The efficacy of lumacaftor/ivacaftor combination therapy was further assessed in 2 multinational multicentre phase 3 trials. The results of these 2 trials were presented in one paper. 19 Together, the studies involved 1108 patients homozygous for Phe508del mutation, aged 12 years or older with an FEV1 between 40% and 90% of normal predicted values. Participants received 24 weeks of placebo, lumacaftor 600 mg daily/ivacaftor 250 mg twice daily, or lumacaftor 400 mg twice daily/ivacaftor 250 mg twice daily. 19 Patients needed a higher dose of ivacaftor than those receiving ivacaftor monotherapy (150 mg twice daily) as lumacaftor induces hepatic metabolism of ivacaftor.27,28 The study showed that those receiving ivacaftor/lumacaftor had an improvement in absolute percent of predicted FEV1 of 2.6% to 4.0% and fewer pulmonary exacerbations. There was also a slight (1%) increase in BMI. Sweat chloride was not reported. Patients in the ivacaftor/lumacaftor group had a higher rate of study withdrawal due to adverse effects, mainly due to bronchospasm, dyspnoea, and liver enzyme elevation. The improvement in FEV1 was modest when compared with other CF treatments, such as inhaled DNAase and nebulised antibiotics. 29 The potential explanation for the modest effect of lumacaftor/ivacaftor is pharmacologic interactions, with in vitro evidence showing that ivacaftor impaired correction of Phe508del CFTR and in vivo evidence showing that lumacaftor induced hepatic metabolism of ivacaftor.27,28 Lumacaftor/ivacaftor combination therapy is marketed as Orkambi (Vertex Pharmaceuticals), and given its modest effects, some countries have funded its use (United States, Europe), whereas others have not (Australia). 6 Other potentially more effective compounds are already being evaluated, some of which are in phase 3 testing. 6
Ataluren
Ataluren promotes ribosomal read-through of premature stop codons, which are the cause of class I mutations (found in 10% of patients with CF). 20 It has also been trialled in Duchenne muscular dystrophy, another disease caused by a premature stop codon. 6 In a placebo control trial, lasting 48 weeks and involving 238 patients aged 6 years or older, with at least 1 class I mutation, ataluren did not improve lung function (−2.5% relative change in FEV1 percent predicted for ataluren vs −5.5% for placebo; P = .12) or decrease the rate of pulmonary exacerbations (1.42 exacerbations for ataluren vs 1.78 exacerbations for placebo; P = .77). 20 It is important to note that the authors reported relative change in FEV1 as opposed to absolute change which was used in trials of ivacaftor and lumacaftor/ivacaftor. In a subgroup of patients not receiving inhaled tobramycin, there was a significant improvement in mean change in FEV1 (−0.7% relative change in FEV1 percent predicted for ataluren vs −6.4% for placebo; P = .008) and pulmonary exacerbations (1.42 exacerbations for ataluren vs 2.18 exacerbations for placebo; P = .006). Aminoglycosides have been known for some time to promote ribosomal read-through, although not effectively, at clinically tolerable doses. The authors suggested that tobramycin is likely to affect ataluren’s effect as they compete for ribosomal binding sites. Given the improvements seen in the subgroup not receiving inhaled tobramycin, a phase 3 study which excludes those receiving inhaled tobramycin is underway (ACT CF TRIAL, NCT02139306). Of note, patients receiving ataluren were found to have elevated creatinine raising the possibility of renal toxicity. 20
Gene therapy
Another strategy for restoring CFTR function is gene therapy. Artificial CFTR genes can be delivered to the lungs either via inhaled therapy or by stem cell transplantation. To date, clinical research has focused on inhaled therapy. 30 A benefit of gene therapy over previously described treatments is that it would help all patients, regardless of CFTR genotype. A limitation of inhaled therapy is that it would not affect the nonpulmonary CF complications. The artificial gene can be transported to the airway epithelium with either a viral or liposomal vector. 30 Initial trials showed that use of a viral vector triggered an inflammatory response which negated any benefit of the gene therapy. 30 A more recent phase 2 trial involving 116 adult patients used a liposomal vector. 21 In this trial, patients were randomised to receive nebulised artificial gene with a liposomal vector or placebo, monthly for 12 months. In a subgroup of patients, success of gene therapy delivery was assessed by measurement of vector-specific DNA and messenger RNA (mRNA) either at bronchoscopy or during nasal studies. A subgroup of patients had either bronchoscopy or nasal studies performed to assess whether the gene therapy was successful from a mechanistic point of view, with both vector-specific DNA and mRNA being assessed. Most of the patients receiving active treatment had detectable vector-specific DNA (bronchoscopy group, 12 of 14; nasal studies, 17 of 17); however, no patients had detectable vector-specific mRNA. The authors felt that the absence of detectable mRNA was likely due to poor assay performance as opposed to a true absence of mRNA. The study showed a modest benefit in the treatment group with a 3.7% relative improvement in FEV1 which was not deemed to be significant. Currently, gene therapy is not being used in routine clinical practice, but there is ongoing research being done in this area.
Where Are We Now?
To date, the only widely used treatment which restores CFTR function is ivacaftor, which helps 4% to 14% of patients with CF with a class III mutation. 31 Combination lumacaftor/ivacaftor treatment is approved for Phe508del homozygous patients in the United States, but not other parts of the world, and the evidence of its benefit is modest at best. 6 However, there are multiple trials underway assessing the efficacy of other medications that target the Phe508del mutation.
Initial trials of ataluren and inhaled gene therapy have not shown them to be substantially beneficial, but there is ongoing research into their use.
Future of Treatments Improving CFTR Function
Although treatments aimed at correcting CFTR function are an exciting development, they raise many questions for both researchers and clinicians involved in CF care when looking to the future.
Existing Treatments Delivered at an Earlier Age
To date, most trials have been conducted in patients aged more than 6 years; however, it is well established that 50% to 70% of patients with CF have irreversible structural lung disease by this point. 32 Treatments restoring CFTR function are unlikely to repair these changes, and hence, the aim should be to give patients these treatments as early as possible, with the ultimate goal being prenatal treatment. 31 Trials of ivacaftor have been completed in the 2- to 5-year-old age group with plans to start trials in newborn patients as well.
One consideration when conducting trials in the less than 6-year-old age group is what outcome measurement will be used. Patients under 6 years cannot reliably perform spirometry, and hence, using FEV1 as a primary outcome is not possible. Potential solutions would include using computed tomographic (CT) scans to assess the evolution of structural lung disease, infant lung function testing (ie, multiple breath washout test) and evaluation of inflammation in the lung (ie, neutrophil elastase measured on bronchial lavage). This combination is already being used to evaluate the evolution of early CF disease by the Australian Respiratory Early Surveillance Team for CF (AREST CF) cohort. 32 There are limitations with the accessibility to these tools, as well as iatrogenic risk with radiation from CT scans and risks of anaesthesia with CT scans and bronchial lavage.
Personalised Medicine
As more treatments emerge, it is possible that there will be more than 1 treatment which can benefit a patient; for example, there could be multiple drugs which correct Phe508del. In this situation, it is unknown how the most beneficial drug for an individual patient could be selected. In addition, it is unclear whether the current approach of standardised dosing for all patients is optimal. There is already evidence of pharmacologic interactions, with medicines commonly used in CF care affecting dosing of ivacaftor. 33 Clinicians will need to develop robust methods for choosing the most beneficial medicine, and optimal dosing if a truly personalised approach to treatments restoring CFTR function is going to be used. One method could be the use of patient-derived organelles, which have been used to assess the effect of combination therapy 34 and have identified patients who may benefit from existing correctors and potentiators even though their CFTR genotype would not predict benefit. 35
Effect on Traditional CF Management
Another unknown is the effect of these novel medicines on traditional CF management. Outcomes in CF have improved significantly due to the significant regimen of treatments patients undertake. 4 Currently, adult patients spend on average 2 hours a day on CF care, and ideal treatments with correct CFTR function would reduce this. 5 There are case reports in which ivacaftor has improved the treatment burden in some children who have regained exocrine pancreatic function after starting treatment and have been able to cease pancreatic enzyme replacement. 36 However, in adults who have irreversible complications, such as bronchiectasis, chronic infection, and CF-related diabetes, the treatment burden has not been reduced. As these treatments are used for younger patients, without established complications, clinicians will have to decide if and when they cease to recommend treatments aimed at preventing complications that are currently considered routine in CF care. It will be particularly difficult to make this decision for treatments where there is no objective indication such as mucolytic therapy and airway clearance. It will be important to maintain a balance between not exposing patients to risk by withdrawing treatments too quickly while reducing treatment burden where possible.
Effect on Newborn and Carrier Screening
These treatments will have an effect on both genetic counselling and newborn screening. In genetic counselling, which can take place during pregnancy planning, during the pregnancy, or after a baby with CF has been born, the information provided to families will have to incorporate up-to-date information related to the mutations involved as they will affect patient’s outcomes. 31 In addition, it will be hard to accurately inform families about the progress being made in this area, without being able to give definite information about what this would mean for their child.
Regarding newborn screening, we should ensure that screening paradigms include mutations responsive to CFTR restorative treatments so that targeted therapy can be initiated as early as possible.
Cost
In addition to newborn screening, another important consideration for health funders will be the significant cost of these novel treatments. For example, ivacaftor costs approximately £182 000 per patient per year, and for treatments with a more modest benefit, such as ivacaftor/lumacaftor, concerns have been raised as to whether the benefits justify the significant cost. 29 Although use of these medications has the potential to reduce spending in other areas, such as hospital admissions, each medicine will have to be assessed individually. A cost-benefit analysis of ivacaftor undertaken in the United Kingdom estimated that for each quality-adjusted life year gained, the cost would be £284 000 to £1 850 000. 37 The authors also estimated the total additional lifetime cost of £1.6 to £1.8 million per patient treated with ivacaftor, as compared with a lifetime cost of treatment without ivacaftor of £400 000. The analysis accounted for the significant reduction in cost of ivacaftor once its patent expires (estimated to be in 2028), with an estimated cost of £20 000 per patient per year. It is important to note that this cost-benefit analysis was undertaken in the United Kingdom and hence may not be applicable to other countries with differing models of health care. In addition, the accuracy of any cost-benefit analysis is limited, given the unknown long-term effect of ivacaftor on current treatments and hence health care costs.
Rigorous cost-benefit analysis will be even more important for any treatments either utilising gene therapy or targeting Phe508del as they will be used by most of the patients with CF as opposed to ivacaftor which is used by approximately 10% of patients with CF. Given the significant cost of these treatments, health care providers may try to identify subgroups of patients who will benefit most from treatments and only fund use of the medications in such patients. There will also be the potential for the development of a divide in care delivered to patients with CF, with one group of patients having access to CFTR restorative treatments and another being denied such treatment due to the cost.
Implications for Development of Other CF Treatments/New Products
These new treatments will undoubtedly affect future research in CF. When new treatments which correct CFTR function are trialled, it will be important that similar outcome measures are used to allow for comparison of effectiveness. As these treatments become part of standard care, they will need to be included in the placebo group in all trials of novel CF therapy. Another consideration, which has already been demonstrated in some trials, is there is likely to be a heterogeneous response to medicines restoring CFTR function. It will be important for researchers to determine what factors lead to a varied response to identify subgroups who may benefit from treatments not shown to benefit a wider population. Potential explanations of a varied response to treatment include extent of existing disease, interactions with other treatments, environmental factors, and effect of CF gene modifiers. 38
Conclusions
Treatments improving CFTR function are an exciting development in CF; some have been referred to as the ‘holy grail’. 29 Many people have worked to ‘cure’ CF and it is unclear what exactly would constitute a cure. For example, some argue that trials should show that they improve lung function; however, as CF causes progressive decline in lung function, others argue that the aim should be to arrest decline and that stabilising lung function should be viewed as a significantly beneficial outcome. In general, a ‘cure’ would lead to patients with CF having normal lung structure and function. Given that the only CFTR restorative treatment, ivacaftor, is currently commenced at an age where patients already have structural lung damage, it is clear we are some way from a cure. There are multiple new treatments being developed, so hopefully all patients with CF will have access to CFTR restorative treatments eventually.
Footnotes
Peer review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totalled 980 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Both authors contributed to the conception, drafting and final review of the manuscript.
