Abstract
Introduction:
Transrectal ultrasonography (TRUS) has long been established as the first-line diagnostic modality for evaluating distal reproductive system obstructions in males. This method provides a valuable anatomical insight but may not fully characterise the functional status of the reproductive organs. Recent advancements, particularly sonoelastography, offer a novel approach for assessing the tissue elasticity and stiffness. Increased testicular stiffness indicates the pathological changes associated with obstructive azoospermia, making it crucial to explore the correlation between duct size and testicular stiffness in the patient population.
Materials and Methods:
This prospective observational study was conducted at a tertiary care hospital in Chennai from March 2021 to December 2023. A total of 50 patients referred for scrotal ultrasound due to azoospermia were included after obtaining informed consent. Each patient underwent both Acoustic Radiation Force Impulse (ARFI) imaging of testes and TRUS.
Results:
The analysis of this study revealed a significant positive correlation between the sizes of the ejaculatory ducts and the ARFI values of the corresponding testis. Specifically, an increase in the size of the right ejaculatory duct was associated with a rise in the ARFI value of the right testis and similar results were also observed for the left side. Additionally, an increase in the testicular volume was noted in cases of obstructive azoospermia.
Conclusion:
This study demonstrated a significant difference in ARFI values between control subjects and those with obstructive azoospermia, indicated a relationship between obstructive azoospermia and increased testicular stiffness. ARFI imaging proved to be a viable noninvasive technique for distinguishing obstructive azoospermia from normal testicular conditions.
Introduction
Azoospermia is defined as the complete absence of sperm cells in semen and is a significant cause of male infertility. It can be classified into obstructive and nonobstructive.[1] Nonobstructive azoospermia often results from the conditions such as primary testicular failure, whereas obstructive azoospermia is typically due to blockages in the reproductive tract and can often be corrected surgically.[2] Distinguishing between these two types is crucial for determining the appropriate treatment approach.[1,2]
Several diagnostic techniques were available to evaluate infertility associated with azoospermia, including testicular biopsy, vasography, hormonal analysis, biochemical marker analysis in semen and various imaging modalities. Among these, transrectal ultrasonography (TRUS) has been conventionally used as the first-line of diagnostic tool for assessing distal reproductive system obstructions in males. TRUS provides a valuable anatomical insight but may not be fully characterise the functional status of the reproductive organs.[2]
Recent advancements in ultrasound technology, particularly sonoelastography, have introduced innovative methods for assessing tissue properties.[3] Sonoelastography allowed for the evaluation of tissue elasticity and stiffness, offering an additional diagnostic information that can enhance the understanding of male reproductive disorders. This technique operates by mechanically exciting tissue with localised, impulsive acoustic radiation forces, leading to the propagation of shear waves. The Virtual Touch Quantification (VTQ) technique combines the radiation forces with an ultrafast imaging sequence to capture the resulting shear wave propagation, providing a quantitative assessment of tissue stiffness.[3]
This study aimed to investigate the correlation between ejaculatory duct sizes and testicular stiffness in patients with obstructive azoospermia, using both ARFI and TRUS to enhance the diagnostic accuracy and to establish the potential therapeutic strategies.
Materials and Methods
This prospective observational study was conducted in the radiology department in a tertiary care hospital, Chennai from March 2021 to December 2023, involving patients referred for scrotal ultrasound due to azoospermia. A total of 50 patients with azoospermia were included based on the hormonal levels and semen analysis reports and also after obtaining informed consent. This study established a clear inclusion and exclusion criteria: Inclusion criteria comprised adult males aged 21–50 years who were diagnosed with azoospermia and willing to provide consent, while exclusion criteria included individuals with acute scrotal conditions, such as torsion, cryptorchidism, extensive testicular microlithiasis and those older than 50 years. Additionally, 50 controlled subjects, matched for age and sex, were recruited to provide comparative data. Each participant underwent both Acoustic Radiation Force Impulse (ARFI) imaging to assess the testicular stiffness and TRUS for anatomical evaluation of the reproductive tract. The resultant data was analysed to identify correlations between ejaculatory duct sizes and ARFI values of the testes, aiming to enhance the understanding of obstructive azoospermia and its implications for male infertility.
Methodology
Patients referred for scrotal sonography who met the inclusion criteria were initially screened for a history of infertility and informed consent was obtained from all the participants. Those with a documented history of infertility who attended for TRUS of the prostate and seminal vesicles showing evidence of obstruction were further subjected to ARFI imaging to assess the testicular stiffness during the same session.
This study was employed the SIEMENS ACUSON 2000 ultrasound system equipped with VTQ software, utilising a 9L4 linear transducer for all imaging procedures. For ARFI measurements, the region of interest (ROI) was carefully positioned on the testis, ensuring that no extra-testicular tissue was included, typically at the centre of the testicular structure.
The measurements were recorded seven times to ensure the reliability, with the highest and lowest values being excluded from analysis to minimise the impact of outliers. The average of the remaining values was then calculated for each testis. Additionally, both ARFI values and TRUS findings for the prostate and seminal vesicles were correlated to hormonal levels and semen analysis reports to establish a comprehensive understanding of the patient’s conditions. This methodological approach aimed to elucidate the relationship between ejaculatory duct size and testicular stiffness in patients with obstructive azoospermia.
Statistical Analysis
All continuous variables were assessed for normality using the Shapiro–Wilk test. If any variable is found to follow a Gaussian distribution, it was expressed as the mean ± standard deviation (SD); otherwise, it was presented as the median with interquartile range. Categorical variables were represented as percentages. To compare continuous variables between groups, an independent samples t-test was employed. Data entry was performed using Microsoft Excel, and subsequent data analysis was conducted using SPSS version 25.0. A significance level of P < .05 was considered statistically significant for all tests conducted. This rigorous statistical analysis plan was designed to ensure the reliability and validity of the study findings.
Results
This study comprised 50 cases of obstructive azoospermia and 50 control subjects, allowing for a comparative analysis of various parameters. The mean age of the cases was 36.22 years (SD ± 7.69), while the mean age of the control was 36.28 years (SD ± 8.14) [Table 1].
In evaluating the echotexture of the testes, we found that all patients (cases and control) exhibited a normal echotexture for both right and left testes. The mean volume of the right testis in cases was 14.82 cm³ (SD ± 4.06), compared to 12.97 cm³ (SD ± 3.12) in controls, indicated that obstructive azoospermia was associated with an increase in testicular size. Similarly, the mean volume of the left testis in cases was 15.35 cm³ (SD ± 3.77). In contrast, controls had a mean volume of 12.96 cm³ (SD ± 2.95), further supporting the conclusion of obstructive azoospermia results in increased testicular volume [Table 1].
Regarding testicular stiffness, the mean ARFI value for the right testis in cases was 1.73 m/s (SD ± 0.55), significantly higher than the control group’s mean of 0.71 m/s (SD ± 0.07). This suggested that the obstructive azoospermia was associated with an increased stiffness in the testes. The left testis showed a similar pattern, with a mean ARFI value of 1.73 m/s (SD ± 0.51) in cases versus 0.73 m/s (SD ± 0.08) in controls. These findings indicated a strong correlation between testicular stiffness (as measured by ARFI values) and the presence of obstruction in the spermatic pathway, and allowed an effective differentiation between obstructive azoospermia and normal conditions [Table 1].
Age, volume and ARFI parameters in obstructive azoospermia in case and control subjects
An increased size of the epididymal head (>12 mm) was observed exclusively in cases, with 23% of the overall study population exhibited this finding, while the remaining 77% had normal-sized epididymal heads (5–12 mm) [Table 2].
Epididymal heads size variations in study population

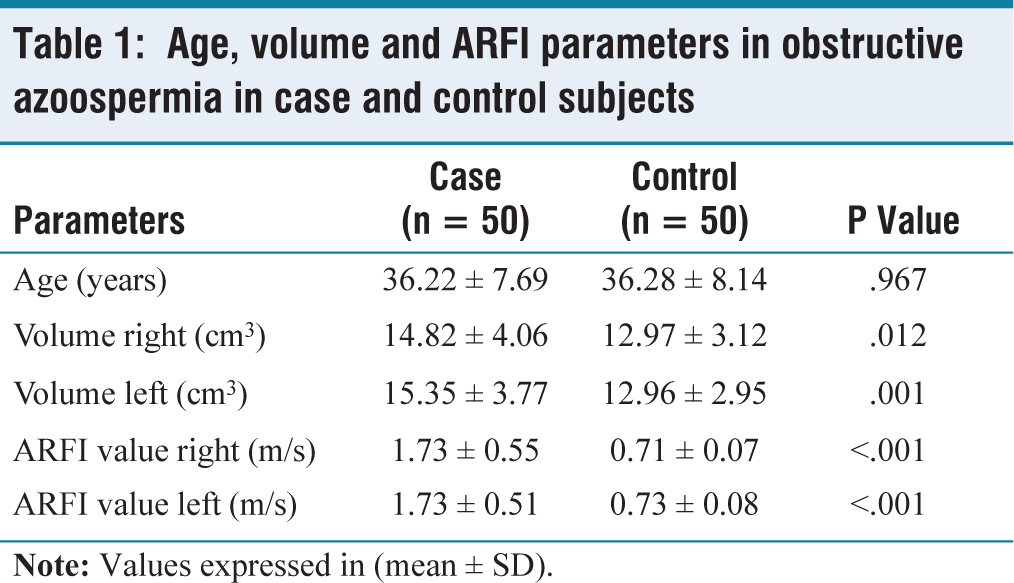
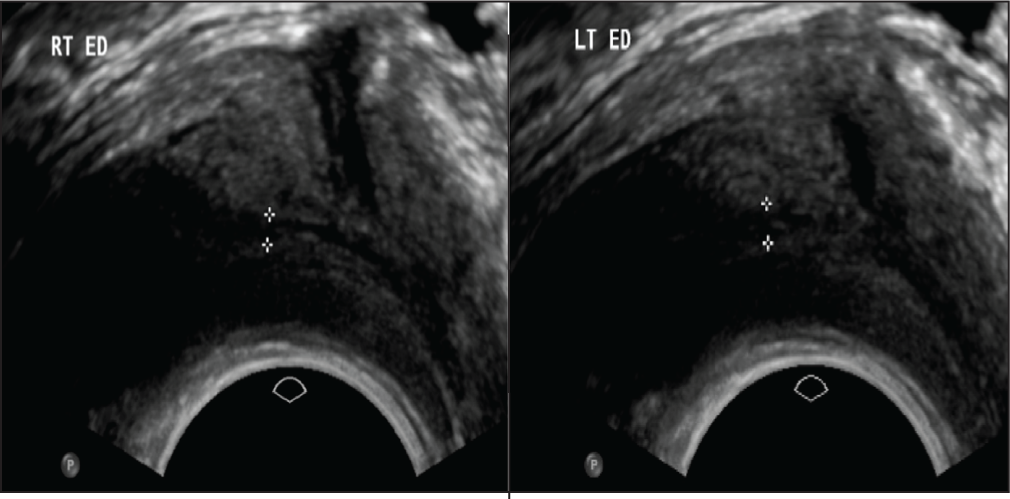
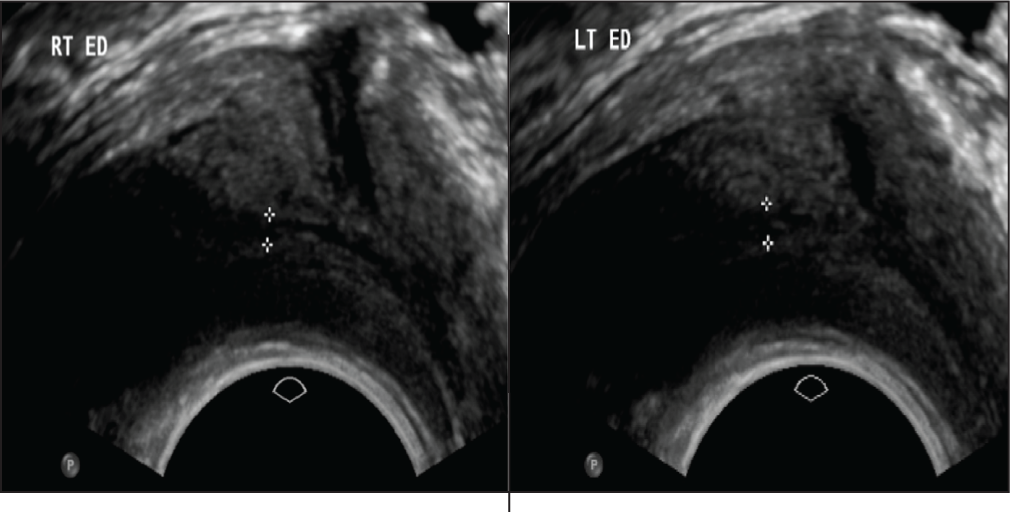
In terms of seminal vesicle assessment, we found that 50% of the total population exhibited the normal seminal vesicles. Among the remaining 50%, 28% were showed enlarged, cystic and thickened right seminal vesicles [Figure 1], 12% were absent and 10% were atrophic. Notably, all controls displayed normal right seminal vesicles, while all cases showed abnormalities. For the left seminal vesicle, the results were similar, with 28% exhibiting enlargement, 12% absent and 10% atrophic [Table 3, Figure 2].
Cross-sectional transrectal image of bilateral thickened seminal vesicles
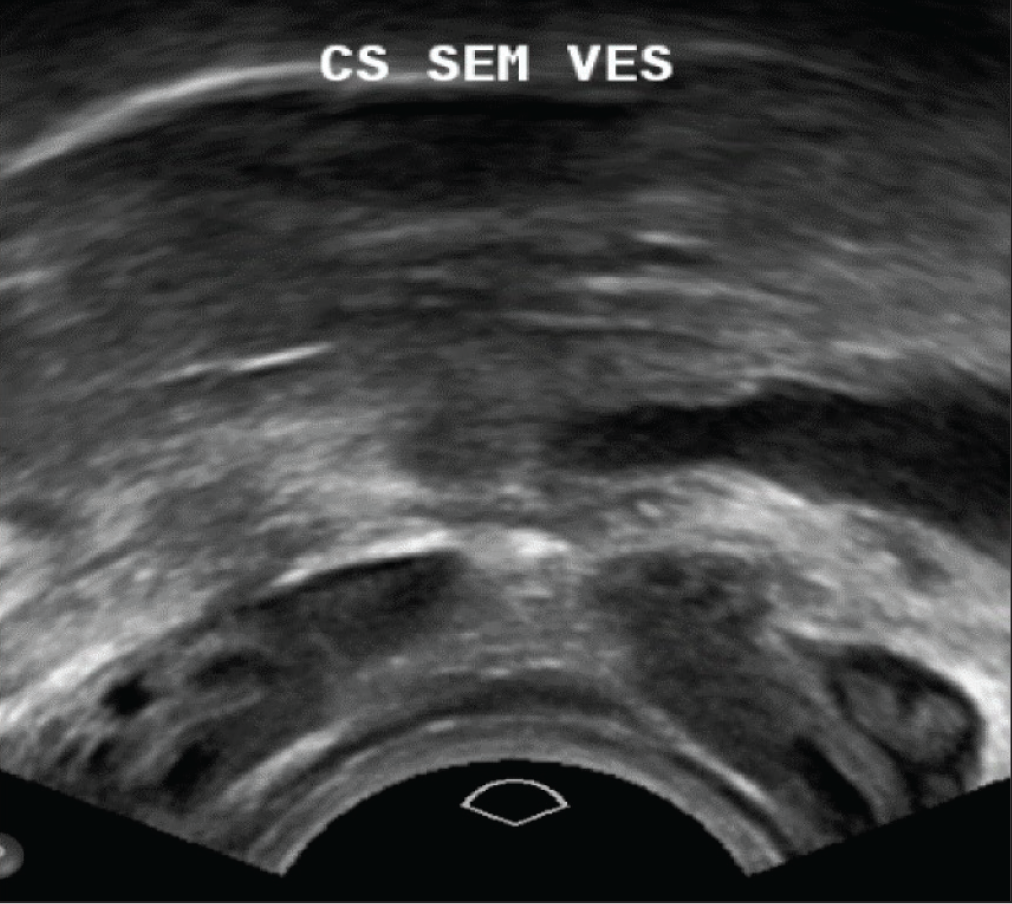
Assessment of right and left seminal vesicles in total populations
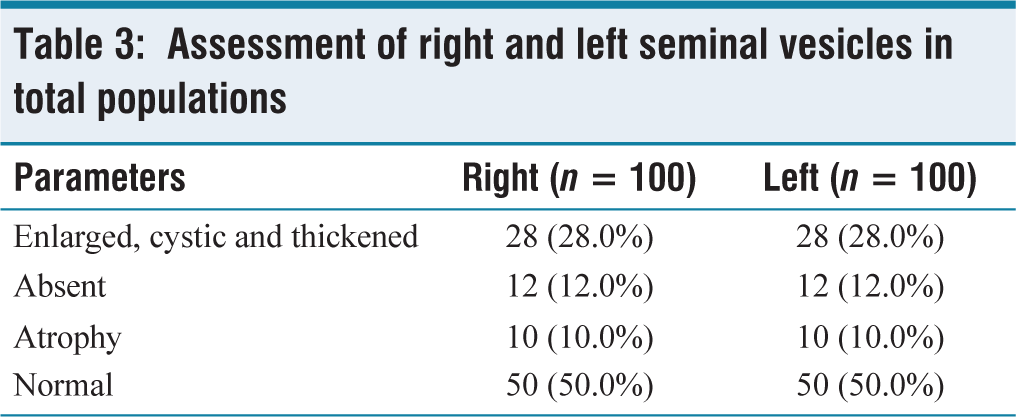
Cross-sectional transrectal image of bilateral enlarged seminal vesicles with thickening and longitudinal transrectal image of right thickened and enlarged seminal vesicles
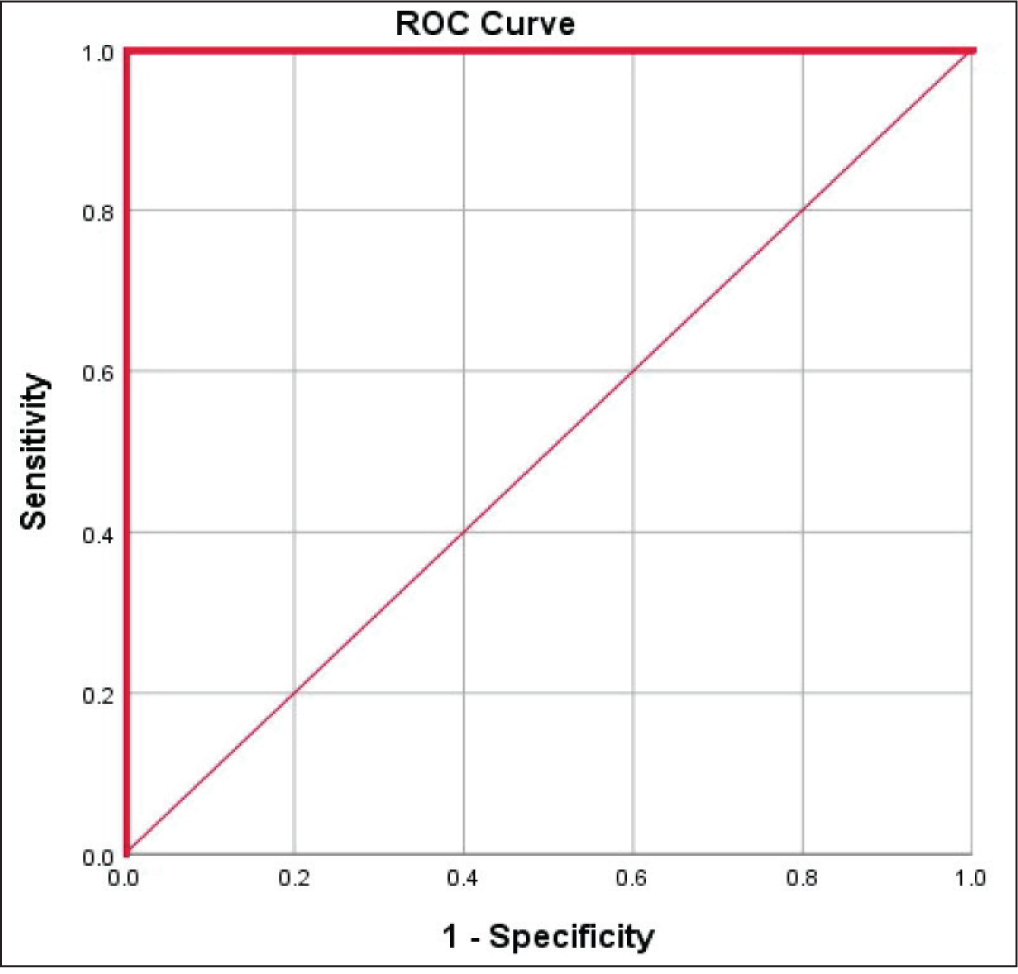
Finally, this study established a cutoff value for ARFI in predicting obstructive azoospermia. The cutoff value for the right testis was 1.01 m/s (range: 0.85–4.0 m/s), with a sensitivity and specificity of 100%. For the left testis, the cutoff was 1.05 m/s (range: 0.85–4.0 m/s), also with a sensitivity and specificity of 100%. These findings underscore the utility of ARFI imaging in diagnosing the obstructive azoospermia [Figure 3].
Sensitivity and specificity of cutoff value for ARFI in predicting obstructive azoospermia
Discussion
In this study, we demonstrated an increased ARFI value (1.73 m/s mean and 0.85–3.5 m/s range) in the population with an evidence of obstructive azoospermia on TRUS, which indicated chronic obstruction to the semen pathway causing inflammatory changes at the testis leading to increased stiffness. On comparison with Du et al.[1], this study showed that the strain ratio significantly differed for nonobstructive and obstructive azoospermia (median: 0.490 and 0.340).
As compared to cases, the ARFI value of the testis in the normal population was observed to be lower (0.60–1.0 m/s). If we include age-related changes in a normal population, this range (0.60–1.0 m/s) may be taken as indicative of normality. Our results of normal testis ARFI value in a fertile population were further supported by a study conducted by D’Anastasi et al.[4].
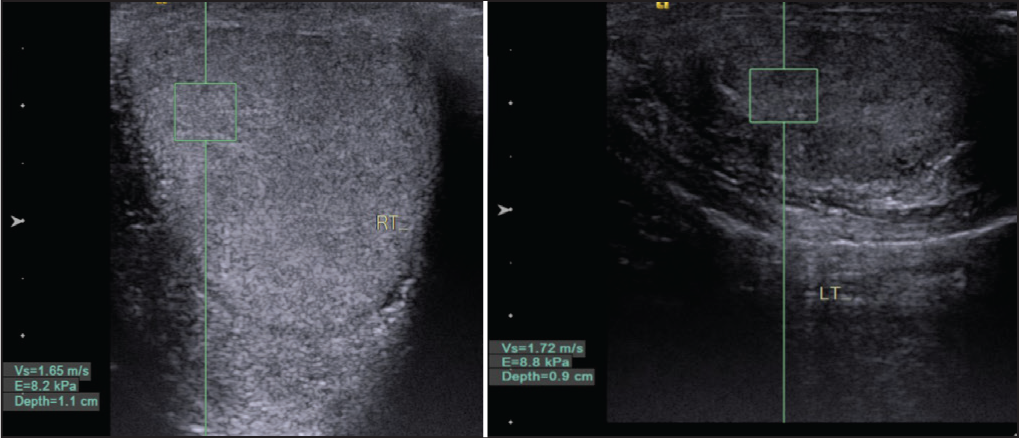
The study by Trottmann et al.[5] showed that real-time shear wave elastography proved feasible in assessing testicular stiffness [Figure 4A and 4B]. Mean shear-wave velocity values were similar in the inferior and superior parts of the testicle (1.15 m/s) and significantly lower in the centre (0.90 m/s). These values were age-independent, similar to our study, and testicular stiffness was significantly lower in the upper pole than in the rest of the testis with increasing volume (P = .007) [Figures 5A and 5B].
(A) Increased right testicular parenchyma ARFI in longitudinal section 1.6 m/sec. (B) Increased left testicular parenchyma ARFI in longitudinal section 1.7 m/sec
(B) Increased right testicular parenchyma ARFI in longitudinal section 1.31m/sec
It is further evident that in patients with obstructive azoospermia, testicular ultrasound is often abnormal. A study performed by Moon et al.[6] found that 86% of cases of obstructive azoospermia had an abnormal ultrasound, with abnormalities associated with obstruction, including tubular ectasia of the epididymal head, tapering of the epididymal tail, absence of the epididymis and the presence of heterogeneous mass along the epididymal course.[6] Finally, various causes of seminal tract obstruction, such as ejaculatory duct pathology [Figure 6] and seminal vesicle pathology, cause a rise in the ARFI value of testicular parenchyma.
A limitation of our study is the absence of histological correlation. However, a biopsy is not possible when studying volunteers with no known testicular pathology. One examiner performed the ultrasound exams, so we did notice inter-observer variability. As ARFI measurements are relatively operator-independent, hence we do not expect a significant difference in results to be obtained by another examiner.
The relatively low number of patients (50) did not allow us to define normal values for every age group. Since testicular stiffness is dependent on age, more subjects would be needed to define normal seminal vesicle wall values for every age group.
Conclusion
ARFI technology has been proven to be a feasible and effective method for evaluating the stiffness of testicular parenchyma, allowing for the differentiation between populations with obstructive azoospermia and those without. The technique is safe, rapid and operator-independent, making it a valuable tool in clinical practice. Our study found a significant association between ARFI values and obstructive azoospermia, indicating that obstructive azoospermia is linked to increased testicular stiffness. Further research with larger sample size may be warranted to establish definitive ARFI value ranges for each group, enhancing the understanding and diagnosis of male infertility related to obstructive conditions.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Institutional Ethical committee approval obtained (1104–2022).
Informed consent
Informed consent has been obtained from the patients.
Credit author statement
MR participated in conceptualisation, data analysis, literature search, investigations, and draft preparation, review and editing. SK and BS participated in conceptualisation, investigation, supervision, data analysis and manuscript editing. NJ participated in methodology and draft editing All the authors reviewed and approved the manuscript.
Data availability
Not applicable.
Use of artificial intelligence
There was no use of artificial intelligence in this research work.
