Abstract
Objectives
To evaluate the pharmacokinetics of a novel sustained-release oral tablet (C-Fence, Inventia Healthcare Limited, Mumbai, India).
Methods
We conducted a randomized, placebo-controlled, parallel-design, 500 mg single-dose pharmacokinetic study of this new preparation in 18 healthy adult human subjects (nine in each group) under fasting conditions. The concentration-time profile and pharmacokinetic parameters of L-ascorbic acid, including Cmax (maximum plasma concentration), Tmax (time to reach Cmax), and AUC0-24h (area under the plasma concentration versus time curve from time 0 h to 24 h) were calculated using baseline-corrected values.
Results
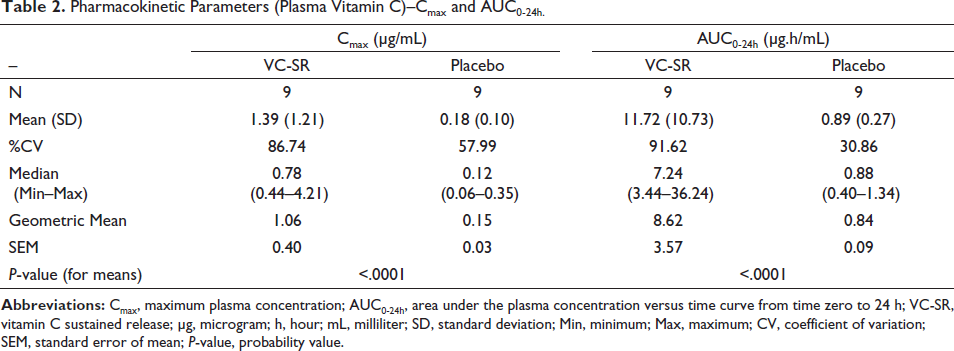
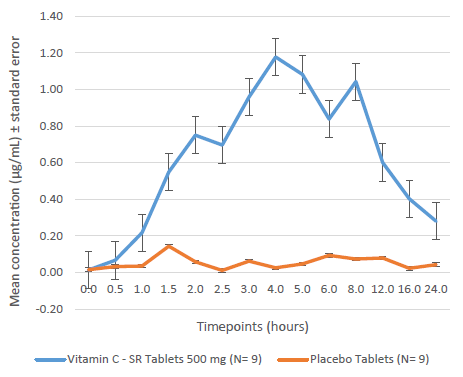
The sustained-release tablets resulted in mean Cmax and AUC0-24h, respectively, of 1.39 ± 1.21 µg/mL and 11.72 ± 10.73 µg.h/mL against 0.18 ± 0.10 µg/mL and 0.89 ± 0.27 µg.h/mL, respectively, in the placebo group. The mean Tmax with the sustained-release tablets was 4.3 ± 2.5 h. At 12, 16, and 24 h from dosing, the concentrations were 0.6, 0.4, and 0.3 µg/mL, respectively, above baseline values.
Conclusion
Novel sustained-release formulations of vitamin C are expected to help achieve plasma vitamin C values above the homeostatic saturation level and result in higher steady-state plasma concentration, which might result in better cellular uptake.
Introduction
Ascorbic acid or, simply, vitamin C, is a water-soluble and thermolabile vitamin, essential for human health, with pleiotropic functions related to its ability to donate electrons.1–3 The body requires vitamin C for normal and healthy physiological functions and metabolic activities.1, 4–6 Vitamin C is crucial in various aspects of the immune system, particularly immune cell function, epithelial barrier function against pathogens, oxidant scavenging activity of the skin, and because of its highly effective antioxidant properties, it potentially protects important biomolecules like proteins, lipids, carbohydrates, and nucleic acids against environmental oxidative stress. 1 Vitamin C is also a cofactor for a family of biosynthetic and gene regulatory monooxygenase and dioxygenase enzymes and helps in the synthesis and metabolism of tyrosine, folic acid, and tryptophan, hydroxylation of glycine, proline, lysine, carnitine, and catecholamines.1, 5 Vitamin C is beneficial in the management of oxidative stress, inflammatory conditions, recovery from stroke and tissue damage, and preventing and treating respiratory and systemic infections.1, 4 It lowers the blood cholesterol levels by facilitating the conversion of cholesterol into bile acids and increases the absorption of iron in the gut. 5 Numerous epidemiologic studies strongly suggest that vitamin C lowers the incidence of and mortality from two of the most prevalent human diseases: cardiovascular disease and cancer. 7
Although an essential nutrient, unlike most mammals, vitamin C cannot be synthesized by humans because of mutation of the terminal biosynthetic enzyme,1, 8 wherein humans lack the enzyme gulonolactone oxidase, required for the biosynthesis of L-ascorbic acid from D-glucose.9, 10 This necessitates the consumption of vitamin C-rich foodstuffs or supplements containing ascorbic acid, to fulfill its requirement. 10 Vitamin C must be retained in the body at relatively high levels to function effectively, 2 but because of the low storage capacity of the body for water-soluble vitamins, requires regular and adequate intake. 1
To assess the vitamin C status and/or prevalence of deficiency in adults, several large epidemiological studies have been conducted worldwide. 11 For example, the European EPIC-Norfolk study carried out in England, National Diet and Nutrition Survey in the United Kingdom, and MONICA study in Glasgow, Scotland revealed a prevalence of deficiency of 1.4%, 14%, and 20%, respectively. 11 A large population-based study characterized vitamin C deficiency in India and reported a deficiency in 74% of adults in North India and 46% of adults in South India, with a higher prevalence of deficiency in men than women, because of low dietary intakes. 11
Most available supplements usually contain ascorbic acid. Other forms of vitamin C supplements include sodium ascorbate; calcium ascorbate; other mineral ascorbates; ascorbic acid with bioflavonoids; and combination products. Only when oral administration of vitamin C is not feasible or when malabsorption is suspected, it may be required to be administered parenterally, by intravenous infusion. 4
Unlike most of the orally administered low molecular weight compounds, the absorption, distribution, metabolism, and excretion of vitamin C in humans is highly complex and differs in pharmacokinetic properties. 4 Vitamin C, primarily exists in two forms in vivo, viz., ascorbate (ASC) and dehydroascorbic acid (DHA), ASC being predominant (>99.9%) because of intracellular recycling of DHA to ASC by most cell types.4, 12 Primary means of membrane transport for most of the low molecular weight compounds is simple diffusion, but vitamin C diffuses at a relatively slow rate even with a concentration gradient, because of its hydrophilic nature. It has been shown that when vitamin C is orally ingested, plasma and tissue concentrations are tightly controlled at least through three mechanisms in healthy individuals: absorption, tissue accumulation, and renal reabsorption. A fourth mechanism-rate of utilization may be important in disease states. 8 Two ascorbate transporters, namely sodium-dependent vitamin C transporter (SVCT) 1 and SVCT2, coded by genes SLC23A1 and SLC23A2, respectively, play a key role in this tight control and underlying mechanisms in the clinical pharmacokinetics and in maintaining more or less constant plasma concentrations of vitamin C. The SVCTs contribute to the majority of intestinal absorption, tissue distribution, and renal reuptake of vitamin C. The absorption takes place in the small intestine, achieving peak plasma concentrations within 120 min to 180 min after ingestion.4, 13
The previously recommended daily allowance (RDA) for vitamin C was 60 mg daily, based on threshold urinary excretion, which increases at higher doses. 14 Current RDA for vitamin C in the United States and Canada is 75 mg for women and 90 mg for men.13, 15 However, literature suggests that minimal intake (i.e., 90 mg per day) may not be optimal, and might cause degenerative diseases and reduced ability to respond to stress. 16 For adult consumption, doses up to 2000 mg/day may be considered safe, above which several potentially toxic effects such as renal impairment, glucose-6-phosphate dehydrogenase deficiency, calcium oxalate nephropathy, etc. have been reported.8, 17 However, a depletion-repletion pharmacokinetic study showed that a daily intake of about 200 mg to 400 mg of vitamin C is sufficient to saturate the blood in healthy individuals,4, 15 and intake of more than 400 mg/day would lead to a homeostatic state with maximal plasma steady-state concentration of 70 µM to 80 µM. It was also reported that after administration of 400 mg vitamin C, 56% to 80% was excreted because of rapid ascorbate clearance because of saturation of renal reabsorption of ascorbate. 16 Thus, ingestion of several smaller doses or a single slow-release dose of vitamin C each day is preferable to a single dose of more than 200 mg.8, 10
Slow-release formulations are designed to release the active ingredients gradually and maintain their therapeutic plasma concentration for an extended period and provide a superior bioavailability.10, 14 Unlike conventional dosage forms which release the vitamin rapidly after administration, the sustained release forms allow for a prolonged and more regular action, thereby decreasing the side effects. 10 Advantages of sustained-release formulations include decreased total amount of dosage, reduced peak and trough fluctuations, and improved compliance, as less frequent administration of the drug is required.10, 18 A slow-release vitamin C formulation can assist in exceeding the homeostatic saturation level to reach a steady-state plasma concentration of about 250 µM, resulting in a prolonged and thus increased accumulated cellular uptake. 4
A novel sustained-release oral tablet can deliver vitamin C slowly for a prolonged period. In this study, we assessed the bioavailability of vitamin C in plasma following oral administration of a single dose of a sustained-release vitamin C tablet (C-Fence, developed by Inventia Health Care Limited, Mumbai, India) in healthy human subjects.
Materials and Methods
Study Design and Population
This was a double-blind, balanced, randomized, single-dose, parallel-arm, placebo-controlled study in healthy, adult, human subjects under fasting conditions. A pragmatic sample size of 18 analyzable subjects was planned for the study. The study was conducted at a single center–SPINOS Life Science and Research Private Limited, Coimbatore, Tamil Nadu, India. Subjects were housed between 16 and 19 March 2021. The randomization sequence was prepared using R-Software R Version 4.0.4 (R Core, New Zealand). Unique randomization codes were also generated using the same computer software. The statistician blinded the samples (bottles containing tablets) by removing the product label and applying preprinted stickers with the unique codes, which served as the only identity to separate one tablet from the other. This ensured blinding of the participants and the study team, except for the study statistician who was responsible for the blinding process.
This study was reviewed and approved by the Research Ethics Committee, Coimbatore-34, Tamil Nadu, India (registration number: ECR/84/Indt/TN/2013/RR-19) and was conducted in accordance with the Guideline for Good Clinical Practice (International Council on Harmonization [ICH]) and ethical principles according to The Declaration of Helsinki. The trial was registered with the Clinical Trials Registry–India (CTRI Number–CTRI/2021/02/031421). Prior to study initiation, all subjects provided informed consent in the presence of the investigator, or the person designated by the investigator. They were briefed on the procedures and potential risks of participating in the study. The complete study-specific informed consent process was carried out under audio-visual surveillance and was recorded.
Eighteen healthy male subjects aged between 18 and 45 years were enrolled in the study. The key inclusion criteria comprised body mass index (BMI) between 18.5 kg/m2 and 30 kg/m2, no evidence of underlying disease, nonsmokers or ex-smokers, and who agreed to abstain from all vitamin C-rich food/fruits for at least 15 days before the study. The key exclusion criteria included allergy or known hypersensitivity to vitamin C or its inactive ingredients, alcohol/drug abuse, existing tinnitus, intake of prescription medications or any vitamin C supplements, any over-the-counter medicinal products, herbal medications, any hormonal agent within two weeks prior to starting of study check-in, and consumption of caffeine/xanthine/tobacco-containing products, alcohol and its products, grapefruit (or its juice), and poppy-containing foods within two days prior to check-in.
Intervention
Vitamin C sustained-release (VC-SR) tablets 500 mg were the investigational product in the study. The subjects were allocated to either VC-SR group or placebo group in a 1:1 ratio, by the investigator, as per the randomization schedule provided by the statistician. The subjects were housed in the clinical facility from at least 36 h prior to dosing to 24 h post-dose. Subjects were served specified standardized meals from the time of check-in. After an overnight fasting of at least 10 h, on Day 1, a single oral dose of either VC-SR tablet 500 mg or placebo tablet was administered with 240 mL of water. Subjects remained in a sitting position for an hour post-dose, and only necessary movement was allowed during this period. Water was restricted for an hour pre- and post-dose.
Measurements
A total of 17 blood samples were collected from each subject, including four pre-dose samples at -12, -2, -1 h, and immediately before dosing and 13 post-dose samples at 0.5, 1, 1.5, 2, 2.5, 3, 4, 5, 6, 8, 12, 16, and 24 h. These samples were analyzed for the pharmacokinetics of plasma L-ascorbic acid. All samples were collected in pre-labeled K2EDTA–vacutainers from the forearm vein using an indwelling cannula. Heparin-lock technique was used to prevent clotting of blood in the indwelling cannula. Before each draw, 0.5 mL of blood was discarded to prevent the saline-diluted blood and heparin from interfering with the analysis. Cannulas were kept in-situ from -12-h sample to 4-h sample.
Vacutainers were kept in a rack with wet ice bath in an upright position until centrifugation and during separation. The samples were centrifuged at 3800 ± 50 rpm for 10 min at 2°C to 8°C to separate the plasma. Plasma (1.5 mL) was added to a pre-labeled polypropylene tube containing 1.5 mL of 10% metaphosphoric acid and centrifuged at 3800 ± 50 rpm for 10 min at 2°C to 8°C and equally transferred into two aliquots for analysis of ascorbic acid in plasma. All of this was conducted under yellow monochromatic light.
A sensitive and selective liquid chromatography-mass spectrometry/mass spectrometry (LC-MS/MS) method to quantify L-ascorbic acid in K2EDTA human plasma over the concentration range 100.6188 ng/mL to 19583.4541 ng/mL was developed and validated. L-ascorbic acid and its internal standard (L-ascorbic acid-13C6) were selectively extracted from 100 µL plasma using the protein precipitation technique. Separation was achieved by reverse-phase liquid chromatography on ZORBAX C8 3.5 µm column in API 4000, AB Sciex, coupled with Shimadzu Nexera UHPLC LC30AD, Autosampler Shimadzu SIL30AC with API 4000, and AB Sciex Detector.
Chromatographic Condition
Chromatographic separations were achieved by using a Zorbax C8 3.5 µm column. The mobile phase consisted of acetonitrile and 0.1% formic acid (pH 4.5) with a ratio of 90:10 v/v. All separations were performed isocratically at a flow rate of 0.5 mL/min and the injection volume was 10 µL/min. The effluent was ionized by negative electrospray ionization mode and detected by mass spectrometry. Chromatograms got separated at the run time of 1.5 min.
Mass Spectrometric Condition
Mass spectrometric data were acquired in negative ion mode ESI-MS parameters having curtain gas flow rate of 20 psi; collision gas flow rate of 4 psi; and ion source gas (Gs1 and Gs2) of 45 psi and 50 psi, respectively. The curtain gas and ion source gases were nitrogen. Data were recorded in MRM mode. LC-MS/MS machine was tuned for 175.030 m/z and 115.020 m/z with respective Q1 and Q3 mass of L-ascorbic acid.
Bioanalytical Method Validation
Calibration Curve
The linearity of the method was evaluated by a calibration curve in the range of 100.58 ng/mL to 19610.64 ng/mL of ascorbic acid. The calibration curve was achieved by plotting the peak area ratios of ascorbic acid and L-ascorbic acid-13C6 by least-squares linear regression analysis.
Accuracy and Precision
Three precision and accuracy batches were carried out to establish the calibration range of 100.58 ng/mL to 19610.64 ng/mL.
Recovery
Recovery of ascorbic acid was evaluated by comparing the mean peak areas of three extracted low-, medium-, and high-quality control samples to the mean peak areas of three neat reference solutions (unextracted). Recovery of the analyte was found to be 94% and the internal standard (L-ascorbic acid-13C6) was observed to be 94%.
Stability
In order to find out the stability of ascorbic acid in human plasma, bench top stability, freeze–thaw stability, auto-injector stability, and long-term stability studies were carried out by using six replicates of the low and high plasma quality control samples. For the bench top stability, frozen plasma samples were kept at room temperature for 24 h before sample preparation. Freeze–thaw stability of the samples was obtained over three freeze-thaw cycles, by thawing at room temperature for 2 h to 3 h and refreezing for 12 h to 24 h for each cycle. Autosampler stability of ascorbic acid was tested by processed and reconstituted low and high plasma QC samples, which were injected 24 h after reconstitution and were compared with freshly prepared QC samples. The long-term stability of ascorbic acid in rat plasma was tested after storage at approximately –70°C for 30 days. For the acceptance criteria of stability, the deviation compared to the freshly prepared standard should be within 15% of the nominal concentration.
Matrix Effect
Matrix effect is the suppression or enhancement of ionization of an analyte by the presence of matrix components in the biological samples. Matrix effect was evaluated by comparing analyte peak areas in six independent blank matrices which passed the selectivity test. Six blank matrices were extracted using the proposed extraction method and spiked with the standard (equal to LQC and HQC concentration) containing internal standard. The standard solution equal to LQC and HQC concentration containing internal standard using diluent (aqueous equivalent) were similarly prepared and the samples were injected in a single batch.
The matrix effect was performed in six different lots of human plasma by taking 100 µL of human plasma and 50 µL of diluent. To the mixture, 300 µL of methanol was added. The blank mixtures were vortexed for 10 min at 2000 rpm and centrifuged at 14000 rpm for 10 min to prepare the extracted blank. The aqueous equivalent solution was prepared by taking 20 µL of water and adding 10 µL of internal standard along with 400 µL of cyanomethane containing 0.1% formic acid. The extracted blank supernatant and aqueous equivalent solution were mixed in a ratio of 1:1. The blank aqueous solution was prepared by mixing 20 µL of water along with 410 µL of cyanomethane and subjected to vortexing. The neat solution and post-extracted solution were prepared by mixing the aqueous equivalent solution with both extracted blank and blank aqueous solution in a ratio of (1:1) solution. Both solutions were vortexed and subjected to LC-MS/MS for analysis. Matrix suppression or enhancement was calculated as follows: 100 × mean peak area of post-extracted sample/mean peak area of neat standard solution. The experiment met the acceptance criteria of %CV being less than 15% of matrices tested and at least 80% of matrices met the above criteria. Therefore, it was concluded that there was no matrix effect because of vitamin C.
The following primary pharmacokinetic parameters were calculated: area under the plasma concentration versus time curve from time zero to 24 h (AUC0–24h) and maximum plasma concentration (Cmax). These were assessed to evaluate the statistical significance of the difference between the treatments. The time to reach Cmax (Tmax) was calculated as a secondary parameter. Elimination parameters were not calculated considering the presence of baseline endogenous vitamin C.
Safety Assessments
Subjects were monitored for safety during the study and till the completion of the study by analyzing the adverse events. Safety assessment was carried out based on clinical observations, laboratory data at the beginning and at the end of the study, and evaluation of the adverse events observed during the course of the study. Clinical examinations and vital sign measurements were performed before check-in, prior to dosing and check-out, and at 1, 2, 6 and 10 h post-dose.
Statistical Analysis
The mean, standard deviation (SD), geometric mean, standard error mean (SEM), coefficient of variation (CV%), minimum, median, and maximum were calculated for Cmax and AUC0-24h and Tmax using SAS® software version 9.4 (SAS Institute Inc., Cary, NC, USA). The results were presented as mean ± standard deviation (median and interquartile range for non-normal data) for continuous variables and count and percentages for categorical variables. The t-test was used for normally distributed data. A P-value of less than 0.05 was considered to claim statistical significance.
Results
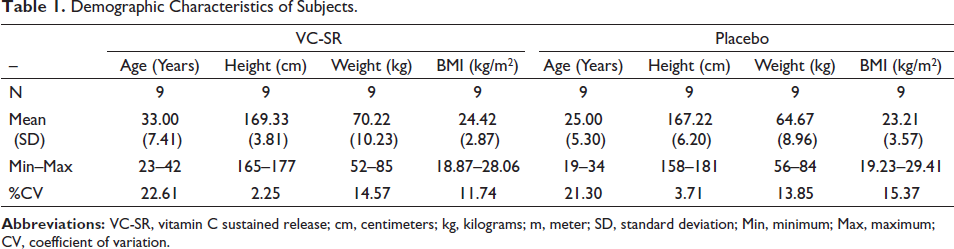
All 18 subjects, assessed for eligibility, were randomized, received the interventions, and completed the study (Figure 1). All the subjects were male and there were no significant differences in the demographics and baseline characteristics between the two groups in terms of age, weight, and BMI (Table 1). No premature unblinding occurred during the study.
Flowchart of Study Subjects (CONSORT Flow Diagram).
Demographic Characteristics of Subjects.
C max and AUC 0-24h of Vitamin C
Pharmacokinetic Parameters (Plasma Vitamin C)–Cmax and AUC0-24h.
Mean Plasma Vitamin C Concentration Over 24 h.
T max of Vitamin C
The mean Tmax (mean ± SD) in the VC-SR group was 4.28 ± 2.53 h. The Tmax data for the placebo treatment were considered irrelevant because the vitamin C concentrations obtained with placebo were almost equal to baseline concentration.
Safety Outcomes
The interventions were well tolerated and there was no adverse event reported in any subject during the study. No adverse clinical signs or derangement in laboratory values were recorded in any subject.
Discussion
Oral delivery, the most widely used route of administration of drugs and supplements, aims to achieve a steady-state blood level of the active ingredient that is therapeutically effective and nontoxic for an optimum period. This is relatively easy for drugs that have conducive pharmacokinetics. For others, this requires the design of a proper dosage form. Sustained-release dosage form is a novel delivery system that is designed to release (liberate) the active ingredients at a predetermined rate over an extended period of time after administration of a single dose. The objective is to maintain a uniform concentration of the active ingredients in blood, leading to better compliance and providing an enhanced clinical output of the product.
The pharmacokinetics of vitamin C is peculiar because its plasma concentration after oral administration is maintained under tight control by at least three mechanisms. While doses less than 100 mg/day of vitamin C resulted in a sigmoidal relationship between dose and concentrations, higher daily doses lead to the achievement of plateau plasma concentrations of 70 µM to 80 µM, and doses above 400 mg/day are believed to provide insignificant increases in plasma concentration, indicating that most of the dose is lost. 19 It has been suggested that multiple daily gram doses of vitamin C can help in exceeding the homeostatic saturation level. Studies have demonstrated that single gram-dose supplements produce two- to three-fold higher peak plasma concentrations, however, these returned to steady-state levels within 24 h after intake. 4 Additionally, oral administration of higher doses of vitamin C is limited by osmotic diarrhea and saturation of absorption, along with an additional risk of hyperoxaluria. Lack of compliance to multiple daily dosing is also a practical issue. Thus, a slow-release formulation can serve as a potential alternative, which can release vitamin C at subsaturation levels for a prolonged period, and thereby effectively increase overall exposure and uptake. 4 Superior bioavailability has been reported with slow-release formulations in studies that have compared the relative bioavailability of vitamin C from different tablet formulations. 8
In this study, the intake of vitamin C was restricted to a minimum for 15 days prior to the study, to minimize the impact of baseline vitamin C values on absorption. The placebo group acted as a control, to rule out the impact of fluctuations in endogenous vitamin C levels over the 24-h period. As expected, the plasma concentrations of vitamin C in the placebo group did not differ much from the baseline values with the peak mean concentration reaching 0.18 µg/mL above baseline; the corresponding concentration in the VC-SR group was about eight times higher than this. The Cmax and AUC0-24h values show that with a 500 mg sustained-release preparation, vitamin C was well absorbed, and the levels of vitamin C were sustained well above the baseline values for the entire 24-h study duration. The average time to achieve maximum levels (mean-1.39 µg/mL) in plasma was about 4.5 h, in contrast to the 2 h to 3 h seen with immediate-release formulations, which indicates the slow release of the active ingredient. More importantly, even at 12, 16, and 24 h post-dose, the plasma concentrations remained much above both the baseline concentrations and the peak concentration in the placebo group. Taken together, these findings confirm the slow and sustained-release characteristic of the tested novel formulation.
The study was limited by not having an immediate-release formulation for a direct comparison of exposure. The parallel design was also a limitation, as a cross-over design would have been useful in restricting the marked variations observed in the values.
Conclusion
The novel sustained-release vitamin C formulation demonstrated that, with a single dose, not only did it lead to slow and effective absorption of vitamin C, but it was also able to maintain the plasma concentrations much above baseline values over a 24-h period with good safety profile. Such formulations can help achieve plasma vitamin C values above the homeostatic saturation level resulting in higher steady-state plasma concentration, which might result in better cellular uptake while enhancing compliance.
Source(s) of Support
The study was funded by Inventia Healthcare Limited, India. The study products and other study materials were provided by Inventia Healthcare Limited, India.
Footnotes
Author contribution
The authors RS and ST conceptualized the study and participated in the study design; RA and ST participated in data interpretation and manuscript preparation; ST and RA participated in the design of the study and collection and interpretation of data; RA participated in data interpretation and manuscript preparation. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
Shah Rajat and Thanawala Shefali are employees of Inventia Healthcare Limited, India. Ramapalaniappan Abiraamasundari is an employee of Spinos Life Science and Research Private Limited, India, the agency contracted with the conduct of the study.
Ethical statement
This material is the authors’ own original work, which has not been previously published elsewhere. The paper is not currently being considered for publication elsewhere. The paper reflects the authors’ own research and analysis in a truthful and complete manner.
Informed consent
This study was conducted after taking written informed consent of all the participants. All the authors provide their consent for this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
