Abstract
Background and Aims:
Colorectal carcinomas are one of the most prevalent malignant neoplasms globally and are characterised by a range of genetic and epigenetic alterations, primarily chromosomal instability and microsatellite instability. This study aims to assess the expression of MMR proteins in patients with colorectal carcinoma and to examine the relationship between MMR status and various histopathological and clinical variables.
Material and Methods:
All biopsy and resection cases of colorectal carcinoma on which MMR was done by IHC were studied from December 2022 to November 2023. Histopathological and immunohistochemical assessments of mismatch repair proteins were done for 153 colorectal carcinoma patients in formalin-fixed paraffin-embedded tumour tissues. Statistical analysis was carried out using SPSS software (IBM, 28.0).
Results:
Out of 153 cases collected, 23 cases (15%) had a loss of MMR expression, and 130 cases (85%) had no loss of MMR protein. Combined loss of MLH1 and PMS2 was found in 16 cases (70%), combined loss of MSH2 and MSH6 was found in five cases (21%) and isolated loss of PMS2 was found in two cases (9%). Adenocarcinoma NOS, mucinous differentiation, medullary pattern, right-sided tumours, female gender and younger age group correlated with loss of MMR expression.
Conclusion:
Molecular study is the first approach for detecting MSI associated with CRC, IHC is a very good alternative to identify microsatellite instability. IHC methods can be used as a reliable tool in the workup for MSI as IHC markers (antibodies to the MMR proteins) are now easily accessible in most laboratories.
Keywords
Introduction
Colorectal carcinoma (CRC) is a leading cause of cancer-related deaths, ranking third in males and second in females.[1,2] While screening has reduced deaths in older populations, there is a concerning rise in incidence among younger individuals. CRC involves various genetic and epigenetic alterations, with 85% from chromosomal instability and 15% from microsatellite instability (MSI).[3] Among MSI-High (MSI-H) cases, 13% are sporadic, and 2%-3% are linked to Lynch syndrome due to germline mutations in mismatch repair (MMR) genes. MSI-H tumours generally show a lower risk for metastasis, necessitating accurate prognostic and therapeutic decisions.[4] Our study evaluated the immunohistochemical expression of mismatch repair genes in CRC cases at a quaternary care centre in Southern India.
Materials and Methodology
The study was conducted with 153 cases of colorectal carcinoma from December 2022 to November 2023. It was a prospective observational study that included colonoscopic biopsies and resection specimens. The study aimed to assess the expression of MMR proteins in colorectal carcinoma by immunohistochemistry and to correlate histopathological and clinical variables with MMR status.
Methodology
Clinical details and treatment history were gathered from the laboratory information system. Tissue was fixed in 10% neutral buffered formalin, processed, paraffin-embedded and 4μ sections were stained with haematoxylin and eosin (H and E). Various histological parameters that were evaluated in resection samples were histological type, histological grade, lymphovascular and perineural invasion, necrosis, presence of mucinous differentiation, signet ring cell morphology, tumour budding, Crohn’s like peritumoral lymphoid response and tumour-infiltrating-lymphocytes-(-TILs). Histological type, histological grade and TILs alone were evaluated on endoscopic biopsies. TNM and anatomic stage/prognostic groupings on the resected sample were done according to AJCC’s eighth edition.
Immunohistochemistry (IHC) testing was done using VENTANA MMR assay for colorectal carcinoma in the VENTANA staining platform using an Optiview DAB immunohistochemistry detection kit and optiview amplification kit. A known positive control tissue or normal colon fixed and processed in the same manner as the patient’s specimens was run to serve as a system-level control. Unequivocal nuclear staining in lymphocytes, fibroblasts, or normal epithelium near the tumour serves as an internal positive control. Antibodies with clones used were MLH1 clone (M1), Mouse monoclonal primary antibody VENTANA, PMS2 clone (A16-4), Mouse monoclonal primary antibody VENTANA, MSH2 clone (G219-1129), Mouse monoclonal primary antibody VENTANA and MSH6 clone (SP93) Rabbit monoclonal primary antibody VENTANA.
IHC was interpreted as either intact or loss of nuclear expression. Intact nuclear expression was considered to be any positive staining of tumour nuclei. It is common for intact staining to be patchy. Intact nuclear expression was semiquantitatively categorised as intact homogenous and intact heterogeneous. An interpretation of loss of staining was considered only in the presence of positive staining in internal control cells, such as adjacent normal epithelial, endothelial, stromal and inflammatory cells.
Statistical Analysis
Descriptive statistics were presented with mean ± SD & frequency (percentage) for the continuous & categorical factors respectively. Chi-square/Fisher’s exact test was used to determine the association between clinical parameters and IHC results. P value <.05 considered as statistical significance. All the statistical analysis was carried out by using SPSS software (IBM, 28.0)
Results
We studied 153 cases of colorectal carcinoma with MMR expression from December 2022 to November 2023. There were 85 males (55.6%) and 68 females (44.4%). Male to female ratio was 1.2:1. The age range was 22-88 years, with a mean age of 59.8 ± 12.9 years. The study included 44 cases in the caecum, 36 in the rectum, 33 in the sigmoid colon, 24 in the ascending colon and eight each in the descending and transverse colon, with more right-sided than left-sided carcinomas. Out of 153 cases, 38 were small biopsies and 115 were resection specimens. Most patients were referrals, so detailed clinical information was not available. One patient had a history of chemotherapy, and another had synchronous tumours in the ascending and sigmoid colon, both with intact MMR expression.
The tumours were primarily adenocarcinoma, not otherwise specified (NOS) (n = 135; 88.2%), followed by mucinous adenocarcinomas (n = 13; 8.5%) and signet ring cell carcinomas (n = 5; 3.3%). In our study, histopathological grades were assessed by morphology: 34 cases were Grade 1 (well-differentiated adenocarcinoma), 105 cases were Grade 2 (moderately differentiated) and 14 cases were Grade 3 (poorly differentiated). No mucinous differentiation was noted in 112 cases, less than 10% mucinous differentiation was seen in 15 cases, 13 cases had 10-50% and 13 cases showed more than 50% mucinous differentiation. A signet ring cell pattern was present in 12 cases (7.8%), and a medullary pattern was found in three cases (2%).
Tumour necrosis was observed in 82 cases, Lymphovascular emboli was noted in 22 cases and perineural invasion was present in 10 cases. TILs were mild in 110 cases, moderate in 31 and marked in 12 cases. Tumour borders were infiltrative in 102 cases and expansile in 13 cases. Tumour budding was low in 70 cases, intermediate in 37 and high in eight cases. The peritumoral lymphoid response or Crohn’s like lymphoid reaction was present in 77 but absent in 38 cases. TNM and AJCC tumour staging (8th edition) was applied to all resected cases. The T-1 category was found in 2 (2.6%) cases, the T-2 category was seen in 56 (48.6%) cases, the T-3 category was found in 49 (41.7%) cases and the T-4 category was seen in 8 (7%) cases. Lymph nodal category showed N0 in 90 (78%) cases, N1 in 16 (13.5%) cases and N2 in 9 (8.5%) cases. Metastasis-M1 was found in 4 (3.5%) cases. Stage I and II tumours were 49 cases each (42.5%), stage III tumours were 15 in number (13.3%) and stage IV tumours were two cases (1.7%).
IHC Results
Out of 153 cases, 23 (15%) showed loss of MMR expression, while 130 (85%) had intact MMR proteins. The combined loss of MLH1 and PMS2 was observed in 16 cases (70%), loss of MSH2 and MSH6 in 5 cases (21%) and isolated loss of PMS2 in 2 cases (9%) [Figure 1]. Heterogenous pattern of expression was noted in all four proteins with MLH1 showing the highest degree of heterogeneity (n = 57), followed by MSH2 (n = 33), MSH6 (n = 31) and PMS2 (n = 31).
IHC results (n = number of cases)
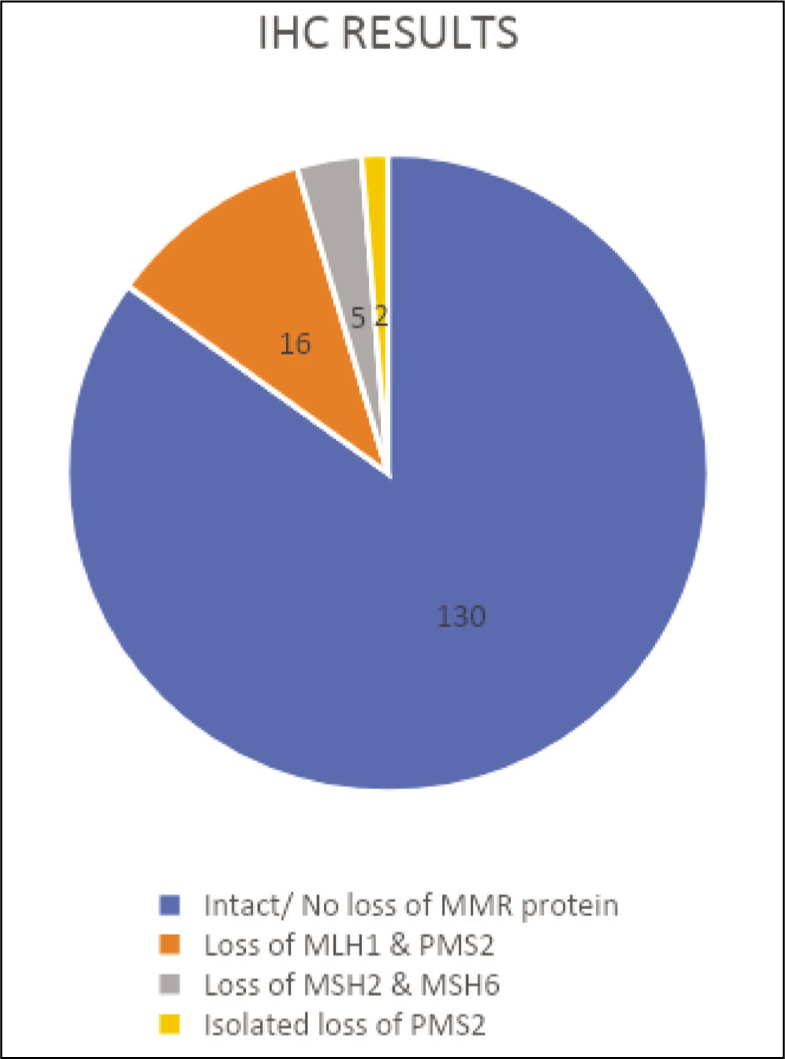
No Loss of MMR Protein
No loss of MMR protein expression was found in 135 cases (85%). About 80% were more than 50 years old, with males constituting 56.9%. The rectum (25.4%) was the most common site, followed by the sigmoid colon (23.1%), indicating that left-sided tumours had more intact MMR expression than right-sided ones. Adenocarcinoma NOS was the predominant histological type (92.3%), with Grade 2 tumours being the most common (67.7%). Mucinous differentiation, signet ring cell differentiation, medullary patterns and tumour heterogeneity were absent. Stage II (38%) and Stage I (33%) tumours were more prevalent in this group.
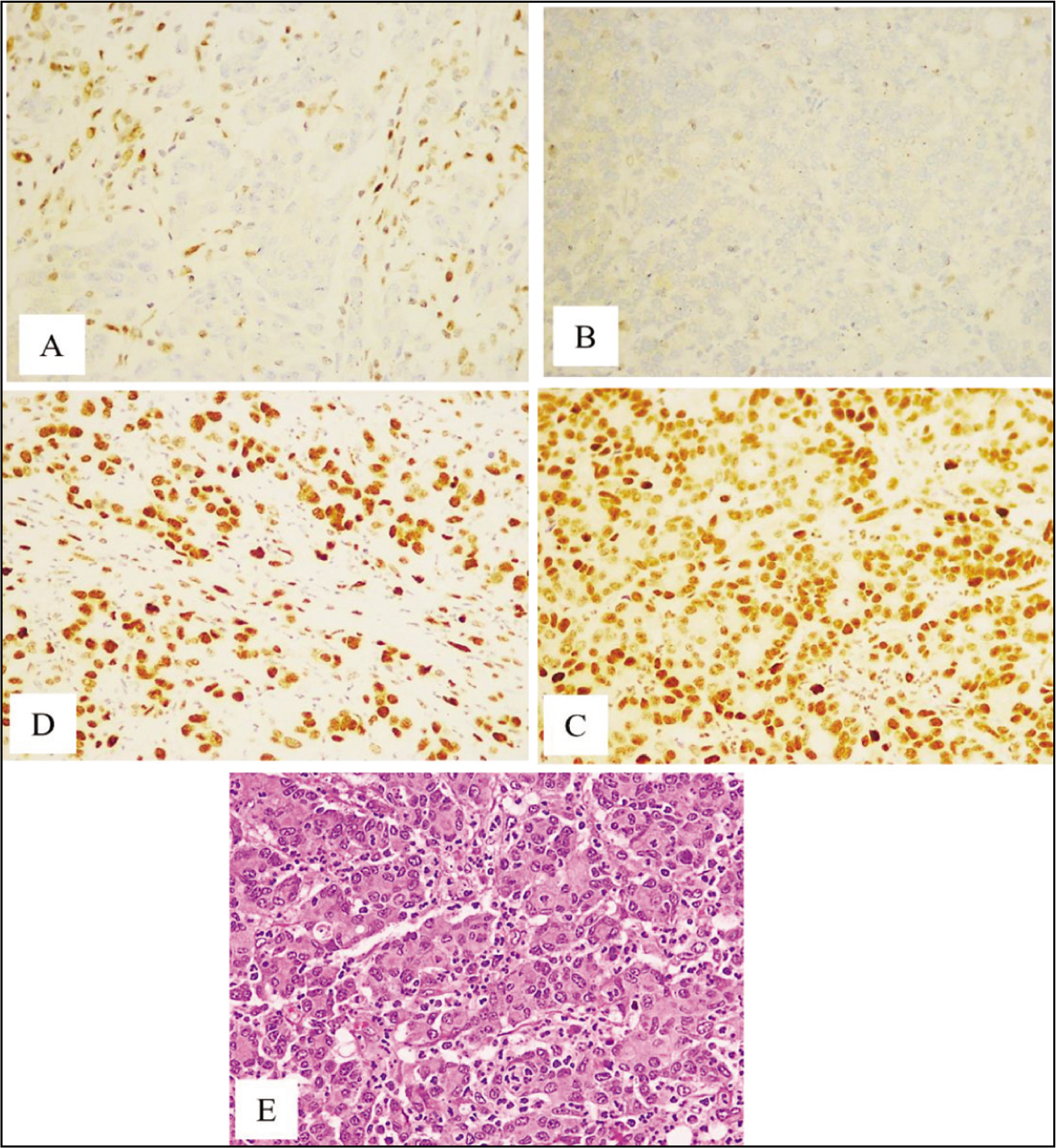
The Combined Loss of MLH1 and PMS2
Loss of MLH1 and PMS2 was observed in 16 cases (10.4%). Among these, 78.6% were patients over 50 years old, and 64.3% were females. They were noted only on the right side of the colon, with the caecum (n = 12) being the most common site, followed by the ascending colon (n = 4). Histologically, Adenocarcinoma NOS comprised 56.25% (n = 9) and mucinous carcinoma 43.75% (n = 7). Grade 2 tumours accounted for 68.75% (n = 11), while Grade 1 tumours made up 31.25% (n = 5). Mucinous differentiation was present in 31.25% (n = 5) of cases, signet ring cell differentiation in 18.75% (n = 3) and a medullary pattern in 7.1% (n = 1). Tumour heterogeneity was noted in two cases. In the analysis of tumour-infiltrating lymphocytes (TILs), 7 cases with mild TILs and six cases with moderate TILs showed a loss of MLH1 and PMS2. No cases with marked TILs exhibited deficiencies in mismatch repair (MMR). Fourteen cases had infiltrative tumour borders, and the rest had expansile borders. For tumour budding, nine cases with low budding and five cases with intermediate budding lost MLH1 and PMS2, whereas only one case was found in the high tumour budding category. Nine cases with a peritumoral lymphoid response lost MLH1 and PMS2, compared to six cases without such a response [Figure 2].
Loss of MLH1 and PMS2. (A) Loss of MLH1(100X), (B) Loss of PMS2 (100×), (C) Intact MSH2 (100×), (D) Intact MSH6 (100×) and (E) Adenocarcinoma with Medullary Phenotype- H&E (100×)
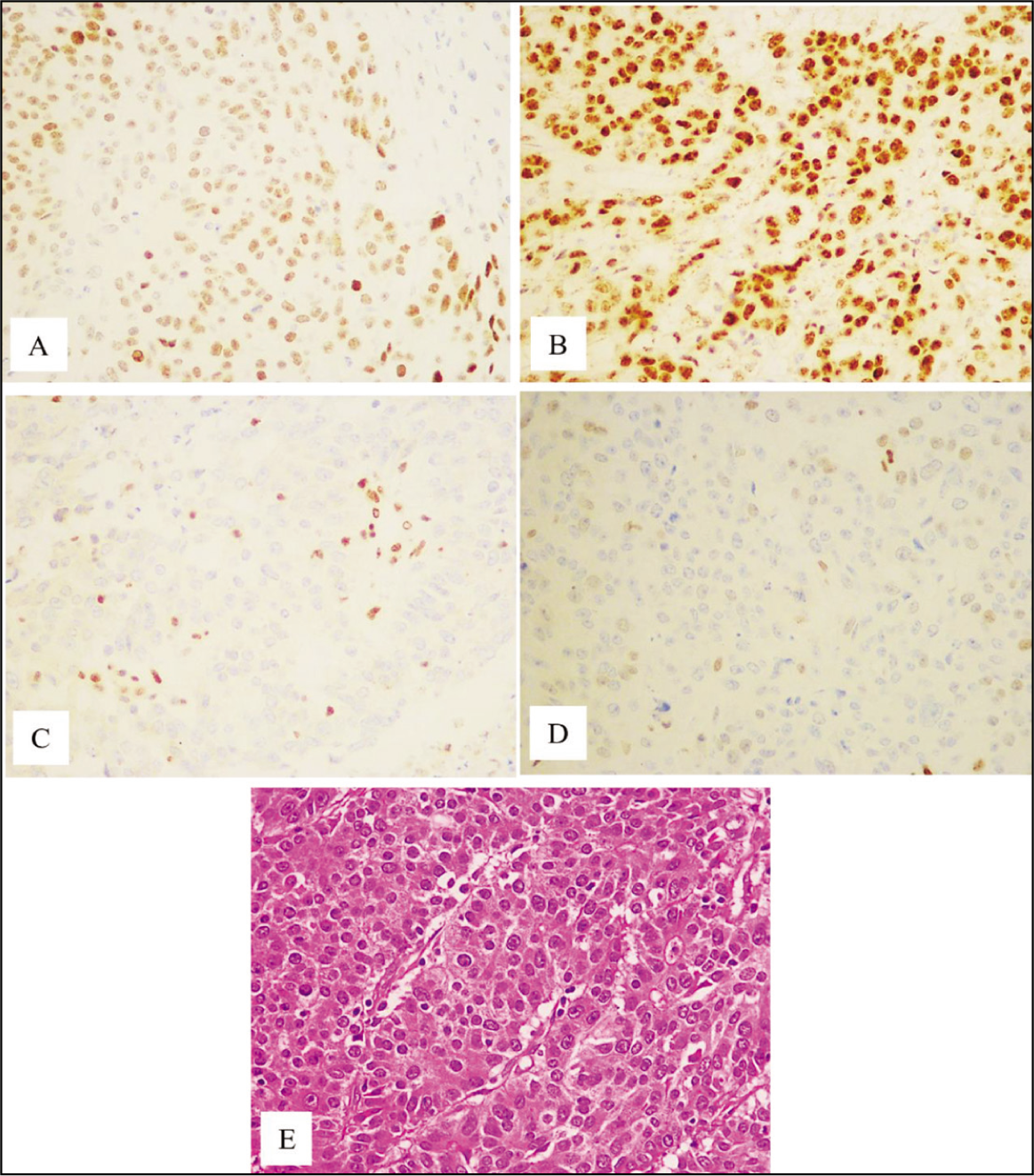
Combined Loss of MSH2 and MSH6
Loss of MSH2 and MSH6 was observed in five cases (3%). Three cases were patients over 50, and two were under 50. Four cases were male and one female. Three cases were located in the sigmoid colon, with one each in the rectum and ascending colon. Four cases were Adenocarcinoma NOS, and one was mucinous carcinoma. Medullary patterns and tumour heterogeneity were absent. Tumour necrosis was present in all five cases. Stage I tumours made up 50% of the cases, with one case each in stage II and stage III. Three cases had moderate TILs, while the rest had mild TILs. Four cases had infiltrative tumour borders. Three cases had low tumour budding, and the rest had intermediate budding. Three cases had a peritumoral lymphoid response [Figure 3].
Loss of MSH2 and MSH6. (A) Intact MLH1 (100×), (B) Intact PMS2 (100×), (C) Loss of MSH2 (100×), (D) Loss of MSH6 (100×) and (E) Adenocarcinoma- H&E (100×)
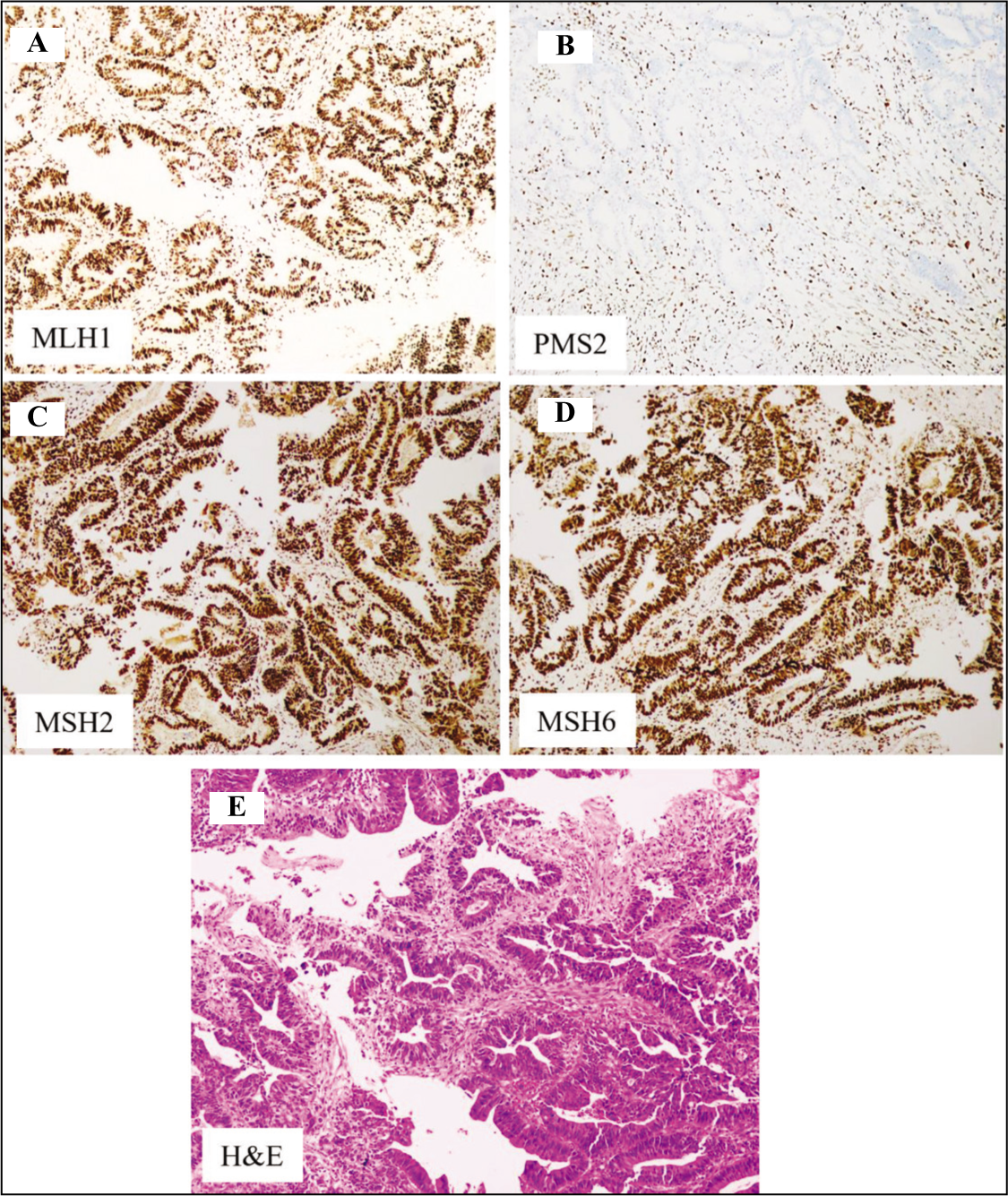
Isolated Loss of PMS2
Isolated loss of PMS2 was observed in two cases (1.3%), with one patient over 50 and the other under 50. Both were females. One case was in the rectum, and the other in the caecum. One tumour was a Grade 2 adenocarcinoma, and the other was a Grade 3 signet ring cell carcinoma. Both were stage II tumours, had mild TILs, infiltrative tumour borders and peritumoral lymphoid response. One case had low tumour budding, while the other had intermediate tumour budding [Figure 4].
Isolated loss PMS2. (A) Intact MLH1 (100×), (B) Loss of PMS2 (100×), (C) Intact MSH2 (100×), (D) Intact MSH6 (100×) and (E) Adenocarcinoma- H&E (100×)
Discussion
The annual incidence of colorectal cancer significantly impacts public health, with familial CRC accounting for 20% and Lynch syndrome for 3.5% of cases.[4] MMR-deficient cells accumulate DNA errors, leading to replication errors and non-functional proteins. MSI-H status indicates a higher likelihood of Lynch syndrome but can also occur without it, requiring validation through germline DNA testing. Identifying defective MMR proteins using immunohistochemistry involves detecting MLH1, MSH2, MSH6 and PMS2 in tumour nuclei. MMR gene products form heterodimer complexes, with MLH1 and MSH2 as primary partners. Mutations in MLH1 and MSH2 also lead to the loss of PMS2 and MSH6. MSI mutations often result from hypermethylation of the MLH1 promoter, leading to gene silencing, particularly in the BRAF pathway, which frequently features BRAF V600E mutations. Isolated PMS2 loss typically indicates a germline PMS2 mutation.[5,6] While molecular methods are the gold standard for microsatellite instability detection, IHC is more accessible, especially in resource-limited settings. IHC is straightforward and cost-effective. However, missense mutations may cause false negatives, so confirming ambiguous IHC results with PCR MSI testing is essential.[7] PCR MSI testing assesses MMR protein functional loss by extracting tumour DNA from formalin-fixed paraffin-embedded tissue and amplifying it with specific primers. It is the most precise method available, with MSI tumours typically showing instability in two or more of five markers.[8] Molecular testing, the gold standard for assessing DNA mismatch repair, requires normal colonic tissue and is more expensive than immunohistochemistry. IHC is quick and cost-effective, with around 95%-97% concordance with PCR testing.[9-11]
In our study, MMR protein expression was intact in 135 cases, with losses of MLH1 and PMS2 in 16 cases, MSH2 and MSH6 in five cases and isolated PMS2 in 2 cases. The frequency of MMR expression loss varies, with most of the studies reporting around 15% in sporadic colorectal carcinoma cases, similar to our study. Chew et al. reported a 21% loss in a study in Singapore, Soliman et al. reported 67% in Egypt and Dolcetti et al. found approximately 15% in Western populations.[12–14] Immunohistochemical studies in our cases revealed both heterogeneous and homogeneous expression patterns, with most cases showing intact, homogeneous expression. Joost et al. classified heterogeneity into two types: intra-glandular heterogeneity, where strong immunoreactive cells mix with negative cells, and zonal loss, marked by staining loss in adjacent glands.[15] Many investigations undertaken in comparison to molecular studies demonstrated heterogeneity of MMR protein expression to be retained and intact.[16,17]
Among 34 patients aged ≤50 years, 8 had a loss of MMR expression. In contrast, among 119 patients aged >50 years, 15 had a loss of MMR expression. Younger patients had a higher loss of MMR protein expression, consistent with the findings from Greenson et al., Jenkins et al., Kim et al. and Hyde et al.[18-21] In our study, there were 85 male patients (55.6%) and 68 female patients (44.4%), indicating a male predominance. Among males, 13% and 17.7% of females had a loss of MMR expression. Our findings showed a greater female predominance in MMR loss, similar to Greenson et al. However, no statistical significance was found regarding gender, aligning with studies by Kumar et al. and Thibodeau et al.[22,23]
In this study, 44 cases were noted in the caecum, 36 in the rectum, 33 in the sigmoid colon, 24 in the ascending colon and eight each in the descending and transverse colon. Right-sided carcinomas were more prevalent than left-sided, consistent with findings from studies like those by O’Brien et al. and Golas et al.[6,24] In the caecum, 27.2% showed a loss of MLH1 and PMS2; the ascending colon had a 16.6% loss, and the sigmoid colon had a 9% loss of MSH2 and MSH6. Although MMR loss was seen more in right-sided carcinoma, it was not statistically significant. Greenson et al. and Hyde et al. also indicated a higher incidence of MMR deficiency in more proximal and right-sided tumours.[18,21]
In our study, tumours were primarily classified into three types: Adenocarcinoma NOS (n = 135; 88.2%), mucinous adenocarcinoma (n = 13; 8.5%) and signet ring cell carcinoma (n = 5; 3.3%). Among Adenocarcinoma NOS, 10.2% showed loss of MMR protein expression, while 53.6% of mucinous adenocarcinomas exhibited loss. In signet ring cell carcinomas, 20% had isolated loss of PMS2. Studies by Reynolds et al. and Zeng et al. reported a statistical significance between the extracellular mucinous component and MMR status, consistent with our findings.[25,26] The medullary pattern was observed in three cases, with two cases showing loss of MMR protein. Hyde et al. also found a link between medullary phenotype and MMR deficiency.[21] Greenson et al. and Hyde et al. found that tumour grade, signet ring cell component, TILs, Crohn-like inflammatory reactions, infiltrative tumour borders and the absence of dirty necrosis are significantly associated with MMR-deficient tumours.[18,21] However, our study did not find a statistical correlation with these histomorphological features.
Tumour-infiltrating lymphocytes and peritumoral lymphoid reaction play a crucial role in mismatch repair protein-deficient colorectal carcinoma by contributing to the immune response against tumours. High levels of CD3+ and CD8+ T cells are associated with a better prognosis and enhanced response to immunotherapy, indicating a robust anti-tumour immune activity.[27] In MMR-deficient tumours, the presence of TILs and peritumoral lymphoid response suggests increased immunogenicity due to accumulated mutations, which influence the tumour microenvironment and can hinder tumour growth.[28-30]
Tumour budding is the formation of small clusters of cancer cells at the invasive edge of tumours. It is associated with a more aggressive tumour behaviour and a greater likelihood of metastasis, which often results in a poorer prognosis. Ryan et al. and Lugli et al. identified in their studies that MMR-deficient CRC patients with higher tumour budding had a worse clinical outcome. No correlation with tumour budding and MMR status was found in their study, as with our experience.[31-33]
Chemotherapy, alone or with a targeted therapy, is the standard treatment for metastatic colorectal cancer. Patients typically start on 5-fluorouracil-based regimens, often combined with anti-VEGF or anti-EGFR therapies. In stage II MSI-high cases, oxaliplatin is preferred over 5-fluorouracil, while in stage IV, immunotherapy can be used alone.[30] Immune checkpoint blockade therapy has advanced significantly, offering long-term responses for some patients. Pembrolizumab monotherapy has shown substantial improvements in health-related quality of life for those with MSI-H/dMMR metastatic CRC, compared to chemotherapy.[34]
In our study, various clinical and histopathological parameters were compared and correlated based on AJCC and CAP protocols. Differences between our findings and those of other studies are primarily due to varying cut-off values selected by different authors worldwide. Many Western studies focused on molecular findings alone or combined with immunohistochemistry, while recent studies correlating MMR protein expression with clinicopathological variables are limited. There are a few limitations to our study, such as, no follow-up was conducted regarding the treatment received and outcome, molecular testing correlation for IHC results of MMR status was not performed and detailed clinical history could not be obtained as most of them are referral cases.
Conclusion
Our study revealed significant correlations between tumour type, mucinous differentiation, medullary pattern and the pathological ‘T’ category with MMR loss. Right-sided tumours, younger patients and females showed greater loss of MMR expression. The most common loss was the combined loss of MLH1 and PMS2. While molecular studies are the primary approach for detecting microsatellite instability in colorectal cancers, IHC serves as a reliable alternative, as MMR antibodies are widely accessible. Regular IHC assessment in the routine histopathological examination is crucial for prognostic and therapeutic decision-making, aiding in the screening for Lynch syndrome in family members and associated tumours. Further research on MMR proteins and the significance of heterogeneity is needed to explore treatment and prognosis outcomes. Artificial intelligence is being explored to predict MSI based on tumour morphology, but these methods are not yet clinically viable.
Footnotes
Acknowledgements
Sivasankari and Logesh from the department of biomedical research.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
AMH-DNB-004/01/23.
Informed consent
Obtained from all patients.
Credit author statement
Dr. Dona-Conception or design of the work, Data collection, Data analysis and interpretation, Drafting the article.
Dr. Mekala: Study supervision, Data collection, Data analysis and interpretation, Drafting the article, Critical revision of the article.
Dr. Archana: Conception or design of the work, Study supervision, Data collection, Data analysis and interpretation, Drafting the article, Critical revision of the article.
All authors reviewed the results and approved the final version of the manuscript.
Data availability
Data supporting the findings of study is available with the corresponding author.
Use of artificial intelligence
The use of artificial intelligence is not relevant to this article, as it does not involve AI technologies or methodologies in its analysis or conclusions.
