Abstract
Background:
Skin diseases have been shown to worsen psychological distress, which, in turn, may be detrimental to treatment outcomes. Both the impact of psychological distress on response to treatment in mycosis fungoides (MF) and the effect of treatments on psychological well-being are unclear.
Objectives:
To evaluate (1) the association between pretreatment psychological morbidity and treatment outcome in early-stage MF and (2) the impact of response to treatment on psychological well-being.
Methods:
This was a prospective cohort study of patients with early-stage MF who started a new stage-directed treatment for their disease. The response was determined using the modified severity-weighted assessment tool, and psychological distress was assessed using the 12-item General Health Questionnaire (GHQ-12) and Penn State Worry Questionnaire (PSWQ). Participants were followed for 1 year.
Results:
In all, 24 consecutive patients were recruited. Objective response rate was 71% (17/24), consistent with existing literature. Prior to treatment, 9 patients (38%) had clinically significant psychological distress on the GHQ-12, while 8 (33%) demonstrated high-level worry on the PSWQ. Of these patients, 6 had pathologic scores on both instruments. Patients with significantly less baseline anxiety/depression on the GHQ-12 responded better to treatment than patients with higher levels (P = .004). In addition, responders’ mean GHQ-12 scores decreased by 39% and their PSWQ scores by 17%, whereas nonresponders’ GHQ-12 scores increased by 93% (P = .042) and their PSWQ scores by 11% (P = .019).
Conclusions:
These findings suggest that (1) baseline psychological distress is associated with worse outcomes in patients with early-stage MF and that (2) effective treatment improves psychological morbidity.
Introduction
Primary cutaneous T-cell lymphomas (CTCLs) are a heterogeneous group of non-Hodgkin lymphomas characterized by clonal accumulation of neoplastic T cells in the skin, with mycosis fungoides (MF) accounting for approximately 50% of cases. 1 MF can profoundly impair patients’ quality of life.2,3 Associated psychological morbidity, in the form of depression, anxiety, and worrying, has been noted.2-4
Skin diseases, such as psoriasis, are known to increase psychological distress, 5 and in a vicious cycle, such distress is associated with a significant, detrimental effect on outcomes. 6 Psoralen plus ultraviolet A photochemotherapy (PUVA) has recently been shown to improve health-related quality of life and psychological well-being in patients with early-stage MF. 7 However, only limited general psychological distress questionnaires have been used for CTCL, while no worry-specific instruments have been used.
This prospective study evaluated (1) the association between pretreatment psychological morbidity and treatment outcome in early-stage (stage IA-IIA) MF and (2) the impact of treatment response on psychological well-being.
Patients and Methods
Study Population and Recruitment
Patients aged 18 years or older with a confirmed diagnosis of early-stage (stage IA-IIA) MF and who started a new stage-appropriate treatment consistent with standard of care were eligible. Exclusion criteria included the use of psychotropic medication concurrently or within the previous 6 months. The study was approved by the institutional research ethics board. All patients provided written informed consent.
Psychometric Measures
Patients completed the 12-item General Health Questionnaire (GHQ-12) 8 and Penn State Worry Questionnaire (PSWQ) 9 every 3 months for 1 year. Given the dual nature of MF as a dermatologic disease and cancer, these tools were selected as they have been validated in dermatology and oncology.
The GHQ-12 is a self-administered instrument that measures distress and detects current non-psychotic psychiatric disorders (anxiety and depression). Answers are scored according to the binary method, where symptomatic responses are allocated a value of 1. The total score is used to differentiate between low-level (GHQ-12 < 3) and clinically significant (GHQ-12 ≥ 3) psychological distress. 10
The PSWQ, a 16-item, self-report tool developed to measure worry, was also administered. It has been shown to possess strong internal consistency and good test-retest reliability. The total score was employed to differentiate between low-level/non-pathological (PSWQ ≤ 60) and high-level/pathological worry (PSWQ > 60). 9
Assessment of Treatment Response
The clinical severity of the MF was measured using the modified skin-weighted assessment tool (mSWAT). Response to treatment in skin was determined according to the standardized clinical endpoints and response criteria for MF. 11
Statistical Analysis
IBM SPSS version 28 was used for statistical analysis. Data were reported as frequency and proportion for discrete variables, and as mean with standard deviation (SD) for continuous variables. Differences between groups were examined using t-tests for continuous data and χ2 tests for discrete data. Statistical significance was considered as P < .05.
Results
In all, 24 consecutive patients were enrolled between July 2016 and April 2020 at our multidisciplinary cutaneous lymphoma clinic. Four individuals were excluded based on predefined exclusion criteria or declined to participate. All 24 patients completed the study. Their mean age was 60.3 years (SD 16.7). In total, 20 patients were male (83%) and 4 were female (17%). The participants had failed an average of 2 prior treatments (SD 1.76) and 4 (17%) had not been treated previously. New treatments initiated at enrolment included topical agents (n = 13), phototherapy (n = 7), methotrexate (n = 1), systemic retinoids (n = 2), and radiotherapy (n = 1). At baseline, 38% of patients (9/24) had clinically significant psychological distress (GHQ-12 ≥ 3), whereas 33% (8/24) reported high-level worry (PSWQ > 60). Of these individuals, 6 had pathologic scores on both instruments. There was no significant difference in baseline mSWAT between patients with psychological morbidity and those without (GHQ-12, P = .396; PSWQ, P = .964).
The objective response rate was 71% (17/24), with complete response in 42% (10/24) and partial response in 29% (7/24), comparable to rates observed in the Prospective Cutaneous Lymphoma International Prognostic Index (PROCLIPI). 12
Patients with significantly less baseline anxiety and depression on the GHQ-12 responded better to treatment than patients with higher baseline levels (P = .004) (Supplementary Table 1). Although a similar trend was seen with lower levels of worry on the PSWQ, the difference was not significant (P = .067). There were no significant differences in other baseline characteristics between responders and nonresponders.
Responders experienced an average reduction in their mSWAT of 91%, whereas nonresponders had a mean increase of 47% (P < .001) (Figure 1). Supporting our hypothesis, responders experienced a significant improvement in anxiety, depression, and worrying, with a mean decrease of GHQ-12 scores by 39% and PSWQ scores by 17%, compared with a mean increase of 93% in GHQ-12 and 11% in PSWQ scores in nonresponders (GHQ-12, P = .042; PSWQ, P = .019) (Figure 2). No responders experienced relapse or progression at any time during the 1 year of follow-up. Only one nonresponder achieved an objective response on a subsequent therapy (PUVA). For the nonresponder group, there was no significant difference in GHQ-12 and PSWQ scores at 1 year compared with when they were determined to have failed their treatment (GHQ-12, P = .67; PSWQ, P = .14).
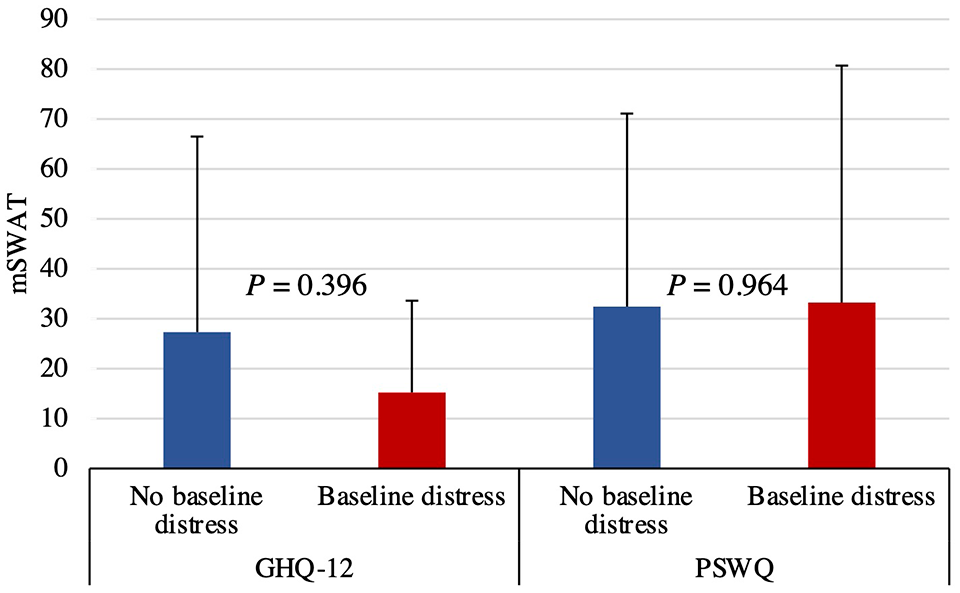
Baseline mSWAT in patients with and without baseline distress. There was no significant difference in baseline GHQ-12 or PSWQ scores in patients with versus without baseline distress. GHQ-12, 12-item General Health Questionnaire; mSWAT, modified severity-weighted assessment tool; PSWQ, Penn State Worry Questionnaire.
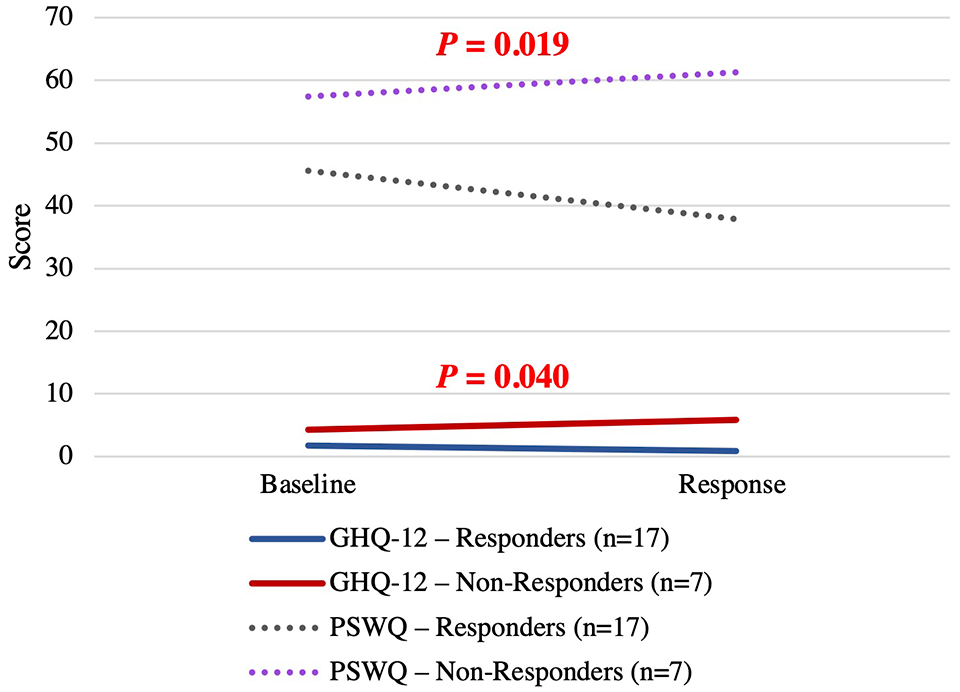
Change in GHQ-12 and PSWQ in objective responders versus nonresponders. The maximum score on the GHQ-12 is 12, and on the PSWQ, 80. GHQ-12, 12-item General Health Questionnaire; PSWQ, Penn State Worry Questionnaire.
Discussion
This prospective study suggests that higher baseline levels of psychological distress correlate with poor response to treatment in patients with early-stage MF, regardless of pretreatment profiles. The lack of any demographic or obvious clinical differences between the objective responders and the nonresponders contributes to the strength of the data. Only one other study to date has evaluated psychological distress in relation to treatment in this population, 7 assessing the impact of PUVA. Our study adds evidence to support the benefit of other treatment modalities on mental health in this population as well as the impact of treatment on worrying. To our knowledge, the PSWQ has not been studied in MF previously. However, given the small sample size, our results should be considered preliminary. Nevertheless, the statistical significance is quite strong for a promising association. It will be important to repeat the study in a larger group, which may also permit a better representation of all treatment modalities and comparisons between them.
The mechanism underlying the relationship between mental stress, MF, and treatment response remains unclear. Such an interaction between psychosocial state and cutaneous health, termed the brain-skin axis, is an expanding research field. Stress and emotional status are known to induce or exacerbate many skin diseases. 13 A bidirectional link has also been suggested since psychiatric diseases occur or worsen in one-third of dermatologic disorders. 13 Psychological distress, including anxiety and depression, has been associated with MF, which can significantly impair the quality of life.3,14 Our results emphasize the frequent psychological comorbidity of early-stage MF, given that pathological distress or worry each found in over 30% of our patients versus 16.7% to 19.0% reported in the general population. 15 This is consistent with previous work showing that worrying, including about the seriousness of the disease and associated mortality, was commonly reported in a predominantly early-stage MF population. 4 Questioning about mood and anxiety/worry may be considered to identify the individuals at risk. Studies to understand whether psychological interventions could improve clinical response rates may be valuable in this population.
We have characterized psychological morbidity using the GHQ-12 and PSWQ in early-stage MF. Significant psychological distress is common in this patient population. Our findings suggest that (1) baseline psychological distress is associated with worse outcomes in patients with early-stage MF and that (2) effective treatment of MF improves psychological morbidity. Larger scale studies are required to validate these findings.
Supplemental Material
sj-docx-1-cms-10.1177_12034754231220913 – Supplemental material for Psychological Distress and Treatment Outcome in Patients With Early-Stage Mycosis Fungoides
Supplemental material, sj-docx-1-cms-10.1177_12034754231220913 for Psychological Distress and Treatment Outcome in Patients With Early-Stage Mycosis Fungoides by Meagan-Helen Henderson Berg, Gizelle Popradi, David Roberge, Tina Petrogiannis-Haliotis and Kevin Pehr in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
IRB Approval Status
Reviewed and approved by CIUSSS West-Central Montreal (16-049, 2017-479).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
