Abstract
Introduction:
NUT carcinoma (NC) is a rare aggressive undifferentiated malignant neoplasm. It can arise from midline and non-midline structures with no predilection for age and gender. NC is due to NUT gene rearrangement (Chromosomal translocation). The diagnosis is established by NUTM1 protein expression by IHC / FISH / NGS RNA sequencing Given the rarity and novelty, herein we discuss our case of NC.
Aim:
To establish a diagnosis of NUT carcinoma and its diagnostic challenges.
Clinical History:
A 23-year-old young male presented with complaints of right neck swelling.
Results:
Histopathology revealed a poorly differentiated malignant neoplasm. Immunohistochemistry showed diffuse positivity for NUTM1 protein, hence was reported as NUT carcinoma.
Conclusion:
It is an aggressive malignant neoplasm with a dismal prognosis. An integrated approach by clinicians, radiologists and pathologists is required for accurate diagnosis and timely management.
Introduction
NUT (Nuclear protein in testis) carcinoma(NC) is an uncommon, undifferentiated aggressive malignant tumour that involves testicular nuclear gene rearrangement and tends to originate from midline anatomical regions and non-midline structures, including renal pelvis, parotid gland, sublingual gland and femur.[1] NC was originally known as Nut midline carcinoma (NMC) because of its location in the midline, upper airways and mediastinum.[2] The WHO reclassified NMC in 2015 as NUT carcinoma (NC). In 1991, mediastinal tumours with t(15;19) translocation were documented. The fusion gene that emerged from this translocation was named as BRD4-NUT in 2003. Numerous other fusion partners subsequently have been discovered such as BRD3, NSD3, ZNF532, ZNF592 and CIC, resulting in the belief that the NUTM1 (NUT midline carcinoma family member 1) may provide the greatest definition for classification, clinical behaviour, treatment alternatives and fusion partner as opposed to immunohistochemistry profile or the tumour appearance. Currently, it is not known what sort of cell NC comes from because patients are commonly diagnosed at an advanced stage of the disease. In many situations, the underlying tumour site is not known and may be a part of the cases of CUP (carcinoma of unknown primary) syndrome, which can account for as much as 5% of malignancies.[3] Under such circumstances, it is easy to hypothesise that NC originated from an abnormal phenotype of germ cells.[4] With a high frequency of patients in their 20s, NC is characterized by a median age of 23 years with no gender predilection.[5] In terms of histology, NC are undifferentiated or poorly differentiated carcinomas with vesicular chromatin, which can be confused with undifferentiated tumours such as poorly differentiated squamous cell carcinoma (SCC) and Ewing sarcoma. Diagnostic confirmation of NC can be achieved by demonstration of NUT rearrangement by IHC/FISH using a split-apart probe or NGS-based RNA-sequencing. Wild-type NUTM1 expression is often restricted to the testes; any expression outside of the testes or in a germ cell tumour could be the result of a pathologic expression of a NUTM1 fusion gene, which is activated by the promoter of its fusion partner. Formalin-fixed, paraffin-embedded tissue sections are regarded as positive when more than 50% of the nuclei show staining. For the diagnosis of NC, IHC staining with this antibody shows an 87% sensitivity and 100% specificity. Thus, World Health Organization’s (WHO) classification of tumours[6] confirms that this pathological test is typically thought to be adequate to diagnose NC tumour tissue. Unfortunately, the prognosis for NC is quite miserable, with a median survival rate of 6-7 months overall, regardless of age and therapeutic targets.[7]
Case Presentation
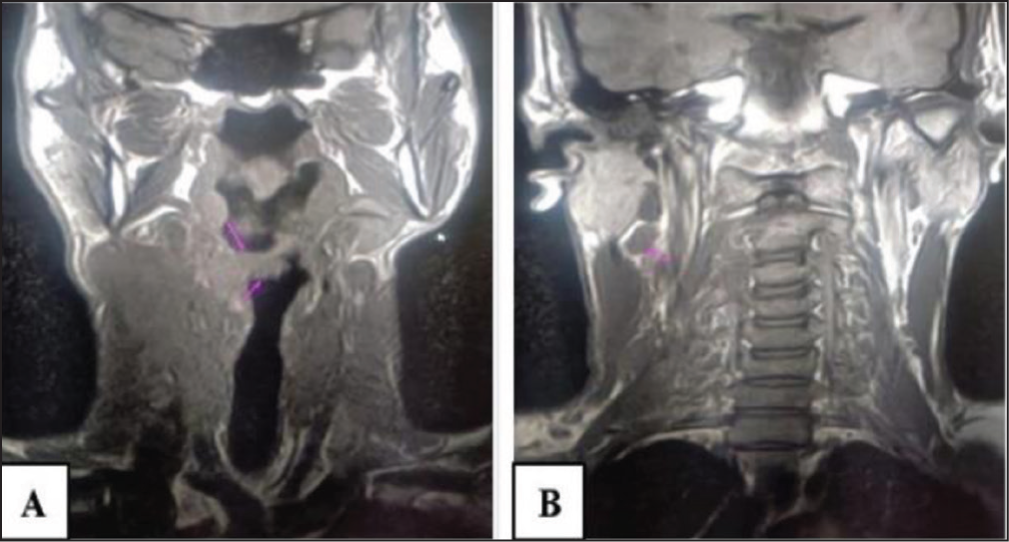
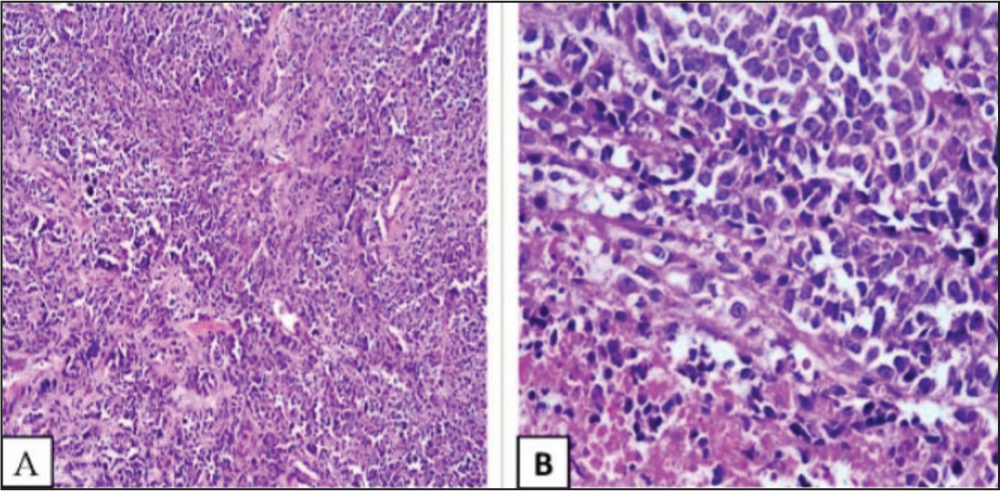
A 23-year-old male presented with complaints of painless swelling in the right side of the neck, gradually increasing in size with difficulty in swallowing. Radiological investigations such as contrast-enhanced CT, MRI and PETCT were performed. A soft tissue mass lesion that extended across the anterior midline and onto the left side was visible on the MRI of the right pyriform fossa (Figure 1), multiple significantly enlarged lymph nodes were seen on both sides of the neck with the largest lymph node in the right supraclavicular region. The possibility of carcinoma was to be considered. PET-CT revealed multiple enlarged lymph nodes on both sides of the neck, with a soft tissue mass lesion in the right pyriform fossa, extending beyond the anterior midline to the left side. The possibility of metastasis was to be considered. A poorly differentiated malignant tumour was suggested by the characteristics found in the right supraclavicular lymph node’s fine needle aspiration cytology; following that, the right supraclavicular lymph node excision biopsy was performed and was sent for frozen section. There was a poorly differentiated malignant tumour in the frozen section and histology report, the morphology showing lymph nodal tissue with effacement of architecture and replaced by sheets and focal perivascular arrangement of large polygonal cells displaying marked pleomorphism, vesicular nuclei with conspicuous nucleoli and moderate eosinophilic cytoplasm. Extensive areas of necrosis and karyorrhectic debris was evident. Perinodal extension was seen. There was no evidence of gland formation or squamoid differentiation (Figure 2).
A&B: MRI of head and neck showing soft tissue mass lesion in right pyriform fossa extending beyond anterior midline to the left side
A, B: Light microscopy showing sheets of atypical cells with no obvious squamous or glandular differentiation (H&E 100x, 400x)
Results
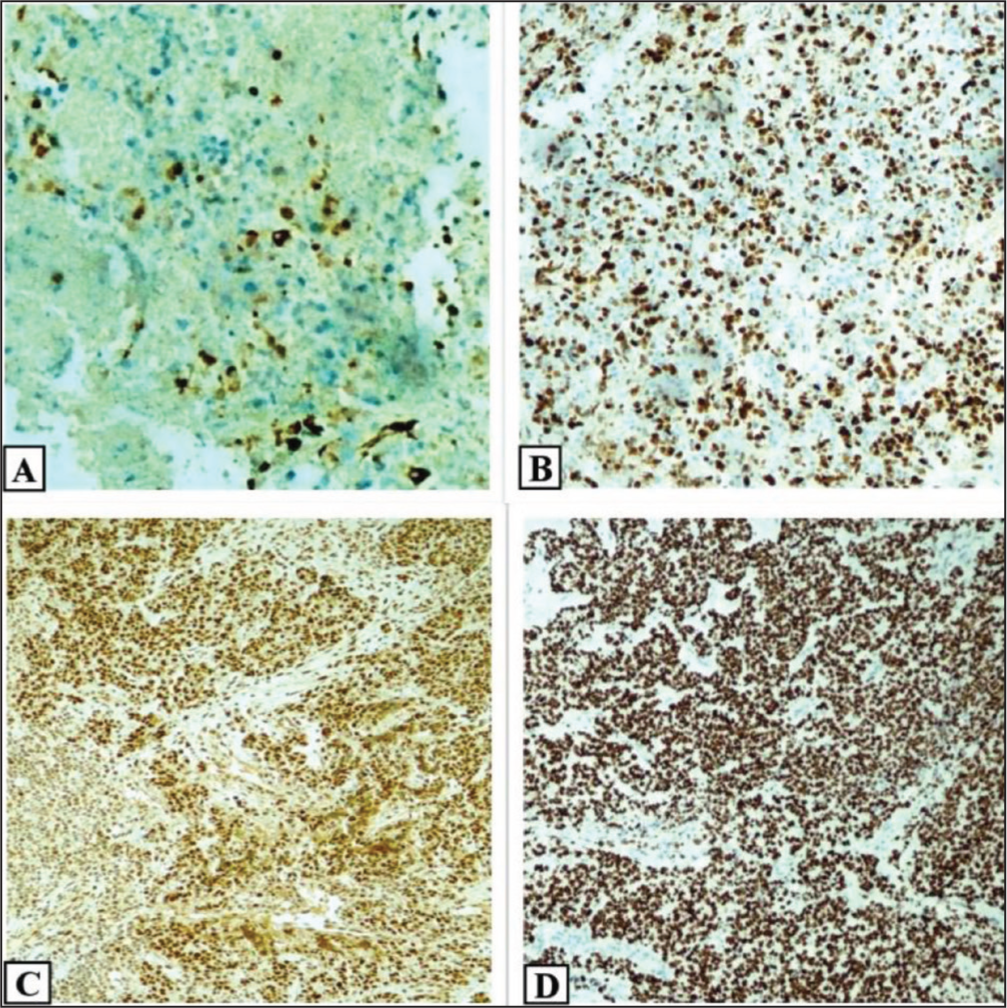
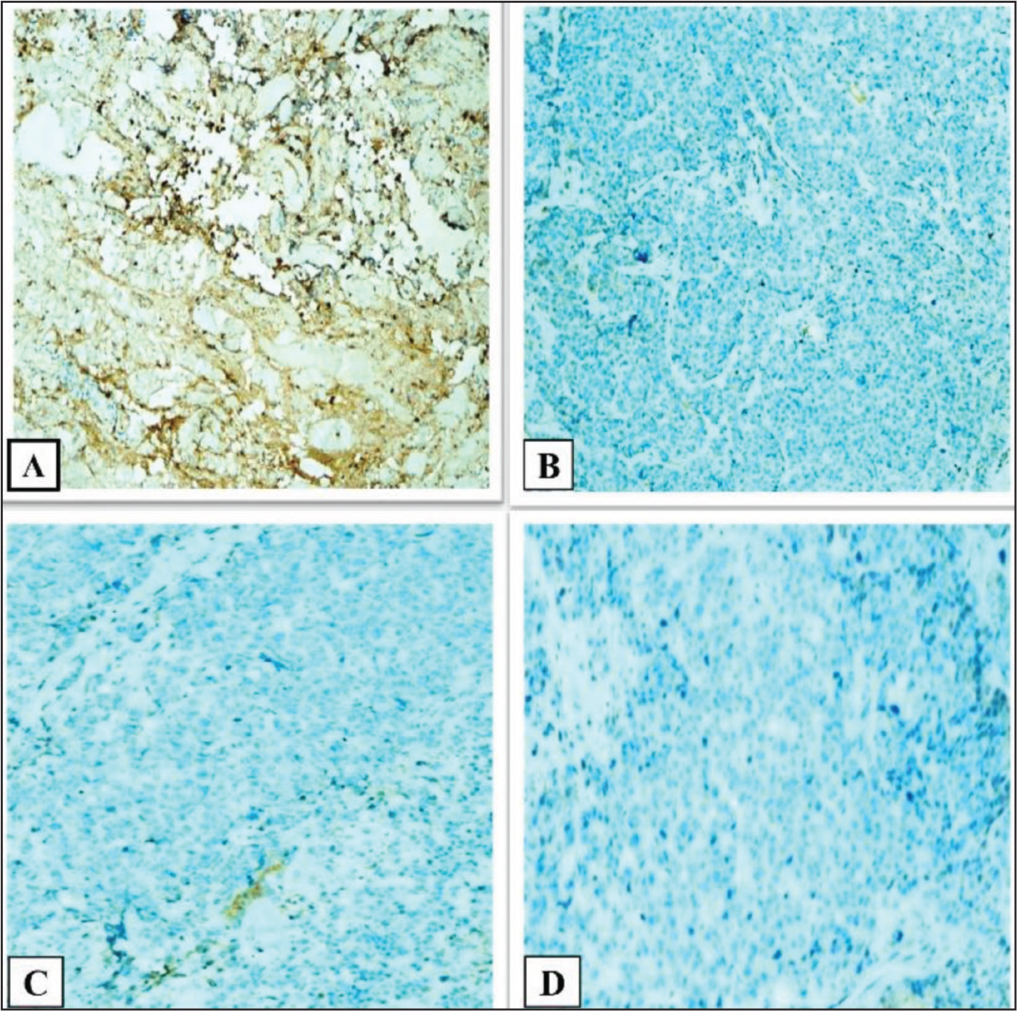
From the histopathology report, the differential diagnosis considered were sinonasal undifferentiated carcinoma, lymphoma, NUT carcinoma, etc. Immunohistochemistry was suggested for further evaluation. A comprehensive panel of markers for immunohistochemistry was used (Table 1). Immunostains were positive for CK and CAM5.2, indicating an epithelial differentiation. There was a diffuse staining pattern with NUT immunostaining. INI1 was retained. Ki67 proliferation index was high (75%-80%) (Figure 3). Squamous differentiation was not evident and the squamous markers (P63, P40, and CK-high molecular weight) were negative (Figure 4). There was negative immunostaining for lymphoma, germ cell tumours, melanoma and lymphatic and vascular tumours. Given the morphology and the immunostaining pattern with NUT antibody, the possibility of NUT carcinoma, metastasis cannot be excluded.. The differential diagnosis which were considered and worked up accordingly has been described in Table 2. Given the rarity of the tumour, molecular studies for BRD3-NUT, BRD4-NUT, Deletion of NUT and NSD3-NUT studies were suggested for confirmation. However, patient could not get the molecular tests done because of financial constraints. After discussion with family members about the prognosis and outcome, the patient was treated with concurrent chemoradiation with 3 cycles of carboplatin and paclitaxel. One month later, he developed stridor and weakness in both lower limbs. Family members requested for chemotherapy. PDL-1(SP 263) immunostain was performed, which showed a TPS score of 4%-5%. Combined chemotherapy in the form of cisplatin and 5-fluorouracil was given along with immunotherapy with Pembrolizumab. However, his disease progressed with ascending sensory loss, motor paralysis and lung parenchymal involvement. So, end of life care was instituted, he was shifted to ICU and was started on Bipap ventilation. The patient had sudden cardiac arrest and succumbed to death.
Panel of Immunohistochemistry Markers
A: CK 7 patchy positive (400x), B: Ki67 75%-80% (100x), C: INI-1 retained (100x) and D: NUT diffuse positivity (100x)
A: LCA negative (100x), B: P63 negative (100x), C: P40 negative (100x) and D: PLAP negative (100x)
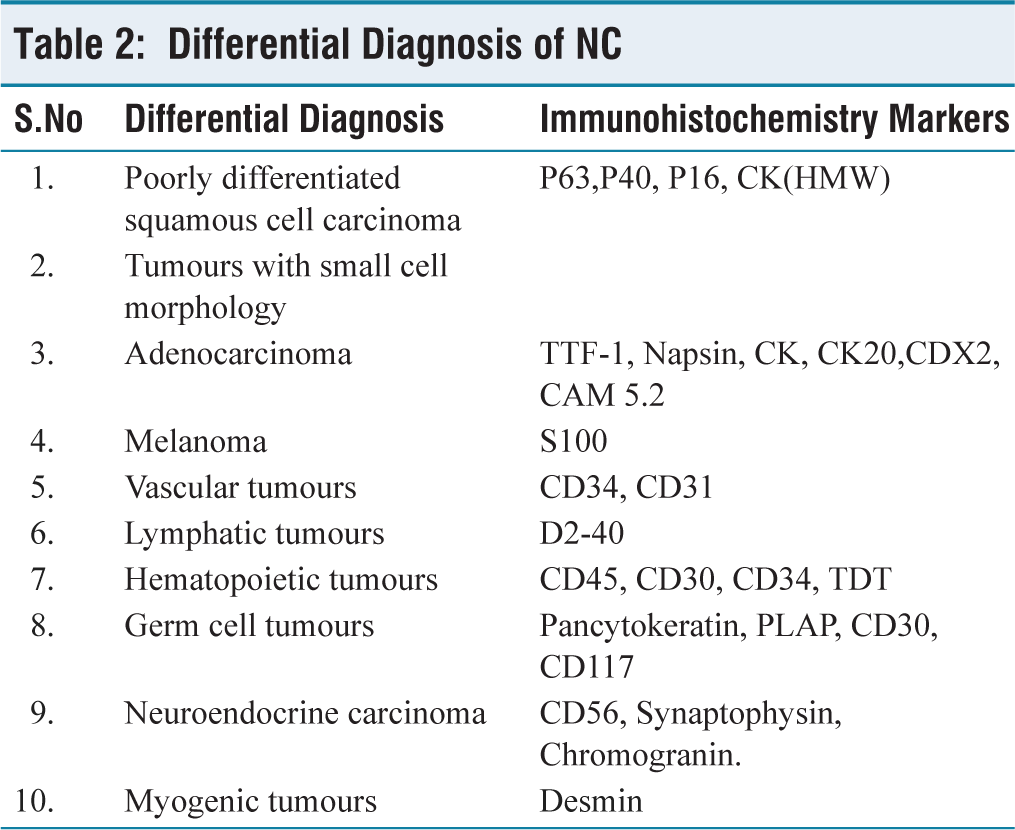
Differential Diagnosis of NC
Discussion
NC is specified by NUT gene rearrangement. The most frequent rearrangement that brings NUT into the frame with BRD4 is t(15:19) translocation. BRD4, a transcriptional activator, advances the BRD4-NUT fusion gene’s expression. The BRD4-NUT fusion was initially described in 2003 by French and associates.[8] The fusion protein BRD4-NUTM1 tethers NUTM1 to acetylated chromatin through the BRD4 bromodomains, which is the suggested mechanism of action. Therefore, HAT EP300 binds to NUTM1 at these chromatin locations, causing the neighbouring chromatin to get acetylated and forming large, active chromatin areas known as mega-domains.[9] Fusions of NUTM1 with BRD4 or BRD3 are more frequent than those with NSD3 or other genes. As a result, pro-differentiation genes located outside of mega-domains are suppressed, while transcriptional activators that promote cellular growth, such as TP63 and MYC, are recruited and expressed. NCs are not associated with carcinoma-related viruses such as EBV and HPV(3). In situ hybridization for these viruses can be used and the identification of the viruses virtually excludes the diagnosis of NC. The complementary role of contrast–enhanced CT, MRI and PETCT is immensely useful for specifying NC tumour burden, presurgical staging, metastasis, vascular invasion and for monitoring therapy response with the potential pitfall of each. Radiological mimics of NC are lymphoma, metastasis and sarcoma.
Morphological clues to diagnose NC
Poorly or undifferentiated malignant neoplasm Monomorphic small to medium-sized cells with vesicular nuclei and clear cytoplasm Abrupt squamoid differentiation Neutrophilic infiltrates Necrosis High mitotic rate
For the following reasons, NUT carcinoma diagnosis might be extremely difficult[10,11]:
Clinical presentation may mimic another neoplasm. By morphology, it can be undifferentiated neoplasm with or without abrupt keratinization. Sometimes, immunohistochemical analysis produces ambiguous data.
Essentially, in a typical clinical setting, NUTM1 FISH and/or NUT immunohistochemistry can be used to make a definitive diagnosis of NUT carcinoma. Hence, in patients (irrespective of age and gender) with sudden onset of symptoms, clinical diagnosis of metastatic carcinoma of unknown primary, histologically poorly differentiated neoplasm with or without squamoid differentiation and non-cutaneous carcinoma with monomorphic appearance, NC needs to be considered.
Diagnosis of NC can be achieved by IHC and/or molecular testing. As the current diagnostic standard for NC is NUT rearrangement, diffuse positivity for NUT protein is a solid indicator of NC as it provides 100% specificity and 87% sensitivity for the diagnosis of NC. NUT rearrangement should be carried out using molecular analysis methods such as FISH or NGS also.[12]
Bishop et al., Henrik et al.[11] and Ulrich M. Lauer et al.[3] emphasized the significance of differential diagnosis of NC. Gasljevic et al. explained aberrant expression of AFP in NC.[10] Hung et al. explained about aberrant expression of TTF-1.[13]
A definitive diagnosis will prompt the physicians for targeted therapies. The standard therapy for NC, a tumour known for its extreme aggressiveness, is still up for debate. Radiation therapy and surgical resection are independent measures of overall survival (OS) and progression-free survival (PFS). The role of radiotherapy is useful in head and neck tumours, but not so in mediastinal primaries. Beesley et al. found vincristine as an effective chemotherapy drug.[14]
Usually, NC presents at late stages, so combined therapy is preferred. The median survival of patients with NC is 6–7 months, occasionally rare cases with extended survival are evident. According to Wang et al., children with salivary gland NUT carcinomas constituted a separate subgroup that had a preference for men and had a higher overall survival rate. Combined clinical, imaging and histopathological examination is required to diagnose NC.
Conclusion
NC is a devastating cancer. Increased awareness amongst pathologists and clinicians is warranted due to its aggressive behaviour demanding a different treatment strategy than conventional poorly differentiated malignant neoplasms.
Footnotes
Acknowledgements
All technical staff of histopathology department, Apollo Hospitals, Chennai.
Consent for publication
Obtained.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
All authors can attest that in the submitted research article, only non-identifiable images are depicted. Approval was obtained from the Institutional Review Board (Ethics Committee) at Apollo Hospitals, Chennai, Tamil Nadu, India. (Institutional Ethics Committee (IEC). Application Number ASH-C-S-057/07-23).
Informed consent
Informed consent has been obtained.
Credit author statement
KM participated in Conceptualization, Methodology, Data curation, Analysis and Investigation, Manuscript preparation and Manuscript Editing.
SN participated in the Data analysis, Investigation, Manuscript preparation & editing.
AS participated in Conceptualization and supervision.
All the authors have reviewed and approved the manuscript.
Data availability
Raw data supporting the findings of this study are available from the corresponding author upon reasonable request.
Use of Artificial Intelligence
AI is not used in this article.
