Abstract
Background:
Metastases from extrathoracic malignancies most commonly spread to the lungs as the second most prevalent site. Detecting metastases can indicate the necessity of chemotherapy or surgical removal. Conversely, ruling out pulmonary metastases is vital for planning a potentially curative treatment, highlighting the importance of understanding the imaging characteristics of lung metastases. This study aims to determine the different patterns of pulmonary metastases in various malignancies.
Materials and methods:
A hospital-based observational study was conducted at Department of Respiratory Medicine, A.J Institute of Medical Sciences and Mangalore Institute of Oncology, Mangalore. The study included 50 consecutive cases of diagnosed extra-thoracic malignancies with intrathoracic metastases attending our hospital. Screening of the cases with primary neoplasms was done as per the American College of Radiology (ACR) Appropriateness Criteria. The data was initially inputted into a Microsoft Excel spreadsheet and subsequently imported into SPSS software, version 24.0, for statistical analysis.
Results:
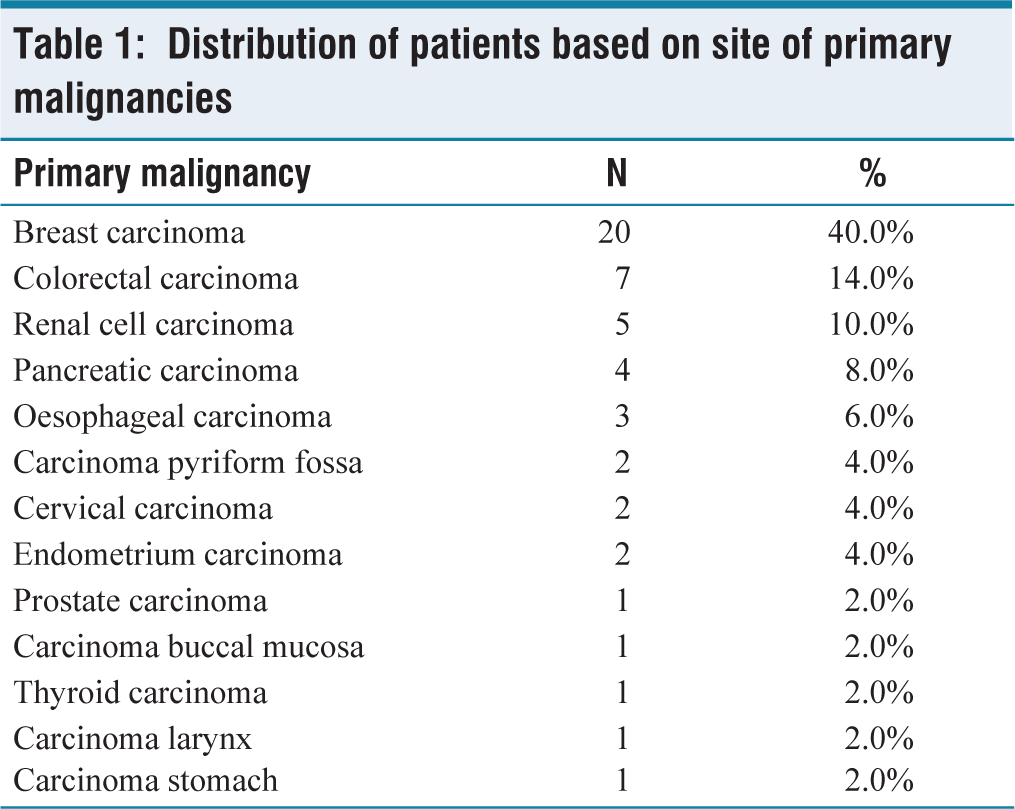
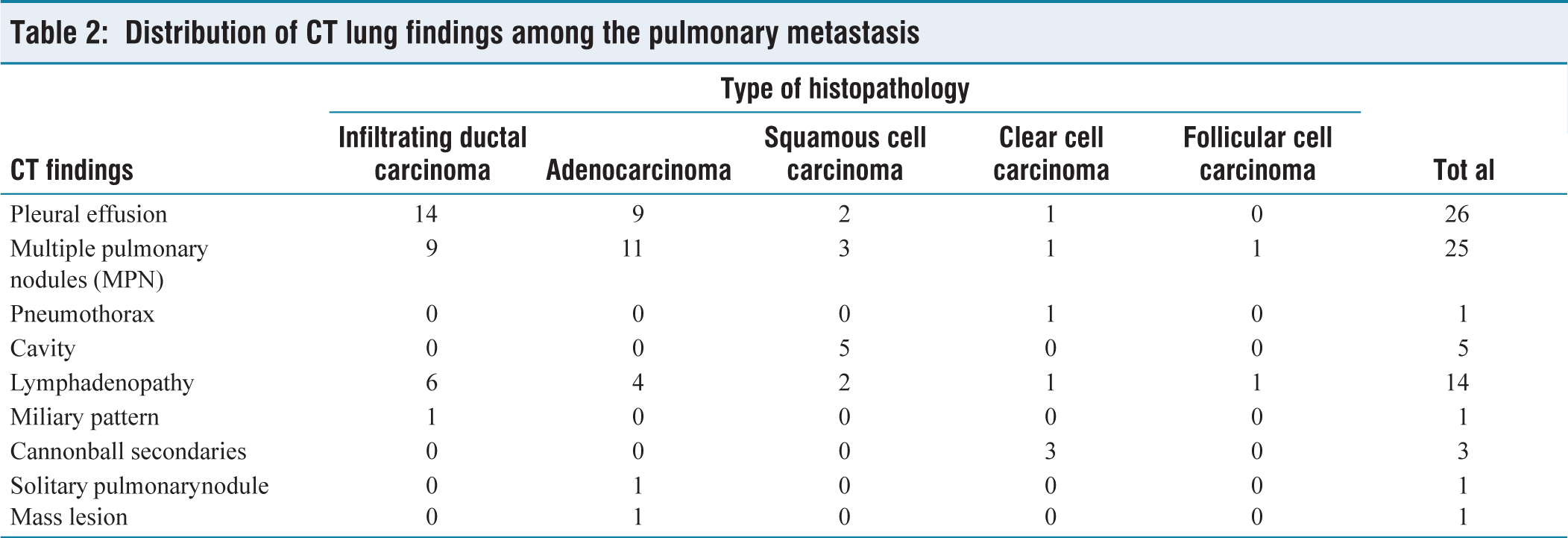
The most common site of primary malignancy was breast carcinoma (40%) followed by colorectal carcinoma (CRC) (14%), renal cell carcinoma (RCC) (10%), pancreatic carcinoma (8%), and oesophageal carcinoma (6%). Most common histopathological subtype of primary malignancy was infiltrating ductal carcinoma (40%) followed by adenocarcinoma (34%), squamous cell carcinoma (14%), clear cell carcinoma (10%), and follicular cell carcinoma (2%). Most common presenting respiratory symptom was breathlessness (54%) and cough (42%). The common findings seen on CT lung were pleural effusion (52%) and multiple pulmonary nodules (50%).
Conclusion:
Recognising the pulmonary metastases pattern is pivotal for early detection and individualised management. Comprehensive patient care, encompassing clinical, imaging, and a multi-disciplinary approach, is vital for improved patient outcomes.
Introduction
Malignancies are the second leading cause of death where the incidence has been increasing in recent decade.[1] As the malignancy progresses, it metastasises to various other organs. Lung metastases are the second most common site of secondary cancer spread from extrathoracic malignancies. They predominantly originate from primary tumours in areas such as the head and neck, breast, stomach, pancreas, kidney, bladder, genitourinary tract, and sarcomas. Notably, approximately 20% of all metastatic diseases are confined to the lungs. The presence of pulmonary metastases in patients with pre-existing extrathoracic malignancies signifies disseminated disease and categorises the patient into stage 4 of the TNM (tumour, node, and metastasis) staging system. This designation typically carries an unfavourable prognosis and necessitates a revised management approach.[2]
The five-year survival rate in patients with extrathoracic malignancies who develop pulmonary metastases is less than 5%. Metastases often render a malignancy non-operable; however, the possibility of resection depends on various factors. In some cases, surgical removal of metastatic lesions is feasible, potentially leading to a significant improvement in the 5-year survival rate, which can reach as high as 30%–40%. Survival also depends on number of metastases, fewer the number better the survival. This makes early diagnosis of metastases important.[3] Histologically in lung metastases, adenocarcinoma can be found in 60%, squamous cell carcinoma 15%, unspecified tumours including small cell and undifferentiated tumours in 25%. Pulmonary metastases differ in form, size, distribution, topography and various other radiological characteristics with primary lung malignancy.[4]
Sometimes pulmonary metastases may be the initial expression of occult primary malignancy. In this setting, primary malignancy can be searched depending upon the characteristic patterns of metastases, histology, age and sex of the patient.[5] The detection of pulmonary metastases holds substantial implications for patient care. The presence of metastatic disease can indicate the requirement for chemotherapy or surgical intervention, while the absence of pulmonary metastases can guide the development of a potentially curative treatment strategy. For healthcare professionals, it is crucial not only to possess a fundamental understanding of the imaging characteristics of lung metastases but also to grasp the underlying mechanisms and physiological processes involved in metastatic dissemination. Additionally, a comprehension of how tumours respond to therapy and adherence to oncologic principles in patient management and follow-up are equally vital aspects of clinical practice in this context. The present study is to understand the patterns of pulmonary metastases which will help in better evaluation and management of various malignancies.
Materials and Methods
A hospital-based, observational study was performed from October 2015 to September 2017 with the diagnosed cases of extrathoracic malignancies with intrathoracic metastases, attending as outpatient/inpatient at Department of Respiratory Medicine, A.J Institute of Medical sciences and Research, Mangalore and Mangalore Institute of Oncology, Mangalore. A total of 50 cases of extrathoracic malignancies attending our hospital were taken for the study after obtaining institutional ethical clearance. The consecutive type of sampling method was used for selecting the participants who satisfied inclusion (patients with extrathoracic malignancies with intrathoracic metastases) and exclusion (patients with primary lung cancer) criteria.
An informed written consent was obtained from all patients before start of the study. The standard hospital protocol was followed for examination and investigation of all study participants. Complete clinical details like symptomology, clinical findings and details of underlying extrathoracic malignancy were recorded. The histopathology of pleural effusion samples were recorded. The screening of the cases with primary neoplasms that are known to metastasise to the lungs was done as per the American College of Radiology (ACR) Appropriateness Criteria.[6] Imaging findings like pattern of involvement, involvement of lobe(s), associated lymphadenopathy, etc. were noted for all the patients in a pre-designed structured proforma.
Statistical Analysis: All the data was initially inputted into a Microsoft Excel spreadsheet and subsequently migrated to SPSS software, specifically version 24.0 from IBM, based in Chicago, Illinois, USA, for comprehensive statistical examination. Qualitative data were represented in terms of frequency and percentage, whereas quantitative data were expressed using the mean and standard deviation (SD) as summary statistics.
Results
The mean age of the study cases was 55.48 years with 68% of them being over 50 years of age. Almost similar distribution was observed with respect to gender with 54% males to 46% females. The history of smoking was given by half of the study subjects (50%), all cases were male patients. The distribution of patients based on primary malignancies was tabulated in Table 1. The most common histopathological subtype of primary malignancy was infiltrating ductal carcinoma (40%) followed by adenocarcinoma (34%), squamous cell carcinoma (14%), clear cell carcinoma (10%), and follicular cell carcinoma (2%). In most cases, primary tumour was present since 1-5 years (62%). Duration of less than 1 year was seen in 36% cases.
Distribution of patients based on site of primary malignancies
The most common presenting respiratory symptom was breathlessness (54%) and cough (42%). Other less common presentations were chest pain (12%) and haemoptysis (4%). No symptoms were present in 34% cases. The new onset respiratory symptom was seen in 40% cases while in majority of the cases the symptoms were present for 1-6 months (58%). The most common findings seen on CT lung was pleural effusion (52%) followed by multiple pulmonary nodules (50%). Other major findings were lymphadenopathy (28%), cavitation (10%) and canon ball secondaries (6%). Multiple pulmonary nodules were most commonly observed in lower lobes in both right and left lungs. The distribution of CT lung findings among the pulmonary metastasis were tabulated in Table 2.
Distribution of CT lung findings among the pulmonary metastasis
Discussion
The identification of pulmonary metastases indeed exerts a profound influence on the management of patients, as its presence implies the necessity for interventions such as chemotherapy or surgical resection. Conversely, the absence of pulmonary metastases serves as a valuable indicator for formulating a more curative strategy directed towards addressing the primary malignancy. To our knowledge, no previous studies were conducted where the entire spectrum of metastatic lung tumours was studied.
Site of Primary Malignancy
The most common primary site of malignancy in present study was breast carcinoma (40%), followed by colorectal carcinoma (CRC) (14%), renal cell carcinoma (RCC) (10%), pancreatic carcinoma (8%) and oesophageal carcinoma (6%). Other less common sites include carcinoma pyriform fossa (4%), cervical carcinoma (4%), endometrial carcinoma (4%), carcinoma prostate (2%), carcinoma buccal mucosa (2%), carcinoma stomach (2%), carcinoma thyroid (2%), and carcinoma larynx (2%) which is comparable with study by Hirakata K et al.[6]
Metastatic dissemination to the lungs represents a prevalent occurrence in the context of malignancy. It has been documented that a substantial proportion, ranging from 20% to 54%, of individuals diagnosed with extrathoracic malignancies exhibit the presence of pulmonary metastases upon post-mortem examination.[7,8] Malignant tumours have the potential to disseminate to the pulmonary region, with virtually any malignancy having the capacity for such metastatic spread. In terms of frequency, the most common sources of such metastases can be delineated, with breast cancer occupying a prominent position, followed by colon, stomach, pancreas, kidney, melanoma, prostate, liver, thyroid, adrenal glands, and male and female genital tracts, listed approximately in descending order of prevalence.[8]
Autopsy studies conducted on breast cancer cases have provided insights into the patterns of metastatic dissemination. These investigations have indicated that widespread metastatic involvement is frequently observed, with a predominant presence in the bones (70% of cases), followed closely by the lungs (66% of cases) and the liver (61% of cases).[9] The prognosis for patients with breast cancer is markedly influenced by the extent of systemic dissemination. Specifically, when examining survival rates, it becomes evident that among patients falling within the stage I-II category, the 5-year survival rate remains notably high, surpassing 70%. However, as the disease progresses to stage III, characterised by regional lymph node involvement, the survival rate experiences a considerable decline, ranging between 20% and 60%. Notably, the most pronounced decrease in survival is observed in stage IV, denoting systemic disease with manifestations such as lung or distant metastasis, where the 5-year survival rate plummets to a mere 5%.[9]
Prior research has yielded valuable information regarding the dissemination patterns of metastasis. These studies have demonstrated that various primary cancers exhibit varying tendencies in terms of metastasising to distinct sites and at differing frequencies.[10,11] At the point of diagnosis, approximately 20% of individuals with CRC have already progressed to metastatic disease, with the liver being the most prevalent site of metastasis, followed by the lung.[12] In the present study, CRC was the second most common tumour responsible for lung metastasis.
RCC is recognised for its diverse spectrum of presentations and the variability in its clinical course. The lung is the commonest site of metastases followed by skeletal system as observed by the studies of Sivaramakrishna et al.[13] and Weiss et al.[14]
Oesophageal cancer (EC) stands out as one of the exceptionally aggressive forms of cancer on a global scale.[15] The most prevalent pattern of metastasis in oesophageal cancer (ECM) typically involves the lymph nodes, lungs, liver, bones, adrenal glands, and brain. The tumour can grow into the mediastinum, possibly to the trachea, to the right bronchus and heart.
Pancreatic cancer, acknowledged as one of the most formidable malignancies, continues to exhibit a grim overall survival rate, whether in cases of metastatic disease or among patients with early-stage disease who have undergone ostensibly curative surgical interventions.[16,17]
Histopathological Subtypes
Most common histopathological subtype of primary malignancy was infiltrating ductal carcinoma (40%) followed by adenocarcinoma (34%), squamous cell carcinoma (14%), clear cell carcinoma (10%), and follicular cell carcinoma (2%).
Studies have shown that by absolute numbers adenocarcinomas far outnumber the other extrathoracic solid tumours that metastasise to lung. The contrary findings seen in present study were due to relatively small sample and increased number of breast carcinoma patients. In present study, adenocarcinoma comprises all the cases of GI malignancies along with 2 out of 3 cases of oesophageal carcinoma and 2 out of 2 cases of endometrial carcinoma.
In our study, all the cases of breast carcinoma were infiltrating ductal carcinoma which was reported by few studies in the literature.[18-21] All the cases of RCC were histopathologically identified as clear cell carcinoma which is in accordance with the results by Sivaramakrishna et al.[13]
Respiratory Symptoms
Most common presenting respiratory symptom was breathlessness (54%) and cough (42%). Other less common presentations were chest pain (12%) and haemoptysis (4%). No respiratory symptoms were present in 34% cases.
Lung metastases may be asymptomatic to begin with. The symptoms associated with lung metastases can vary depending on factors such as the number of tumours and their specific locations within the lungs. Other health conditions can cause the same symptoms as lung metastases. Nevertheless, when pulmonary metastatic lesions develop later in the course of advanced extrapulmonary cancer, the clinical presentation frequently revolves around the manifestations of advanced or terminal malignant disease, coupled with the signs and symptoms attributed to the primary cancer site. Respiratory symptoms generally included cough, breathlessness, recurrent chest infections, haemoptysis and chest pain or discomfort in the chest.
The lymphangitic spread of cancer into the lungs typically manifests as a sudden and rapid onset of breathlessness at rest, sometimes accompanied by a dry cough. This particular presentation is often observed in patients with a documented history of carcinoma, most frequently originating from sites such as the breast, stomach, pancreas, and prostate. Additionally, the presence of endotracheal and endobronchial metastases can give rise to symptoms like a new-onset cough, breathlessness, and on occasion, haemoptysis (coughing up blood) and chest pain.
Fujii et al.[22] reported a case of a breast cancer patient with solitary pulmonary metastasis. The patient was presented with dyspnoea at rest and dry cough as the main symptoms along with anorexia and weight loss. Gao et al.[23] observed in their study that cough was the most common (60%) complaint associated with secondary lung tumours from invasive ductal carcinoma of breast.
Radiological Findings
CT scan stands out as the most sensitive imaging modality for the detection of pulmonary metastases due to its exceptional spatial and contrast resolution, along with the advantage of minimal superimposition with adjacent structures such as bones and vessels. When compared to chest X-rays (CXR), CT scans excel in identifying a larger number of nodules, including those smaller than 5 mm in size. The ACR recommends that, in the initial screening for pulmonary metastasis in patients with confirmed extrathoracic malignancies, CXR should be the starting imaging modality. However, for individuals with bone and soft-tissue sarcomas, malignant melanoma, and head and neck carcinomas, a chest CT scan should be the primary imaging approach. In the case of patients with primary kidney or testicular cancers, the decision to perform a chest CT scan should be influenced by the presence of metastatic disease elsewhere in the body.[2]
In present study multiple pulmonary nodules were seen in half of the cases. The nodules were most commonly observed in lower lobes in both right and left lungs. Hirakata et al.[6] and Davis et al.[7] have showed that most common pattern of metastases to the lungs is multiple pulmonary nodules and they located in lower lobes of both lungs. In the context of secondary pulmonary nodules, haematogenous nodules are characterised by their random distribution. Macroscopically, these nodules tend to have well-defined borders, and their placement within the lung is haphazard in relation to the lung’s natural structure. Typically, they are situated towards the outer regions of the lung, particularly showing a preference for the lower lobes due to the higher blood flow in these areas.
A miliary pattern is characterised by the presence of numerous small nodules, typically measuring around 1-3 mm in size. This pattern can be observed in various malignancies, including vascular tumours like thyroid carcinoma, RCC, as well as, adenocarcinomas and sarcomas. In some instances, larger metastases may also be visible, particularly in cases of RCC, testicular carcinoma, and colon carcinoma. It’s worth noting that in the current study, all cases exhibiting cannonball secondaries were specifically associated with clear cell RCC.
Pleural effusion is one of the common metastatic patterns. The effusions often tend to be massive, recurrent and associated with breathlessness. This pattern is associated with extensive underlying lung and systemic metastases. Malignant effusions account for more than 50% of exudative pleural effusions. Around 20% of newly diagnosed pleural effusions in adults, as identified through chest radiography, are attributed to malignancies. The probability of a pleural effusion being malignant tends to rise with the patient’s age and the size of the effusion, with larger effusions carrying a higher likelihood of being associated with malignancy.[24-26] A significant majority, up to 80%, of malignant pleural effusions are attributed to metastatic adenocarcinoma, which includes breast carcinoma. A smaller percentage of these effusions can be traced back to lymphoma. In present study, malignant pleural effusion was observed in 52% cases with common sources being infiltrating ductal carcinoma of breast and adenocarcinoma of CRC.
Metastatic pulmonary nodules have the potential to develop cavities, particularly in cases of squamous cell carcinoma, adenocarcinoma, transitional cell carcinoma, and sarcoma.[24,27-32] In the present study cavity was seen in 10% cases and all of these cases were associated with squamous cell carcinoma. Seo et al.[33] stated that squamous cell carcinomas are most commonly present as cavity (69%) which is comparable to our study. The unproven histology of pulmonary metastasis was the main limitation of our study.
Conclusion
The recent observational study delves into pulmonary metastasis patterns in patients with extrathoracic malignancies, emphasising the grave nature of lung metastases, often indicating advanced stage 4 diseases with an unfavourable prognosis. The analysis showed a mean patient age of 55.48 years, predominantly older individuals, with a slight male dominance. Notably, half of the male subjects were smokers, suggesting a possible correlation between smoking and lung metastases, although this study does not confirm causality.
Breast carcinoma emerged as the primary malignancy causing lung metastases, trailed by colorectal and RCC. This trend stresses the need for continuous monitoring of patients with these primary tumours for potential lung involvement. Despite the prevalence of respiratory symptoms like breathlessness and cough, a significant 34% were asymptomatic, highlighting the need for periodic imaging for metastatic disease detection. Radiological evaluations revealed pleural effusion and multiple nodules as frequent findings. Interestingly, metastatic presentations on imaging differed based on the primary tumour’s histopathology; squamous cell carcinomas often showed cavities, while clear cell carcinomas displayed cannonball secondaries.
In essence, pulmonary metastasis presents diversely, necessitating tailored diagnostic and treatment plans. Recognising these patterns is pivotal for early detection and individualised management. Comprehensive patient care, encompassing clinical, imaging, and a multi-disciplinary approach, is vital for improved patient outcomes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Institutional ethical committee approval number
A.J. Institute of Medical Sciences and Research Centre - AJEC/REV/100/2015-16 dated 17.10.2015.
Informed consent
Obtained from all the participants in the study.
Credit author statement
All authors contributed equally in the manuscript.
Data availability
Data is included in the manuscript.
Use of artificial intelligence
The use of artificial intelligence is not relevant to this article, as it does not involve AI technologies or methodologies in its analysis or conclusions.
