Abstract
Background and Aims:
Peroxidase deficiency is one of the commonly inherited phagocytic defects. It has been also found to exist as a transient phenomenon in association with some clinical conditions. But that it can interfere and cause erroneous automated differential WBC count is something which is not commonly known.
Materials and methods:
Complete blood counts were analysed using Advia 2120i and Coulter DXH 900 haematology analysers and manually reviewed on peripheral blood smear stained with Leishman stain.
Results:
Peroxidase deficiency in neutrophils and eosinophils resulted in them getting counted as monocytes by the analyser causing pseudomonocytosis, pseudoneutropenia and pseudoeosinopenia. These were detected by a slide review and by reanalysing the samples on an analyser which worked on a different principle.
Conclusion:
There is a need to confirm monocytosis given by analysers working on the peroxidase principle with an alternate method. This will prevent needless medical investigations for pseudoneutropenia, pseudoeosinopenia and persistent monocytosis, thus preventing unwarranted mental agony and financial burden to patients. It also helps to save the laboratory’s reputation. A careful review of instrument flags not only helps reach an accurate result but sometimes they can also aid in the diagnosis of a rare potential genetic disorder like MPO deficiency.
Introduction
Myeloperoxidase (MPO) is an enzyme belonging to a group of heme protein enzymes. The other related enzymes in the family are eosinophil peroxidase and lactoperoxidase.[1,2] The protein is found in the azurophilic granules of White Blood Cells such as polymorphonuclear leucocytes, monocytes and eosinophil, and also in tissue macrophages, Kupffer cells in the liver, microglia, granules of neurons, pyramidal system and the hippocampus.[1,3] On activation of the leucocytes, degranualtion occurs resulting in the release of MPO.[4] MPO utilises hydrogen peroxide and halides, resulting in catalysation and formation of reactive oxygen intermediates which attribute to its microbicidal properties.[4,5]
MPO deficiency may be genetic (primary) inherited as an autosomal recessive disorder or may be acquired as a result of other conditions (secondary). Secondary MPO deficiency may be seen in patients with end-stage kidney disease, cardiovascular disease, diabetes, myelodyspalstic syndromes, degeneration of joints, leukaemia, etc.[1,6,7] MPO deficiency is known to activate the development of a disease but is also likely to be the result of the primary disease.[1] Patients with MPO deficiency have been found to be more susceptible to fungal infections, especially candidal infections.[4,8] EPO deficiency is a rare abnormality characterised by decreased or absent peroxidase activity and reduced volume of the granule matrix with no known clinical significance.[9] It is caused by a mutation in EPX gene. Isolated deficiency of eosinophil peroxidase deficiency without concomitant myeloperoxidase deficiency is rare.
For the ADVIA 2120i haematological analyser, the test principle involves a combination of peroxidase activity and nuclear density analysis (PANDA). The peroxidase method involves the intracellular staining of neutrophils, eosinophils and monocytes which are MPO positive and which, on interaction with 4-chloro-naphthol and hydrogen peroxide, form a dark precipitate, which is then measured.[4,10,11] The analyser also has an inbuilt flag for MPO deficiency and a mean peroxidase activity index. MPXi defines the neutrophil MPO staining of the patient in comparison to the predetermined neutrophil archetype.[4,12] The normal range of MPXi index is 10 to 10. Studies have revealed that MPXi is reduced in patients with MPO deficiency and has resulted in the false flagging of monocytosis.[4,13]
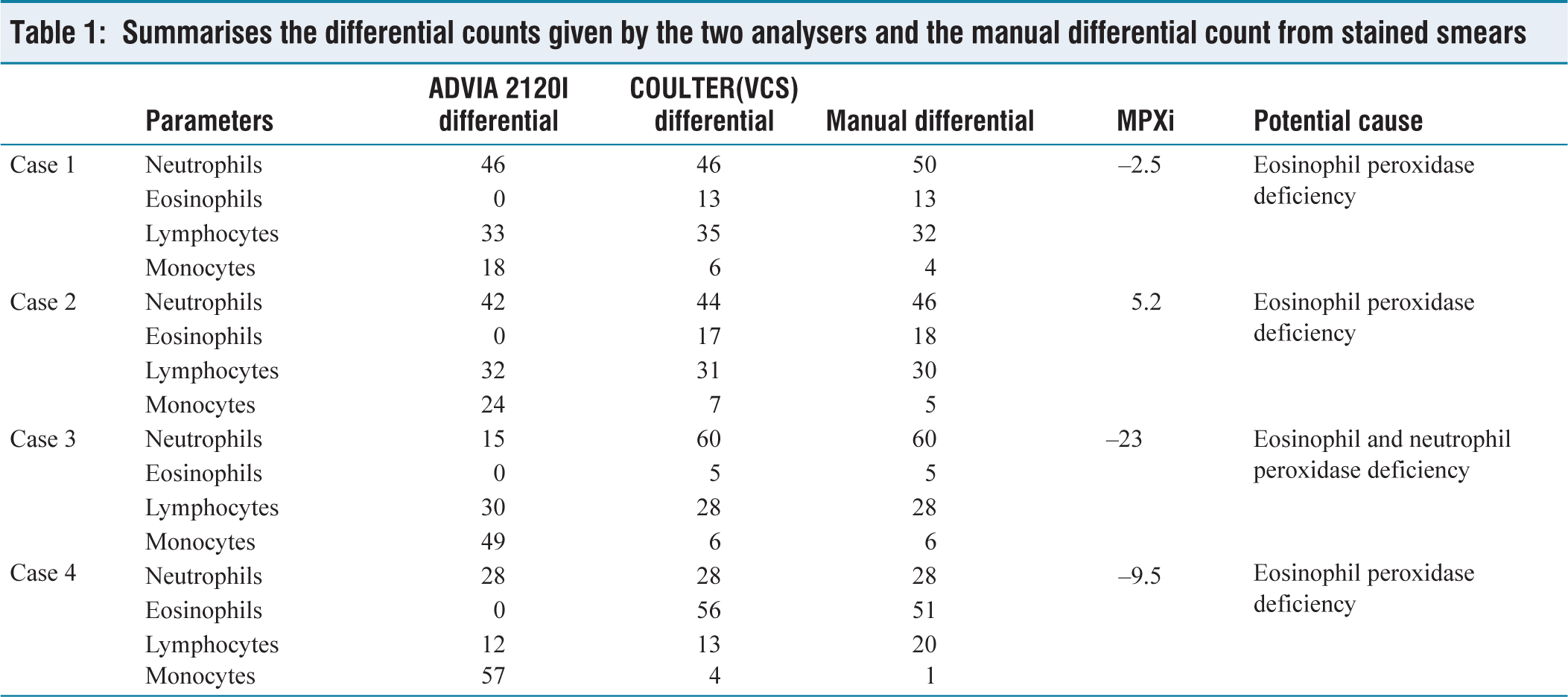
Here we present a series of four patients who were erroneously flagged monocytosis by ADVIA 2120i. The peripheral blood smear examination revealed a different differential count. This was further corroborated by running these samples on another haematology analyser that uses VCS (Volume, Conductivity and Scatter) to determine the differential counts. The differential counts given by this analyser were similar to counts given by manual enumeration on peripheral blood smears [Table 1]. None of these cases had actual monocytosis.
Summarises the differential counts given by the two analysers and the manual differential count from stained smears
Materials and Methods
Complete blood counts were analysed using Advia 2120i and Coulter DXH 900 haematology analysers and manually reviewed on peripheral blood smear stained with Leishman stain.
Results
Case 1, Case 2 and Case 4 analysis by Advia 2120i revealed monocytosis, which on analysis by Coulter and manual examination of peripheral blood smear identified it as eosinophilia. These blood samples were falsely flagged as monocytosis due to eosinophil peroxidase deficiency. In Case 3, Advia flagged monocytosis, which on further examination by Coulter and manual examination of peripheral blood smear identified them as having predominantly neutrophils and eosinophils, thus classifying this patient as having a combined neutrophil and myeloperoxidase deficiency.
Clinical significance of these deficiencies in these patients could not be ascertained.
Discussion
Many studies have identified myeloperoxidase deficiency as the cause of the misquantification of eosinophils and neutrophils as monocytes.[4,13] We also observed that in all the patients, the MPO-deficient neutrophils and eosinophils were falsely flagged as monocytes, resulting in pseudomonocytosis, psudoneutropenia and pseudoeosinopenia. Cases of eosinophilia were missed. It required a review by peripheral blood smear examination to give a correct differential count. None of these patients were found to have monocytosis. Results released without confirmation by an alternate method would have compromised patient safety and would have resulted in the loss of reputation for the laboratory.
Studies have revealed that MPXi is reduced in patients with MPO deficiency and has resulted in false flagging of monocytosis.[4,13] Cells with lower MPXI scores are detected as monocytes with higher scores as neutrophils. We observed a significantly low MPXi value in only one of our cases (Case 3).
MPO is an essential enzyme that is present in the azurophilic granules of leucocytes, mainly neutrophils, eosinophils and monocytes and possesses microbicidal properties attributed to its ability to generate hydrogen peroxide by an oxygen-dependent respiratory system.[4,14]
MPO deficiency is the common inherited phagocytic defect that impairs microbicidal killing, thereby attributing patient’s susceptibility to infections, of which the most common is the candidal infection.[13,15] However the observed frequency is low in patients with MPO deficiency. In our cases, none of the patients presented with an underlying infection. Clinical significance of MPO deficiency could not be ascertained.
Conclusion
We conclude from our observations that there is a need to confirm monocytosis given by analysers working on the peroxidase principle with an alternate method like smear review or reanalysis on an analyser doing WBC differentials on a separate principle. This will prevent needless medical investigations for pseudoneutropenia, pseudoeosinopenia and persistent monocytosis, and would largely reduce mental agony and financial burden for patients.
A careful review of instrument flags not only helps to reach an accurate result but sometimes they can also aid in the diagnosis of a rare potential genetic disorder like MPO deficiency.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author Mamta Soni is a member of the Editorial Board of Apollo Medicine. The author did not take part in the peer review or decision-making process for this submission and has no further conflicts to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this.
Institutional ethical committee approval
Not applicable.
Informed consent
Not applicable.
Credit author statement
Study conceptualized, designed, conducted and manuscript reviewed and revised by Dr. Mamta Soni (author).
Manuscript written by Co-author Dr. Supraja Sundaram.
Data availability
Available.
Use of artificial intelligence
Nil.
