Abstract
Background and Aims:
Large unstained cells (LUCs) are peroxidase negative mononuclear cells which include large lymphocytes in healthy individuals, peroxidase negative blasts, plasma cells and atypical lymphoid cells. This study aims to assess the causes for elevated LUC% in ADVIA 2120i by rapid peripheral smear review.
Materials and Methods:
All consecutive anticoagulated blood samples with LUC% >4%, from June 2022 to March 2024, were processed in ADVIA 2120i, and demographic factors, LUC parameters and peripheral smear observations were documented under six diagnostic disease categories. Statistical analysis was performed using SPSS software (IBM, 28.0).
Results:
The median LUC count of 0.5 × 10 3 cells/µL with an inter-quartile range of 0.2–2.3 × 10 3 cells/µL of blood was observed in haematolymphoid neoplasms. There was a significant overlap of %LUC values among different categories. The majority (69.6%) had % LUC between 4.1% and 9.9%. Marked elevations were observed in AML, CML, ALL and infections.
Conclusion:
Absolute LUC count could be used as a cost-effective and readily available marker to identify a case of clinically suspicious haematolymphoid neoplasm with LUC count more than or equal to 0.5 × 10 3 cells/µL of blood in conjunction with other complete blood count parameters. However, reflex peripheral smear testing is mandatory in the current setting for confirmation.
Introduction
Large unstained cells (LUCs) are large peroxidase-negative mononuclear cells due to the absence of myeloperoxidase-containing granules in their cytoplasm, and are larger than lymphocytes. In healthy individuals, LUC are mainly large lymphocytes. Still, other cells such as peroxidase-negative blasts, hairy cells, atypical and reactive lymphocytes, atypical lymphoid cells, plasma cells, peroxidase-negative neutrophils and CD34+ve hematopoietic stem cells are also included.[1] Flowcytometry-based ADVIA 2120i auto-analyser estimates the LUC parameters in anticoagulated blood samples, which are a part of WBC differential in the PEROX channel. LUC parameters have been the subject of decades’ worth of studies evaluating their correlation with other inflammatory biomarkers.[2] Our study observed the peripheral blood cells causing elevations in %LUC in various disease conditions, and attempted to correlate peripheral blood smear findings with clinical variables.
Materials and Methodology
This prospective observational study was conducted from June 2022 to March 2024. Blood samples with LUC% more than 4% were included in the study. Clotted, haemolysed and lipemic blood samples, samples with LUC% less than or equal to 4% were excluded. We aimed at observing the reasons behind the increase in %LUC and note the cells which fall under this category, and to correlate clinical and other laboratory parameters with peripheral blood smear findings.
Methodology
Clinical details were obtained from the laboratory information system. EDTA-anticoagulated whole blood samples were processed using the ADVIA 2120i autoanalyser, with reflex peripheral blood smear review for cases showing %LUC > 4%, even when other CBC parameters were within normal range. The peripheral smear slides were stained with Leishman stain, examined and both total and differential cell counts were calculated. Any abnormal increases or decreases in cell-counts, and atypical cells identified were documented.
Patients were classified into six diagnostic groups, which included chronic medical disease, infections, haematolymphoid neoplasms, visceral malignancy, multiple co-existing diseases of different groups and no specific clinical disease. Demographic details, LUC parameters and peripheral smear observations were compared under each diagnostic category.
Statistical Analysis Plan
Descriptive statistics were presented with frequency (percentage) & Mean (SD) for the categorical & continuous factors respectively. Median (IQR) was presented while the LUC parameters followed non-normal distribution. Kruskal Wallis test was used to determine the significant difference in LUC% and LUC count in each diagnostic group. Chi-square/Fisher’s exact test was used to determine the association between two independent categorical factors. A P < .05 indicates statistical significance. All the statistical analyses were done by using SPSS software (IBM, 28.0).
Results
A total of 500 consecutive cases which met the inclusion criteria were included. Age range of study population was widely distributed and included four months—89 years with a mean age of 45.6 ± 23.6 years. 95 (19%) were paediatric cases and 405 (81%) were adult cases. 335 (67%) were males and 165 (33%) were females. Most common clinical presentation was pancytopenia (26%), followed by pyrexia of unknown origin (15%). A total of 10% of cases were known haematolymphoid neoplasms under follow-up.
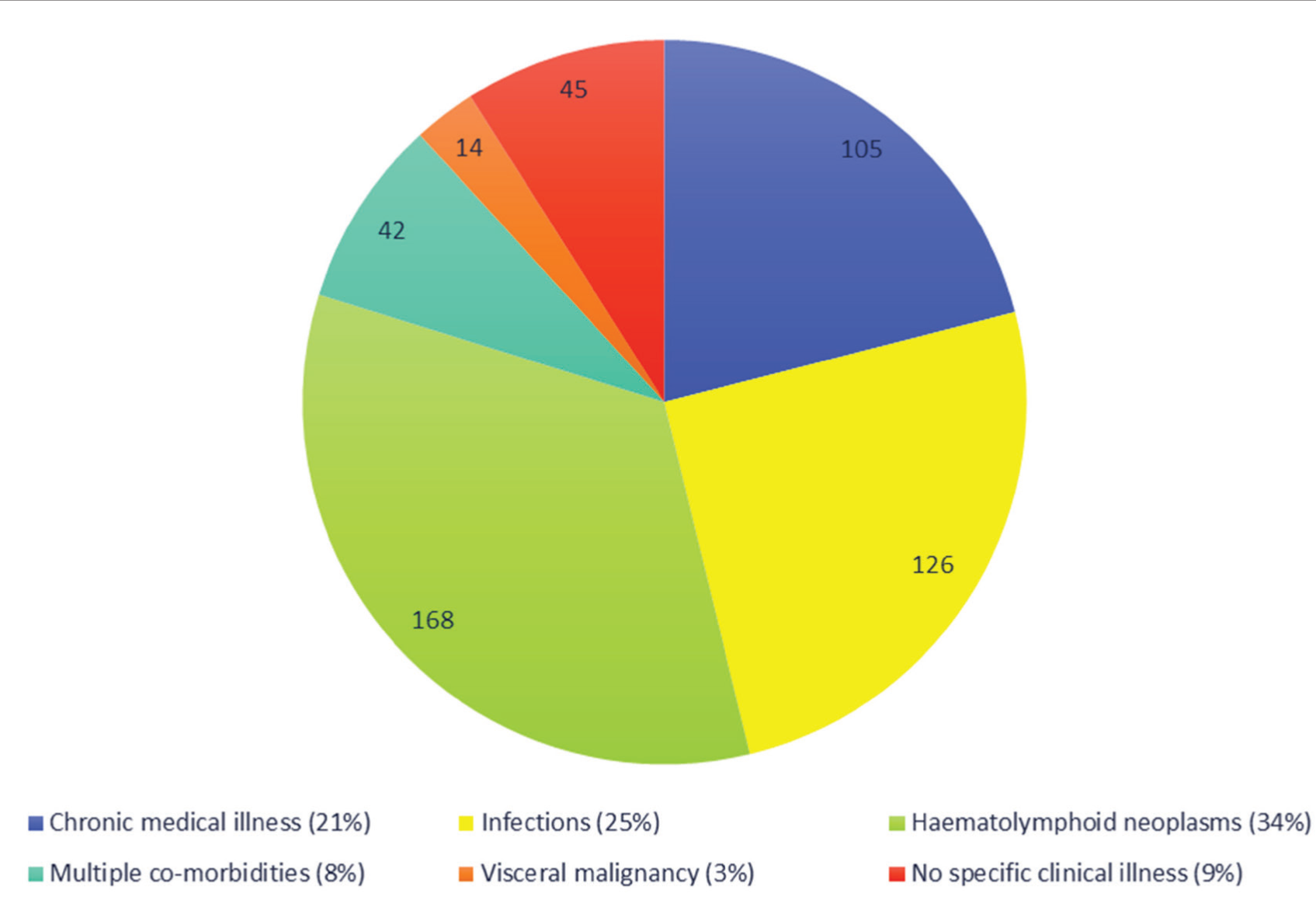
Haematolymphoid neoplasms represented 34% of cases followed by infections (25%), chronic medical disease (21%) and visceral malignancy (3%). A total of 8% had co-existence of diseases of various groups, while 9% had no identifiable diagnostic disease [Figure 1].
Graphical representation of diagnostic categories in study population
LUC Parameters
The average LUC% in the paediatric population (n = 95) was 9.3% ± 6.1% with a median of 7.1% and, in the adult population (n = 405), it was 11.1% ± 12% with a median of 6.4%. Similarly, average LUC count in paediatric population was 0.6 ± 0.5 × 10 3 cells/µL of blood with median of 0.4 × 10 3 cells/µL of blood and, in adult population, the average was 1.9 ± 6.1 × 10 3 cells/µL of blood with a median of 0.4 × 10 3 cells/µL of blood. There was no statistical significance between LUC parameters and the age group of the patients.
%LUC was divided into six arbitrary groups: 4.1%–9.9%, 10%–19.9%, 20%–29.9%, 30%–39.9%, 40%–49.9% and greater than or equal to 50%. Approximately 69.5% cases in paediatric population were clustered in subgroup 1 (%LUC = 4.1%–9.9%), and none of the cases had %LUC >/ = 50%. In adults, subgroup 1 (%LUC = 4.1%–9.9%) had 69.8% cases followed by 17.1% in subgroup 2. Eleven cases in the adult population had %LUC >/ = 50%.
Peripheral Blood Smear Results
Peripheral smear revealed following observations: 38% (n = 190) had lymphocytosis, 33.8% (n = 169) had monocytosis, 23.8% (n = 119) had blasts, 7.8% (n = 39) had plasma cells and 6.2% (n = 31) had atypical lymphocytes. Less common findings include, smudge cells (4%, n = 20), eosinophilia (4.2%, n = 21), hypo-granular neutrophils (4.4%, n = 22), basophilia (3%, n = 15), neutrophilia (2.8%, n = 14), neutropenia (0.8%, n = 4), trophozoites (0.4%, n = 2), hyper segmented neutrophils and lymphopenia (0.2%, n = 1, each). Notably, 13.4% (n = 67) had no peripheral smear abnormalities.
Blood smears in the paediatric population showed monocytes (48.4%), lymphocytes (44.2%) and blasts (6.3%) with 7.4% cases having normal blood smears. In adults, lymphocytes were most common (36.5%) followed by monocytes (30.4%), blasts (27.9%), plasma cells (9.6%) and atypical lymphocytes (7.4%). Fourteen point eight percent of blood smears were normal in the adult population.
Frequency of haematolymphoid neoplasms were as follows: 29.1% (n = 44) AML, 16.6% (n = 25) plasma cell dyscrasia, 14.6% (n = 22) lymphomas, 11.9% (n = 18) Chronic lymphocytic leukaemia, 9.9% (n = 15) Acute lymphoblastic leukaemia, 6% (n = 9) Chronic myeloid leukaemia, 6% (n = 9) Myeloproliferative neoplasms, 6% (n = 9) Myelodysplastic neoplasms [Figure 2]. Majority were adults (98.2%) with a male predominance (69.6%). Seventy percent of atypical lymphoid cells, 69.2% of lymphocytes and 67.8% of blasts fell in this disease group. Notably, 13.23% of normal smears belonged to this group.
Percentage of most observed peripheral blood cells in each morbidity group (X-axis: Diagnostic disease category; Y-axis: percentage of population)
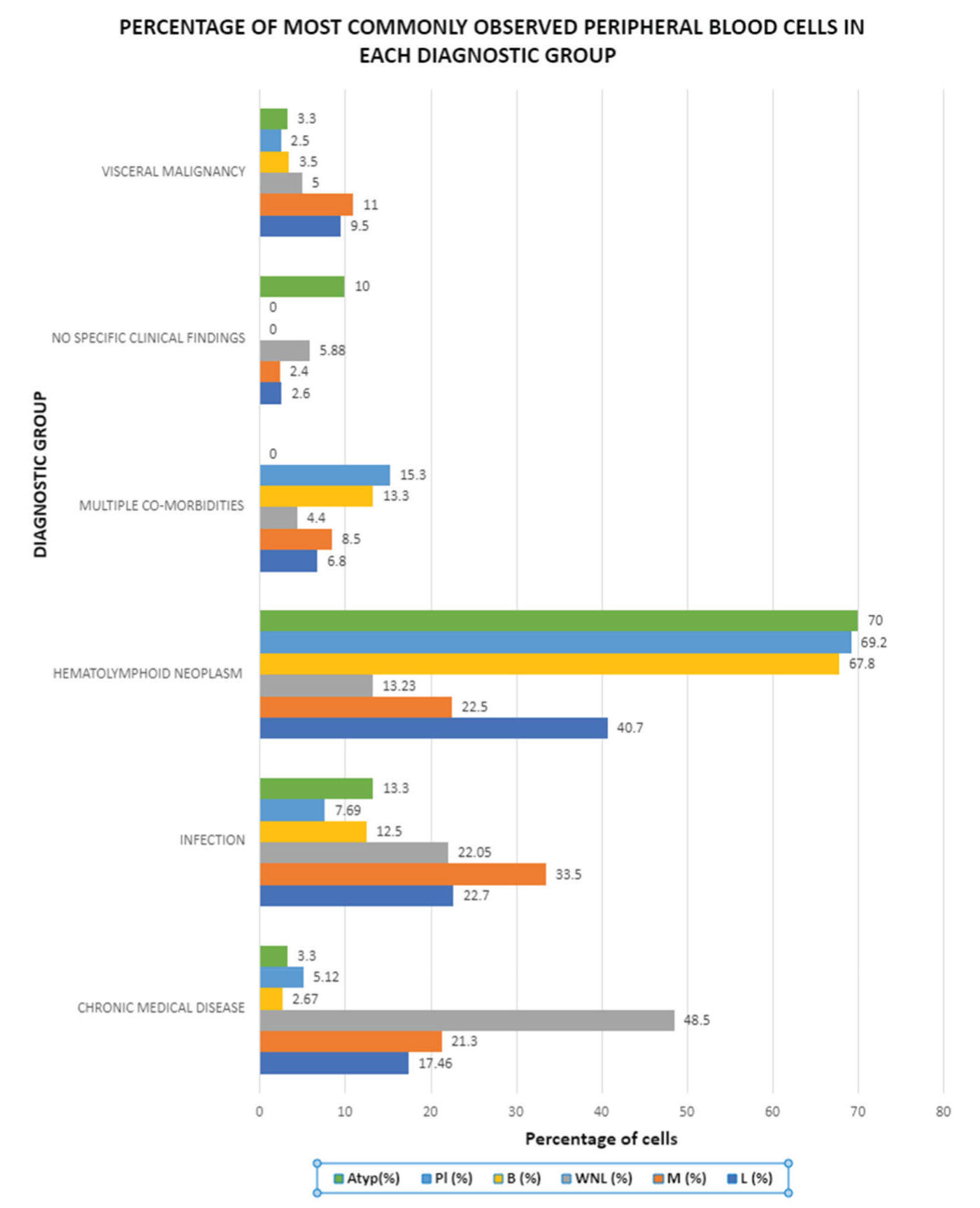
Of 105 chronic medical disease patients, majority were Type 2 diabetes mellitus (66.7%) and %LUC falling between 4.1% and 9.9%. Infections were more common in children (66.6%) with two cases of bacterial sepsis exhibiting highest %LUC of 49% and 41.5%, respectively. Visceral malignancies accounted for the least number of cases (3%) with one case exhibiting %LUC = 50%, which was a case of lung adenocarcinoma undergoing chemotherapy. No specific illness was diagnosed in 9% of cases and was attributed by children with monocytosis (46.3%) and lymphocytosis (29.3%) in peripheral smears. Eight point four percent had multiple co-existing diseases with plasma cells (15.3%), blasts (13.3%), monocytes (8.5%) and lymphocytes (6.8%) in circulation [Figure 3].
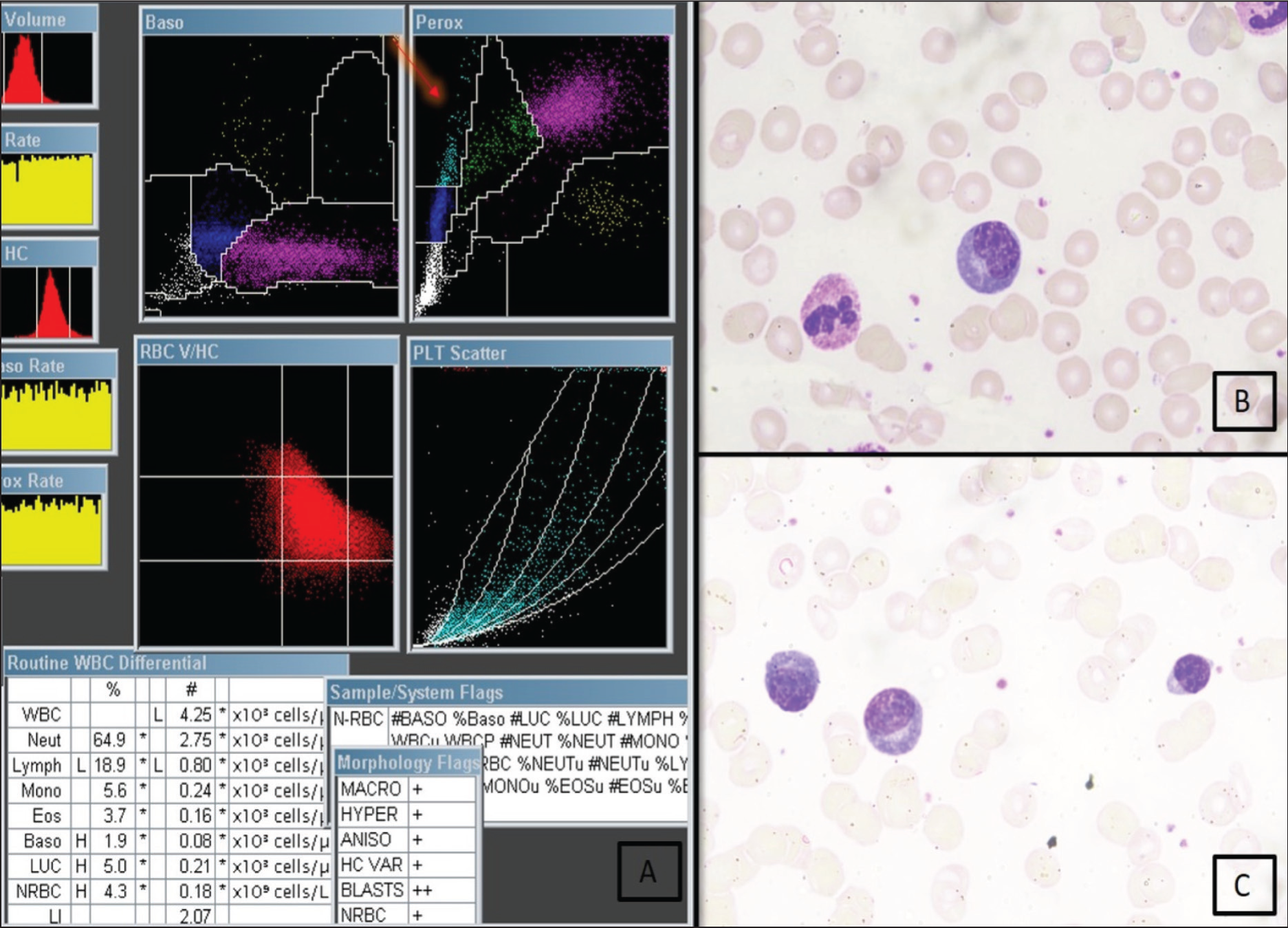
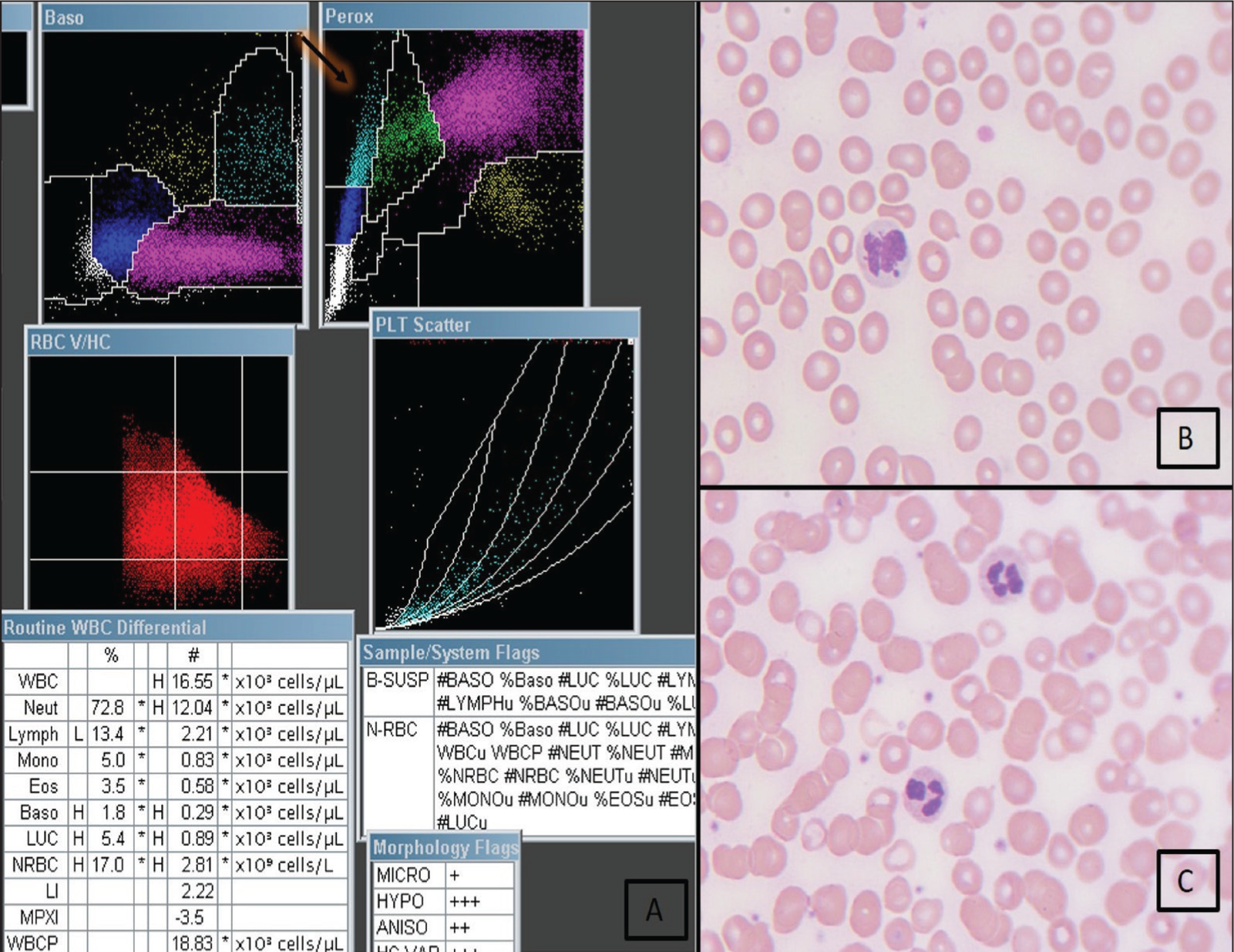
A case of viral infection. (A) PEROX scattergram showing an absolute LUC of 0.21 × 10 3 cells/µL of blood (B) and (C) (40×) Reactive lymphocytes in peripheral smear
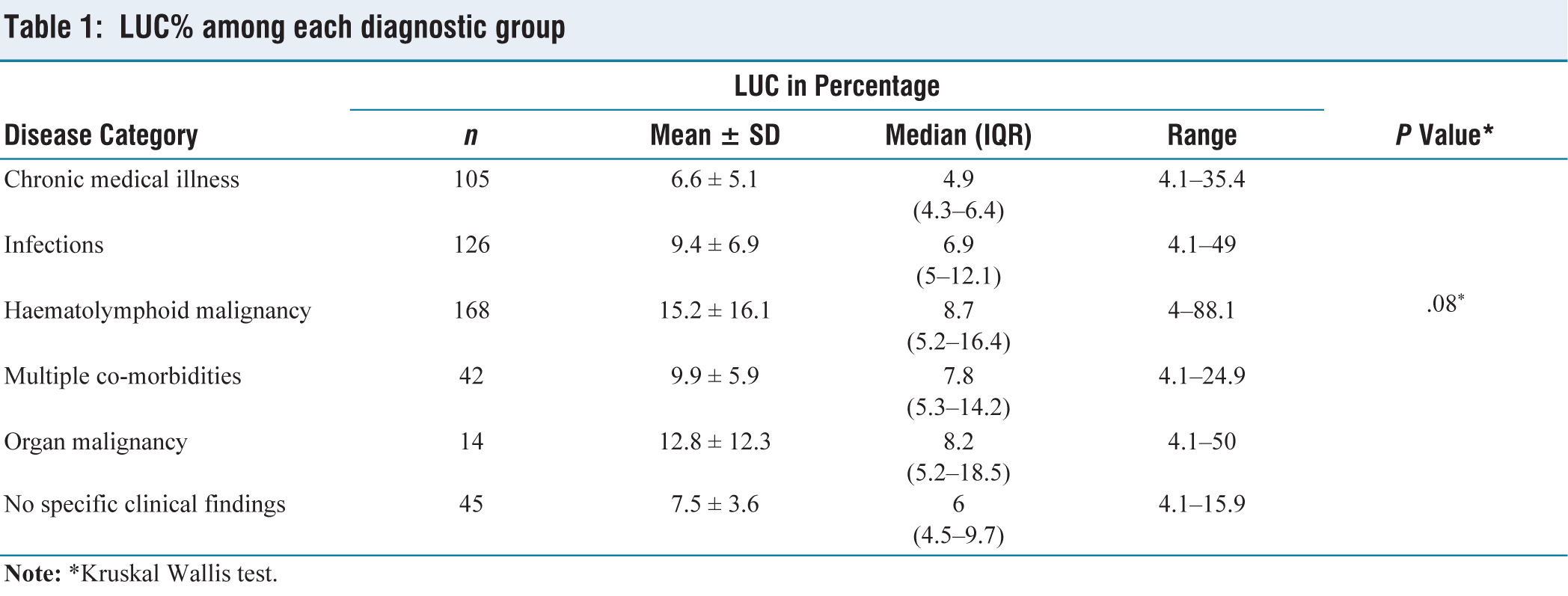
The highest mean and median %LUC were noted in haematolymphoid neoplasm group (15.2% ± 16.1% and 8.7%, respectively), while the lowest were in chronic medical disease group (6.6% ± 5.1% and 4.9% (4.3%–6.4%), respectively). However, the differences in mean and median values among the diagnostic disease groups were not statistically significant [Table 1].
LUC% among each diagnostic group
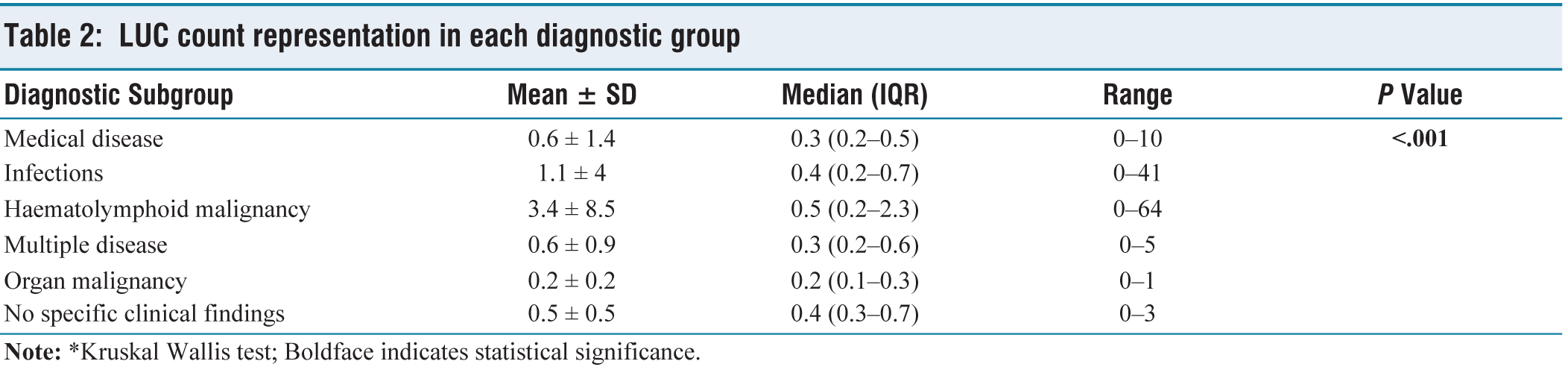
The highest mean and median absolute LUC count was noted in the haematolymphoid neoplasm group [e.g., Figures 3–5] (mean = 3.4 ± 8.5 × 10 3 cells/µL of blood and median = 0.5 × 10 3 cells/µL of blood (0.2–2.3)), while the lowest were in visceral malignancy group (mean = 0.2 ± 0.2 × 10 3 cells/µL of blood and median = 0.2 × 10 3 cells/µL of blood (0.1–0.3)). The difference in median between haematolymphoid neoplasm group and other disease groups was proven to be statistically significant [Table 2].
A case of Acute lymphoblastic leukaemia. (A) PEROX scattergram showing an absolute LUC count of 0.57 × 10 3 cells/µL of blood. (B) Circulating lymphoblasts (20×). (C) Lymphoblasts with round to oval, medium to large sized nucleus with fine chromatin, occasional prominent nucleoli and scant basophilic cytoplasm (40×)
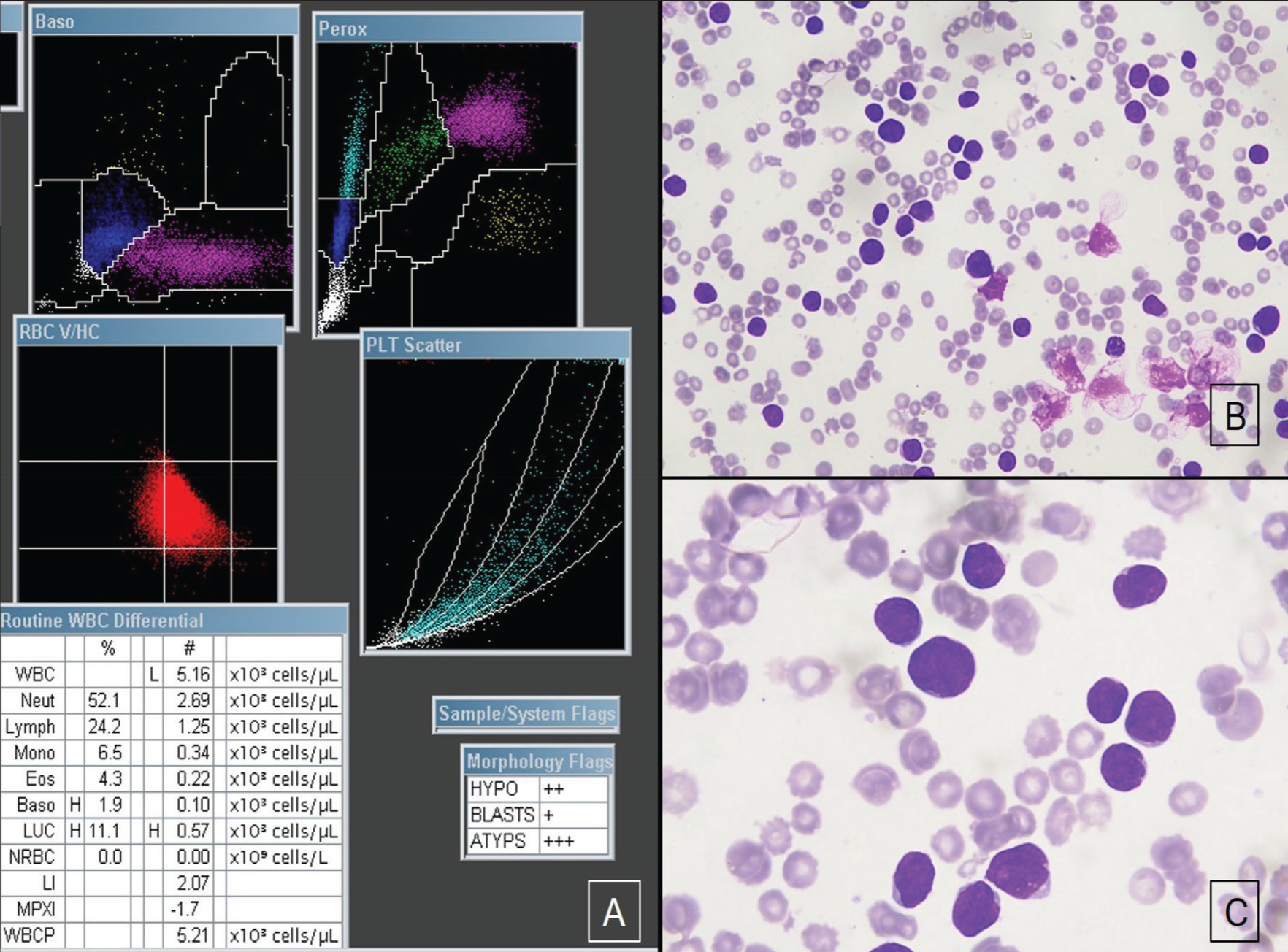
A case of myelodysplastic neoplasm. (A) PEROX scattergram showing an absolute LUC count of 0.89 × 10 3 cells/µL of blood. (B and C) (40×): Hypo-granular neutrophils in peripheral smear
LUC count representation in each diagnostic group
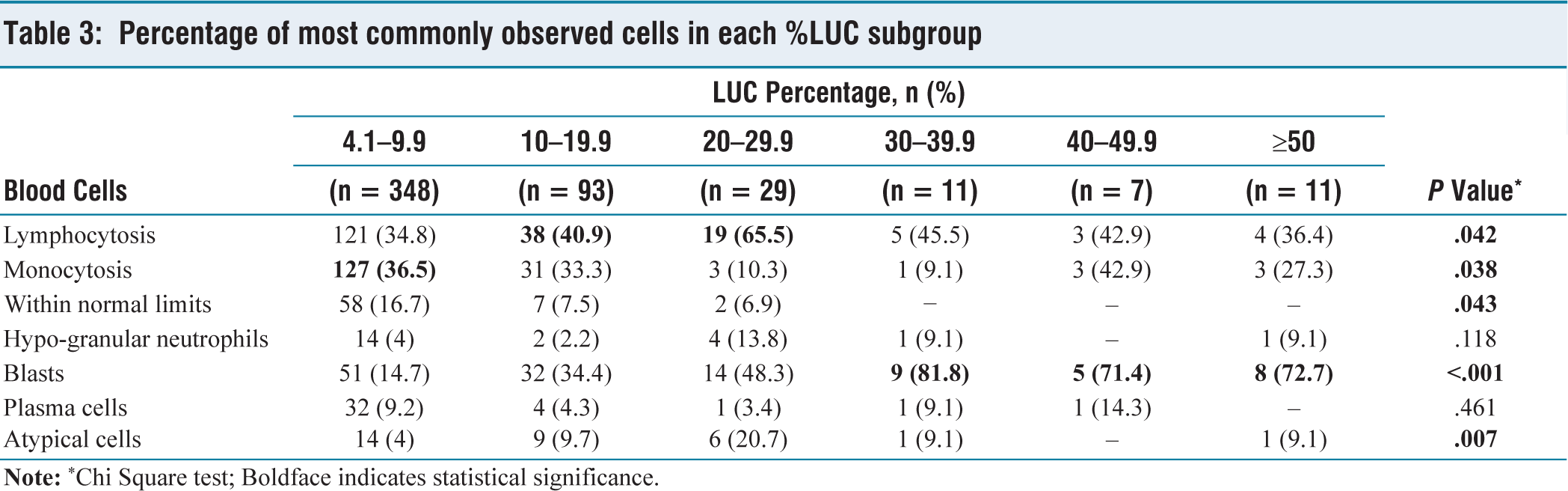
In LUC% subgroup 1 (4.1%–9.9%), monocytes, lymphocytes, blasts, plasma cells and hypo-granular neutrophils were most observed. Notably, blasts were most common when %LUC was greater than 30%, which was statistically significant [Table 3].
Percentage of most commonly observed cells in each %LUC subgroup
Discussion
Combination of CBC components such as neutrophil-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR) and monocyte-to-lymphocyte ratio (MLR) have been investigated as markers of inflammation.[3-6] In addition to these, LUC parameters, which include %LUC and LUC count, have also been investigated for their potential role as inflammatory biomarkers, markers to assess successful stem cell transplants and tumour biomarkers in suspicious leukaemia and lymphomas.[7-9] Although high throughput technologies are available for the correct identification of the clinical disease, a complete blood count serves as a primary tool to arrive at a differential diagnosis cost-effectively.
In our study, there were 405 adults and 95 paediatric cases, with a male predominance in both groups. Infections were more common in children, and haematolymphoid malignancies were more common in adults. No specific clinically detectable disease was seen in 9% of the study population, which was majorly attributed to children (n = 48). The clinical presentation was non-specific, with pancytopenia and pyrexia of unknown origin being more common.
From our observation, the median LUC count of 0.5 × 10 3 cells/µL with an inter-quartile range of 0.2–2.3 × 10 3 cells/µL of blood was observed in haematolymphoid neoplasms, which were found to be higher than the median LUC count of other diagnostic categories, and this difference was found to be statistically significant (P < .001). There was a significant overlap of %LUC values among different categories, and no statistical correlation was obtained. The majority (69.6%) had %LUC between 4.1% and 9.9%. Marked elevations were observed in AML, CML, ALL and infections.
Reactive mature lymphocytes, monocytes, lymphoblasts, myeloblasts, monoblasts, atypical lymphocytes, hypogranular neutrophils, basophils and plasma cells were the most commonly encountered peripheral blood cells in the study population. Each of these cells has variable MPO staining characteristics.
Historically, MPO activity has been considered to be the only unambiguous differentiating marker of myeloblasts that is exclusive to the myeloid lineage. Due to its simplicity of use and interpretation, MPO cytochemistry has shown to be the most effective method for identifying the myeloid lineage of blasts in terms of both specificity and general acceptability. More than or equal to 3% of blast cells exhibiting positive staining in a peripheral blood smear is considered a positive MPO reaction. Less than 3% MPO staining is considered a negative MPO reaction.[10]
Most of the myeloblasts are MPO-positive. Previous studies have shown that AML with minimal differentiation has strongly MPO-negative myeloblasts, which mimic lymphoblasts.[11] Hence, MPO-negative myeloid blasts in AML without differentiation can be scattered in the LUC area, attributing to LUC% elevation.
In a study by Vainchenkar W et al., strong MPO-positive myeloblasts were noted in AML with maturation and Acute promyelocytic leukaemia.[12] In our study, six cases of AML with maturation showed predominantly circulating myeloblasts with LUC% >4% that had MPO positivity in flow-cytometric analysis. Rosniak et al. have described that those myeloid cells with partial or complete MPO deficiency were clustered to monocytic or LUC area in the scattergram.[13] These findings suggest that a small percentage of myeloblasts with scant or absent myeloperoxidase in their cytoplasm in these cases could have been clustered onto the LUC area, attributing to elevations in LUC%.
Similar to a study by Roh et al., in a few AML cases of our study, hypogranular neutrophils were seen in peripheral circulation, which were likely to be counted as monocytes or LUCs due to decreased myeloperoxidase index.[14]
In AML with myelomonocytic and monocytic differentiation, monocytes are part of the neoplastic process. These monocytes and myeloblasts with scant granularity are counted as LUCs. Our study implies that, if the %LUC is more than 50% in a case of leukemic presentation, possibility of AML with monocytic, myelomonocytic differentiation and AML with dysplasia is to be considered in the differential diagnosis.
Lymphocytes, atypical lymphoid cells, plasma cells, lymphoblasts and basophils are invariably MPO-negative cells. Similarly, elevated mature lymphocytes were noted predominantly in haematolymphoid neoplasms (40.74%) and infections (22.75%) in this study.
All the CML cases in our study presented in the chronic phase with a median LUC count of 0.4 × 10 3 cells/µL of blood. The majority had a %LUC between 10% and 19.9%. Basophils and myeloid blasts were the most common peripheral blood cells observed in circulation. In a previous study by Peterson et al. and others, it was observed that blasts in a substantial proportion of CML blast crisis (BC) cases were negative for MPO, even though roughly 70% of CML BC cases were myeloid, and BC follows a previous phase that exhibits florid myeloproliferation.[15,16] Similarly, in a study by Mona et al., 154 out of 161 CML cases had circulating MPO negative cells in circulation, and seven cases had weak MPO staining (4%–20%), which suggests that circulating CML blasts in all phases have an intrinsic MPO negativity or weak positivity.[17-21] Similarly, basophils have metachromatic granules in their cytoplasm, which are negative for MPO staining. %LUC >4% in CML cases of our study can be explained by clustering of circulating MPO negative myeloblasts and basophils in the LUC area in PEROX scattergram.
Myelocytes produce MPO; Neutrophils do not produce MPO; instead, they store it. Neutrophil granules in acute bacterial sepsis release MPO. Hence, hypogranular neutrophils are seen in sepsis patients’ blood. Also, neutrophil dysfunction is brought on by cytokine storm. As a result, these hypogranular neutrophils appear as large, unstained cells or monocytes distributed to the left of the PEROX cytogram.[22]
In viral infections and chronic inflammatory states, such as diabetes, hypertension, autoimmune diseases and chronic kidney disease, monocytes and reactive lymphocytes were markedly increased in circulation, as they are mediators of inflammation. In these systemic illnesses, immune infiltration by T cells and B cells into target tissues and release of cytokines and chemokines sets a chronic low-grade localised and systemic inflammation. In contrast, short-chain fatty acids modulate gene expression of monocytes and reduce inflammation. It also regulates the release of regulatory T cells and thereby suppresses the function of inflammatory T cells.[23] This forms the basis for chronic active inflammation in the above-mentioned metabolic diseases.
In autoimmune diseases, the predominance of leukopenia, along with peripheral blood monocytosis and lymphocytosis, was observed. Neutrophilia is the characteristic feature of RA, indicating active joint disease,[24] but the current study showed no such findings.
Limitations
For assessment, we have not included a completely healthy control cohort or cohort with %LUC</ = 4%, for definitive comparison of pathological versus non-pathologic samples. Also, treatment history was not included as a part of the study.
Conclusion
Our study is a large-scale prospective study, which includes a spectrum of clinical disease. Absolute LUC count could be used as a cost-effective and readily available marker to identify a case of clinically suspicious haematolymphoid neoplasm with LUC count more than or equal to 0.5 × 10 3 cells/µL of blood in conjunction with other complete blood count parameters. However, reflex peripheral smear testing is mandatory in the current setting for confirmation. The characteristic LUCs noted in circulation were reactive lymphocytes, monocytes, blasts including lymphoblasts, myeloblasts, monoblasts, plasma cells, hypogranular neutrophils, basophils and atypical lymphoid cells. Myeloblasts are myeloperoxidase reactive cells but depending upon the maturation, these cells are counted as LUCs in the PEROX scattergram. But the questionable nature of cells in AML with maturation displaying elevated LUC% warrants further investigation.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author Mamta Soni is a member of the Editorial Board of Apollo Medicine. The author did not take part in the peer review or decision-making process for this submission and has no further conflicts to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
The study was approved by the Institutional Ethical Committee under approval number AMH-DNB-013/05-22.
Informed consent
Not applicable.
Credit author statement
Study designed, conducted and manuscript written by Dr Dhivya.
Study conceptualized, guided and manuscript reviewed and revised by Coauthor Dr Mamta Soni.
Data availability
Available on request.
Use of artificial intelligence
Nil.
