Abstract
The Expert Panel for Cosmetic Ingredient Safety (Panel) reviewed the safety of 7 ethers and esters of ascorbic acid, which collectively function as antioxidants, skin-conditioning agents, skin protectants, fragrance ingredients, and skin bleaching agents in cosmetic products. The Panel reviewed relevant data relating to the safety of these ingredients, and concluded that the ethers and esters of ascorbic acid are safe in the present practices of use and concentration, as described in this safety assessment.
Introduction
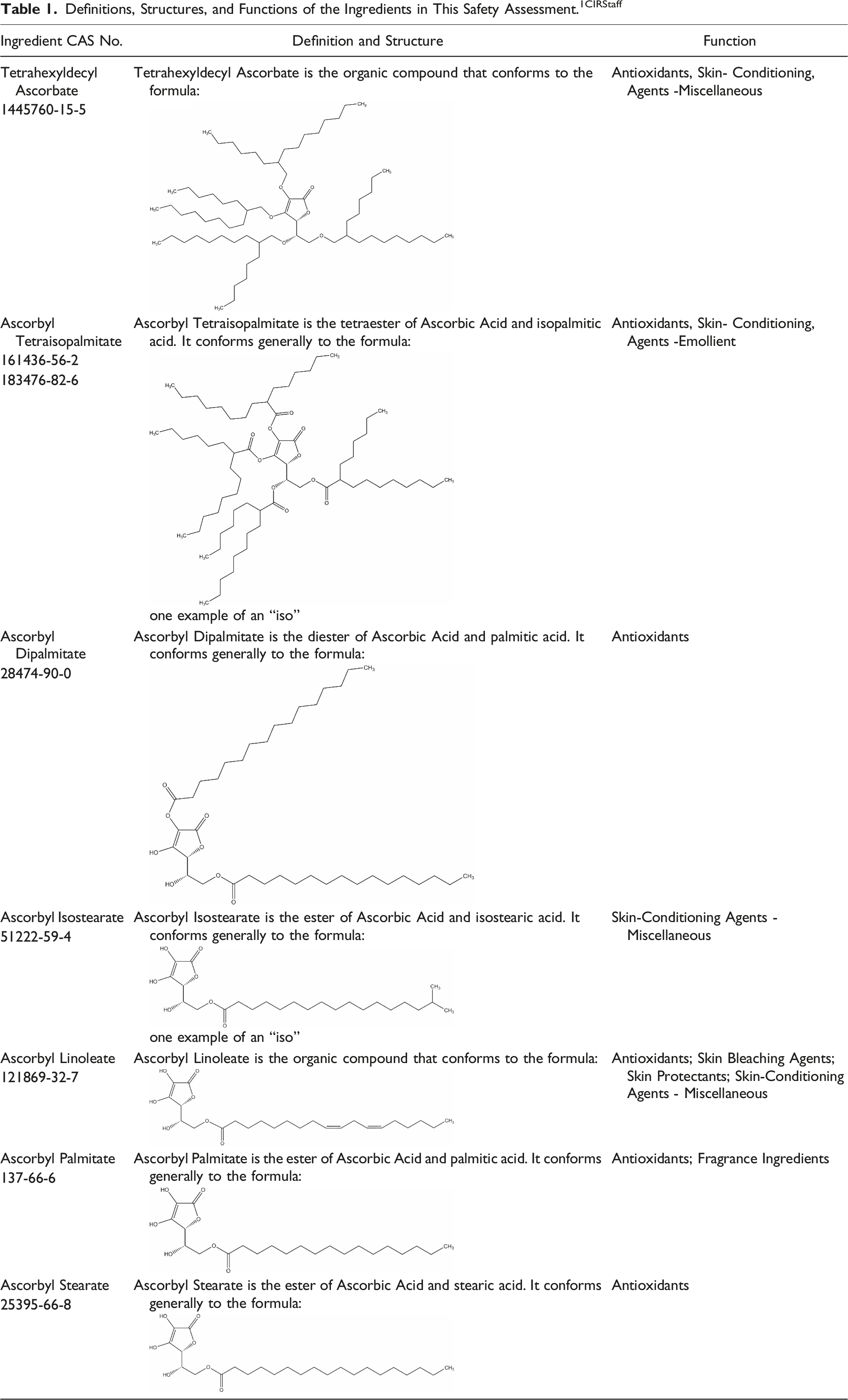
The safety of the following 6 esters and 1 ether of ascorbic acid as used in cosmetics is reviewed in this safety assessment: Tetrahexyldecyl Ascorbate Ascorbyl Isostearate Ascorbyl Linoleate Ascorbyl Tetraisopalmitate Ascorbyl Palmitate Ascorbyl Dipalmitate Ascorbyl Stearate
According to the International Cosmetic Ingredient Dictionary and Handbook (Dictionary), the functions of these ingredients in cosmetic products include: antioxidants; skin-conditioning agents; skin protectants; fragrance ingredients; and skin bleaching agents. 1 Ascorbyl Palmitate is the only ingredient with an additional function of fragrance ingredient, and Ascorbyl Linoleate is the only ingredient with an additional function of skin bleaching agent; however, functioning as a skin bleaching agent is not a cosmetic use and, therefore, the Panel did not evaluate safety for that use.
The Panel has evaluated the safety of Ascorbyl Palmitate, Ascorbyl Dipalmitate, and Ascorbyl Stearate in cosmetics, and issued a final report in 1999 with the conclusion that these ingredients are safe in the present practices of use. 2 The safety of these 3 ingredients is reevaluated in the current safety assessment, taking into consideration data that have been identified in the published literature since the original report was published. Additionally, it is possible that data on these 3 ingredients may be useful for evaluating the safety, particularly for certain toxicity endpoints, of one or more of the remaining 4 ingredients in the current safety assessment. These 4 ingredients, Tetrahexyldecyl Ascorbate, Ascorbyl Isostearate, Ascorbyl Linoleate, and Ascorbyl Tetraisopalmitate, are reviewed for the first time in this safety assessment.
Chemistry
Definition and General Characterization
The ingredients in this report are all alkylated or acylated derivatives of Ascorbic Acid. Ascorbic Acid, also known as vitamin C (an antioxidant), is a normal constituent of human skin, and is concentrated in the dermis and epidermis.3,4 Fatty-alkyl and fatty-acyl derivatives thereof are lipophilic ingredients.
Chemical and Physical Properties
Properties of Ethers/Esters of Ascorbic Acid.

Method of Manufacture
Fatty acyl ascorbyl derivatives may be synthesized via acylation of vitamin C by direct means (with fatty acids or small chain esters) or enzymatically (eg, with certain lipases).
5
The enzymatic process may be preferred because it is easier to control regioselectivity. Polar organic solvents, ionic liquids, and supercritical fluids have been successfully used as reaction media (Figure 1). Ascorbate derivatives, wherein R, independently in each case, is hydrogen, fatty-alkyl or fatty-acyl.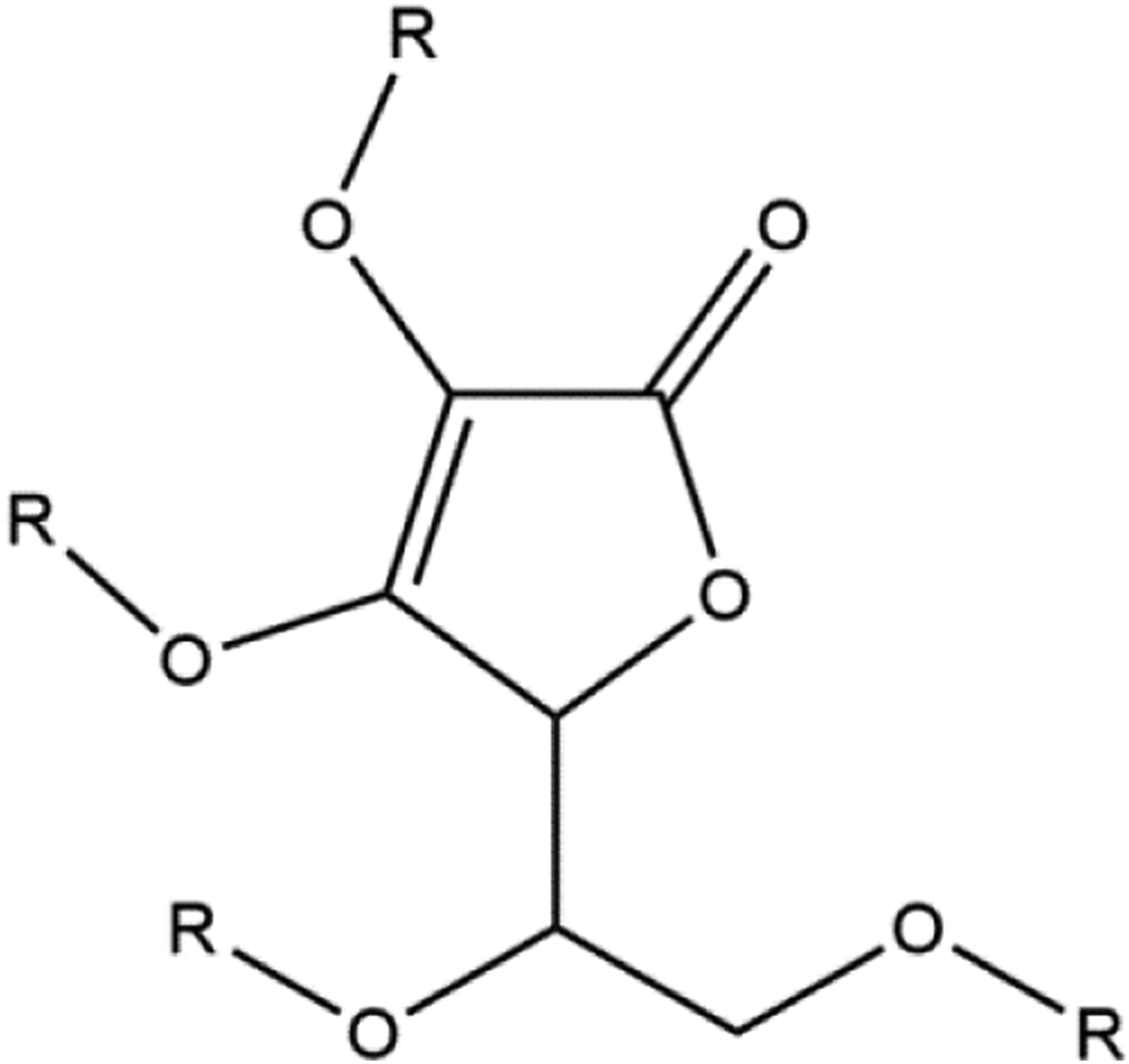
Ascorbyl Palmitate
Ascorbyl Palmitate is prepared by condensing palmitoyl chloride and ascorbic acid in the presence of a dehydrochlorinating agent such as pyridine. 6
Ascorbyl Stearate
Ascorbyl Stearate is produced by the reaction of l-ascorbic acid and stearic acid. 7
Composition/Impurities
Ascorbyl Palmitate and Ascorbyl Stearate
The National Formulary (NF) states that Ascorbyl Palmitate must contain between 95% and 100.5% of C22H38O7, based on the dried weight.
8
Depending on the method of manufacture, Ascorbyl Palmitate could contain stearic acid, because palmitic acid samples contain large quantities of stearic acid. Likewise, Ascorbyl Stearate could contain palmitic acid. When dried, Ascorbyl Stearate contains not less than 93% of
The following limits for impurities in Ascorbyl Palmitate are stated in the Food Chemicals Codex: lead (no more than 2 mg/kg) and residue on ignition (no more than 0.1% sulfated ash). 10 Additionally, the following limitations have been stated in a cosmetics industry specification for Ascorbyl Palmitate: sulfate ash (0.1% minimum), arsenic (as As) (3 ppm maximum), and lead (as Pb) (20 ppm maximum). 2 Another cosmetics industry specification has indicated a 2 ppm maximum limitation for arsenic (as As) in Ascorbyl Stearate. 2
According to the Joint Food and Agriculture Organization of the United Nations (FAO)/World Health Organization 11 (WHO) Expert Committee on Food Additives, 2 mg/kg is the limit for lead in both Ascorbyl Palmitate and Ascorbyl Stearate as food additives. 11
Use
Cosmetic
Note: Because each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure type uses may not equal the sum total uses.
NR = Not Reported; Totals = Rinse-off + Leave-on + Diluted for Use Product Uses.
Not specified whether a powder or spray, so this information is captured for both categories of incidental inhalation.
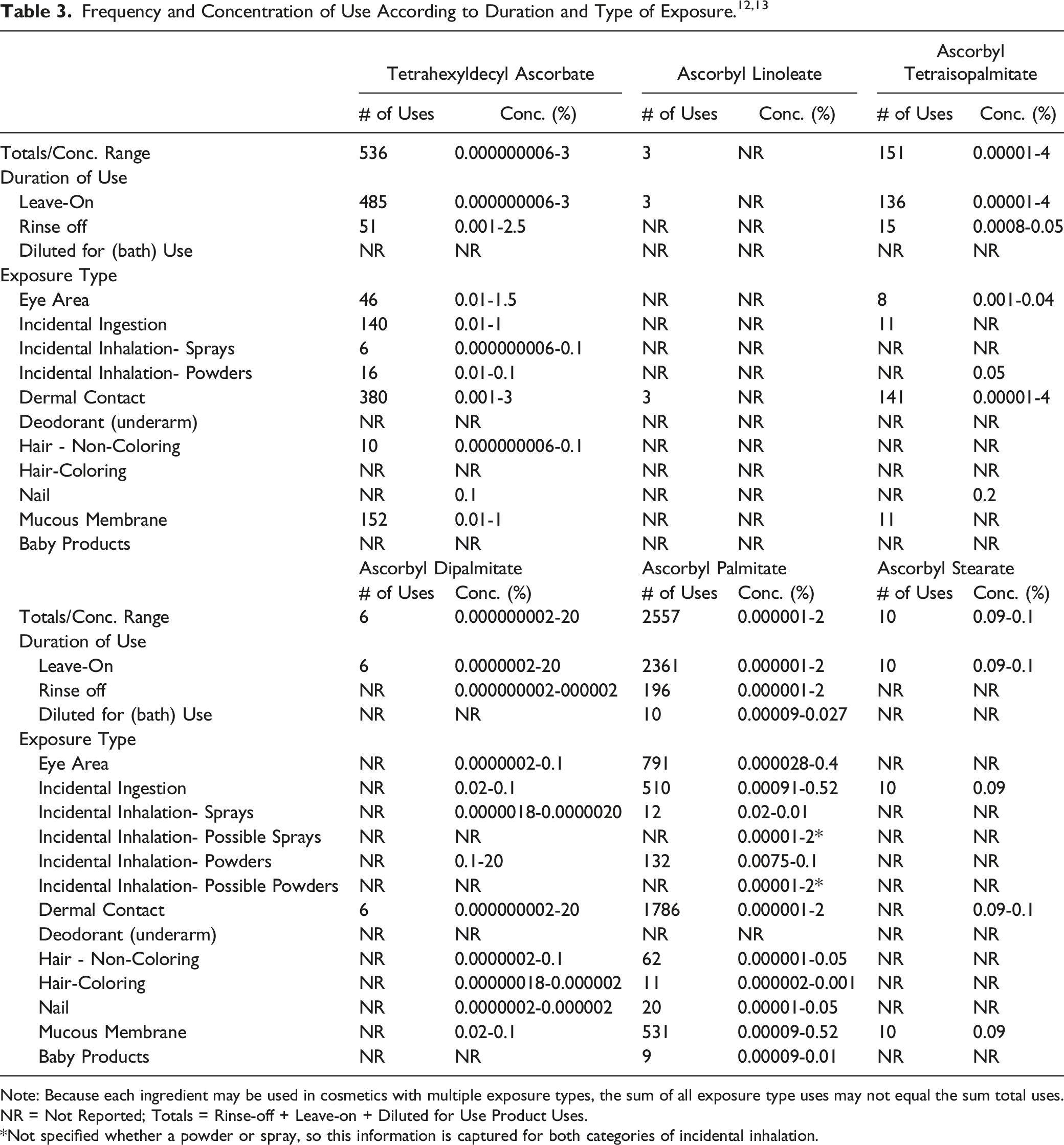
According to 2017 VCRP data, the greatest reported use frequency is for Ascorbyl Palmitate (2557 formulations, mostly in leave-on products), followed by Tetrahexyldecyl Ascorbate (536 formulations, mostly leave-on products) (Table 3). 12 The results of a concentration of use survey provided in 2016 indicate that Ascorbyl Dipalmitate has the highest maximum concentration of use; it is used at concentrations up to 20% in leave-on products (face powders). The maximum concentration of use in rinse-off products is being reported for Tetrahexyldecyl Ascorbate (concentrations up to 2.5% in shaving cream) (Table 3). 13
Cosmetic products containing the ethers/esters of ascorbic acid may be applied to the skin and hair or, incidentally, may come in contact with the eyes and mucous membranes (eg, Tetrahexyldecyl Ascorbate at maximum use concentrations up to 1.5% in eye lotions). Additionally, the following ingredients are being used in lipstick products that may result in incidental ingestion: Tetrahexyldecyl Ascorbate (at maximum use concentrations up to 1%), Ascorbyl Dipalmitate (at maximum use concentrations up to 0.1%), Ascorbyl Palmitate (at maximum use concentrations up to 0.52%), and Ascorbyl Stearate (at maximum use concentrations up to 0.09%). Products containing these ingredients may be applied as frequently as several times per day and may come in contact with the skin or hair for variable periods following application. Daily or occasional use may extend over many years.
The following ingredients are being used in products that, upon use, may result in incidental ingredient inhalation exposure: Tetrahexyldecyl Ascorbate (in hair sprays [aerosol] at maximum use concentrations up to 0.00000006% and in tonics, dressings, and other hair grooming aids [aerosol] at maximum use concentrations up to 0.1%), Ascorbyl Dipalmitate (in perfume at maximum use concentrations up to 0.000002% and in hair sprays [pump sprays] at maximum use concentrations up to 0.0000018%), and Ascorbyl Palmitate (in fragrance preparations at maximum use concentrations up to 0.045%, hair sprays [pump sprays] at maximum use concentrations up to 0.048%, body and hand products at maximum use concentrations up to 0.0025%, and in suntan products at maximum use concentrations up to 0.01%). In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 μm, with propellant sprays yielding a greater fraction of droplets/particles below 10 μm, compared with pump sprays.14,15,15-17 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, they would not enter the lungs) to any appreciable amount.14,15,15
The following ingredients are being used in face powders: Tetrahexyldecyl Ascorbate (at maximum use concentrations up to 0.1%), Ascorbyl Tetraisopalmitate (at maximum use concentrations up to 0.05%), Ascorbyl Dipalmitate (at maximum use concentrations up to 20%), and Ascorbyl Palmitate (at maximum use concentrations up to 0.1%). Conservative estimates of inhalation exposures to respirable particles during the use of loose powder cosmetic products are 400-fold to 1000-fold less than protective regulatory and guidance limits for inert airborne respirable particles in the workplace.18-20
Tetrahexyldecyl Ascorbate is included in the Australian Inventory of Chemical Substances (AICS) with the following conditions of use: “(1) for cosmetic use only, (2) for dermal use only, (3) the concentration is not to exceed 1%, and (4) it is not to be included in topical products intended for use in the eye.” 21
The ingredients reviewed in this safety assessment do not appear on the list of substances that are prohibited in cosmetic products that are marketed within the European Union and are not subject to any restrictions relating to their use in these products. 22
Noncosmetic
Ascorbyl Palmitate and Ascorbyl Stearate
Ascorbyl Palmitate and Ascorbyl Stearate have been approved by FDA as preservatives in margarine, with a concentration limit of 0.02% either individually or in combination. 23 FDA has determined that Ascorbyl Palmitate is generally recognized as safe for use as a preservative in food for human consumption. 24
The European Food Safety Authority (EFSA) has issued a scientific opinion on the safety and efficacy of vitamin C (ascorbic acid, sodium ascorbate, calcium ascorbate, Ascorbyl Palmitate, sodium calcium ascorbyl phosphate, and sodium ascorbyl phosphate) as a feed additive for all animal species. 25 The EFSA concluded that vitamin C, in the form of ascorbic acid and its calcium and sodium salts, Ascorbyl Palmitate, sodium calcium ascorbyl phosphate or sodium ascorbyl phosphate, is safe for all animal species. The EFSA also stated that setting a maximum content in feed and water for drinking is not considered necessary.
Ascorbyl Palmitate has been approved by FDA for use as an inactive ingredient in approved drug products (oral, rectal, and dermal). 26
According to the Environmental Protection Agency (EPA), residues of Ascorbyl Palmitate are exempt from the requirement of a tolerance when used in accordance with good agricultural practice as an inert ingredient in pesticide formulations applied to growing crops or to raw agricultural commodities after harvest. 27 The EPA has also determined that Ascorbyl Palmitate is exempt from the requirement of a tolerance when used in accordance with good agricultural practice as an inert ingredient in pesticide formulations that are applied to animals. 28
Ascorbyl Tetraisopalmitate
In the Republic of Korea, Ascorbyl Tetraisopalmitate, glabridin, (−)-α-bisabolol, arbutin, niacinamide, ascorbyl glucoside, and ethyl ascorbyl ether are the main ingredients used in cosmetics reported to function as whitening agents. 29 vitamin C, or L-ascorbic acid, has the ability to inhibit the activity of tyrosinase.30,31 Vitamin C is unstable and easily oxidized when exposed to air or light. To overcome this effect, Ascorbyl Tetraisopalmitate and other vitamin C derivatives have been introduced. 31
Toxicokinetics Studies
Dermal Penetration
In Vitro
Ascorbyl Palmitate
In a study involving guinea pigs, the topical application of Ascorbyl Palmitate resulted in dermal penetration. Ascorbic acid content in skin, liver, and blood increased 8-, 7-, and 4-fold when compared to control animals. In a study involving guinea pigs with scurvy, [14C]Ascorbyl Palmitate was applied topically. Ascorbic acid concentrations in the skin, liver, kidneys, and blood were 4-8 times greater when compared to the control. 2
According to one source,
A study was performed to examine the suitability of several colloidal systems for Ascorbyl Palmitate skin delivery using pig ear skin and Franz diffusion cells. 35 The receptor fluid consisted of 0.9% sodium chloride containing 0.5% polyoxyethylene (20) oleyl ether and 0.01 M sodium sulfite. One gram of a self-microemulsifying system (mixture of oil and surfactants with water), water-in-oil microemulsion, and liquid crystal loaded with Ascorbyl Palmitate (at 1% or at maximum solubilization concentration) was applied to the skin surface. After 6 h, the formulation was removed and the skin surface was cleaned. The epidermis was separated from the dermis by heat treatment, and Ascorbyl Palmitate was extracted with methanol. The results of Ascorbyl Palmitate skin deposition showed relatively high concentrations of Ascorbyl Palmitate delivered to skin layers, especially to the epidermis, whereas, no Ascorbyl Palmitate was found in receptor fluid. The highest solubilization capacity for Ascorbyl Palmitate was determined for the oil and surfactant mixture alone. The greatest extent of skin permeation was observed for liquid crystal loaded with 1% Ascorbyl Palmitate. Among the phase transition systems tested, liquid crystal was selected as the best potential carrier for Ascorbyl Palmitate. Additionally, the results in a more recent publication suggest that a lecithin-based liquid crystalline system with a lamellar structure could be used as a physiologically acceptable dermal delivery system for Ascorbyl Palmitate. 35
Animal
Ascorbyl Dipalmitate
According to an abstract of a Korean publication, the percutaneous absorption of 6 different oil-in-water cream bases containing 4% Ascorbyl Dipalmitate was studied using rabbits. 36 Details relating to the test protocol and animals tested are not stated in this abstract. The concentration of ascorbic acid in the urine varied depending on the characteristics of the cream bases that were tested. The absorption of ascorbic acid was increased and sustained with the cream bases containing branched chain esters of fatty acids instead of natural oils. The level of excretion of ascorbic acid in the urine was high for the cream base with nonionic surfactants and a small quantity of natural oils.
Human
Troxerutin (a flavonol drug), Ascorbyl Palmitate, and alpha-tocopheryl succinate were incorporated in 10 mg of a gel containing hydroxypropylcellulose, butylhydroxytoluene and ethyl alcohol (95%). 37 Alpha-tocopheryl succinate (100 mg) and Ascorbyl Palmitate (10 mg) were previously dissolved in ethyl alcohol, and troxerutin (30 mg) was previously dissolved in distilled water (0.8 mL). The gel (200 mg) was applied to 25 cm2 of the forearm of 5 volunteers (2 women and 3 men) for 45 min. The stratum corneum was then removed using 12 strips of transparent adhesive tape. The experiment was repeated 3 times (on same forearm area), and each was carried out after a recovery period of 2 weeks. The gel (200 mg) served as the control, and was tested according to the same procedure. Test results indicated that, at 45 min post-application, Ascorbyl Palmitate and the 2 other substances had penetrated into the epidermis, and were found up to the 10th strip. Looking at the cumulated percentage of the 3 substances according to the strips, more than 80% of the total dose of troxerutin and α-tocopheryl succinate, and more than 90% of the total dose of Ascorbyl Palmitate was found in the stratum corneum.
Computational Analyses/Predictions
Ascorbyl Tetraisopalmitate
To simulate Ascorbyl Tetraisopalmitate absorption through human skin, Ascorbyl Tetraisopalmitate percutaneous absorption through the hair follicle infundibulum (important route of absorption into the hair follicle of human skin) was modeled and compared with the stratum corneum membrane. 38 This comparative study was performed via computer simulation by molecular dynamics (with Martini force field). The infundibulum membrane model was constructed to reflect the lipid composition of the human epidermis. The composition of the simulated infundibulum membrane was as follows: phosphatidylcholine (17%), phosphatidylserine (17%), phosphatidylethanolamine (18.8%), phosphatidylinositol (6.9%), sphingomyelin (9.8%), cholesterol (24.4%), cholesterol sulfate (1.5%), and ceramide type II (4.6%). The composition of the simulated stratum corneum membrane was: fatty acids with 24 carbons (39%), cholesterol (36%), and ceramide type II (25%).
A single Ascorbyl Tetraisopalmitate molecule penetrated the infundibulum membrane in approximately 320 nanoseconds (ns). In the case of 3 molecules, the first molecule penetrated in the first 10 ns and the other 2 molecules combined with each other and penetrated in 100 ns. When the number of Ascorbyl Tetraisopalmitate molecules was increased to 9, structural changes in the molecule attributed to clustering of groups was observed. Two molecules combined and penetrated the membrane together in the first 10 ns, 4 other molecules grouped together (∼22 Ångstroms [Å]) and penetrated in 30 ns, and the last 3 molecules (∼19 Å) penetrated in 110 ns. The authors noted that the structural changes were probably related to the solvent (water) and the hydrophobicity of Ascorbyl Tetraisopalmitate. Another observation was that the Ascorbyl Tetraisopalmitate molecules penetrated the first layer of the bilayer membrane and remained in that position for at least 1000 ns of simulation. 38
These results suggested that a high concentration of Ascorbyl Tetraisopalmitate molecules accelerated penetration. The Ascorbyl Tetraisopalmitate molecule was found to have more affinity for the stratum corneum than for the infundibulum, and a straight penetration pathway was observed for up to 600 ns in the stratum corneum simulation. Penetration followed a lateral pathway in the infundibulum. 38
Penetration Enhancement
Ascorbyl Palmitate
Permeation tests of ibuprofen (formulated in Ascorbyl Palmitate coagel (5% w/v) or ascorbyl laurate coagel (5% w/v), or suspended in isopropanol) through excised skin of hairless mice (Strain MF1- hr/hr/Ola, Nossan Srl, Correzzana, Milano) were performed using Franz-type cells. 39 At temperatures higher than the critical micellar temperature, 6-O-ascorbic acid alkanoate aqueous suspensions turn into either micellar solutions or gel phases, depending on the length of the hydrophobic chain. Upon cooling, coagels (liquid-crystal structures) are formed.39,40 Results for the amount of ibuprofen in each vehicle that permeated (mg/cm2 ± standard error of the mean) the skin after 20 h were: 2.10 ± 0.25 (in isopropanol), 0.83 ± 0.21 (in ascorbyl laurate), and 0.47 ± 0.05 (in Ascorbyl Palmitate). Ascorbyl Palmitate did not enhance the skin penetration of ibuprofen in this study.
Absorption, Distribution, Metabolism, and Excretion
In Vitro
Ascorbyl Palmitate and Ascorbyl Dipalmitate
According to an abstract of a Japanese publication, The enzymatic hydrolysis of Ascorbyl Palmitate and Ascorbyl Dipalmitate was studied using guinea pig tissue homogenates (mixture of small intestine and pancreas homogenates). 41 Details relating to the test protocol were not included in the abstract. The yields of ascorbic acid were 80% with Ascorbyl Palmitate and 20% with Ascorbyl Dipalmitate
Animal
Oral
Ascorbyl Palmitate and Ascorbyl Dipalmitate
Guinea pigs were dosed orally with Ascorbyl Palmitate (dissolved in sodium taurocholate solution), and hydrolysis by homogenates of the liver, pancreas, and intestines was reported. Approximately 80% of the Ascorbyl Palmitate was hydrolyzed to free ascorbic acid in the small intestines and pancreas. Guinea pigs were also dosed orally with Ascorbyl Dipalmitate (∼20% in sodium taurocholate solution), and hydrolysis to free ascorbic acid by homogenates of the small intestines and pancreas was reported. In another experiment in this study, guinea pigs were dosed orally with Ascorbyl Palmitate (the equivalent of 20 mg ascorbic acid). Greater amounts of ascorbic acid were excreted at 0-24 h than at 24-48 h. The same results were reported following the oral dosing of guinea pigs with Ascorbyl Dipalmitate (the equivalent of 20 mg ascorbic acid). A difference in body retention or availability of Ascorbyl Palmitate and Ascorbyl Dipalmitate was found, due to differences in the extent and rate of hydrolysis of the 2 esters. 2
Other Routes
Ascorbyl Palmitate
A study was performed to determine the occurrence of Ascorbyl Palmitate hydrolysis in brain tissue using 2 male Wistar rats. 42 Ascorbyl Palmitate (in dimethylsulfoxide (DMSO)) was injected into an internal carotid artery at a dose of 75 mg per rat (dose volume = 0.3 mL). The animals were killed 15 min post-injection, and the brain tissue was extracted with chloroform/methanol and chromatographed using thin-layer chromatography.
Spots of Ascorbyl Palmitate were traceable in the hemispheres ipsilateral to the intracarotid Ascorbyl Palmitate injection side and in the contralateral hemispheres. The Rf (retention factor) of the spots corresponding to the brain samples of Ascorbyl Palmitate-injected rats was nearly identical to that of the standard spot. The Rf was 0.465 for the Ascorbyl Palmitate standard, ipsilateral and contralateral samples in the first rat, and was 0.470 and 0.460 for the ipsilateral and contralateral samples, respectively, in the second rat. These results indicated that Ascorbyl Palmitate resisted hydrolysis in the rat brain, because it penetrated the blood brain barrier and was retained principally in brain tissue as an intact molecule. No conclusion could be drawn as to the amount of Ascorbyl Palmitate that permeated through the blood brain barrier or the fraction that underwent hydrolysis in the brain. 42
Toxicological Studies
Acute Toxicity Studies
Oral
Ascorbyl Dipalmitate
An LD50 of >5000 mg/kg was reported for Ascorbyl Dipalmitate (15% suspension) in a study involving rats. 7
Ascorbyl Palmitate
In an acute oral toxicity study involving rats, the lowest effect level (decreased body weight) for Ascorbyl Palmitate was determined to be 2500 mg/kg. An LD50 of 2000 mg/kg was reported for Ascorbyl Palmitate (33.3% suspension) in an acute oral toxicity study involving mice. 2
Ascorbyl Stearate
The acute oral toxicity of Ascorbyl Stearate was evaluated in a study involving rats. Adverse effects were not observed over the range of doses tested, 100 mg/kg to 3000 mg/kg. 2
Ascorbyl Tetraisopalmitate
The acute oral toxicity of an Ascorbyl Tetraisopalmitate trade name material was evaluated using Wistar rats of the Crl:(WI)BR strain (5 males, 5 females). 43 A single oral dose (2000 mg/kg) of the test substance was administered by gavage to each animal. Dosing was followed by a 15-d observation period and necropsy. There were no clinical signs of toxicity and none of the animals died. Additionally, there was no evidence of organ toxicity at gross necropsy. The reported oral LD50 was >2000 mg/kg.
Computational Analyses/Predictions
The EFSA established a program for the reevaluation of approved antioxidant food additives to help address concerns about the continued safety of food additives. 44 The aim of this research was to predict the toxicity of antioxidant food additives using an in silico methodology for a preliminary evaluation of safety. The in silico prediction was conducted for the following endpoints: acute toxicity (LD50), genotoxicity, carcinogenicity, reproductive toxicity, chronic toxicity (no-observed-effect level (NOEL)), acceptable daily intake (ADI) value, and the toxicity of metabolites. The applied software products used were Toxtree, TEST, Admet Predictor, and the Organization for Economic Co-operation and Development (OECD) QSAR Toolbox. It was noted that many researchers validated the prediction methods used and assessed the accuracy and robustness of each platform. NOEL predictive values were calculated from the predicted value of the ADI, and these NOELs were used to predict long-term toxicity. The antioxidant metabolites were predicted using the cytochrome p450 method. The in silico method predictions for Ascorbyl Palmitate and Ascorbyl Stearate appear in this section and other sections of the report under this subheading.
Ascorbyl Palmitate
Using the in silico methodology noted above, Ascorbyl Palmitate was predicted to be a moderately toxic compound. 44
Ascorbyl Stearate
Using the in silico methodology, Ascorbyl Stearate was predicted to be a slightly toxic compound. 44
Short-Term Toxicity Studies
Ascorbyl Palmitate
In a 63-d oral feeding study involving female mice, signs of toxicity were not observed for Ascorbyl Palmitate at doses up to 3000 mg/kg/d. 2
Chronic Toxicity Studies
Oral
Ascorbyl Palmitate
Ascorbyl Palmitate (0.25% in diet) was fed to rats in a chronic oral feeding study. The rats received Ascorbyl Palmitate at oral doses of ≤2500 mg/kg/d for 728 d. Toxic effects were not observed at doses of 125 or 1000 mg/kg/d, but decreased body weight was observed at doses of 2500 mg/kg/d and greater. Oxalate stones were observed in 2 of 8 rats dosed with 2500 mg/kg/d. 2
Groups of 10 rats were fed Ascorbyl Palmitate (2% and 5% in feed) for 9 months. Significant growth retardation was observed at a dietary concentration of 5%; also, 2 of 10 rats had bladder stones and hyperplasia of bladder epithelium and 1 rat had nephritis. Slight growth retardationwas observed in the 2% dietary group. In a 2-yr feeding study, groups of 8 rats were fed 2% or 5% Ascorbyl Palmitate (equivalent to 424 mg/kg and 1060 mg/kg, or 0.05% and 0.25% of total diet) in a heat-treated lard diet. Decreased growth rate was reported at the higher dose; also, 2 of 8 rats had oxalate stones after feeding for 9 mo. 2
Computational Analyses/Predictions
Ascorbyl Palmitate
Using the in silico methodology noted above, the NOEL for long-term toxicity of Ascorbyl Palmitate was calculated to be 916 mg/kg/d. 44
Ascorbyl Stearate
Using the in silico methodology, the NOEL for long-term (duration not defined) toxicity of Ascorbyl Stearate was calculated to be 834 mg/kg/d. 44
Developmental and Reproductive Toxicity Studies
Computational Analyses/Predictions
Ascorbyl Palmitate
Using the in silico methodology noted above, Ascorbyl Palmitate was predicted not to be a reproductive toxicant. 44
Ascorbyl Stearate
Using the in silico methodology, Ascorbyl Stearate was predicted not to be a reproductive toxicant. 44
Data From Prior CIR Safety Assessments
In addition to the reproductive toxicity in silico predictions for Ascorbyl Palmitate and Ascorbyl Stearate, the Panel proposed that safety data for analogous compounds previously reviewed by the CIR could be used in a weight of evidence approach, and that these data should be referenced in this safety assessment. The analogous compounds comprise ascorbates, fatty acids derivatives, acyl glycerols, fatty acyl/alkyl saccharides, and sorbitan esters. The developmental and reproductive toxicity data summaries for these analogous compounds may be found in the original CIR reports.45-56
Genotoxicity Studies
In Vitro
Ascorbyl Palmitate
The genotoxicity of Ascorbyl Palmitate (dissolved in 0.067 M potassium or sodium sulfate buffer at pH 7) was evaluated in the Ames test at doses of 0.01 to 3.3 mg/plate using the following Salmonella typhimurium strains: TA1535, TA1537, and TA1538. Doses >3.3 mg/plate were toxic to the bacteria. Test results were negative. The genotoxicity of Ascorbyl Palmitate (dissolved in 0.067 M potassium or sodium sulfate buffer at pH 7) was also evaluated in the tryptophan reversion assay, and was tested at doses of 0.01 to 3.3 mg/plate. Test results were negative. 2
Ascorbyl Tetraisopalmitate
The genotoxicity of an Ascorbyl Tetraisopalmitate trade name material (in ethanol) was evaluated in the Ames test using Salmonella typhimurium and Escherichia coli (bacterial strains not stated) with and without metabolic activation. 43 Doses of the test substance up to 1000 μg/plate were tested. The positive controls in this study were not stated. The test substance was non-genotoxic, with and without metabolic activation, over the range of doses tested. The negative and strain-specific positive control values were within the background historical ranges associated with the laboratory where the test was performed.
Computational Analyses/Predictions
Ascorbyl Palmitate
Using the in silico methodology noted earlier, Ascorbyl Palmitate was predicted to be a genotoxic compound. 44
Ascorbyl Stearate
Using the in silico methodology, Ascorbyl Stearate was predicted to be a non-genotoxic compound. 44 The reason for the difference in the genotoxicity prediction for Ascorbyl Palmitate vs Ascorbyl Stearate was not stated.
Carcinogenicity Studies
Oral
Ascorbyl Palmitate
The carcinogenicity of Ascorbyl Palmitate (2% in the diet) was evaluated using groups of 12 female CF-1 mice. Mice (6/group) were fed test or control diet for 2 wk. The remaining 6 mice of one group were injected subcutaneously (s.c.) with 10 mg/kg azoxymethanol (induces focal areas of dysplasia [FADs]) in the colon in saline once weekly for 6 wk. Six mice of the other group were injected with saline (same procedure). Ascorbyl Palmitate was nontoxic. The administration s.c. of azoxymethanol induced proliferation of colonic epithelial cells and the expansion of the proliferative compartment, as well as formation of FADs. There were no FADs in control mice or those fed Ascorbyl Palmitate. 2
Co-Carcinogenicity
Oral
Ascorbyl Dipalmitate or Ascorbyl Stearate
The co-carcinogenicity of Ascorbyl Dipalmitate or Ascorbyl Stearate was evaluated using F344 male rats. The rats were initiated with N-butyl-(4-hydroxybutyl)nitrosamine (BBN) and administered 5% Ascorbyl Dipalmitate or 5% Ascorbyl Stearate. There were no lesions of the liver or kidneys in rats of the test or control group. 2
Anti-Carcinogenicity Studies
In Vitro
Ascorbyl Palmitate
A study was performed to determine whether derivatives of ascorbic acid increase tumor cell death, caused by hyperthermia, to further improve cancer treatment. 57 Hyperthermia is a potent cancer treatment that inhibits the growth of tumor cells. The study was performed using human tongue squamous carcinoma cells (HSC-4). For the examination of carcinostatic activity, cells previously cultured for 24 h were suspended in culture medium. A test solution of Ascorbyl Palmitate (100 μM) was placed in a test tube and the solvent was evaporated by jet flow of nitrogen gas. Culture medium was then added to the residue and sonicated to become homogenously emulsified. The cell suspensions and test substance were mixed in a glass sample bottle. Hyperthermic treatment involved incubation of the cell suspension for 60 min at a temperature of 37°C or 42°C in a water bath. The suspension was then maintained by sequential culture for 24 h (at 37°C). Carcinostatic activity was evaluated using a crystal violet staining assay; cell morphology was observed under a phase-contrast microscope.
The cell viability of control cultures (at 37°C) was considered to be 100%, but was reduced to 57.3 ± 2.7% at 42°C (P < 0.0001). Treatment with Ascorbyl Palmitate at 37°C yielded a cell survival rate of 86.8 ± 5.7%. At 42°C, treatment with Ascorbyl Palmitate decreased cell viability to 42.0 ± 2.1% (P < 0.0001). The authors noted that the carcinostatic activity of Ascorbyl Palmitate was markedly increased with hyperthermia. 57
Animal
Dermal
Ascorbyl Palmitate was applied topically to mice at doses of 4 or 5 μmol twice weekly. Ascorbyl Palmitate (5 μmol) was administered twice weekly to previously initiated mice. Topical application caused inhibition of 12-O-tetradecanoylphorbol-13-acetate (TPA)-induced tumor production and DNA synthesis in mouse epithelial cells; 60-70% inhibition was observed at the 4 μmol dose. Ninety-one percent of tumors were inhibited per mouse at a dose of 5 μmol. When Ascorbyl Palmitate (0.017 mmol) was injected s.c. into mice, the growth of sarcoma 180 was inhibited. 2
Inhibition of Tumor Promotion
Ascorbyl Palmitate
The effect of Ascorbyl Palmitate (in acetone vehicle) on the induction of epidermal ornithine decarboxylase (ODC) activity, epidermal hyperplasia (epidermal thickness), skin edema, and skin tumor promotion by 1,8-dihydroxy-3-methyl-9-anthrone (chrysarobin, an anthrone tumor promoter) was tested using female SENCAR mice. 58 Many cellular and biochemical changes have been associated with tumor promoters and the process of tumor promotion, such as sustained hyperplasia and the induction of ODC activity. For the analysis of edema and hyperplasia, groups of 4 mice were treated for 4 wk (once weekly) with acetone, chrysarobin, or chrysarobin plus Ascorbyl Palmitate (4 μmol). After the final treatment with chrysarobin, the mice were killed at 24 h to measure edema, or, at 48 h for histological analysis of hyperplasia. Ascorbyl Palmitate (4 μmol) inhibited (24% inhibition; P < 0.05) the induction of edema by chrysarobin (220 nmol). This dose of Ascorbyl Palmitate (4 μmol) also inhibited (26% inhibition; P < 0.05) the induction of epidermal hyperplasia by chrysarobin (220 nmol).
In the tumor induction experiment, groups of 30 mice were initiated with dimethylbenzanthracene (DMBA) (25 nmol) and, 2 wk later, received once weekly treatments of chrysarobin (220 nmol). Ascorbyl Palmitate (0.2 mL in acetone; 1 μmol or 4 μmol doses) was applied to a shaved area of the back 5 min before the application of chrysarobin. The number and incidence of papillomas were recorded weekly. Promotion was continued until the average number of papillomas per mouse reached a plateau in all groups. Ascorbyl Palmitate inhibited chrysarobin-induced tumor promotion at 1 μmol and 4 μmol, but the researchers noted that there was no clear dose-response relationship. At 27 wk of promotion with chrysarobin (220 nmol per mouse), 1 μmol and 4 μmol Ascorbyl Palmitate reduced the average number of papillomas per mouse by 48% and 44%, respectively. Additionally, the number of papillomas per mouse in both groups receiving Ascorbyl Palmitate was significantly lower (P < 0.01) than the tumor response in the group that was promoted with chrysarobin alone. 58
Ascorbyl Palmitate was applied topically according to the procedure in the tumor induction experiment (ODC assay). At 60 h after chrysarobin application, mice were killed by cervical dislocation and the dorsal skin was surgically removed. Epidermal scrapings from groups of 4 mice were pooled, homogenized, and centrifuged. ODC activity in the soluble supernatant was determined by measuring the release of [14C]CO2 from
Ascorbyl Stearate
Human glioblastomas (gliomas) are characterized as highly invasive and rapidly growing brain tumors. 59 A study was performed to determine the in vitro effect of Ascorbyl Stearate on cell proliferation, transformation, apoptosis and modulation of expression of insulin-like growth factor-I receptor (IGF-IR) in human glioblastoma multiforme (T98G) cells. Ascorbyl Stearate showed significant inhibition of fetal bovine serum and human recombinant insulin-like growth factor-I (IGF-I)-dependent cell proliferation in a dose-dependent manner. Treatment of T98G cells with 50, 100, and 150 μM Ascorbyl Stearate for 24 h slowed down the cell multiplication cycle, with significant accumulation of cells at the late S/G2-M phase of the cycle. Ascorbyl Stearate treatment (100 μM) reversed the transformed phenotype, as determined by clonogenicity in soft agar, and also induced apoptosis of T98G cells. These changes were said to have been associated with a significant decrease in IGF-IR expression in a dose- and time-dependent manner when compared to untreated controls. These data clearly demonstrated that Ascorbyl Stearate had antiproliferative and apoptotic effects on T98G cells, probably through modulation of IGF-IR expression and the facilitation of programmed cell death.
Pancreatic cancer is an aggressive tumor with short median survival, and is associated with a high mortality rate. 60 A study was performed to evaluate the effects of Ascorbyl Stearate on pancreatic cancer. The treatment of human pancreatic carcinoma cells with Ascorbyl Stearate (50-200 μM) resulted in a dose-dependent inhibition of cell proliferation. Ascorbyl Stearate slowed down the cell cycle, accumulating human pancreatic carcinoma epithelial-like cells (PANC-1 cells) in late G2-M phase. Furthermore, Ascorbyl Stearate treatment (150 μM) markedly inhibited growth in soft agar and facilitated apoptosis of PANC-1 cells, but not human pancreatic ductal adenocarcinoma cells (Capan-2 cells). These effects were accompanied by a significant reduction in insulin-like growth factor 1 receptor (IGF1-R) expression, when compared to untreated controls. Capan-2 cells, the least responsive to Ascorbyl Stearate treatment, did not overexpress the IGF1-R. These results demonstrated the efficacy of Ascorbyl Stearate in inhibiting the growth of pancreatic cancer cells.
The effect of Ascorbyl Stearate (25-150 μM) on human ovarian epithelial cancer cells (OVCAR-3 cells) was studied. 61 Treatment with Ascorbyl Stearate caused a dose-dependent inhibition of cell proliferation. The antiproliferative effect was due to the arrest of cells in the S/G2-M-phase of the cell cycle. Treatment of OVCAR-3 cells with Ascorbyl Stearate also inhibited phosphatidylinositol-3-kinase and protein kinase B (PI3K/AKT) activity. The presence of a constitutively active AKT protected OVCAR-3 cells from the effects of Ascorbyl Stearate, suggesting that this ester targets the PI3K/AKT pathway. The administration of Ascorbyl Stearate by gavage induced involution of human ovarian carcinoma xenografts in nude mice. These studies indicate that the antiproliferative effect of Ascorbyl Stearate on ovarian epithelial cancer cells is associated with decreased PI3K/AKT activity, and point toward the PI3K/AKT signaling pathway as a target for this drug. Data from another study indicated that the anti-proliferative activity of Ascorbyl Stearate on ovarian cancer cells was due in part to G2/M (G2/M phase of the cell cycle) arrest modulated by means of a tumor protein p53-dependent pathway. 62
Other Relevant Studies
Promotion of Lipid Peroxidation
Ascorbyl Palmitate
A study was performed to determine the antioxidative properties of Ascorbyl Palmitate using human keratinocyte cultures. 63 The fatty acid analog cis-parinaric acid (cPA) was used to quantify lipid peroxidation. This fluorescent fatty acid analog integrates into membranes, where it is readily oxidized because of its extensive unsaturation. As oxidized cPA loses fluorescence, relative levels of lipid peroxidation can be determined. Keratinocytes treated with 10-100 μM Ascorbyl Palmitate prior to UVB irradiation showed increased loss of fluorescence. At the 100 mM dose, there was significant loss of cPA fluorescence, vs UVB-irradiated cells not exposed to Ascorbyl Palmitate (P < 0.05), with little residual fluorescence detectable. UVB-induced increases in lipid peroxidation in the absence of Ascorbyl Palmitate were readily detected (P < 0.05 vs nonirradiated cells). Cells pretreated with 100 or 300 mM Ascorbyl Palmitate for 30 min prior to irradiation showed dose-dependent increases in mean fluorescence values. At the 300 mM dose, Ascorbyl Palmitate pretreatment resulted in significant increases in lipid peroxidation when compared to UVB irradiation only (no Ascorbyl Palmitate).
Levels of reactive oxygen species were determined using the fluorescent probe dihydrorhodamine (DHR). Keratinocytes were loaded with DHR, pretreated with 1-25 μM Ascorbyl Palmitate, and exposed to UVB. Ascorbyl Palmitate effectively inhibited DHR oxidation in a dose-dependent manner, indicating its antioxidant potential. Thus, the authors noted that Ascorbyl Palmitate reduced cellular levels of reactive oxygen species after UVB irradiation. 63
Furthermore, the treatment of keratinocytes with Ascorbyl Palmitate inhibited UVB-mediated activation of epidermal growth factor receptor, extracellular regulated kinases 1 and 2, and p38 kinase because of its ability to prevent reduced glutathione depletion and scavenge hydrogen peroxide. However, Ascorbyl Palmitate strongly promoted UVB-induced lipid peroxidation, c-Jun N-terminal kinase activation, and cytotoxicity. The authors noted that the lipid component of Ascorbyl Palmitate probably contributes to the generation of oxidized lipid metabolites that are toxic to epidermal cells. They also noted that the data in this study suggest that, despite its antioxidant properties, Ascorbyl Palmitate may intensify skin damage following physiologic doses of UV radiation. 63
Dermal Irritation and Sensitzation Studies
Irritation
Animal
Ascorbyl Dipalmitate
The skin irritation potential of Ascorbyl Dipalmitate (10% aqueous) was evaluated in a modified Draize test using occlusive patches that were applied for 24 h. The test substance was non-irritating to intact skin of rabbits. 2
Ascorbyl Palmitate
The skin irritation potential of Ascorbyl Palmitate (10% aqueous) was evaluated in a modified Draize test using occlusive patches that were applied for 24 h. The test substance was non-irritating to intact skin of rabbits. 2
Ascorbyl Tetraisopalmitate
The skin irritation potential of an Ascorbyl Tetraisopalmitate trade name material was evaluated according to OECD Guideline 404 using 3 New Zealand white rabbits. 43 The test substance (0.5 mL, no vehicle) was applied for 4 h, under a semiocclusive patch, to 10 × 15 cm2 of skin on one flank. Following application, the animals were observed for up to 72 h. Any evidence of skin irritation (reactions not specified) was fully reversible within 2 d. Effects other than irritation were not observed. The test substance was classified as a non-irritant (primary irritation index (PII) = 0.3, classified as negligibly irritating).
Sensitization
Animal
Ascorbyl Tetraisopalmitate
The guinea pig maximization test was used to evaluate the skin sensitization potential of an Ascorbyl Tetraisopalmitate trade name material. 43 The test substance (undiluted) was applied to 10 guinea pigs during induction (epidermal application and intradermal injection) and the challenge phase (epidermal application). Five guinea pigs served as negative controls (substance applied not stated). Following intradermal injection (0.1 mL into scapular region), reactions were assessed 24 h and 48 h later. For the epidermal application, a 2 × 3 cm2 non-woven patch containing 0.5 mL of the test substance, secured with an elastic bandage, was applied for 24 h to the flank. Again, reactions were assessed at 24 h and 48 h post-administration. There was no evidence of irritation during the induction phase. Skin sensitization was observed in 8 of 10 animals, but not in any of the 5 negative control animals. Scaling (at application site) was observed in one animal. The skin sensitization rate was 80%, and the test substance was considered to have strong sensitization properties.
Human
Ascorbyl Dipalmitate
In another study, the skin sensitization potential of a face powder containing 20% Ascorbyl Dipalmitate (tested at a concentration of 30%; therefore, effective concentration = 6%) was evaluated in the maximization assay using 25 healthy male and female adult volunteers. 64 Patches were applied to the upper outer arm, volar forearm, or the back of each subject. During induction, 0.25% aqueous sodium lauryl sulfate (SLS, 0.1 mL) was applied under an occlusive patch (15 mm disk) for 24 h. After patch removal, the test substance (0.1 mL) was applied to the same site and the site was covered with an occlusive patch for 48 h or 72 h (72 h, when placed over a weekend). This sequence was repeated for a total of 5 induction applications. The challenge phase was initiated after a 10-d non-treatment period. A single 48-h challenge application (occlusive patch) of the test substance was made to a new site on the opposite arm, forearm, or side of the back. The challenge application was preceded (1 h before challenge patch application) by 1 h of pretreatment with 5% aqueous SLS (0.1 mL) under an occlusive patch. Challenge reactions, if observed, were scored at 1 h and 24 h post-removal of the patch. There were no instances of contact allergy either at 48 h or 72 h after challenge patch application in any of the 25 subjects tested. It was concluded that the face powder did not possess detectable contact sensitivity potential. 64
Ascorbyl Palmitate
The skin sensitization potential of Ascorbyl Palmitate (0.2% in eye cream) was evaluated in the maximization test using 26 male and female subjects. The test substance (0.1 mL) was applied for 48 h to the arm, forearm, or back under occlusive patches during induction (5 applications total). Challenge reactions at a new site were scored at 48 h and 72 h. There were no adverse reactions or signs of sensitization. In another study, the skin sensitization potential of Ascorbyl Palmitate (1%, 3%, and 5% in petrolatum) was evaluated in a modified Draize assay using 106 subjects. Ten induction applications under occlusive patches (in Finn chamber) were made to the scapular back. A challenge patch was applied for 48 h to new site. Reactions were scored at 96 h. Seven 1+ reactions (at 1% concentration) were observed in one subject and five 1+ reactions (at 5% concentration) were observed in one subject. There were no reactions to the 3% concentration. Ascorbyl Palmitate was classified as non-sensitizing at concentrations of 1% to 5%. 2
Ascorbyl Tetraisopalmitate
The skin irritation and sensitization potential of an Ascorbyl Tetraisopalmitate trade name material was evaluated in a human repeated insult patch test involving 102 male and female subjects. 65 A 2 × 2 cm2 occlusive patch containing 0.2 mL or 2 g of the test substance (diluted to 10% in silicone) was applied for 24 h to the infrascapular region of the back (to the right orleft of the midline). The procedure was repeated for a total of 9 consecutive 24-h applications. Applications were made on Mondays, Wednesdays, and Fridays for 3 consecutive weeks. Reactions were scored approximately 24 h after patch removal. The challenge phase was initiated following a 10- to 14-d non-treatment period. A challenge patch containing the same amount of the test substance was applied to a previously untreated site. Reactions were scored at 24 h and 48 h post-application. There was no evidence of adverse reactions during the course of the study. The Ascorbyl Tetraisopalmitate trade name material (10% dilution in silicone) was classified as a non-irritant and non-sensitizer.
Phototoxicity
Animal
Ascorbic Acid
Ascorbic Acid (10%) was applied topically to pigs. Treated skin was protected from UVB damage (as measured by erythema and sunburn cell formation) and UVA-mediated phototoxic reactions. The number of sunburn cells reduced by 42% when compared to controls (20% propylene glycol (v/v) with 0.5% hydroxypropylcellulose). 2
Ocular Irritation Studies
Animal
Ascorbyl Dipalmitate
The ocular irritation potential of Ascorbyl Dipalmitate (10% aqueous) was evaluated in a modified Draize test. The test substance was instilled (0.1 mL) into the conjunctival sac. Ocular irritation was not observed. 2
Ascorbyl Palmitate
The ocular irritation potential of undiluted Ascorbyl Palmitate was evaluated in a modified Draize test. The test substance was instilled (0.1 mL) into the conjunctival sac. The test substance was minimally irritating. 2
Clinical Studies
Case Reports
Ascorbyl Tetraisopalmitate
A 54-yr-old non-atopic woman presented with a skin reaction 2 d after the initial application of an anti-aging skin care product. 66 The reaction began on the face, and spread to the arms and pre-sternum. Patch testing of the product and ingredients from various test series was performed using patch test chambers, and reactions were scored according to the guidelines of the International Contact Dermatitis Research Group (ICDRG) on days 2, 4, and 7. The patient had positive patch test reactions to methylchloroisothiazolinone (100 ppm), thiomersal, and the product. The results of a repeated open application test (ROAT) with the product became positive after 3 d. Patch testing with the ingredients of the product revealed a strong positive reaction to Ascorbyl Tetraisopalmitate (20% in liquid paraffin); negative results were reported for the 20 control subjects patch tested with this ingredient.
An 83-yr-old man was treated for atopic dermatitis with a non-steroidal over-the-counter (OTC) moisturizer that contained Ascorbyl Tetraisopalmitate. 67 Acute contact dermatitis that spread rapidly to the limbs and trunk was observed on application day 14. Patch tests (chambers) were performed according to ICDRG guidelines; reactions were scored on days 2 and 4. Patch test results for Ascorbyl Tetraisopalmitate (0.05%; dose/cm2 not stated) were positive (++ reaction) on day 4. The patient refused any additional investigation.
Other Clinical Reports
Tetrahexyldecyl Ascorbate
In a double-blind manner, a newly formulated vitamin C complex containing 10% ascorbic acid (water soluble) and 7% Tetrahexyldecyl Ascorbate (lipid soluble) in an anhydrous polysilicone gel base was applied to one-half of the face of 10 subjects. Polysilicone gel base was applied to the opposite side. 68 Clinical evaluation for inflammation was performed prior to the study and at weeks 4, 8, and 12. Biopsies of both sides of the face, sampling a treated area as well as a control area, were performed at week 12 in 4 patients. Inflammation of the skin was assessed as present or absent, and the presence or absence of an inflammatory infiltrate in biopsy specimens was evaluated as well. No patients were found to have either clinical or histologic evidence of inflammation on either side of the face. The average epidermal thickness of the treatment side was 51.8 μm, while that of the gel-base side was 48.1 μm. The Grenz zone collagen measurements averaged 52.5 μm on the treatment side and 35.5 μm on the gel-base side, indicating new collagen formation. One patient showed no difference in epidermal thickness between the treatment and gel-base sides, while 3 patients showed an increase on the treatment side.
A clinical trial on a moisturizer was conducted using 37 female subjects. 69 The composition of the moisturizer was as follows: Astragalus membranaceus root extract, a peptide blend including palmitoyl tripeptide-38, standardized rosemary leaf extract (ursolic acid), ubiquinone (coenzyme Q10), and Tetrahexyldecyl Ascorbate. The subjects were instructed to apply the moisturizer twice per day, and were evaluated at baseline and after 4, 8, and 12 wk of product use. A vehicle control was not used in the study. Digital photography was used to document changes in appearance. At weeks 8 and 12, statistically significant (P < 0.001) improvement in the following parameters was reported: fine lines, wrinkles, clarity/brightness, visual roughness, tactile roughness, evenness of skin tone (redness), evenness of skin tone (hyperpigmentation), and overall appearance. Levels of improvement for the parameters evaluated ranged from 89% to 100%, with most of the improvement in the 97% to 100% range. The moisturizer was well-tolerated by the panelists, ie, there was no statistically significant increase in scores for tolerability parameters at all time points, when compared to baseline scores. Details relating to the tolerability parameters evaluated were not included.
A single-center study was performed to assess the efficacy and tolerance of a dual-product regimen containing a 0.5% retinol treatment and an anti-aging moisturizer containing 30% Tetrahexadecyl Ascorbate. 70 In addition to encapsulated retinol, the 0.5% retinol treatment contained bakuchiol and Ophiopogon japonicas root extract. In addition to 30% Tetrahexyldecyl Ascorbate, the anti-aging moisturizer also contained vitamin E and coenzyme Q10. The dual-product regimen was used over a 12-wk period by 44 women who had mild-to-moderate facial hyperpigmentation and photodamage. At the baseline visit, the subjects were instructed to apply the anti-aging moisturizer to the entire face once per day in the morning after cleansing. For the first 2 wk of the study, the subjects were instructed to apply the 0.5% retinol treatment to the entire face every other evening. After the 2-wk period, the subjects were instructed to apply the retinol treatment every evening. Tolerability parameters were assessed, at baseline and weeks 4, 8, and 12, and included: erythema, dryness, scaling, burning, stinging, and itching. Use of the dual-product regimen resulted in a statistically significant increase (worsening) in clinical grading scores for dryness on the face at weeks 4 (15% of the subjects) and 8 (13% of the subjects) when compared to baseline scores. However, this change did not persist to the week 12 time point. No statistically significant changes from baseline were detected for the following at weeks 4, 8, and 12: erythema, scaling, burning, stinging, or itching.
Ascorbyl Tetraisopalmitate
A clinical test was performed to clarify the effect of a Ascorbyl Tetraisopalmitate cream on UVB-induced skin pigmentation. 71 Twenty-two males and females with characteristic Japanese photo-skin type II or III were enrolled in the study. The inner side of the upper arm was used for testing as the site of sun-protected skin. The subjects were exposed to 1.5 minimal erythema dose (MED) of solar-simulated light. Following exposure, a cream containing 3% Ascorbyl Tetraisopalmitate or vehicle only (oil-in-water type cream) was topically applied to the UV-irradiated area immediately after irradiation. After 1, 2, and 3 wk, intensities of pigmentation were evaluated with L* value (parameter for lightness of skin; measured with chromameter) measurement. Based on visual scoring, statistically significant differences between vehicle-treated areas and Ascorbyl Tetraisopalmitate-treated areas were reported 1 wk after UVB irradiation (P < 0.05). ΔL*-values (L* of each week - L* before UV-irradiation) of Ascorbyl Tetraisopalmitate-treated areas were significantly lower than those of vehicle-treated areas at 1 wk and at 2 wk after UVB irradiation (P < 0.05). It was concluded that the topical application of a 3% Ascorbyl Tetraisopalmitate cream suppressed pigmentation after UVB irradiation.
Ascorbyl Palmitate
Ascorbyl Palmitate was applied to the skin, damaged by ultraviolet radiation, of 5 subjects. 72 In the first experiment, areas of the skin (forearm) were either left unprotected or had been treated topically with 3% Ascorbyl Palmitate (in a lecithin gel base) prior to UVB exposure (1 to 3 times the MED). When compared to untreated skin, either the absence of erythema or decreased erythema was observed after pretreatment with 3% Ascorbyl Palmitate before UVB exposure. In the second experiment, erythema was produced by UVB exposure in the range of 1 to 2 times the MED and the skin was treated topically with a 5% Ascorbyl Palmitate lotion 3 h later. When compared to untreated skin, the UVB-induced erythema resolved approximately 50% sooner in skin treated with the lotion.
Summary
The safety of the following 7 ingredients, ie, alkylated or acylated derivatives of ascorbic acid, in cosmetics is evaluated in this safety assessment: Tetrahexyldecyl Ascorbate, Ascorbyl Isostearate, Ascorbyl Linoleate, Ascorbyl Tetraisostearate, Ascorbyl Palmitate, Ascorbyl Dipalmitate, and Ascorbyl Stearate. The reported functions of these ingredients in cosmetic products include: antioxidants; skin-conditioning agents; skin protectants; fragrance ingredients; and skin bleaching agents. Ascorbyl Palmitate is the only ingredient with an additional function of fragrance ingredient, and Ascorbyl Linoleate is the only ingredient with an additional function of skin bleaching agent; however, functioning as a skin bleaching agent is not a cosmetic use and, therefore, the Panel did not evaluate safety for that use.
According to 2017 VCRP data, the greatest reported use frequency is for Ascorbyl Palmitate (2557 formulations, mostly in leave-on products), followed by Tetrahexyldecyl Ascorbate (536 formulations, mostly leave-on products). The results of a concentration of use survey provided in 2016 indicate that Ascorbyl Dipalmitate has the highest maximum concentration of use; it is used at concentrations up to 20% in leave-on products (face powders). The maximum concentration of use in rinse-off products is being reported for Tetrahexyldecyl Ascorbate (concentrations up to 2.5% in shaving cream).
Skin penetration data from in vitro studies indicated that Ascorbyl Palmitate was delivered mainly to the epidermis, but was not found in the receptor fluid. Ascorbic acid levels in pig skin were not significantly increased after Ascorbyl Palmitate was applied to skin positioned in a semi-occlusive chamber. In an in vivo percutaneous absorption study involving rabbits, 6 different oil-in-water cream bases containing 4% Ascorbyl Dipalmitate were applied to the skin. The concentration of ascorbic acid in the urine varied depending on the characteristics of the cream bases that were tested.
Ascorbyl Palmitate (10 mg in ethyl alcohol) penetrated into the epidermis after dermal application to human subjects. When the absorption of Ascorbyl Tetraisopalmitate through human skin was simulated using stratum corneum and infundibulum membrane models, the Ascorbyl Tetraisopalmitate molecule was found to have more affinity toward the stratum corneum than toward the infundibulum.
In a skin penetration enhancement study, the amount of ibuprofen that penetrated the skin after 20 h was dependent upon the vehicle that was used. Values for ibuprofen in isopropanol and Ascorbyl Palmitate vehicles were 2.10 ± 0.25 mg/cm2 and 0.47± 0.05 mg/cm2, respectively.
Ascorbic acid was detected in the urine of guinea pigs at 24 h after oral dosing with Ascorbyl Palmitate or Ascorbyl Dipalmitate. Ascorbyl Palmitate resisted hydrolysis in the brain of rabbits after injection into the internal carotid artery. It penetrated the blood brain barrier and was retained principally in brain tissue as an intact molecule.
An acute oral LD50 of >2000 mg/kg was reported in a study involving rats. There were no clinical signs of toxicity, and none of the animals died. Using in silico methodology, Ascorbyl Palmitate and Ascorbyl Stearate were predicted to be moderately toxic and slightly toxic compounds, respectively.
Using in silico methodology, the NOEL for long-term toxicity was calculated to be 916 mg/kg/day for Ascorbyl Palmitate and 834 mg/kg/d for Ascorbyl Stearate.
Ascorbyl Palmitate and Ascorbyl Stearate were not predicted to be reproductive toxicants using in silico methodology.
Ascorbyl Tetraisopalmitate was non-genotoxic in the Ames test, with and without metabolic activation. Using in silico methodology, Ascorbyl Palmitate was predicted to be a genotoxic compound, whereas Ascorbyl Stearate was not predicted to be a genotoxic compound.
Ascorbyl Palmitate (1 μmol and 4 μmol) inhibited chrysarobin-induced tumor promotion in female SENCAR mice, but the authors noted that there was no good dose-related effect. At concentrations of 1 μmol and 4 μmol, Ascorbyl Palmitate reduced the average number of papillomas per mouse by 48% and 44%, respectively. Furthermore, the number of papillomas per mouse in both groups of 30 mice treated with Ascorbyl Palmitate was significantly lower (P < 0.01) than the tumor response in the group that was promoted with chrysarobin alone.
The treatment of HSC-4 cultures with Ascorbyl Palmitate (100 μM) at 37°C yielded a cell survival rate of 86.8 ± 5.7%. At 42°C, cell viability decreased to 42.0 ± 2.1% (P < 0.0001). Thus, the carcinostatic activity of Ascorbyl Palmitate was markedly increased with hyperthermia. Ascorbyl Stearate (doses up to 150 μM) had antiproliferative and apoptotic effects on T98G cells, probably through modulation of IGF-IR expression and the facilitation of programmed cell death. The treatment of human pancreatic carcinoma cells with Ascorbyl Stearate (50-200 μM) resulted in a dose-dependent inhibition of cell proliferation. Ascorbyl Stearate (25-150 μM) also caused a dose-dependent inhibition of cell proliferation in human ovarian epithelial cancer cells.
The pretreatment of human keratinocytes with Ascorbyl Palmitate (300 mM) prior to UVB irradiation resulted in significant increases in lipid peroxidation, when compared to UVB irradiation only. In another experiment in the same study, human keratinocytes were loaded with DHR, pretreated with 1 to 25 μM Ascorbyl Palmitate, and exposed to UVB. Ascorbyl Palmitate effectively inhibited DHR oxidation in a dose-dependent manner, which was indicative of its antioxidant potential. Ascorbyl Palmitate reduced cellular levels of reactive oxygen species after UVB irradiation. Ascorbyl Palmitate also strongly promoted UVB-induced lipid peroxidation, c-Jun N-terminal kinase activation, and cytotoxicity. It was noted that the lipid component of Ascorbyl Palmitate probably contributes to the generation of oxidized lipid metabolites that are toxic to epidermal cells.
An Ascorbyl Tetraisopalmitate trade name material (0.5 mL) was non-irritating to the skin of 3 rabbits. In the maximization test, application of the same material (undiluted) to the skin of 10 guinea pigs resulted in strong sensitization (skin sensitization rate of 80%). In an HRIPT involving 102 subjects, an Ascorbyl Tetraisopalmitate trade name material (10% in silicone) was a non-irritant and non-sensitizer.
The skin sensitization potential of a face powder containing 20% Ascorbyl Dipalmitate (tested at a concentration of 30%; therefore, effective concentration = 6%) was evaluated in the maximization assay using 25 healthy male and female adult volunteers. It was concluded that the face powder did not possess detectable contact sensitivity potential.
In a study involving 5 human subjects, either the absence of erythema or decreased erythema, compared to untreated skin, was observed after pretreatment with 3% Ascorbyl Palmitate before UVB exposure. When the same subjects were exposed to UVB, followed by topical treatment with 5% Ascorbyl Palmitate lotion, the UVB-induced erythema resolved approximately 50% sooner when compared to untreated skin.
When a newly formulated vitamin C complex containing 10% ascorbic acid and 7% Tetrahexyldecyl Ascorbate was applied to the face of 10 patients for 12 wk, there was neither clinical nor histologic evidence of inflammation; however, there was evidence of new collagen formation. In a clinical trial involving 37 subjects, a moisturizer containing Tetrahexyldecyl Ascorbate (concentration not stated) was said to have been well-tolerated. The moisturizer was applied twice per day for 12 wk.
A single-center study involving 44 subjects was conducted to assess the efficacy and tolerance of a moisturizer containing 30% Teterahexyldecyl Ascorbate. The product was applied over a 12-wk period. No statistically significant changes from baseline were detected for the following tolerability parameters: erythema, scaling, burning, stinging, or itching. A clinical test involving 22 subjects was performed to clarify the effect of a cream containing 3% Ascorbyl Tetraisopalmitate on UVB-induced skin pigmentation. The cream was applied following UVB irradiation. Pigmentation intensity was evaluated for up to 3 wk post-application. It was concluded that topical application of the cream after UVB irradiation resulted in the suppression of pigmentation.
Discussion
A study reported that Ascorbyl Palmitate strongly promoted UVB-induced lipid peroxidation in human keratinocyte cultures, and the author suggested that Ascorbyl Palmitate may intensify skin damage by this mechanism following exposures to UV radiation. However, the Panel characterized the results of this study as an artifact of an irrelevant model, and disagreed with the author’s interpretation of the results. Furthermore, the results of this study were not consistent with the results of a clinical study in which topical application of Ascorbyl Palmitate prior to UVB exposures resulted in decreased or no erythema (3% Ascorbyl Palmitate cream) or enhanced resolution of UVB-induced erythema (5% Ascorbyl Palmitate cream).
The Panel also reviewed the results of a study in which computational chemistry analyses were conducted to predict the toxicity of some of the esters of ascorbic acid. They agreed that the results of these analyses appeared to be consistent with the safety profile for Ascorbyl Palmitate and Ascorbyl Stearate, although an older methodology was used and few details of the analyses were presented. The Panel found that the information provided was insufficient to explain why the predictions differed for these 2 ingredients, and determined that the predictions were not very helpful for informing the safety assessment.
The Panel noted the absence of developmental and reproductive toxicity animal data on ethers and esters of ascorbic acid from this safety assessment, but agreed that pertinent data on analogous chemicals from prior Panel safety assessments (i.e., on ascorbates, ingredients with fatty acyl chains, acyl glycerols, fatty acyl/alkyl saccharides, and sorbitan esters) can be used to support the safety of these ingredients in relation to any concerns about developmental and reproductive toxicity.
In addition to the reproductive toxicity in silico predictions for Ascorbyl Palmitate and Ascorbyl Stearate, the Panel proposed that safety data for analogous compounds previously reviewed could be used in a weight of evidence approach, and that these data should be referenced in this safety assessment. The analogous compounds comprise ascorbates, fatty acids derivatives, acyl glycerols, fatty acyl/alkyl saccharides, and sorbitan esters.
An undiluted Ascorbyl Tetraisopalmitate trade name material was a sensitizer in the guinea pig maximization test. However, the Panel was not concerned about the sensitization potential of the ingredients in this safety assessment, given the negative human skin sensitization data on a 10% dilution of the trade name material and negative results for a face powder tested at a concentration of 6% Ascorbyl Dipalmitate in a human maximization test.
Because the several functions of Ascorbyl Linoleate in cosmetic products include use as a skin bleaching agent, the Panel emphasized that Ascorbyl Linoleate must not have this effect at use concentrations in cosmetic products.
Finally, the Panel discussed the issue of incidental inhalation exposure, as some of the ethers and esters of ascorbic acid are used in cosmetic sprays and could possibly be inhaled. For example, Tetrahexyldecyl Ascorbate is being used in tonics, dressings, and other hair grooming aids (aerosol) at maximum use concentrations up to 0.1%. Additionally, the Panel noted that droplets/particles from spray cosmetic products would not be respirable to any appreciable amount. Furthermore, droplets/particles deposited in the nasopharyngeal or bronchial regions of the respiratory tract present no toxicological concerns based on the chemical and biological properties of these ingredients. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic effects. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at the Cosmetic Ingredient Review (CIR) website http://www.cir-safety.org/cir-findings
Conclusion
The Expert Panel for Cosmetic Ingredient Safety concluded that the following 7 ethers and esters of ascorbic acid are safe in cosmetics in the present practices of use and concentration described in this safety assessment: Tetrahexyldecyl Ascorbate Ascorbyl Isostearate* Ascorbyl Linoleate Ascorbyl Tetraisopalmitate Ascorbyl Palmitate Ascorbyl Dipalmitate Ascorbyl Stearate *Not reported to be in current use. Were the ingredient in this group not in current use to be used in the future, the expectation is that it would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.
Author Contributions
Johnson, W. contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted manuscript; Boyer, I.J. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Heldreth, B. contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted manuscript, and critically revised manuscript; Bergfeld, W.F., Belsito, D.V., Hill, R.A., Klaassen, C.D., Liebler, D.C., Marks, J.G., Shank, R.C., Slaga, T.J., and Snyder, P.W. contributed to conception and design, contributed to analysis and interpretation, and critically revised manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Author’s Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.