Abstract
Objectives
Brain injury resulting from an ischemic stroke affects cognitive performance by disrupting the hippocampus. Several processes are involved in brain injury progression, including inflammation, glutamate excitotoxicity, and modulated brain peptide systems such as the melanocortin system. Reports show that quercetin exerts neuroprotective activity. This study investigates quercetin’s role in the cognitive function of ischemic stroke-induced mice and the possible mechanisms involved.
Materials and Methods
ICR mice were used. The left unilateral common carotid artery occlusion was conducted for 4 h to induce an ischemic stroke in the mice. Quercetin 50, 100, and 200 mg/kg were administered to separate groups intraperitoneally for 7 days. Cognitive function was examined using the T-maze test. The hippocampal mRNA expressions of NR2A, NR2B, melanocortin 4 receptor (MC4R), pro-opiomelanocortin precursors (POMC), and nuclear factor 2 (Nrf2) were examined using reverse transcription-polymerase chain reaction.
Results
It was found that stroke disrupted cognitive function. Quercetin administration ameliorated cognitive impairment. Quercetin attenuated the stroke-induced decrease in MC4R mRNA expression. Moreover, quercetin suppressed the stroke-induced increase in the hippocampal mRNA expression of NR2A.
Conclusion
Quercetin ameliorates cognitive deficits and normalizes impaired hippocampal melanocortin and glutamatergic signaling in ischemic stroke-induced mice.
Introduction
Stroke is the world’s second-most dominant cause of mortality and one of the top three causes of disability and mortality. There are two types of stroke: hemorrhagic and ischemic. A recent study reveals that there are more than 12 million stroke events and more than 6 million stroke deaths each year. An ischemic stroke causes approximately 60% of stroke cases. Ischemic strokes are caused by a sudden blockage of blood flow to the brain. 1 The brain is an organ that is susceptible to oxidative damage because it requires large amounts of oxygen, high levels of unsaturated fats, and deficiencies in anti-oxidative defense mechanisms. 2 A decreased blood flow caused by an ischemic stroke leads to hypoxia. This condition causes cell death in various brain areas, especially in the hippocampus. 3
The hippocampus is a brain area that plays an important role in learning and memory. The presence of damage, lesions, and inhibition in the hippocampus decreases the ability to process information into memory traces. Memory impairment is one of the conditions that can occur after a stroke, meaning that a person cannot learn new information or recall previously obtained information. 4 In a stroke, synaptic changes occur, which impair the ability to process memories. 5
At the molecular level, stroke conditions cause glutamate toxicity in neurons. This condition happens because of the failure of the electron transport process through the Na+/K+ pump, causing depolarization and resulting in efflux K+ and Ca2+ influx. High amounts of Ca2+ in cells trigger the over-release of the neurotransmitter glutamate in the synaptic gap that activates N-methyl-D-aspartate (NMDA) receptors. This condition causes oxidative stress and cell damage, leading to apoptosis and necrosis. The NMDA receptor consists of NR1 and NR2 subunits. 6 It has been reported that the NR2 subunit plays an important role in synaptic plasticity and memory functions. 7 NR2A- and NR2B-containing receptors are located in synaptic and extrasynaptic membranes in the hippocampus. 8 Overexpression of the NR2 subunit has been associated with cognitive impairment in patients with neurologic deficit conditions.9, 10
The melanocortin system is reportedly involved in nerve protection. The system involves several peptides, such as α-melanocyte-stimulating hormone (α-MSH), which is the cleavage product of the precursor protein pro-opiomelanocortin precursors (POMC). The melanocortin 4 receptor (MC4R) is a subtype of melanocortin receptor predominantly expressed in the central nervous system in the basal area of the ganglia, cerebral cortex, and hippocampus. The binding of α-MSH to MC4R exhibits protective, anti-inflammatory, and anti-apoptotic effects in brain ischemia. Furthermore, increasing MC4R mRNA expression has been associated with improved neuronal recovery.11, 12 Stroke causes oxidative stress, which leads to apoptosis. The nuclear factor 2 (Nrf2)-associated erythroid 2 pathway plays a critical role in decreasing the effects of oxidative stress. Nrf2 is a sensor and regulator of oxidative stress. 13 Nrf2 becomes a crucial defense regulator against endogenous stresses by coordinating basal activity and stress to trigger numerous cytoprotective genes. 14 Nrf2 has been known to have an important role in brain injury.15–17 Taken all together, NMDA, melanocortin, and Nrf2 may play a vital role in neuronal recovery after an ischemic stroke.
Quercetin (3,5,7,3’,4’-pentahydroxyflavone) is a flavonoid compound widely contained in natural ingredients such as cranberries, apples, cherries, onions, tea, and broccoli. Quercetin has been empirically used as a supplement or therapy due to its biological activities, including antibacterial, antidiabetic, anti-inflammatory, and antioxidant. Quercetin has neuroprotective effects by activating the Nrf2 pathway and attenuating neuroinflammation.18, 19 Furthermore, quercetin is known to improve memory in Alzheimer’s disease models. 20 However, there is a lack of evidence showing the effect of quercetin on the cognitive deficit in the ischemic stroke mice model. Thus, this study aimed to determine the role of quercetin in the cognitive function of ischemic stroke-induced mice. The involvement of several genes regulating hippocampal NMDA, melanocortin, and Nrf2 systems, such as NR2A, NR2B, MC4R, POMC, and Nrf2 mRNA expression in the hippocampus, was investigated in this study.
Materials and Methods
Animal and Experimental Design
The experiments were performed on ICR male mice (6–8 weeks old). Before the experiment, all mice were maintained in their housing for 1 week under standard laboratory conditions in terms of temperature (25°C ± 2°C), humidity (60% ± 10%), and 12 h of light/dark cycle, with free access to food and water. After the adaptation period, pre-training using the T-maze test was performed on all mice. Then, the mice were operated on to induce an ischemic stroke. Cognitive function tests were performed several times after stroke induction, and on day 14, the mice were sacrificed to obtain the hippocampus (Figure 1). Five mice per group were counted based on the previous study in producing mouse stroke models and by using the calculation for value “E”.12, 21 The sample size was increased to eight mice per group to anticipate the number of animals suffering from death after stroke induction. 22 Forty mice were divided into five groups: sham group (mice were operated without arterial blockade), stroke group, and stroke groups treated with quercetin at 50 mg/kg, 100 mg/kg, and 200 mg/kg, respectively. According to previous studies, quercetin has pharmacological effects in rodents at doses between 50 and 200 mg/kg.12, 23 Therefore, this study used three doses from the range of 50, 100, and 200 mg/kg to examine the dose-dependent effect of quercetin on stroke-induced cognitive impairment. The ethics committee of the Faculty of Veterinary Medicine, Airlangga University, has approved all experiment protocols, and ethical clearance decree No. 2.KE.060.06.2021 has been obtained.
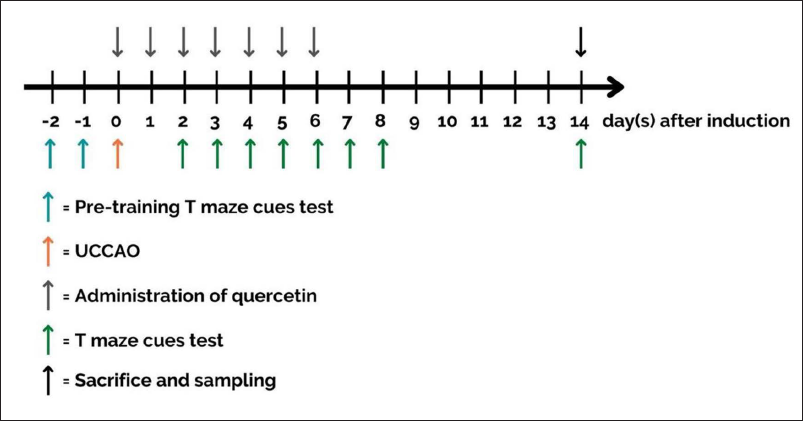
Mouse Model of left Unilateral Common Carotid Artery Occlusion (lUCCAO)
Mice were anesthetized with ketamine (80 mg/kg) and xylazine (10 mg/kg). The animals were turned supine and fixed to the surgical table using adhesive tape. The hair in the neck was shaved, and a small incision was made approximately 1–2 cm long in around 5 mm left side from the midline of the neck. The left common carotid artery was separated from connective tissue and then blocked by a bulldog clamp for 4 h. After 4 h, the bulldog clamp was removed.
Drug Treatment
Quercetin (Tokyo Chemical Industry, Tokyo, Japan) was dispersed in 5% Tween 80. The sham and stroke groups were given 5% Tween 80 (vehicle). The treatment groups were given 50, 100, and 200 mg/kg of quercetin, respectively. The vehicle and quercetin were injected intraperitoneally 30 min after reperfusion, followed by daily injection for 6 days.
T-Maze Test Using Rewarded Alternation
The T-maze test using rewarded alternation was performed according to Anggreini et al.’s methodology, with some modifications. 24 The mice were briefly placed in one arm of the T-maze and then allowed to move freely to explore either the right or left arm. Skimmed milk was placed on the right arm of the T-maze as a reward for the mice’s success in remembering the exercises during pre-training. Mice that chose the right arm received a score of one, while mice that chose the left arm received a score of zero. The mice were fasted for 5 h before performing the test. The T-maze apparatus was cleaned with a 10% acetic acid solution before each mouse was tested. Pre-training was performed before stroke induction with the T-maze using rewarded alternation for 2 min with five repetitions. Then, the T-maze using rewarded alternation was performed with five replications on days 2, 3, 4, 5, 6, 7, 8, and 14 after stroke induction (Figure 1).
Reverse Transcription Polymerase Chain Reaction
The reverse transcription-polymerase chain reaction (RT-PCR) was used to determine the relative expression of NR2A, NR2B, MC4R, POMC, and Nrf2 mRNA in the hippocampal area. The hippocampus was extracted from the mice’s brains, then frozen in liquid nitrogen and kept at −80°C in the refrigerator. The RNA of each sample was isolated using the Total RNA Purification Kit (Jena Bioscience, Germany). The results of RNA isolation were calculated using the QuantusTM Fluorometer. The GoScript™ Reverse Transcription System (Promega, USA) was used to synthesize cDNA. Specific primers (Table 1) and GoTag® Green Master Mix (Promega, USA) were used to perform PCR on a thermal cycler as follows: pre-denatured for 1 min at 95°C, denatured for 30 s at 95°C, annealed for 30 s at 55°C, and extended for 1 min at 72°C, for a total of 35 cycles. Electrophoresis on a 2% agarose gel was used to examine the PCR result. Agarose gel was analyzed using ImageJ software (National Institutes of Health, USA). The beta-actin expression measurement was done on the same cDNA samples used for each target gene measurement and in the same amplification steps. The same bands of beta-actin were used to normalize the measurement results of each target gene’s expression.
The Sequence of Primer Used for RT-PCR Analysis of the Hippocampus Sample.

Data Analysis
Data were analyzed using Graph-Pad Prism software version 9.0. A two-way analysis of variance (ANOVA) method presents T-maze test data on the average percentage of correct choice. The relative mRNA expression data were presented as the mean intensity value ± SEM using the one-way ANOVA test, followed by multiple comparisons between groups using Tukey’s post hoc test. The difference between groups is considered significant if p < 0.05.
Results
Effect of Quercetin on Cognitive Function of Ischemic Stroke-induced Mice
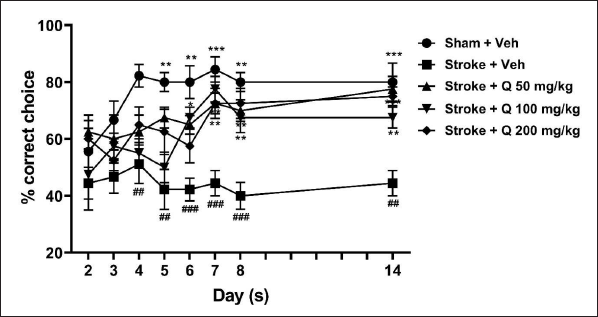
Ischemic stroke induction significantly decreased the percentage of correct choices made by mice compared to the sham group. In the T-maze experiment, the decrease in the percentage of correct choices remained low, around 40%, after 14 days. Quercetin 50, 100, and 200 mg/kg significantly ameliorated the stroke-induced decrease in the percentage of correct choices (Figure 2; two-way ANOVA; F(4,37) = 23.08; p < 0.001).
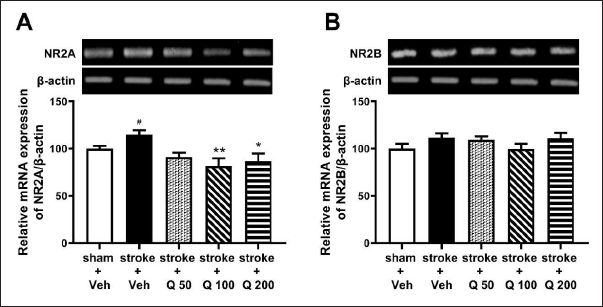
Effect of Quercetin on the Hippocampal mRNA Expression of NMDA Receptor Subunits in Ischemic Stroke-induced Mice
In the RT-PCR analysis, it was found that stroke induction notably increased hippocampal NR2A mRNA expression. The results showed that quercetin 100 and 200 mg/kg, but not 50 mg/kg, significantly decreased the NR2A mRNA expression in ischemic stroke-induced mice (Figure 3A; one-way ANOVA; F(4,25) = 4.571; p < 0.01). Meanwhile, ischemic stroke conditions and quercetin administration did not provide a significant change in the expression of NR2B mRNA (Figure 3B; one-way ANOVA; F(4,25) = 1.437; p > 0.05).
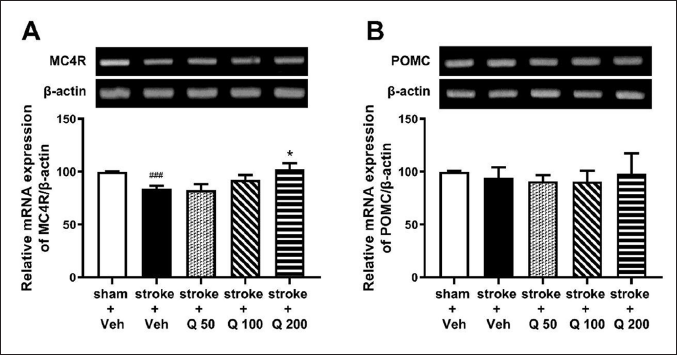
Effect of Quercetin on MC4R and POMC mRNA Expression in the Hippocampus of Ischemic Stroke-induced Mice
Figure 4A shows that an ischemic stroke lowered the hippocampal mRNA expression of MC4R. Quercetin administration dose-dependently attenuated the decrease in the mRNA expression of MC4R in the hippocampus of ischemic stroke-induced mice (Figure 4A; one-way ANOVA; F(4,25) = 4.317; p < 0.01). Meanwhile, hippocampal POMC mRNA expression did not change significantly after ischemic stroke induction or after quercetin administration (Figure 4B; one-way ANOVA; F(4,25) = 0.1426; p > 0.05) (Figure 4B).
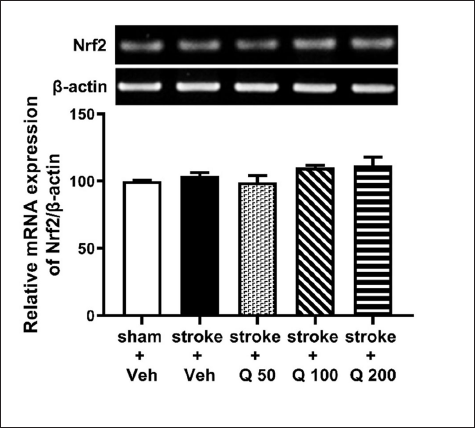
Effect of Quercetin on Nrf2 mRNA Expression in the Hippocampus of Ischemic Stroke-induced Mice
Figure 5 shows that the ischemic stroke condition did not significantly affect the expression of Nrf2 mRNA in the hippocampus. Furthermore, quercetin did not significantly change the Nrf2 mRNA expression in the hippocampus of stroke-induced mice (Figure 5; one-way ANOVA; F(4,25) = 2.118; p > 0.05).
Discussion
Ischemic stroke is defined as a sudden loss of blood flow to the brain, resulting in the loss of neurological function. Cell death is considered a major cause of neurological decline. Cell death mechanisms in cerebral ischemia include oxidative or nitrosative stress, excitotoxicity, ion imbalance, and apoptosis. 25 It is known that the stroke animal model is generally made through the focal occlusion in the middle carotid artery by a nylon suture protruding via the internal carotid artery. However, stroke induction using other techniques, such as UCCAO, was previously reported as having a successful effect on motor and cognitive impairment.26, 27 Previous research shows the adequate features of lesions induced by the UCCAO model. Furthermore, the model has been confirmed to cause neuronal damage in the striatum and hippocampus.28, 29
The T-maze paradigm using rewarded alternation was used to assess cognitive performance on days 2–8 and day 14 following stroke induction. The percentage of correct choices achieved by the T-maze test using rewarded alternation illustrates the outcomes of cognitive performance in mice. On days 4–14, there were notable differences in cognitive function between the sham and stroke groups. The stroke group showed a lower percentage of correct choices than those in the sham group, indicating a stroke-induced cognitive deficit. A previous study demonstrated that cognitive function measurements using the T-maze in healthy groups would reach a correct choice above 80%. In comparison, the group with lesions in the hippocampus is generally unable to achieve the correct choice above 60%. 30 Thus, the present study demonstrates a reliable model of ischemic stroke-induced cognitive impairment.
The positive control group is usually added to compare the outcomes of the test substance and clinically approved drugs. However, there is no currently approved drug for ischemic stroke that specifically works as a neuroprotectant as well as improves cognitive deficits during stroke. Therefore, the present study focuses on the attenuation effect of quercetin compared to the untreated/vehicle-treated group. The present study showed that administration of quercetin at 50, 100, and 200 mg/kg led to considerable improvements in the cognitive function of stroke-induced mice starting from day 6 compared to the untreated stroke group. Interestingly, this protective effect was persistently maintained on days 7–14, when quercetin administration had been discontinued since day 6. This finding supports previous research reporting that quercetin has a 45% protective effect against ischemia-induced brain infarction in mice. 31 These results indicate the importance of quercetin treatment in the early phase after ischemic stroke occurrence to provide a significant effect on cognitive function.
Research into NMDA receptor subunits as a target for stroke therapy has excellent implementation prospects, seeing that glutamate excitotoxicity mediated by NMDA receptors plays a crucial role in many aspects of brain damage, especially in ischemic conditions. 6 Therefore, the present study examined the NMDA receptor subunits 14 days after a stroke. The current study confirms that NR2A mRNA expression increases in the hippocampus of stroke-induced mice. Meanwhile, NR2B mRNA expression in the hippocampus did not significantly change in stroke conditions. A study by Heurteaux et al. shows that forebrain ischemic conditions in experimental animals cause fluctuations in NR2A and NR2B mRNA expression. NR2A and NR2B mRNA expression decreased over the first 24 h after an ischemic stroke, then increased for the next 48 h. 32 A previous study also demonstrated that the cells had an apoptotic pattern aligned with NR2A and NR2B mRNA expression in the hippocampus. 33 The present study demonstrates an increase in hippocampal NR2A mRNA expression, which indicates a high rate of cell apoptosis in the hippocampus of stroke mice. This result is associated with ischemic conditions resulting in excess glutamate release in the synapse gap. The release of glutamate activates the post-synapse ionotropic NMDA receptors that bind to glutamate. This binding causes massive entry of Ca2+ into cells, leading to neuronal damage.34, 35 In contrast to stroke mice, the current study found that a seven-day quercetin treatment reduced the expression of NR2A mRNA in the hippocampus. However, a significant change did not occur with NR2B mRNA expression in the hippocampus. These results suggest the ability of quercetin to ameliorate NMDA subunit receptor overactivity caused by glutamate imbalance in stroke conditions. These results are in line with previous studies showing evidence that quercetin reduces glutamate-induced Ca2+ influx so that mitochondria do not release apoptotic signals that prevent neuronal cell death.36, 37
It is well known that melanocortin activity is involved in the neuroinflammatory process in the brain. The present study found that MC4R mRNA expression in the hippocampus of the stroke mice decreased compared to the sham group. On the other hand, Pomc mRNA expression in the stroke group did not significantly change compared to the sham group. These results are consistent with previous studies that showed MC4R mRNA expression decreases in the injured brain area in hypoxic-ischemic conditions. 12 As previously described, hippocampal POMC/MC4R circuit stimulation is important for rescuing synaptic impairment that causes cognitive deficits. 38 In line with the behavioral performance of stroke mice, this finding may suggest that disruption of the hippocampal POMC/MC4R circuit is involved in causing cognitive deficits in ischemic stroke-induced mice.
The stroke group given quercetin 200 mg/kg, but not those given 50 or 100 mg/kg, showed a significant increase in MC4R mRNA expression in the hippocampus compared to the stroke mice. Meanwhile, quercetin did not change Pomc mRNA expression. This result suggests that quercetin’s effect on MC4R may involve repairing hippocampal tissue damage and cognitive deficits due to an ischemic stroke by encouraging the activation of the melanocortin system through increased hippocampal MC4R mRNA expression. The melanocortin system plays a crucial role in brain damage prevention. Activation of the postsynaptic MC4R increases the number of mature dendritic spines in the hippocampus, which improves synaptic plasticity. 39 Activation of the MC4R promotes microglia/macrophage activation and neutrophil infiltration in stroke patients, as well as downregulation of ionized calcium binding adaptor molecule 1, tumor necrosis factor-α, and interleukin-1β expression. 11 Furthermore, MC4R activation has been associated with neuron protection in the hippocampus after an ischemic attack by decreasing inflammatory and apoptotic responses, promoting antioxidant defense, and reducing mitochondrial damage.40, 41
Excessive production of free radicals causes oxidative damage that leads to chronic diseases such as cancer, ischemia, atherosclerosis, Alzheimer’s, diabetes, and liver fibrosis. Nrf2 is one of the most studied biomarkers because of its essential role in providing neuroprotective effects.13, 42 The current study showed that Nrf2 was normalized in the hippocampus of mice 14 days after ischemic stroke induction. In addition, treatment with quercetin did not significantly change the expression of Nrf2 mRNA in the hippocampus. Besides its effect on Nrf2 expression, the role of quercetin in providing a protective effect in ischemic stroke conditions may be related to its ability to increase Nrf2 translocation from the cytoplasm to the nucleus. 43 Quercetin is known to disrupt the cytoplasmic interaction of kelch-like ECH-associated protein 1 and Nrf2, causing Nrf2 to be pushed to the nucleus and activate cytoprotective genes. The activation of cytoprotective genes could reduce the impact of oxidative stress by maintaining mitochondrial homeostasis. The postulated effect of oxidative stress is the regulation of mitochondrial number and function, which leads to the inhibition of degenerative brain processes that slow that cognitive deficit. 44 After ischemia, oxidative stress and neuroinflammation activities cause brain damage, particularly in the hippocampus. Hence, using quercetin as an antioxidant is still rational for preventing brain injury, especially in the early phase after a stroke occurs, as it causes a rapid increase in the concentration of oxidants in the brain.45, 46
Conclusion
Quercetin, a potent antioxidant, could be useful for brain injury prevention, particularly in the early stages of stroke. The findings indicated that quercetin improves cognitive function in ischemic stroke-induced mice. It is suggested that the mechanism of cognitive attenuation involves the increase in hippocampal MC4R mRNA expression and the decrease in hippocampal NR2A mRNA expression.
Footnotes
Acknowledgments
The authors would like to thank the Ministry of Education, Culture, Research, and Technology of Indonesia for the PDUPT Grant 2021 for Dr. Chrismawan Ardianto. Grant No: 4/E1/KP.PTNBH/2021; 387/UN3.15/PT/2021.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Ministry of Education, Culture, Research, and Technology of Indonesia for the PDUPT Grant 2021 for Dr Chrismawan Ardianto. Grant No: 4/E1/KP.PTNBH/2021; 387/UN3.15/PT/2021.
Ethical Approval
The ethics committee of the Faculty of Veterinary Medicine, Universitas Airlangga has approved all experiment protocols and ethical clearance decree No. 2.KE.060.06.2021 has been obtained.
