Abstract
>Background:
Curcumin, a well-known phytocompound in turmeric, has demonstrated anti-cancer properties. However, its therapeutic efficacy is often limited. Additionally, Epidermal Growth Factor Receptor (EGFR) is frequently overexpressed in various cancers, making it a promising target for novel drug development. This study aims to utilise in silico analysis to identify potential anti-cancer agents within the phytocompounds of Curcuma longa (turmeric) that target the EGFR protein. The objective of this research is to investigate the binding interactions between curcumin and other Curcuma longa phytocompounds with the EGFR protein through computational modelling.
Methods:
Curcumin and paclitaxel (standard drug) were chosen as ligands and retrieved from PubChem. The researchers obtained the 3D structure of the EGFR protein from the Protein Data Bank (PDB) and prepared it for docking simulations. To assess curcumin’s drug-likeness and potential safety profile, online tools were utilised: SWISSADME analysed its ADMET (absorption, distribution, metabolism, excretion, and toxicity) properties, and ProTox-II evaluated its predicted toxicity. Finally, molecular docking simulations using Pyrx software were performed to predict how curcumin interacts with the EGFR protein. This computational approach helps predict how well a small molecule (ligand) like curcumin binds to a larger molecule (protein) like the EGFR receptor.
Results:
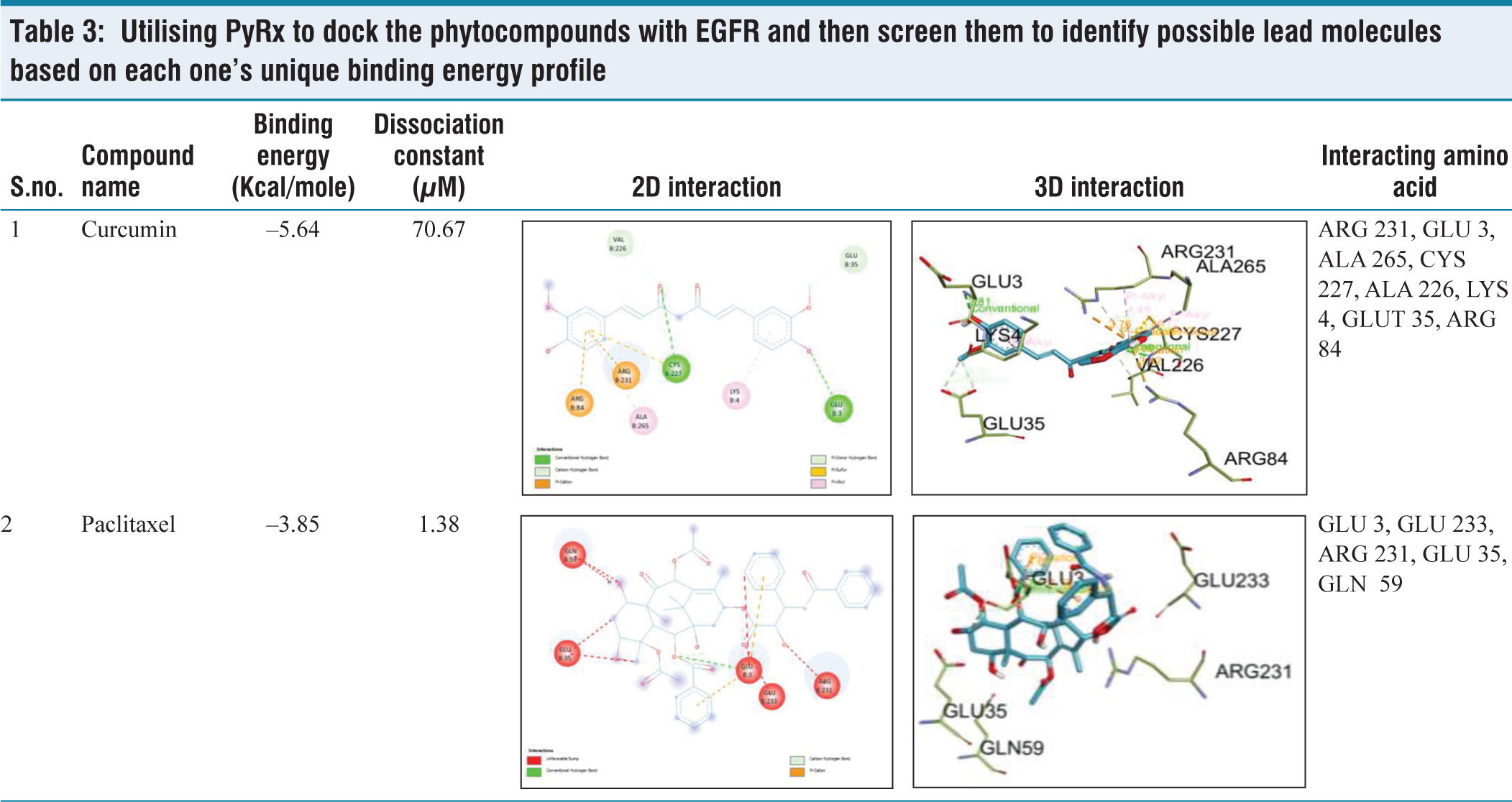
In silico analysis predicted favourable drug-like properties and a potentially safer profile for curcumin compared to paclitaxel. Curcumin exhibited high gastrointestinal absorption, adhered to Lipinski’s rule, and lacked predicted hepatotoxicity or carcinogenicity. The molecular docking binding energy for curcumin was –5.64 as compared to –3.85 for paclitaxel.
Conclusion:
This in silico investigation identified the potential for curcumin to target the EGFR protein, a promising strategy for cancer treatment. Curcumin displayed favourable drug-like properties and a potentially safer profile compared to the reference drug paclitaxel. However, in-vitro and in-vivo experiments are crucial to validate these findings and assess curcumin’s efficacy as a potential anti-cancer agent.
Introduction
Cancer develops in humans as a result of abnormal cell growth secondary to genetic alterations that disrupt normal cellular pathways and functions. Cancer is the second leading cause of mortality worldwide.[1] The commonly available chemotherapeutic agents for cancer treatment have several drawbacks, including the development of resistance to existing drugs, toxicity of the therapeutic agents, lack of awareness for alternative treatment approaches, and the absence of a specific curative drug. These drawbacks have led researchers to search for novel compounds with anti-lung cancer properties.[2] Researchers are focusing on natural sources to discover new bioactive compounds.[3] Traditional medicine practitioners in India, Southeast Asia and China have long recognised the health benefits of Curcuma longa L., or turmeric. Curcumin, the active ingredient in turmeric, was initially used to treat cholecystitis all the way back in 1937.[3] Curcumins ability to inhibit tumour cell proliferation and migration, as well as inhibit angiogenesis, induce apoptosis, and increase sensitivity to anti-tumour therapy, is mediated by modulation of molecular level targets such as micro-RNAs, transcription factors, genes and cytokines linked to apoptosis and proliferation.[4,5] Curcumin is widely utilised for the treatment of many different disorders, such as gastrointestinal illness,[6] liver cirrhosis,[7] cardiovascular illness,[8] diabetes,[9] and cancer, because of its excellent pharmacological action.[10] Increasing research indicates that curcumin can slow the spread of lung cancer cells by altering their epigenome, preventing cell division, and causing apoptosis.[11] These findings suggest that curcumin could be very useful in the treatment of cancers, however, our current knowledge of the drug’s functional mechanism prevents us from using it more widely.
Materials and Methods
Ligand Selection
Curcumin and the standard drug paclitaxel were chosen as ligand and control, respectively, for this investigation. Simple data format (SDF) ligand structures were downloaded from the PubChem server (
Target Protein Preparation
A protein data bank (
ADMET (Absorption, Distribution, Metabolism, Excretion, and Toxicity) Studies
An ADMET study is required to determine the pharmacodynamics properties of the chemical. To determine the active pharmaceutical ingredient (ADME) properties of natural substances and pharmaceuticals that are most likely to be suitable matches, the website SWISSADME (
Toxicity Analysis
Predictive toxicity analysis was done by the online tool ProTox-II (
Molecular Docking
The process of molecular docking was a strong computational approach used to anticipate how receptors and ligands will interact. Molecular docking was performed on the Pyrx. ChemBioOffice 15.1 (Waltham, MA, USA) and the X-ray crystal structure database (PDB) were used to construct curcumin’s 3D structure. Before starting the docking, we added polar hydrogen atoms, removed structural water molecules and co-crystal ligands, assigned the AMBER7 FF99 field atomic charge to protein atoms, and carried out a termini treatment.
Results
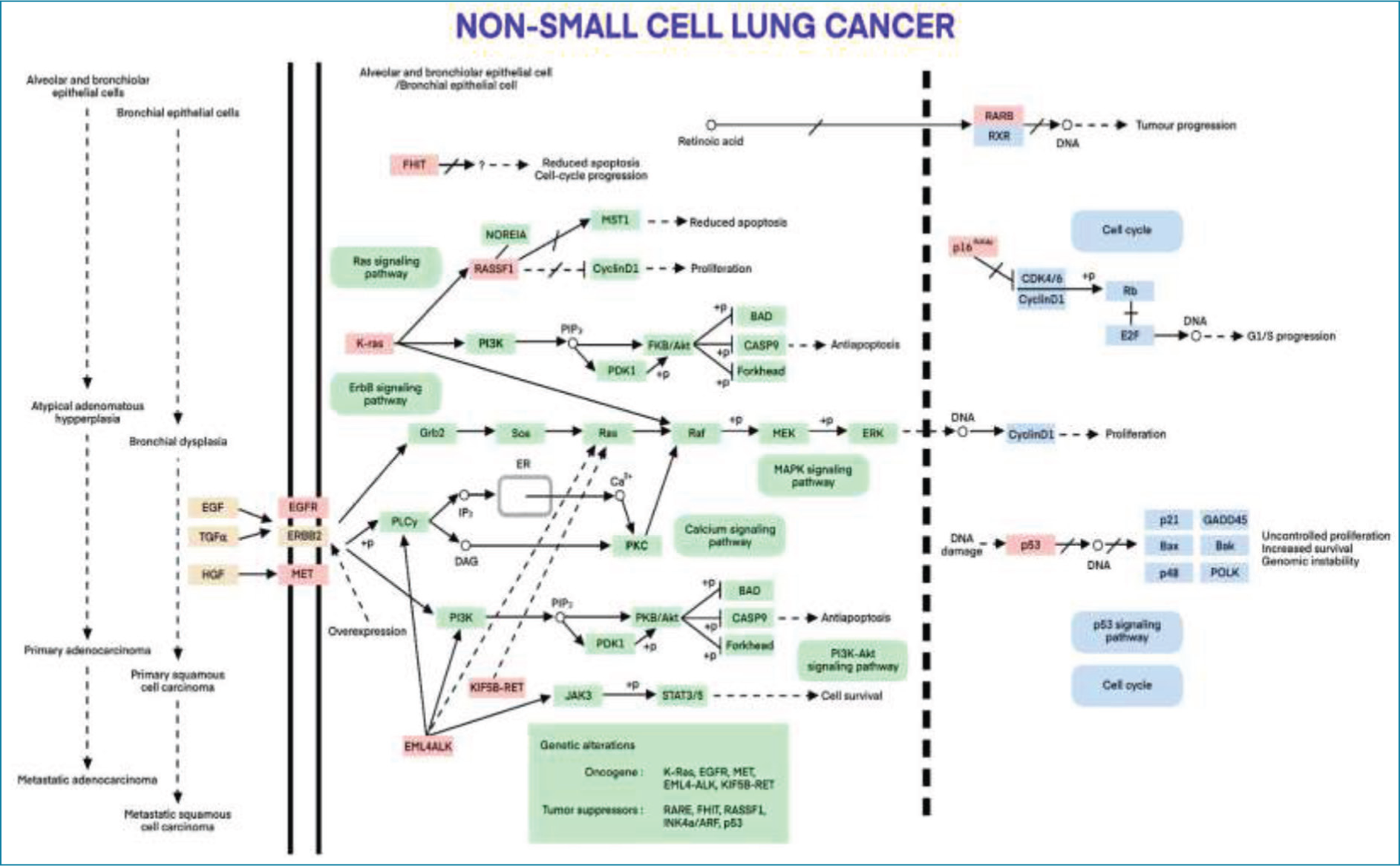
In this study, we selected curcumin compounds and analysed their ADME properties like gastrointestinal absorption, Lipinski rule and CYP family enzyme inhibitors properties which are shown in Table 1. Apart from this we also checked its toxicity analysis through ProTox-II and its activity is shown in Table 2. Molecular docking analysis shows that curcumin has better interaction with EGFR as compared to the standard drug, the binding energy has been shown in Table 3. Figure 1 shows the KEGG pathway of EGFR in small-cell lung cancer.
SwissADME web server evaluation of the curcumin’s ADME properties found in Curcuma longa

Toxicity profile based on ProTox-II-predicted values of paclitaxel and curcumin; active indicates some toxicity based on predicted data, while inactive indicates no toxicity found

Utilising PyRx to dock the phytocompounds with EGFR and then screen them to identify possible lead molecules based on each one’s unique binding energy profile
The KEGG pathway of EGFR in small cell lung cancer
ADMET Analysis
The online platform SWISSADME was used to evaluate the ADMET profile of curcumin. The results in Table 1 indicate that curcumin possesses favourable drug-like properties:
High gastrointestinal absorption: This suggests curcumin has the potential to be well-absorbed by the body after oral administration.
Compliance with Lipinski’s rule of five: Curcumin adheres to this rule, which is a set of guidelines commonly used to predict drug-likeness based on molecular properties.
No inhibition of major CYP enzymes: The analysis indicates that curcumin is unlikely to interfere with the activity of key Cytochrome P450 (CYP) enzymes involved in drug metabolism. This could minimise potential interactions with other medications metabolised by these enzymes.
Toxicity Analysis
The in silico toxicity evaluation using ProTox-II provided insights into the potential safety profile of curcumin compared to paclitaxel (a standard chemotherapeutic drug) as shown in Table 2. While both compounds exhibited predicted immunotoxicity (potential to affect the immune system), some key differences were observed:
Hepatotoxicity: Curcumin showed no predicted toxicity to the liver, whereas paclitaxel did.
Carcinogenicity: Neither curcumin nor paclitaxel exhibited predicted carcinogenicity (cancer-causing potential) based on this analysis.
Mutagenicity: Curcumin did not show mutagenic potential (ability to cause genetic mutations), while paclitaxel did.
Cytotoxicity: Curcumin was predicted to be non-cytotoxic (not toxic to cells), whereas paclitaxel showed predicted cytotoxicity, which is consistent with its known mechanism of action as an anti-cancer drug.
Discussion
The search for less toxic alternative treatments for lung cancer is the need of the hour. Lung cancer has been shown to exhibit dysregulation of a number of biochemical pathways, which may be targets for therapeutic intervention. These pathways include NF-kB activation, EGFR and PI3/AKT/mTOR signalling, STAT3 expression, the MAP kinase cascade, and VEGF-mediated angiogenesis.[12,13] While targeted medicines have shown some encouraging results, the limited clinical response observed with single-agent biologic therapy may be partly explained by the intricate interactions among different signalling pathways.[14] Curcumin is a natural product that is considerably non-toxic and has a variety of inhibitory effects on a number of different pathways involved in carcinogenesis. Curcumin may prove to be an appropriate adjuvant therapy for head and neck cancers that are resistant to currently available treatments due to its broad-reaching mechanism of action and lack of systemic toxicity.[15] In addition, curcumin has also shown some anti-tumour benefits in the treatment of lung cancers in preclinical studies.[4] The findings of the current in silico study point towards a favourable ADME and toxicity profile of curcumin in addition to its potential anti-cancer action by targeting the EGFR, a target implicated in several cancers, such as non-small cell lung cancer.
Conclusion
This in silico investigation identified potential for curcumin to target the EGFR protein, a promising strategy for cancer treatment. Curcumin displayed favourable drug-like properties and a potentially safer profile compared to the reference drug paclitaxel. According to the findings of this in silico study, curcumin shares a compatible structure with the EGFR target, which contributes to its high affinity as compared to the standard drug. This was demonstrated by the fact that it had higher free energy and dissociation value. However, in-vitro and in-vivo experiments are crucial to validate these findings and assess curcumin’s efficacy as a potential anti-cancer agent.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
Ethical permission was not applicable for this article, as this is a review article drafted from various research articles and not from patients directly.
Patient consent
Not applicable as this project is an in silico study.
Credit author statement
Mishra HP – Idea, conceptualization, supervision, writing draft, approved final draft.
Suvvari TK – Resources, project administration, writing draft and revision of draft, approved final draft.
Grover S – Data collection, formal analysis, writing draft and revision of draft, approved final draft.
Mohapatra C – Resources, validation, writing draft and revision of draft, approved final draft.
Patra PK – Resources, validation, writing draft and revision of draft, approved final draft.
Data availability
Data is available and can be provided on request.
Use of artificial intelligence
No generative AI or AI-assisted tools was used for this project.
