Abstract
Introduction:
Cutaneous lymphomas represent the second most common extra-nodal non-Hodgkin lymphoma. Mycosis fungoides (MF) is the most common primary cutaneous lymphoma. In this study, we aim to estimate the prevalence of cutaneous lymphomas and to differentiate early MF from reactive cutaneous lymphoid hyperplasia (CLH) of the skin.
Materials and Methods:
We retrospectively studied all biopsy and resection specimens with a high index of clinical and histomorphological suspicion for cutaneous lymphoma for 10 years. We classified all the cases according to the World Health Organisation’s (WHO) 2022 classification of haematopoietic and lymphoid neoplasms. Statistical analyses were carried out using the software SPSS Statistics v23.0.
Results:
Out of 33 lymphoma cases, MF was the most common, followed by subcutaneous panniculitis-like T-cell lymphoma (SPLTCL). There were 13 males (39.3%) and 20 females (60.7%). Patches were the most common skin lesion, accounting for 78.7% of the cases, and presented as multiple lesions (81.8%), with the majority affecting the extremities (33.3%). Epidermotropism and basal tagging of lymphocytes, dermal fibrosis, and convoluted nuclear membranes were significant in early MF. Spongiosis correlated negatively with MF. T-cell receptor rearrangements were observed in 12 cases of MF.
Conclusion:
MF, the most common cutaneous lymphoma, is increasingly common in younger people. Histological variables such as epidermotropism, basal tagging of lymphocytes, convoluted nuclear membranes, and dermal fibrosis, along with clonal T-cell receptor rearrangements, aid in correct diagnosis and early therapeutic decision-making.
Keywords
Introduction
Cutaneous lymphomas account for 19% of extra-nodal non-Hodgkin lymphoma,[1] making them the second most common site for EN-NHL, with the gastrointestinal tract being the most common.[2] They are either primary or secondary, presenting as a part of systemic disease. Approximately 65% of cutaneous lymphomas originate from T cells, unlike nodal lymphomas, where B-cell lymphomas predominate.[3] According to surveillance, epidemiology, and end results (SEER) data from 2009 to 2018, the incidence of primary cutaneous T-cell lymphomas (CTCL) in the western population was 8.55 per million people, showing a 0.61% increase in annual incidence.[4] Mycosis fungoides (MF) is the most common form of primary CTCL.[5] Diagnosing MF can be challenging in its early stages due to scant tumoural infiltrate and the overlap of clinical and pathological features with cutaneous lymphoid hyperplasia (CLH), previously known as pseudo-lymphoma of skin.[6] The National Comprehensive Cancer Network guidelines recommend a biopsy of suspicious skin lesions with subsequent evaluation in terms of dermatopathology, immunohistochemistry, and clonality analysis by molecular studies. This study aims to assess the prevalence of cutaneous lymphoma at a tertiary care centre in South India and to differentiate early MF from reactive CLH based on clinicopathological features.
Materials and Methodology
This retrospective descriptive study was conducted at the Department of Histopathology and Cytology, Apollo Hospitals, Chennai, for 10 years, from 1st January 2014 to 30th April 2024, on all consecutive skin biopsy and resection specimens sent to the lab, both in-house and referral cases. Cases with a high index of clinical and histomorphological suspicion for lymphoma and cases with appropriate clinical history were included. We excluded skin specimens received for non-lymphoid malignancies, as well as specimens that were either over-fixed or under-fixed. This study was approved by the Institutional Ethical Committee-Biomedical Research.
Methodology
Clinical details were retrieved from the lab information system. Histopathological examination was done on haematoxylin- and eosin-stained sections of the specimen fixed in 10% Neutral buffered formalin. Immunohistochemistry was performed by the VENTANA Benchmark XT platform, employing OptiView DAB along with relevant positive and negative controls. An appropriate panel of antibodies was used wherever deemed necessary. We performed molecular studies for clonality assessment using short tandem repeat by capillary electrophoresis.
Predefined histopathological features were recorded. The epidermal features were hyperkeratosis, parakeratosis, acanthosis, epidermal atrophy, epidermotropism, Pautrier micro abscess, basal tagging of lymphocytes, peri-lymphocyte halo, basal vacuolar change, and spongiosis. Dermal features were patterns of lymphocyte distribution, fibrosis, pigment incontinence, polymorphous inflammatory infiltrate, and lymphoid follicle formation. The size of the lymphocyte, the nuclear membrane contour, and large cell transformation (defined as >25% of infiltrate composed of large atypical lymphoid cells) were also assessed. The histopathological and immunohistochemical diagnoses rendered were based on the World Health Organisation’s (WHO) Haemato-lymphoid Tumours of Skin 4th and 5th Editions. Clinical staging was based on the tumour node metastasis blood (TNMB) scoring system.
Statistical Analysis
We used Microsoft Excel to store and structure the data for statistical analysis using SPSS Statistics v23.0 software. Chi-square tests and analysis of variance tests were used to analyse the results. P value ≤ .05 was considered statistically significant. The qualitative variables were described as mean ± standard deviation.
Results
Out of 10,834 skin biopsy samples sent, 64 cases with histological suspicion of lymphoma were included in this study. The study consisted of 33 established cases of cutaneous lymphoma and 31 cases of CLH.
Cutaneous Lymphoma
The age range of the lymphoma population was 19-82 years, with a peak incidence occurring between 20 and 30 years of age (mean of 42.8 ± 18 years and median of 37 years). The cohort consisted of 13 males (39.3%) and 20 females (60.7%). All the patients were adults. The majority of cases (78.7%) presented as patches, and most had multiple lesions (81.8%). At presentation, 54.5% of cases were asymptomatic, and 27.3% had itching as their primary symptom. Extremities were the most commonly involved site (33.3%), followed by the trunk (30.3%) and multiple contiguous sites (27.3%). Head and neck involvement was very rare (9.1%). The mean duration for diagnosis was 11.46 ± 16.9 months (median of four months). All patients were of Indian ethnicity.
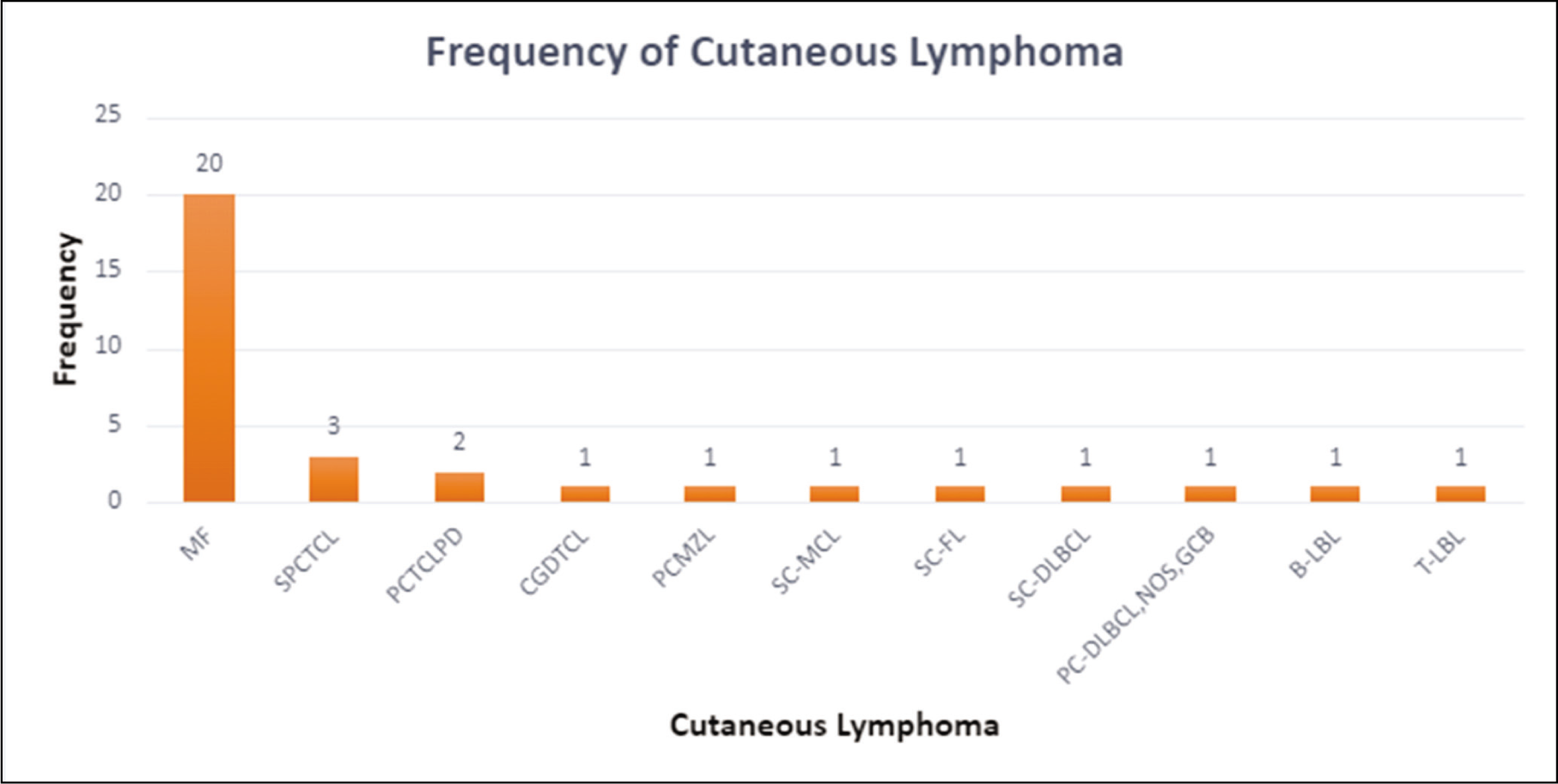
Primary CTCL accounted for 78.8% of cases and included MF (n = 20, 60.6%), subcutaneous panniculitis-like T-cell lymphoma (SPLTCL) (n = 3, 9.1%), CD30+ lymphoproliferative disorders (n = 2, 6.1%), and cutaneous gamma delta TCL (n = 1, 3%). Cutaneous B-cell lymphoma in this study comprised primary cutaneous marginal zone lymphoma (3%, n = 1), Primary cutaneous large B-cell lymphoma, NOS, non-germinal cell type (3%, n = 1), Secondary cutaneous involvement by systemic diffuse large B-cell lymphoma (3%, n = 1), Mantle cell lymphoma (3%, n = 1), and nodal follicular lymphoma (3%, n = 1) [Figure 1]. Immature haematopoietic malignancies included one case of T-cell lymphoblastic leukaemia/lymphoma (3%) and one case of B-lymphoblastic leukaemia/lymphoma (3%).
Distribution of lymphoma with cutaneous presentation
B-lymphoblastic leukaemia with cutaneous involvement was presented in a 46-year-old male patient. The majority of the lesions were located in the extremities and manifested as multiple patches. A diffuse dermal infiltrate with an intervening Grenz zone was characteristic, with expression of CD79a, Tdt, HLA-DR, CD19, CD20, CD10 and CD22.
T-LBL with cutaneous involvement presented as a single patch involving the lower extremity in a 70-year-old female. Bone marrow showed 70% atypical lymphoid cells. The lymphoid cells expressed Tdt, CD3, and CD5 markers. The patient was started on systemic chemotherapy, but later, the patient died of the disease. The review diagnosis of cutaneous lymphomas was concordant with the initial primary diagnosis.
Clinicopathological Profile of MF and CLH
The peak incidence of both MF and CLH was observed in the age group of 21-30 years of age (35% in MF and 29% in CLH). In MF, there was a slight male predominance with an M: F ratio of 1.58:1, and in CLH, the M: F ratio was 1:1.85. Patients initially presented with patches (75% in MF and 74% in CLH), and most of them presented as multiple lesions in both groups. 65% of MF cases and 51.61% of CLH cases were asymptomatic at presentation. Itching was noted as a presenting symptom in a subset of both MF (35%) and CLH (32.26%). The mean duration for diagnosis of MF was 15.5 ± 19.93 months, and the mean duration for diagnosis of patients with CLH was 9.48 ± 16.7 months. Statistical analysis revealed no discernible variations in the clinical presentations of MF and CLH.
Histological Evaluation of MF and CLH
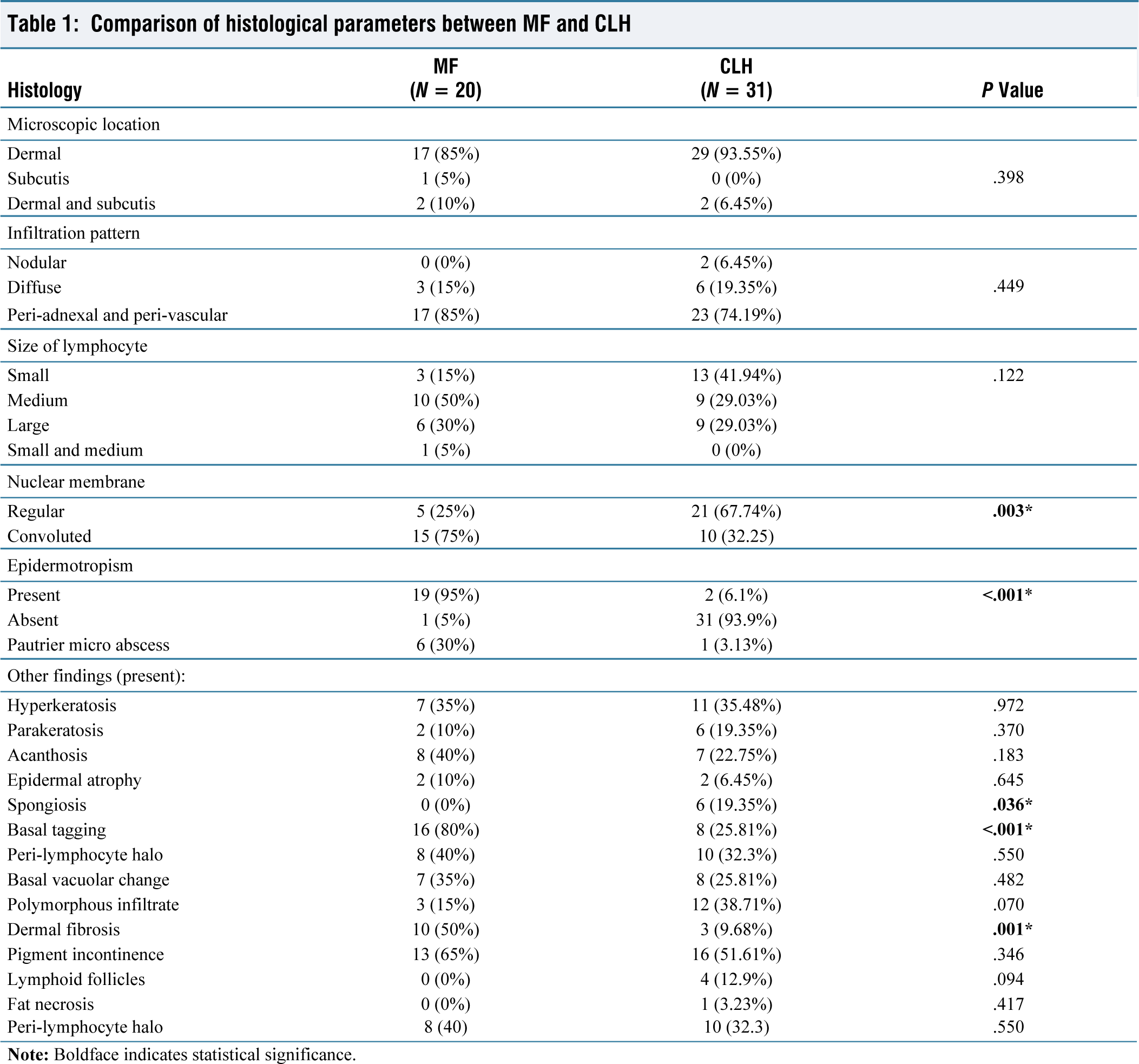
On histological assessment, lymphoid infiltrates were predominantly located in the dermis in 85% of MF and 93.5% of CLH cases. The infiltration pattern for both groups was peri-adnexal and perivascular (85% in MF and 74.2% in CLH), followed by a diffuse pattern (15% in MF and 19.35% in CLH). In MF, the atypical lymphoid cells were predominantly of medium size (n = 10, 50%), whereas in CLH, they were of small size (n = 13, 41.93%). None of the cases showed large cell transformation.
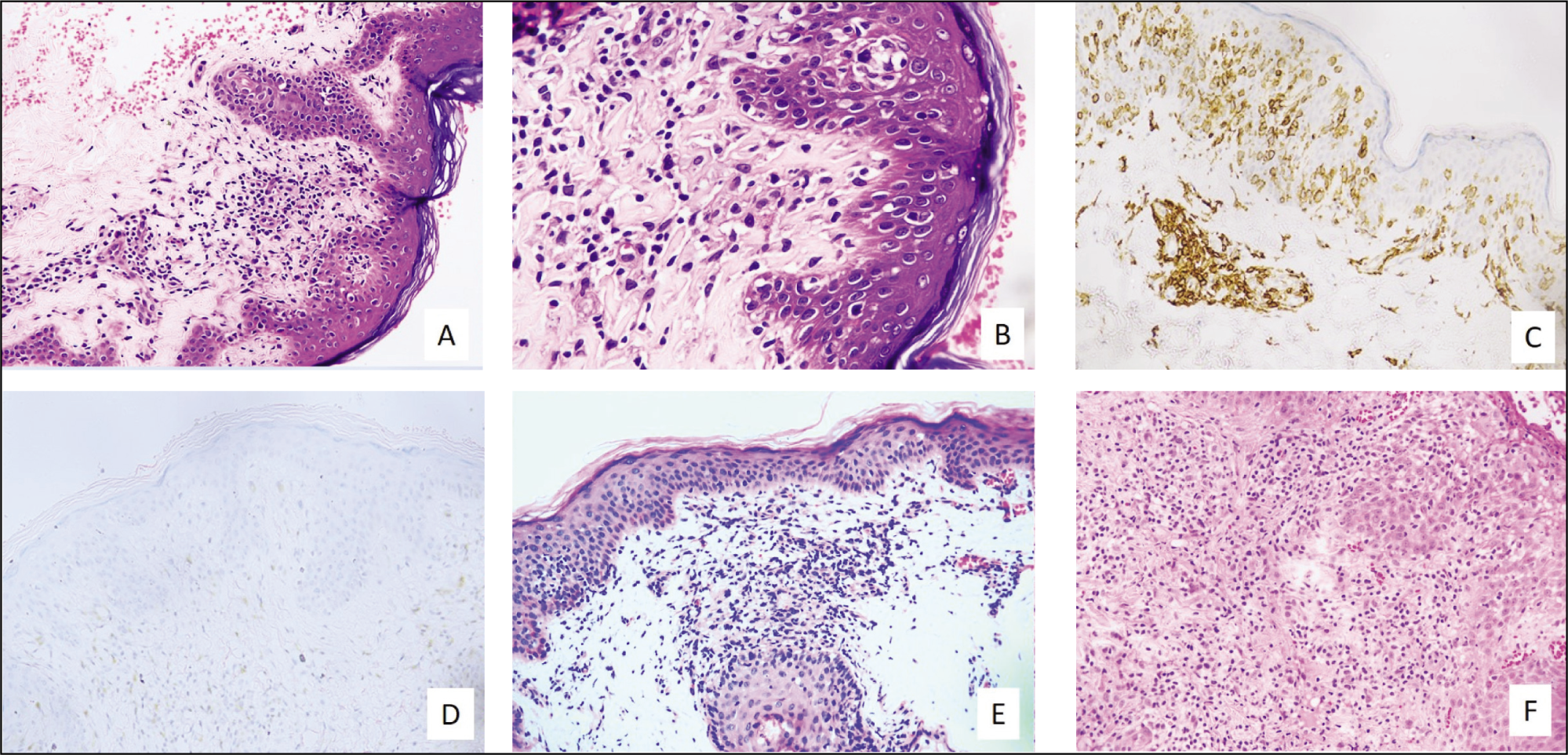
Significant differences were noted in the epidermotropism and basal tagging of lymphocytes, the contour of the nuclear membrane, spongiosis, and dermal fibrosis between the two groups. Epidermotropic lymphocytes (95%), basal tagging (80%), convoluted nuclear membrane (75%) and dermal fibrosis (50%) were predominant in MF, whereas spongiosis was noted in none of the MF cases [Figure 2A & B]. Spongiosis (19.35%) and regular nuclear membrane (67.74%) were significant in CLH cases [Figure 2E & F]. Reactive lymphoid follicles with germinal centres were present in four of the CLH cases. Histological variables such as hyperkeratosis, parakeratosis, acanthosis, atrophy, peri-lymphocyte halo, basal vacuolar change, polymorphous inflammatory infiltrate, pigment incontinence, and fat necrosis did not show significant differences between MF and CLH (Table 1).
(A and B) Mycosis fungoides (H&E stain): (A) Atypical dermal lymphocytic infiltrate with epidermotropism and dermal fibrosis. (B) Basal tagging of lymphocytes. (C) Diffuse CD4 expression (IHC). (D) Loss of CD7 expression (IHC). (E and F) Cutaneous lymphoid hyperplasia ((H&E stain)
Comparison of histological parameters between MF and CLH
IHC and Molecular Profile of MF and CLH
In MF patients, CD3 and CD4 were positive in 100% of cases [Figure 2C]. The expression of pan T-cell markers in MF was heterogeneous: CD2 was intact in 55% of cases and absent in 45%, CD5 was intact in 65% and absent in 35%, and CD7 was intact in only 15%, with the majority (85%) showing loss of expression [Figure 2D]. Fifty percent of cases showed positivity for CD8. CD30, CD20, PD1 and CD56 were completely negative. Most cases (85%) exhibited a low Ki67 proliferation index. In CLH, 77.42% of cases expressed CD3, implying T-cell phenotype. CD20 expression was present in 9.7% of cases. The expression of CD3 and pan T-cell markers did not show any statistically significant differences between MF and CLH patients.
Twelve of the 20 patients in the MF subgroup who had TCRG analysis showed clonal TCRG rearrangements. In the CLH subgroup, seven of the nine patients who had clonality testing showed polyclonal TCRG, one had an IGH gene rearrangement, and one was polyclonal for IGH rearrangement. Follow-up of the CLH case with IGH rearrangement showed no progression to lymphoma. In 8 of 20 MF cases where TCRG analysis was not performed, repeat biopsies were done to confirm the diagnosis. All MF patients were diagnosed at an early stage (T1/T2, N0, M0), with no evidence of blood and bone marrow involvement at the initial presentation.
Discussion
In this study, we calculated the frequency and distribution of cutaneous lymphomas in our institute from January 2014 to April 2024 using WHO (2022) and WHO-EORTC 2018 updated classification. A total of 33 cases of cutaneous lymphomas were studied. The incidence of PCL (87.9%) in our study was higher than secondary cutaneous involvement by nodal lymphomas. CTCL (78.2%) was predominant, and CBCL (15%) was relatively less common. Gabor et al. reported that 83% of primary cutaneous lymphomas were CTCL, with higher rates observed in Asian and South American countries.[7] MF, the most common CTCL, had an incidence of 5.42 per million people and an annual incidence rate of 1.34%.[4] MF (60.6%) was the most common PCL in our study. CD30+ primary cutaneous T-cell lymphoproliferative disorders (PCTLD) had the highest incidence (19%) among East European countries.
Rare CTCL, such as SPLTCL and cutaneous gamma delta TCL (PCGD), were more commonly observed in the Asian population, attributing to higher frequencies of HAVCR2 germline alterations.[1,7] In our study, SPLTCL (9.1%) was the second most common CTCL, and PCTLD was the third most common. Precursor lymphoid neoplasms were rare. The presentation of B-LBL in our study was consistent with the findings of a previously published study by Alhaji et al.[8] In contrast to our research, T-lymphoblastic leukaemia typically affects young adults and manifests as widespread cutaneous nodules in the head, neck, and trunk, along with bone marrow, mediastinal and nodal involvement.[9]
While MF typically affects elderly adults,[7] our study primarily observed both MF and CLH in young adults aged 20-30 years.[10,11] We can attribute the higher incidence in younger patients to early diagnosis and greater physician accessibility.[12] Consistent with previous studies, there was a slight male predominance among MF patients, but the differences were not statistically significant. In both MF and CLH, lesions were predominantly patches, presenting with multiple lesions.[11,13] Plaques were the most common skin lesion in studies by Kadher et al. The trunk was the most common site involved, followed by extremities.[10]
Epidermotropism is a well-established diagnostic feature of MF; it was observed in 95% of MF cases in our study.[14] The nuclear membrane of lymphocytes in MF often appears convoluted, indented or cerebriform in shape.[15,16] The presence of lymphocytes in the basal layer of the epidermis is a key diagnostic feature of MF. A study by Saira Fatima et al. suggests that dermal fibrosis is a characteristic feature of CTCL, aiding in its diagnosis.[11] Similarly, our study observed a significant degree of dermal fibrosis in patients with CTCL. Spongiosis is typically found in patients with CLH.[15,17]
IHC results showed CD3 and CD4 positivity in all MF cases, along with patchy to complete loss of pan T-cell markers such as CD7, CD2 and CD5. Although there were no significant differences when compared to CLH,[4] these findings were consistent with previously published studies.
TCRG analysis of 12 MF patients in our study revealed rearrangements in all the tested cases. Hence, TCRG analysis by high-throughput technologies and PCR has a remarkable ability to establish the clonality of early MF.[18] However, clonal expansions of T lymphocytes have been observed in various benign inflammatory dermatoses.[19] Also, early MF is difficult to diagnose due to the confounding spectrum of clinical manifestations, and hence, multiple biopsies are recommended.
Management of CTCLs is individualised and ranges from skin-directed topical applications, such as steroids and immunotherapy in T1/T2 stage MF, to phototherapy and external beam radiation therapy in extensive skin diseases. Systemic therapies in advanced stages include oral retinoids and chemotherapy.[20]
Limitations
The limitations of our study include its retrospective study design and lack of adequate treatment and follow-up data, which restricted the ability to identify potential correlations.
Conclusion
Our study concludes that the incidence of primary CTCL is increasing among younger populations. Histological variables such as epidermotropism, convoluted nuclear membrane, basal tagging of lymphocytes, and dermal fibrosis, along with immunohistochemistry and clonal T-cell receptor gene rearrangements, aid in distinguishing early MF from reactive CLH. This integrated approach is crucial for accurate diagnosis and informed therapeutic decision-making, as early MF are linked to better clinical outcomes.
Footnotes
Acknowledgements
We acknowledge Dr Shivashankari, Research Assistant, and Mr Logesh, Biostatistician, from the Research Department, Apollo Hospital, Chennai, for their contributions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Ethical approval was obtained from Institutional Ethical Committee – Biomedical Research.
Informed consent
Informed consent has been obtained from all the patients who met the eligibility criteria.
Credit author statement
ND participated in data acquisition, literature search, investigations, data analysis, and manuscript preparation.
RAK participated in conceptualization, investigation, supervision, data analysis, and manuscript editing.
Both the authors reviewed and approved the manuscript.
Data availability
Data presenting the study’s findings is available with the corresponding author.
Use of artificial intelligence
The use of artificial intelligence is not relevant to this article, as it does not involve AI technologies or methodologies in its analysis or conclusions.
