Abstract
Recent research has shown a strong correlation between gut dysbiosis and Alzheimer’s disease (AD). The purpose of this review is to investigate the relationship between gut dysbiosis, immune system activation, and the onset of AD and to examine current breakthroughs in microbiota-targeted AD therapeutics. A review of scientific literature was conducted to assess the correlation between gut dysbiosis and AD and the various factors associated. Gut dysbiosis produces an increase in harmful substances, such as bacterial amyloids, which makes the gut barrier and blood-brain barrier more permeable. This leads to the stimulation of immunological responses and an increase in cytokines such as interleukin-1β (IL-1β). As a result, gut dysbiosis accelerates the progression of AD.
The review highlights the connection between gut dysbiosis and AD and the potential for microbiota-targeted therapies in AD treatment.
Pictorial Abstract
Introduction
Multiple strains of bacteria, lower eukaryotes, archaea, and viruses live in gut and are linked to human health and disease. 1 Microbes in the gut live in communities and interact with one another through exchanging genetic and molecular material. 2 In a healthy body, these microorganisms maintain the pH of the stomach, as a result, they provide a protective barrier against infections. 3 Probiotics are bacteria that have a positive impact on the health. These beneficial bacteria aid in the absorption of nutrients and the prevention of inflammation. Microbiota can aid in the breakdown of specific nutrients, which are then digested by host cells, some of these nutrients have been connected to brain function. 4 As a result, gut flora produces amino acids (tryptophan and gamma-aminobutyric acid [GABA]) as well as monoamines (dopamine, serotonin, and histamine), which act as neurotransmitters or precursors in the brain 5 (Figure 1). These neuroactive chemicals can influence the enteric nervous system (ENS) as well as the central nervous system (CNS). The microbiota also regulates immunological and metabolic activities, as well as providing nutrition and energy to the host via vitamin production. Through the gut–brain axis (GBA), the immune system communicates with the CNS via cytokines, neurotransmitters, hormones, neuropeptides, and growth factors. According to scientific evidence, the gut microbiota can affect circulating cytokine levels, which can affect a range of brain processes. 6
The Effect of Gut Microbiota’s Metabolites on CNS.
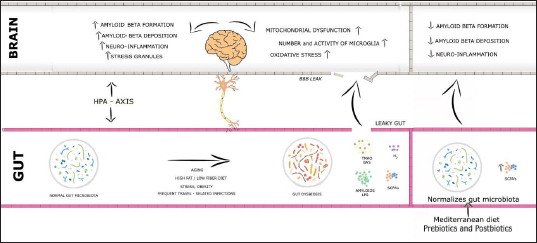
The human foetal intestine is free of microorganisms. The colonization of microorganisms and their diversity occurs as the age progresses. Dietary changes, antibiotic exposure, and infections lead to dysbiosis which result in obesity, type-2 diabetes, allergies, metabolic syndrome, inflammatory bowel disease, and neurological disease. The gut barrier consists of the intestinal mucosa, gut bacteria, mucosal immune cells, and other epithelial-derived compounds. Leaky gut syndrome (LGS) occurs as a result of dysbiosis. Dysbiosis causes the release of hazardous substances such as bacterial amyloids, lipopolysaccharide (LPS), trimethylamine N-oxide (TMAO) and bacterial bile acids (BA), which leads to LGS characterized by more permeable gut barrier and blood–brain barrier (BBB). Resulting in systemic and neuroinflammation as well as cerebellar and hippocampal dysfunction. Importantly, individuals with CNS problems had increased intestinal permeability, which resulted in immunological activation due to hazardous compounds leaking from the gut into the circulation. While dysbiosis has been shown to influence the course and symptoms of various neurological diseases, there is emerging evidence that medicines to restore gut barrier integrity and microbiota can improve the clinical course and symptoms of various neurological diorders. 7
Methodology
A search was conducted from 2002 to 2022 using PubMed and Google Scholar to gather information on the relationship between gut dysbiosis and Alzheimer’s amyloidopathy. “gut microbiota,” “gut–brain axis,” “Alzheimer’s disease,” “dysbiosis,” and “amyloidopathy” are some of the terms utilized. Research and review articles were both included, although only works published in English were taken into consideration (Figure 2).
Methodology.
Gut–Brain Axis
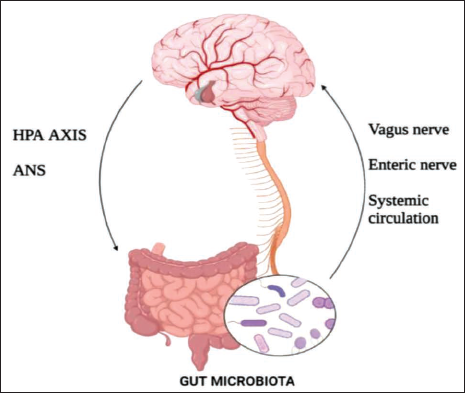
The GBA is a two-way communication system that links the CNS to the ENS (Figure 3). Afferent signals originating in the lumen and communicated to the CNS via enteric, spinal, and vagal pathways, as well as efferent signals from the CNS to the gut membrane, are controlled by the sympathetic and parasympathetic limbs of the autonomic system. 8 Efferent fibres, which make approximately 10–20% of all fibres, carry the message down from the brain to the gut through the vagal efferent. While the vagal afferents transmit signals up from the gut membrane to the brain via afferent fibres, which make up 80–90% of all fibres. The vagal afferent pathways play a part in the stimulation and control of the hypothalamus–pituitary–adrenal axis (HPA), which involves coordinating the organism’s defensive responses to a variety of stresses. 9 Elevated systemic proinflammatory cytokines stimulate the HPA axis, causing the hypothalamus to release corticotropin-releasing factor (CRF), which in turn triggers the pituitary gland to secrete adrenocorticotropic hormone (ACTH), resulting in the release of cortisol from the adrenal glands. Cortisol activates the peripheral immune response by activating the immune cells like macrophages. 10
This Diagram Shows GBA. It Involves the Autonomic Nervous System (ANS), Hypothalamic–Pituitary–Adrenal Axis (HPA), Enteric Nervous System (ENS) and Systemic Circulation.
In the GBA, the gut microbiota plays a key role. Changes in the bacterial makeup of the gut dysbiosis have an influence on brain function and vice versa. The interaction and activation of pattern recognition receptors (PRRs) by gut microbiota metabolites, notably Toll-like receptors (TLR) 2 and 4, has an impact on CNS and ENS development and function. 11 Gut barrier permeability is maintained by a healthy gut microbiota, which regulates commensal-related inflammatory responses and supports a balance between immunity and harmful effects. However, exposure to bacterial toxins, medications, or viral infections can alter the intestinal barrier function, leading in gut membrane translocation. When immune cells in the intestinal mucosa or the ENS are unintentionally exposed to bacteria, microbe-derived metabolic products are produced, which might provoke an immunological response. It includes the release of inflammatory cytokines and stimulation of the vagal nerve. According to studies, a tight BBB and gut integrity is linked to a healthy gut microbiota. Increased BBB and gut permeability are caused by dysbiosis. As a result of a disturbed gut barrier and BBB, microbe-derived metabolites or germs, antigens, or activated immune complexes leak from the gut to the systemic circulation and then to other tissues, including the brain parenchyma. An interdependent network of interaction loops involving the gut barrier, altered microbiota, the BBB, and the immune system can explain a wide range of neurodegenerative and neuropsychiatric disease pathologies. 12
Experiments in germ-free animals have led to several conclusions on the role of the gut microbiota in neural development. 13 The CNS and gut microbiota development happens together in the first three years of life, so there is a high chance of interaction between them. If the foetus’ gut is sterile, the new-born’s first interaction with the microbial will happen during birth. Bacterial exposure is essential for maintaining a healthy systemic immune system. 14 Vagal innervation is a critical bi-directional neuronal link that transmits sensory and motor innervations from the brain to the stomach. The quantity and density of potential efferent terminals are higher in the early post-natal period, although axons from the dorsal motor nucleus and nodose ganglion cell bodies migrate to intestine destinations throughout foetal development. Vagal nerves, which innervate numerous regions of the digestive system, are regulated by the gut microbiota. As a result, the gut microbiome influences the transitory impulses sent by vagal nerves to growing axons. 15 If the HPA axis appears to develop normally during childhood, the child will respond appropriately to pressures as a teenager and adult. As a result, the hyporesponsive phase of the HPA is maintained. The axis may become hyperactive if this period of development is disrupted, for example, by poor parental care or abuse. 13 It has the potential to induce brain neurodegeneration. According to recent research, the microbiota controls neurogenesis. Neuronal growth and survival are regulated by brain-derived neurotrophic factor (BDNF), a plasticity-related protein. Germfree mice had lower BDNF expression in the brain and amygdala than normal mice. 16 Adult mice treated with antibiotics, which deplete the microbiota, experience a significant reduction in neurogenesis. Antibiotics administered to new-borns can alter their microbiota, predisposing them to the development of inflammatory and metabolic illnesses. 17 This impact appears to be mitigated by exercise or the use of a probiotic cocktail. 18 These findings reveal a direct link between the microbiota and neuronal development. Hormones and neurotransmitters produced by microbes have influence on cognitive ability via vagus nerve. Gut dysbiosis causes the development of potentially toxic compounds, which affect gut and BBB permeability and cause cognitive impairment through neuronal inflammation. The prefrontal cortex, which governs cognitive function, is directly affected by microbes (Figure 4).
The Pathways Through which the Microbes Regulate Cognitive Functions.
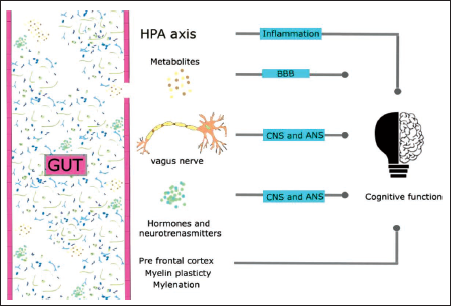
The Correlation Between Gut Dysbiosis and Onset of Alzheimer’s Disease
Alzheimer’s Disease (AD) is a progressive neurodegenerative disease marked by impaired cognition and a build-up of amyloid-beta peptides in the brain. According to a recent study, the gut microbiome composition of Alzheimer’s patients shows an increase in Escherichia/Shigella and a reduction in E. rectale strains. 19
Gut Dysbiosis
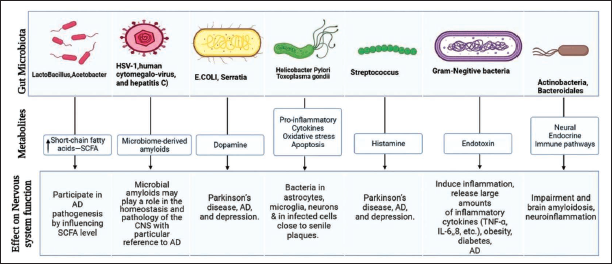
Gut dysbiosis results in an increase in harmful molecules (such bacterial amyloids, LPS, and TMAO) and a decrease in beneficial substances (like short-chain fatty acids [SCFAs] and H2), causing the gut membrane and BBB to become more permeable. 20 Bacterial amyloids, TMAO and LPS can trigger peripheral immune responses, resulting in elevated oxidative stress levels both centrally and peripherally 19 (Figure 5).
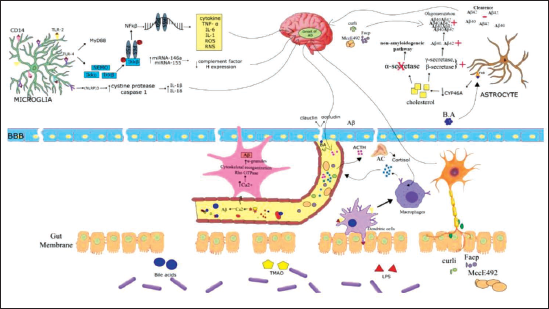
Bacterial LPS stimulates the NFκB signaling pathway by binding to microglial cell receptors (TLR2, TLR4, CD14) in the hippocampus area. 21 When NFκB is activated, the levels of proinflammatory miRNA-146a and miRNA-155 elevate, leading to complement factor H being downregulated, resulting in neurodegeneration and inflammatory signaling. 22 When LPS binds to TLR4, it activates the NEMO complex, causing subunit IκB to be phosphorylated and the p50–p65 NFB dimer to be released. After that, the NFκB dimer is translocated to the nucleus, where it binds to RNA response elements, causing the release of pro-inflammatory cytokines, chemokines, reactive oxygen, and nitrogen species. LPS activates the inflammasome via the microglial receptor protein (NLRP3) causing the cysteine protease caspase-1 to activate and the proinflammatory cytokines interleukin (IL)-1 and IL-18 to be produced. 23 This LPS and TLR cascade leads to the onset of AD. LPS also binds to TLR receptors on dendritic cells and macrophages, causing the release of proinflammatory cytokines. These cytokines enter the brain through the circulatory system or activate the HPA axis. When the HPA axis is stimulated by inflammatory cytokines or bacteria’s toxic compounds, corticotrophin-releasing factor is generated. This triggers the release of cortisol in the systemic circulation by stimulating the pituitary gland to produce ACTH, which acts on the adrenal cortex. Cortisol acts on macrophages to increase the release of cytokines. 24
TMAO promotes platelet hyperactivity by increasing the release of calcium ions from intracellular reserves, which activates Rho GTPase and cytoskeleton reorganization, resulting in Aβ production. Aβ produced in platelets enters the circulation and travels to the brain. In the brain, Aβ spreads from neuron to neuron in the same manner as prion molecules do.
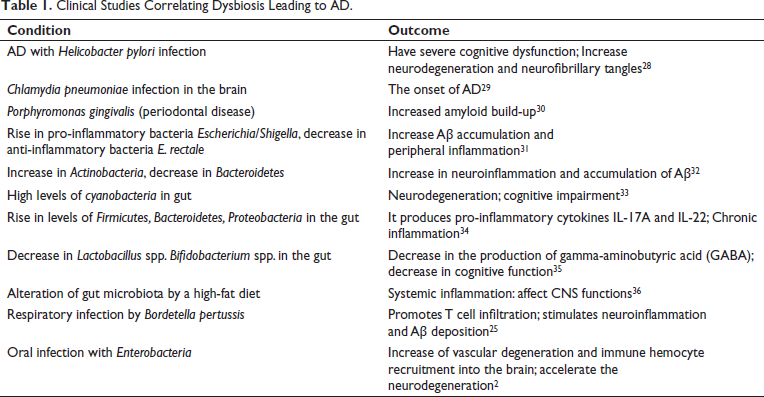
Bacteria-derived BA breach the leaky gut membrane, enter the bloodstream, and interact with the BBB proteins claudin and occludin, causing the disruption of the BBB’s tight junction. BA inhibits the expression of the cholesterol-metabolizing enzyme CYP46A via interacting with the farnesoid X receptor present in astrocytes. 25 This causes an increase in cholesterol, which serves as a host for β and γ secretase. As a result, mono fibrils develop, which lead to Aβ oligomerization and, eventually, Aβ accumulation. 26 Cholesterol inhibits the α-secretase and non-amyloid pathways as well. Bacterial amyloids Curli, Facp, and MccE492 cross the gut membrane and cause A oligomerization in the brain. 27 As a result, gut dysbiosis promotes the onset of AD by increasing Aβ formation and deposition, mitochondrial dysfunction, neuroinflammation, and stress granules. Several clinical studies correlating dysbiosis leading to AD are shown in Table 1.
Clinical Studies Correlating Dysbiosis Leading to AD.
Various Factors Associated with Dysbiosis and AD
Age
Preclinical and clinical studies have shown a link between age-related dysbiosis and AD. Indeed, changes in the makeup of the gut microbiome have been linked to aging, with an increase in proteobacteria and a reduction in probiotics like bifido-bacterial. 37 SCFAs, which are neuroprotective substances, will be diminished as age progresses. 38
Microbial Infection
A viral or bacterial infection has been linked to the onset of AD. H. pylori infection causes dysbiosis. When compared to non-infected patients, chronic H. pylori infection was found to promote the production of inflammatory factors and was connected to a poorer Mini-Mental State Exam (MMSE) score. Furthermore, blood levels of A1−40 and A42 are higher in AD with H. pylori, Borrelia burgdorferi, and Chlamydia pneumoniae infection. In neuroblastoma cells, exposure to H. pylori filtrate causes tau hyperphosphorylation similar to that seen in AD tau pathology. 39
Obesity
The modification/alteration of the microbial pattern in the colon and the progressive increase in body weight have a direct relationship. This complex process is controlled by hereditary variables, which are aided by excessive caloric intake, the use of food additives and sweets, and the administration of other products (antibiotics) that cause dysbiosis. In recent years, obesity and dysbiosis have been linked to neurodegenerative disorders. The Firmicutes:Bacteroidetes ratio (number of cells/mL) increased in obesity. A strong correlation was determined in vivo between Akkermansia muciniphila and an inflammatory response in relation to obesity. In AD, increase in Rikenellaceae and a decrease in Allobacillum and Akkermansia were determined. 40
Periodontal Infection
Prolonged periodontal inflammation has been linked to changes in the gut microbiome as well as an increase in the host’s overall inflammatory response. Normally oral bacteria will maintain a physiological balance, but changes in this can lead to dysbiotic periodontal bacteria populations and periodontitis. Many systemic diseases, such as atherosclerosis, diabetes, and obesity, have been linked to periodontitis. Periodontitis can release bacterial pathogens such LPS, flagellin, peptidoglycan, and other pro-inflammatory substances, which are risk factors for AD. 41
Physical Activity
Exercise has been shown to alter the microbiota composition. Several observational studies reported that a greater firmicutes to bacteroidetes ratio has been associated to a higher VO2 max (maximum oxygen uptake). 42 Women who exercised for at least 3 hours per week had a higher amount of butyrate-producing bacteria, particularly Akkermansia muciniphila, which aids in weight loss. 43 Regular physical exercise lower the incidence of dementia or AD in persons with mild cognitive impairment, which is mediated by increased gut microbial diversity. Moderate physical activity has been shown to increase brain volume in the elderly. However, lack of physical exercise in middle age and the elderly has been related to an increased risk of AD. 44
Stress
Stress has been found to affect health via altering gut microorganisms. It can affect composition of gut bacteria in a variety of gut areas and habitats. 45 Stress can increase gut barrier permeability, resulting in a leaky gut, allowing bacteria to enter the bloodstream and triggering inflammatory cascades. Stress has a significant impact on make up the composition and function of microbiota, and microbial composition modulates stress responses. 46 An imbalance in this reciprocal interaction can promote the onset of AD. Microbiota signals are sent to brain and peripheral tissues, which release glucocorticoids. Although these hormones are produced in the adrenal cortex, they can also be produced in the gut via extra-adrenal glucocorticoid synthesis, where the enzyme 11b-hydroxysteroid dehydrogenase type 1 produces active glucocorticoids, corticosterone, or cortisol from inactive 11-oxo derivatives. 47 Apart from pituitary ACTH, glucocorticoids are also produced from neuropeptides, cytokines, and even from bacterial ligands. So, the gut microbiota also takes part in steroidogenesis. Furthermore, the HPA axis has an impact on microbiome development and function. The altered microbiota influences the stress response by regulating the HPA axis. In the infected condition, there is an increase in c-FOS levels (which is responsible for the anxious mood) and an alteration in BDNF levels. Treatment with probiotics restores these changes. 48
Environmental Toxins
Toxins in the environment may be another unnoticed concern that harms and disrupts gut microbiota. Smoking can change the gut microbiome by raising Bacteroides–Prevotella species. 49 Other environmental toxins’ impacts on gut microbiota and mental health should be researched further. Studies show toxic metals, pesticides, and metal-based nanoparticles play a role in Tau protein phosphorylation in the brain and Aβ peptide formation in the gut by causing dysbiosis. Lifestyle also contributes to dysbiosis, which can trigger severe immune response, neuroinflammation, and metabolic diseases such as insulin resistance. As age progresses, the body’s ability to handle environmental toxins decreases, damaging the microbiota permanently and indirectly causing cognitive decline. 50
Treatments for Normalizing Dysbiosis
Diet
Diet is a crucial element in regulating the composition of gut flora. People can maintain a healthy microbial flora by a well-balanced diet. Certain foods and dietary patterns can modify the composition of gut microbiome. Dietary modifications have an immediate impact on the gut microbiome. Food has been shown to alter the overall structure of the gut microbiome by around 60%. 51 The phrase Mediterranean diet (MD) was coined by Ancel Keys in 1960, and it is now one of the most researched and well-known dietary patterns in the world, which is having a direct influence on maintaining a healthy gut microbiome (Figure 6).
Mediterranean Diet (MD) Composition and its Effects on the Gut Microbiome.
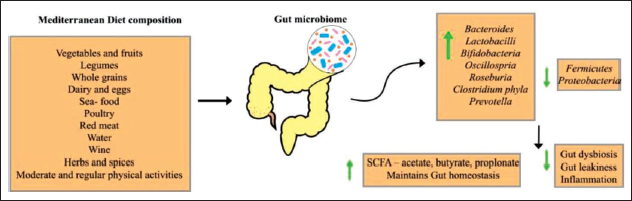
MD consist of vegetables, fruits, legumes, whole grains, seafood, poultry, red meat, water, wine, herbs and spices. Along with MD diet, regular and moderate physical activity is recommended. MD aids in the reduction of gut dysbiosis and aids in maintenance of gut homeostasis. MD corrects gut dysbiosis by raising the number of beneficial bacteria such as Bacteroides, Lactobacilli, Prevotella, Bifidobacteria, Oscillobacteria, Roseburia, and Clostridium phyla, all of which produces SCFA and have anti-inflammatory properties. It also reduces harmful bacteria like fermicutes and proteobacteria. Dietary components of MD like vitamins (e.g., D, E, C, beta-carotene, B12, B6, and folic acid), omega-3 fatty acids, minerals (e.g., zinc and magnesium), and nutraceuticals (e.g., curcumoids, acetyl-carnitine, gingko biloba, tea, epigallocatechin-3-gallate, caffeine, phytoestrogens, garlic, and resveratrol) have been investigated for their antioxidant, anti-inflammatory, and vasodilating properties. It is reported that the ingredients of MD can help improve cognitive impairment in AD patients. The Western diet has an undesirable effect on gut microbiome, it lowers the SCFA levels and causes leaky gut. 52
The MD is a promising treatment for AD through altering gut flora. According to previous research reports, MD slows the progression of AD within 1.5–3.5 years. 53 Detailed study is required to unfold correlation of diet and its influence on the microbiota–gut–brain axis, as well as whether diet-induced changes in microbiota result in changes of the brain function. 54
Probiotics
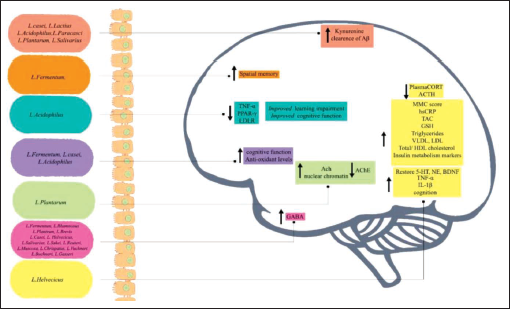
If probiotics such as Lactococcus, Streptococcus, Enterococcus, Bacillus clausii, and Enterococcus faecium are consumed in adequate amounts, it will help to keep the integrity of the gut membrane. 55 Probiotics aid to enhance CNS function, 56 by increasing the beneficial bacterial composition. 57 Figure 7 illustrates the impact of Lactobacillus strains on AD pathophysiology.
The Effect of Strains of lactobacillus on AD Pathophysiology.
Prebiotics
Prebiotics are beneficial to human health because they are used by host bacteria. 58 It modulates the immune system and metabolism of the host. 59 Prebiotics are a group of dietary components that include resistant starch, insulin, fructooligosaccharides, and xylooligosaccharides. 60 Resistant starch helps to produce SCFA. Fructooligosaccharides are effective in preserving the richness and strength of the microbial population, reducing neuronal death, brain tissue inflammation, regulation of neurotransmitter production, and downregulating tau and Aβ1-42 expression in the brains of AD rat models. 61 It protects APP/PS1 transgenic mice against AD via changing the glucagon-like peptide-1 (GLP-1) and GLP-1 receptor pathway in the gut microbiome. 62 The xylan hydrolysis product, xylooligosaccharides, provides a substrate for the commensal bacterial population. SCFA is produced during the fermentation of xylooligosaccharides, which improves gut epithelial health and regulates metabolic activities. 63
Fecal Microbial Transplant
Fecal Microbial Transplant (FMT) is a transmission of pre-screened donor stool into the intestine of the patients to restore gut microbial composition and function. 64 Although the appropriate quantity of stool weight for FMT has yet to be determined, 50–60 g of stool is advised for each session. At 4°C, the faeces can be kept for up to eight hours. The faeces are dissolved in 150 mL of normal saline solution. After that, the mixture is filtered. The finished faeces should be labeled appropriately and kept at –80°C. It should be thawed in a warm water bath at 37°C on the day of the infusion and injected within four hours of thawing. FMT can be given directly to the colon by colonoscopy, or more often via flexible sigmoidoscopy or an enema. 65 For AD patients with Clostridium difficile infections, this sort of therapy has been widely accepted. FMT has been demonstrated to improve cognition and reduce Aβ accumulation in the brains of AD rats. Hazan et al., described a case study of an 82-year-old AD patient who had a single FMT from his 85-year-old wife in 2020, yet his symptoms of C. difficile infection disappeared two months after the treatment. In addition, the patient’s MMSE score improved from 20 (moderate cognitive impairment) to 26 (normal cognitive function) after two months of FMT. 66
Conclusion
A significant and active microbial community dwells in the gut. The gut microbiota plays a key role in food digestion, immunopotentiation, and preventing dangerous pathogens from colonizing the gastrointestinal tract. Several preclinical and clinical studies reported that the gut dysbiosis effects GBA function, which may play a role in the neuropathogenesis of CNS diseases. Dysbiosis can promote neuroinflammation by increasing inflammatory cytokines and bacterial metabolites, which can change gut and BBB permeability. Results in the entry of bacterial BAs, LPS, and bacterial amyloids in to the brain. Causes the imbalance in cholesterol pathway, activation of immune responses, release of inflammatory mediators, and aid in the accumulation of beta amyloids. Restoring the gut microbiome to a composition similar to that observed in healthy adult humans will greatly decrease the course of neurodegeneration by regulating immune response and amyloidogenesis. Diet, prebiotics, probiotics and faecal microbial transplantation can reduce the progression of AD by restoring gut microbiota.
Summary
This article summarizes the pathophysiology of gut dysbiosis and the production of hazardous substances such as bacterial amyloids, LPS, TMAO, and bacterial BA. Production of gut dysbiosis which leads to onset of AD. Various factors like age, obesity, physical activity and so on, which leads to gut dysbiosis is also discussed. The article also summarizes about the treatment approaches to normalize the gut flora.
Footnotes
Acknowledgement
Some of the figures used in this article are adapted and modified from BioRender.com.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not Applicable
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Informed Consent
Not Applicable
Abbreviations
AD, Alzheimer’s disease; BA, bile acids; MMSE, Mini-Mental State Exam; LPS, lipopolysaccharide; TMAO, trimethylamine N-oxide; IL-1β, interleukin-1β; GABA, gamma-aminobutyric acid; CNS, central nervous system; LGS, leaky gut syndrome; ENS, enteric nervous system; CRF, corticotropin-releasing factor; ACTH, adrenocorticotropic hormone; PRRs, pattern recognition receptors; BBB, blood–brain barrier; HPA axis, hypothalamic-pituitary-adrenal axis; BDNF, brain-derived neurotrophic factor; MD, Mediterranean diet; FMT, fecal microbial transplant.
