Abstract
Secukinumab, a fully human monoclonal antibody, is a biological agent that targets interleukin-17A. Secukinumab is used in the management of the common dermatological entity - plaque-type psoriasis and its various types, namely psoriatic arthritis, hypertrophic palmoplantar psoriasis and generalized pustular psoriasis. Other less common indications of this popular interleukin -17A inhibitor, secukinumab include ankylosing spondylitis, rheumatoid arthritis, systemic lupus erythematosus, Familial Mediterranean fever, and tumor necrosis factor receptor-associated periodic syndrome (TRAPS). This review article was written by referring to various review articles, original articles, and some books related to highly regarded databases, such as the Web of Science, PubMed, and Scopus. The keywords explored during review of literature consisted of combinations of the following words: human monoclonal antibody, IL-17A, and biologicals. The authors with this in-depth review hope to explore the working of this versatile biological, Secukinumab, especially as a silver lining in dermatology.
Introduction
Secukinumab is a fully human monoclonal antibody that targets interleukin-17A and hence is helpful in the management of psoriasis, psoriatic arthritis, and ankylosing spondylitis. In the upcoming era of biologicals, secukinumab has secured a top spot in the therapeutic options available for psoriasis, with its negligible adverse effects and high tolerability. The fact that it shows a good response has been beneficial to both the dermatologist and the patient, paving the way for many more biologicals to be added to the already available resources. This review explores the various aspects of this simple biological, secukinumab in detail.
Discovery
It is a fully recombinant human immunoglobulin G1/kappa monoclonal antibody made from Chinese hamster ovary cells. Novartis Pharmaceutical Corporation in Switzerland discovered this drug. The first publication was a Phase I trial published in 2010. 1 The USA Food and Drug Administration (FDA) approved secukinumab (under the brand name, Cosentyx) in January 2015 to treat patients with moderate to severe plaque-type of psoriasis. Secukinumab was the first interleukin-17A inhibitor to be approved. FDA approved the treatment of adults presenting with ankylosing spondylitis and psoriatic arthritis in January 2016. A Label update was agreed to be included for the management of moderate to severe scalp psoriasis in February 2018. 2
Action of IL-17A
Secukinumab acts by preventing the interaction of Interleukin-17A (IL-17A) with the IL-17 receptor by targeting IL-17A. IL-17A is produced from various cell types of both innate and adaptive immune responses like neutrophils, macrophages, mast cells, T-helper 17, and cytotoxic T-cells. 3 It occurs naturally in inflammatory and immune pathways. The frequency of interleukin-17 (IL-17) producing cells is markedly increased in the subchondral bone marrow of the facet joints in ankylosing spondylitis.
Mechanism of Action of Secukinumab
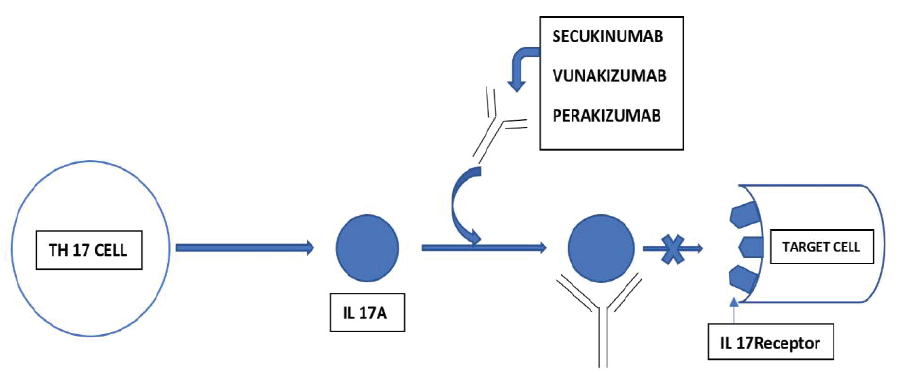
It specifically binds to IL-17A (Figure 1) and neutralizes it, thereby preventing the interaction with IL-17 receptors, which are found on fibroblast-like endothelial cells, keratinocytes, synoviocytes, osteoblasts, and chondrocytes 4 and thus the release of chemokines, pro-inflammatory cytokines, and tissue damage mediators are inhibited. Hence, in certain autoimmune and inflammatory diseases, the contribution of IL-17A is reduced because of the usage of Secukinumab. An adequate amount of this drug reaches the skin and reduces local inflammatory markers. There is a tremendous reduction of erythema, induration, and desquamation in plaques of psoriasis after treatment with Secukinumab.
Mechanism of Action of Secukinumab.
Pharmacokinetics
Intravenous (IV) and subcutaneous (SC) dosing were done in healthy volunteers to evaluate the pharmacokinetics of Secukinumab. Peak drug concentration was achieved between five and six days after a single SC dose (150 mg or 300 mg). A twofold increase in peak concentrations after repeated monthly dosing with a steady-state reached after 20 weeks. The average bioavailability of subcutaneous secukinumab was around 73%, similar to immunoglobulin G1 human monoclonal antibodies. 5 Metabolism of secukinumab is via intracellular catabolism, which is then broken down into amino acids and small peptides, which is the reason for limited drug interactions. In addition, hepatic metabolizing enzymes are not involved in eliminating monoclonal antibodies, thus decreasing the chances of interactions with other drugs. 6 The mean elimination half-life was roughly about 27 days in volunteers and psoriatic patients. The process of drug elimination is time and dose-dependent and unaffected by race, sex, and disease severity. Various factors, including age (more than or equal to 65 years), renal impairment, and hepatic impairment, do not influence the metabolism and excretion of Secukinumab. 7
USFDA Approved Indications
Hypertrophic palmoplantar psoriasis
Moderate to severe psoriasis
Generalized pustular psoriasis
Ankylosing spondylitis
Psoriatic arthritis
Secukinumab is referred to as “a new kid on the block” by recent literature. The efficacy for psoriatic arthritis management, plaque psoriasis, and ankylosing spondylitis have been demonstrated by many clinical trials. Secukinumab is administered in psoriatic patients who are only partially responsive or completely unresponsive to topical or systemic therapy, as well as when standard modes of therapy are contraindicated or result in adverse events. In unresponsive or partial responsive severe psoriatic patients, combination treatment of biologic agents with other routine systemic drugs has been proposed to improve psoriasis treatment outcomes.
Off Label Indications
Systemic lupus erythematosus, Rheumatoid arthritis, Tumor necrosis factor receptor-associated periodic syndrome (TRAPS), and familial Mediterranean fever.
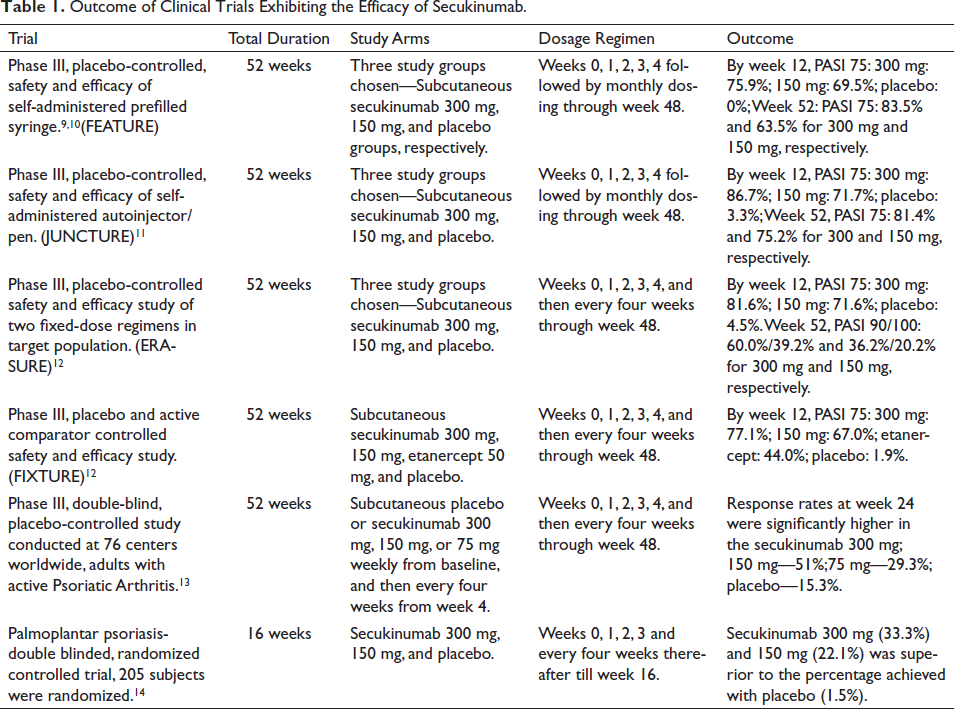
Efficacy of the Drug
Outcome of Clinical Trials Exhibiting the Efficacy of Secukinumab.
Availability of the Drug
It is easily available in the market. The cost of 150 mg/mL of secukinumab is around ₹ 15,000 to ₹ 20,000 per vial in India.
Administration
It is available as a powder (lyophilized) in a vial of 150 mg or in a sensory-ready pen that needs to be stored at a temperature between 2°C and 8°C. The prefilled syringe or pen should be kept for about 20 min to 30 min until the room temperature is reached before administration. The drug is mixed with 10 mL distilled water followed by slow stirring for the powder to dissolve and should be kept aside for 10 min. There should not be any residual particles or discoloration in the solution. The drug should be administered immediately. A specialist physician should administer the drug with routine monitoring of the condition of the disease. The sites such as the thigh, upper arm, and abdomen are chosen for subcutaneous administration of the drug were previously injected sites, inflamed, tender, indurated, bruised, or lesional skin should be avoided. For plaque psoriasis, the recommended dosage is two 150 mg, that is, 300 mg, which is injected subcutaneously at weekly intervals for the first four weeks (for a total of five doses), followed by 300 mg every month 15 and the same dosing is approved for psoriatic arthritis.
Clinical Uses
Secukinumab in Psoriasis
Because of the inhibition of the effects of IL-17A, which is a pro-inflammatory cytokine, there is the standardization of immune function and histology of skin as it interferes with psoriasis disease pathways. 16 Secukinumab neutralizes IL-17A without neutralizing interleukin-17F at curative doses used for psoriasis. In comparison to other therapies, this target gives fewer side effects. The recommended dosage includes two 150 mg, that is, 300 mg, given subcutaneously at weekly interval for the first four weeks, followed by 300 mg every month. 7
Secukinumab in Psoriatic Arthritis
The recommended dose includes
With loading dose—It is 150 mg at weekly interval followed by every month.
Without loading dose—It is 150 mg every month. 17
In case of any persisting activity of the disease, a dose of 300 mg should be considered once a month. Secukinumab may also be given with or without the drug methotrexate.
Secukinumab in Ankylosing Spondylitis
Subcutaneous secukinumab is indicated for the active management of patients with ankylosing spondylitis who failed to respond to conventional therapy. The recommended dosage is 150 mg given at a weekly interval for the first four weeks and every fourth week after that, and on a monthly schedule, if given without any administration of the loading doses. 18 A different site should be chosen for the administration of the drug rather than the previously injected site. Discontinuation of the drug must be done if there is no response after the completion of treatment for 16 weeks. In some partially responsive patients, the continuation of treatment beyond the course of 16 weeks may result in improvement of the condition. 19
Secukinumab in Pregnancy
There is limited data available relating to the use of secukinumab in pregnant women. 15 In a recent study, there were no developmental anomalies found in infants of pregnant monkeys even after administering the drug secukinumab. It was administered during the period of organogenesis with a dose of approximately 30 times the recommended dosage for humans. The possibility of miscarriage and birth deformity after administration of secukinumab is unknown. 20
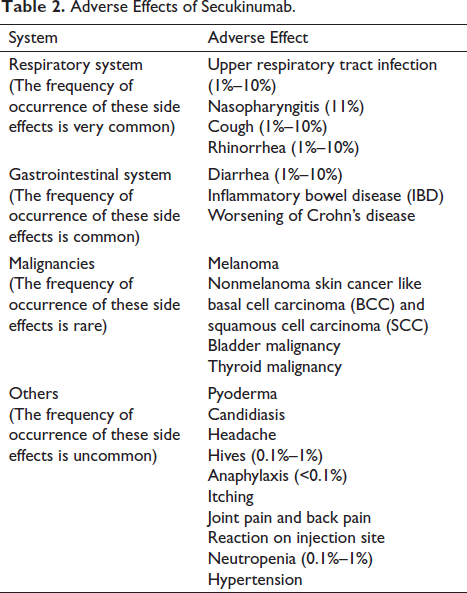
Adverse Effects
Warnings and Precautions
Infections: Infections such as upper respiratory tract infection (URTI), mucocutaneous infections with candida, and nasopharyngitis were seen at a high rate in secukinumab-treated patients. Hence, secukinumab increases the risk of infection, and careful monitoring of the patient is warranted, both prior to and after postadministration of the agent. 22
Tuberculosis patients: Patients must be screened for tuberculosis (TB) infection before initiating the treatment. Secukinumab must not be given to patients with active TB. Management for latent tuberculosis must be initiated before the administration of secukinumab. It is imperative to assess the risk:benefit ratio and consider anti-tubercular therapy before administrating Secukinumab in patients with active, latent or past history of tuberculosis. Regular monitoring of the symptoms of active TB infection must be monitored in patients receiving secukinumab. 23
During lactation: It is not known whether secukinumab is excreted via human milk or systemically absorbed following ingestion.
There is no such data available to date following the effect of secukinumab on a breastfed child.
Inflammatory bowel disease (IBD): Exacerbations were observed in a few cases treated with secukinumab for psoriatic arthritis, plaque psoriasis, and ankylosing spondylitis. Hence, caution must be taken while prescribing secukinumab to patients with IBD. 24
Hypersensitivity reactions: Anaphylaxis and urticaria were observed in patients during clinical trials. If any reaction occurs following the drug administration, further administration should be stopped, and proper therapy must be started.
Risk of hypersensitivity in latex-sensitive individuals: Allergic reaction is observed in latex-sensitive individuals as the cap of secukinumab sensory ready pen, and secukinumab prefilled syringe contains rubber latex.
Vaccinations: All age-appropriate immunization should be completed before planning secukinumab. Live vaccines must not be administered in patients receiving secukinumab. In patients receiving secukinumab, nonlive vaccines might not show an immune response necessary to prevent the disease. There is no proper data currently available on the effect of COVID vaccination on patients treated with secukinumab. 25
Monitoring
Baseline Monitoring in Patients Receiving Secukinumab
Complete blood count (CBC), erythrocyte sedimentation rate (ESR), C-Reactive Protein (CRP), liver function test (LFT), renal function test (RFT) and urinalysis, serum electrolytes, antinuclear (ANA) and anti-dsDNA antibodies, hepatitis screening, HIV screening, testing for tuberculosis such as chest radiograph, tuberculin skin testing, and QuantiFERON Gold test must be done prior to the administration of secukinumab. Pregnancy test must be done on women of childbearing age.
Ongoing Monitoring
Effect of treatment with secukinumab is assessed by scores such as Dermatology Life Quality Index (DLQI), Psoriasis Area and Severity Index (PASI) initially three months and then every six months. CBC is repeated at three months and then every six months thereafter while the patient is on the drug. Renal function tests, LFT, urinalysis, and serum electrolytes evaluations are done at three months from the initiation of the treatment and then every six months. 26
Safety Profile
The risk of candidial and staphylococcal infections is enhanced as secukinumab interferes with the normal immune response.
Few studies mentioned that pregnancy during secukinumab therapy has no risk of abortion or congenital anomalies. Hence, compulsory contraception is not necessary. Studies have stated a few adverse effects if administered during the third trimester of pregnancy because of the drug transfer through the placenta. Hence, the drug is contraindicated during pregnancy and lactation. 27
Carcinogenic potential is another possible adverse effect as a few reports have stated the occurrence of cancers such as melanoma, nonmelanoma skin cancers, bladder cancer, and thyroid cancer.
Conclusion
Secukinumab is considered to be an efficient IL-17A inhibitor, a biologic agent used to manage psoriasis, psoriatic arthritis, and ankylosing spondylitis. It is confirmed to be superior to ustekinumab and etanercept. 28 Secukinumab is known for its rapid rate of response to treatment in psoriatic patients. Improvement is observed within four weeks of treatment and maintained response throughout the treatment process. Secukinumab also shows tremendous results in the management of other types of psoriasis, including psoriatic arthritis. Regarding the safety profile, it is generally well-tolerated and is safer compared to other antipsoriatic biologic and nonbiologic drugs. 29 To summarize, secukinumab provides a better therapeutic response in various forms of psoriasis and psoriatic arthritis and is considered to be an important addition to our psoriasis therapeutic armamentarium.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.