Abstract
Background and Aims:
Given the absence of a universally accepted non-invasive and objective clinical tool to predict a patient’s response to stereotactic radiosurgery for Trigeminal Neuralgia (TN), this research endeavoured to appraise the potential of Diffusion Tensor Imaging as a predictive tool in managing TN.
Objective:
This study aimed to assess microstructural changes in the trigeminal nerve following CyberKnife radiosurgery (CKRS) for TN using diffusion tensor imaging (DTI) and to correlate these changes with clinical outcomes.
Methods:
Twenty-eight TN patients treated with CKRS underwent pre and 4-month post-treatment Three-Tesla magnetic resonance imaging scans with DTI metrics, including fractional anisotropy (FA), axial diffusivity (AD), radial diffusivity (RD) and mean diffusivity (MD), were collected. Clinical outcomes were assessed using the Barrow Neurological Institute Pain Intensity Score (BNI-PIS) at fourth, sixth and ninth months post-CKRS.
Results:
Twenty-four patients reported good pain relief (responders), while four patients did not experience adequate relief (non-responders). Responders exhibited significantly lower FA (mean drop of 13.8%, P < .001) and lower RA (mean drop of 16.1%, P < .001) compared to non-responders. RD, AD and MD remained constant across the study cohort.
Conclusion:
DTI metrics, particularly FA and RA, at 4 months post-CKRS, emerged as predictors of long-term treatment effectiveness for TN. These findings suggest the potential utility of DTI in evaluating microstructural changes in the trigeminal nerve post-CKRS and predicting clinical outcomes.
Background and Introduction
French physician Nicholas Andre coined the term ‘tic douloureux’ or ‘Trigeminal Neuralgia’ in 1756.[1] Trigeminal Neuralgia (TN) is characterised by paroxysmal lancinating or electrical shock-like episodes of neuropathic pain that are normally restricted to one side of the face along the course of affected trigeminal nerve. Periods without pain are frequent, and the duration might range from a few days to several years. The trigeminal nerve’s mandibular and maxillary branches are most often impacted.[2] According to the International Association for the Study of Pain, TN is defined as sudden, unilateral, severe, brief, stabbing, recurrent episodes of pain in the distribution of one or more branches of trigeminal nerve.[3] Medical management is the initial approach for the treatment of TN and typically consists a cocktail of, Carbamazepine, Gabapentin, Pregabalin and Baclofen.[4] Response to Carbamazepine may be helpful to differentiate TN from other orofacial pain syndromes. It is common for patients with TN to become refractory to medical management over time. Patients with TN refractory to medical management are considered for non-ablative or ablative treatments. Microvascular decompression (MVD) represents a non-ablative surgical procedure aimed at addressing the underlying origin of TN with neuro-vascular conflict, initially introduced by Taarnhoj in 1952 and subsequently refined by Jannetta.[5] On the other hand, stereotactic radiosurgery (SRS) is a non-invasive ablative approach inducing gradual nerve lesioning. Initial utilisation of SRS for TN dates back to 1953 when eminent Neurosurgeon Dr. Lars Leksell introduced this technique, achieving positive outcomes in two patients and publication of this data in medical literature did not occur until 1971.[6] However, identifying treatment responders remains a challenge to this day. This study aims to explore diffusion tensor imaging (DTI) as a tool for predicting long-term pain relief post-SRS. Such predictive metrics could enhance TN management, guiding medication adjustments effectively.
Methods
Participants
This prospective observational study included 28 patients diagnosed with TN out of which 11 (39%) were males and 17 (61%) were females. Age of the study participants ranged between 35 and 78 years with a mean age of 57 years. These patients met the following inclusion criteria: (a) age more than 30 years, (b) classic or idiopathic TN with cutaneous and oral triggers: TN1 defined by the presence of paroxysmal pain alone and TN2 defined by paroxysmal pain along with constant pain in the background for more than 50% of the time, (c) medically refractory primary TN not tolerating prescribed pain medications due to side effects namely drowsiness, double vision, nausea, dizziness, (d) treatment with (CyberKnife radiosurgery [CKRS]) and no previous surgical procedures for TN, (e) availability of magnetic resonance imaging (MRI) and DTI scans at 4 months post-treatment and (f) minimum of 9 months of clinical follow-up Patients with the following conditions were excluded from the study: (a) trigeminal autonomic cephalgias: cluster headache, paroxysmal hemicranial headache, (b) short-lasting unilateral neuralgiform headache attacks with conjunctival injection and tearing (SUNCT syndrome), (c) short-lasting unilateral neuralgiform headache attacks with cranial autonomic symptoms (SUNA), (d) glossopharyngeal neuralgia: patients who presented with pain in tongue, mouth and throat and the pain was triggered by chewing, talking and swallowing, (e) pain relapse after microvascular decompression and one session of radiosurgery, (f) post-herpetic neuralgia and (g) temporo-mandibular joint pain syndromes.
Clinical Outcome Measures
Clinical outcomes were assessed prospectively in patients with a minimum 9-month follow-up post-CKRS as per the study design [Figure 1]. At baseline pre-CKRS, fourth, sixth and ninth months post-CKRS, clinical variables and medication history were documented. Pain intensity was measured using the Barrow Neurological Institute Pain Intensity Score (BNI-PIS) scale: Class I: No pain, no medication, Class II: occasional pain, no medication, Class III: some pain, managed with medication, Class IV: some pain, not well controlled by medication and Class V: severe pain, no relief. Responders achieved BNI-PIS Classes I-III within 4 months post-treatment, indicating excellent pain response for TN. Non-responders experienced no response or recurrence of severe pain (BNI-PIS Classes IV and V) within 4 months.
Study design
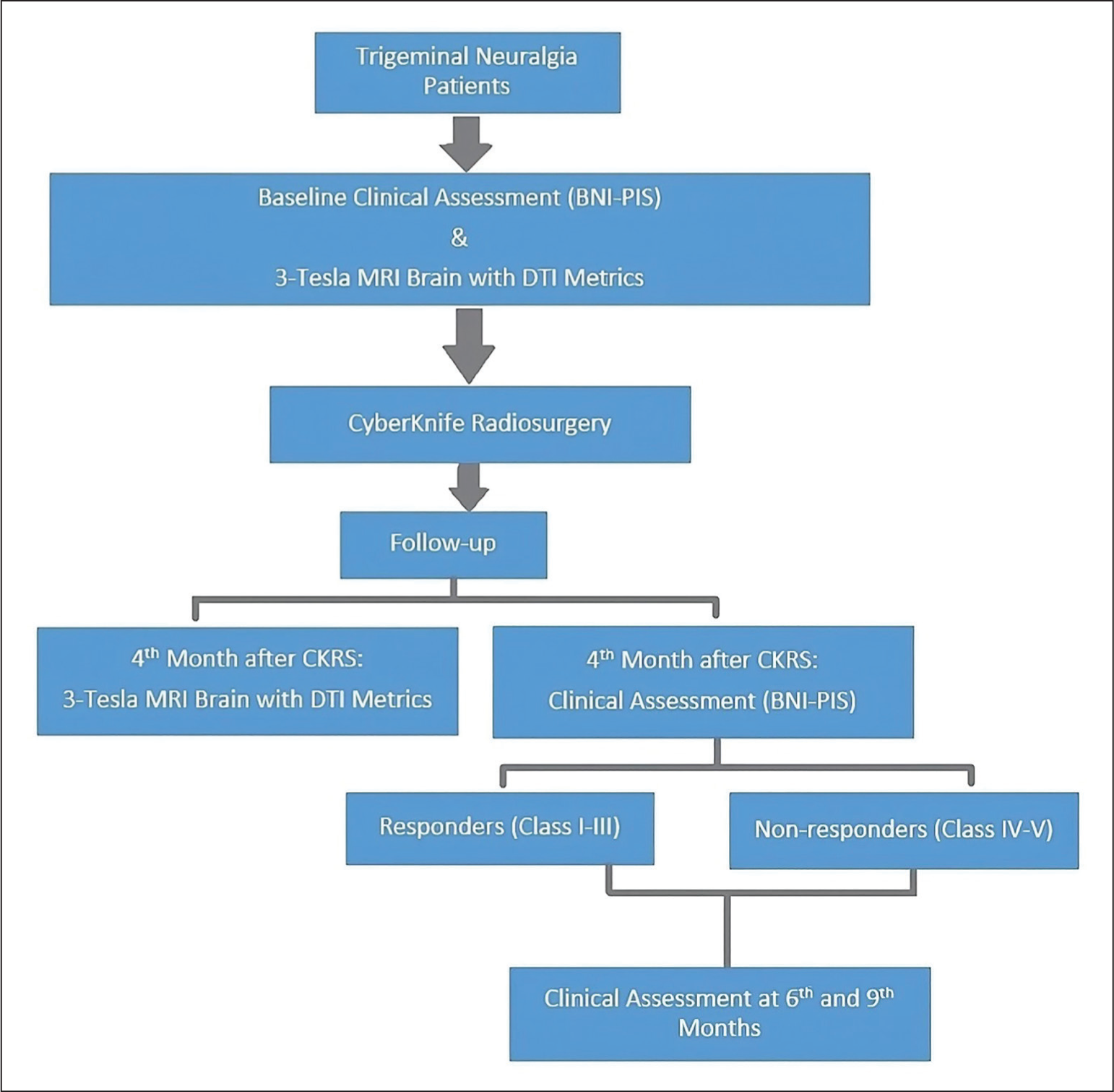
CyberKnife Radiosurgery
All patients underwent treatment with CyberKnife-G4 Robotic Radiosurgery System. They were immobilised in a supine position with a frameless double-reinforced thermoplastic cast. Imaging included plain computed tomography (CT) scans and Three-Tesla MRI sequences. Target volume of trigeminal nerve dorsal root entry zone (DREZ) within the pre-pontine cisternal space was contoured in the MultiPlan Treatment Planning System. Mean maximum voxel dose (Dmax) delivered to the affected DREZ of trigeminal nerve was 80.44 Gy (range: 68.96-84.70 Gy) respecting dose constraints for critical structures in proximity, especially the brainstem. Prescription protocols were adjusted individually based on the pre-pontine cisternal space, where a wider space would provide freedom to escalate doses (not beyond 85 Gy). Mean Dmax to the Brainstem was 24.06 Gy, mean Dmax to the ipsilateral temporal lobe was 25.1 Gy and mean Dmax to the ipsilateral auditory apparatus was 2.47 Gy as illustrated (Table 1). Six mega-voltage (6 MV) unflattened photon beam energy was used, with ray-tracing and Monte Carlo dose calculation algorithms. Multiple stereotactic non-coplanar beams were used for treatment, shaping the beams with a 5-mm fixed collimator. Online imaging guidance with 6D skull tracking ensured minimal motion errors during treatment.
CyberKnife Radiosurgery Dosimetry
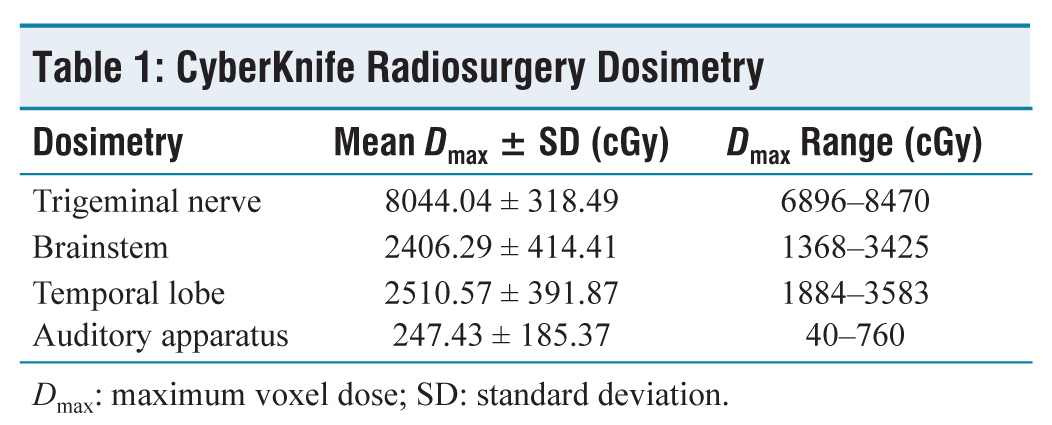
Dmax: maximum voxel dose; SD: standard deviation.
MRI Acquisition and Processing
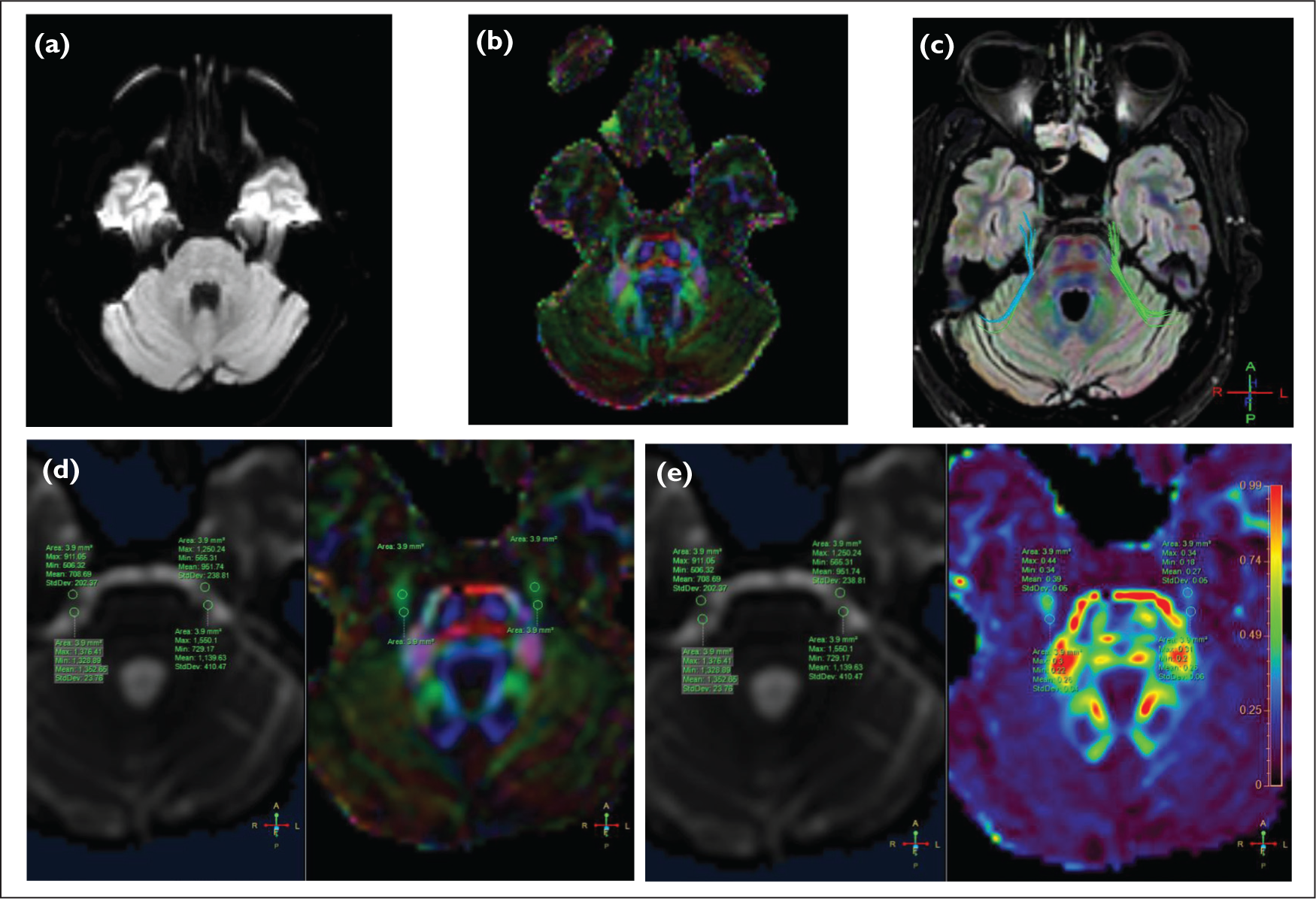
MRI with acquisition of DTI metrics was conducted at baseline prior to CKRS and again at fourth month after CKRS. As per clinical imaging protocol, each patient underwent a Three-Tesla Philips Ingenia Elition-X MRI brain scans at baseline prior to CKRS and again at fourth month after CKRS. DTI metrics were estimated using in-built DTI FiberTrakTM from Philips Healthcare, which produced scalar maps including fractional anisotropy (FA), radial diffusivity (RD), axial diffusivity (AD) and mean diffusivity (MD) [Figure 2]. These DTI metrics were measured with FA and RA as scalar values ranging between 0 and 1. AD, MD and RD measured in unit mm2/sec Moreover, DTI FiberTrakTM aided in the visualisation of MR images and the superimposition/linear registration of T1-weighted FSPGR anatomical MR images into the diffusion space for each patient. The accuracy of radiosurgical shots was confirmed by observing T1-Gado enhancement over the affected side DREZ in the post-CKRS MRI scan.
(A) Raw unprocessed DTI after acquisition. (B) Processed DTI image using DTI FiberTrakTM showing the principle diffusion map. (C) Processed DTI image showing fibre tractography of bilateral trigeminal nerves. (D) Extraction of fractional anisotropy (FA) at the DREZ of bilateral trigeminal nerves. (E) Extraction of relative anisotropy (RA) at the DREZ of bilateral trigeminal nerves
Statistical Analysis
Descriptive statistics were presented with mean ± standard deviation (SD) and frequency (percentage) for the continuous and categorical factors, respectively. Median (interquartile range [IQR]) was presented for the continuous factors while it skewed. Normality of the data was checked by using the Shapiro–Wilk test. Paired t test/Wilcoxon signed rank test was used to find out the significant changes between pre- and post-treatment values. Friedman test was used to find out significant changes in the BNI-PIS score over follow-up clinical visits. A value of P <.05 was considered statistically significant. All analysis was carried out by using statistical software SPSS (IBM, 28.0). Tests statistical significance will be decided as follows: (a) P >.05 is not significant, (b) P < .05 is significant and (c) P < .01 is highly significant
Results
Patient Demographic and Disease Characteristics
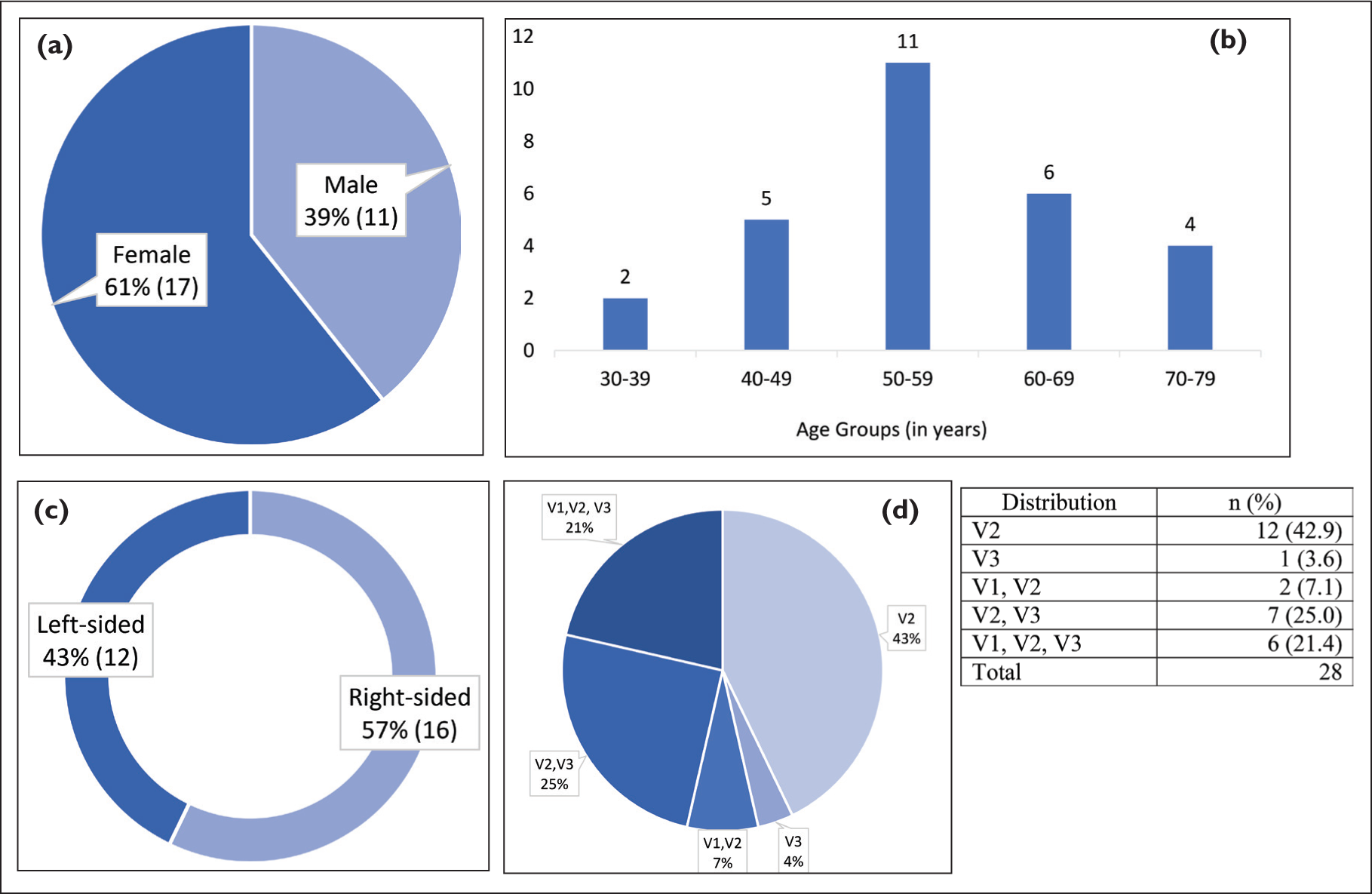
Of the 28 participants with patient demographics and disease characteristics summarised in [Figure 3]. We identified 24 patients who reported good pain relief (BNI-PIS Classes I-III) 4 months after CKRS and were labelled as responders. Fifteen females and nine males with a mean age of 58.45 ± 3.58 years SD were in the responder group. There were four non-responders reporting BNI-PIS Classes IV and V, 2 males and 2 females with a mean age of 48.25 ± 5.59 years SD.
(A) Gender distribution. (B) Age group distribution. (C) Laterality of affected side. (D) Pain distribution over the region of affected trigeminal nerve
Clinical Assessment
During the first follow-up of participants at the fourth month after CKRS, it was found that four people had reported no significant pain relief (BNI-PIS Class IV or V). While rest of the study participants reported pain relief to treatment with a statistically significant P < .001 [Table 2]. This response was recorded and sustained over subsequent follow-ups at the sixth month and ninth months after completion of CKRS with a significant P < .001 [Table 2]. Participants who were classified as responders also reported reduction in number of pain medication used by them for pain relief which was statistically significant (P < .001).
Responders and Non-responders at First Follow-up 4 Months Post-CKRS
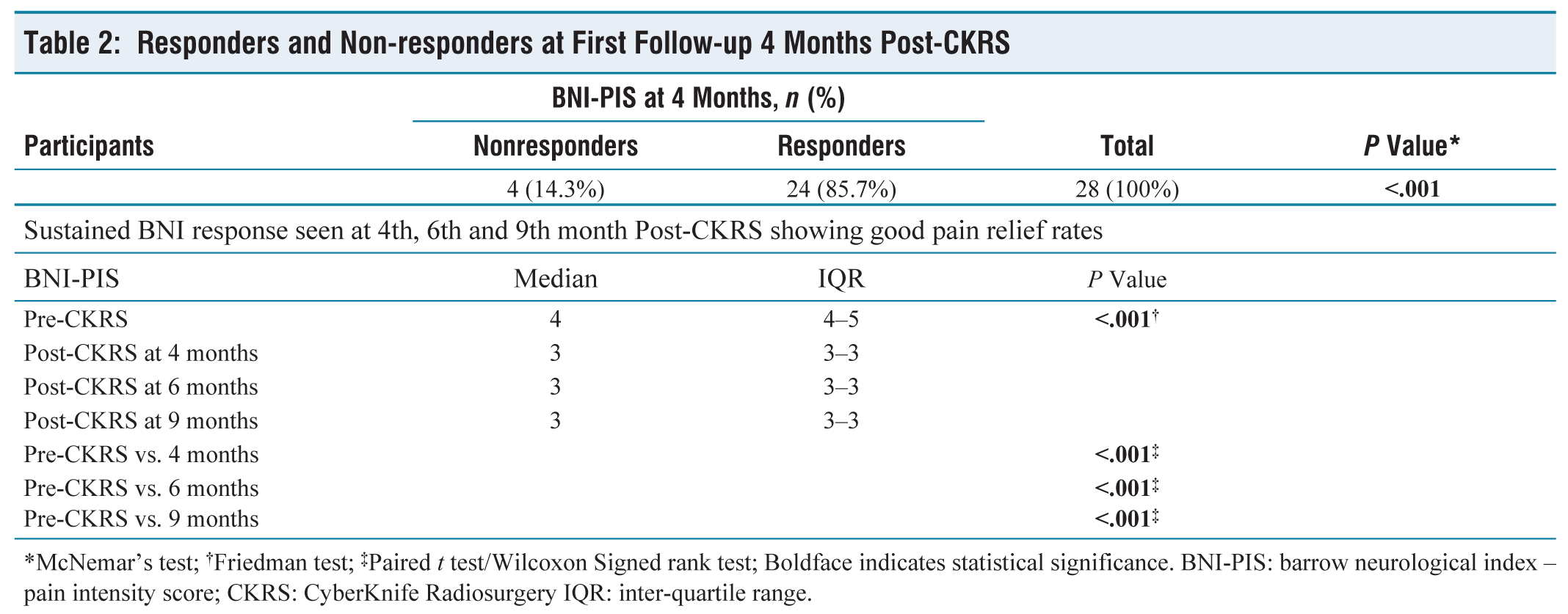
*McNemar’s test; †Friedman test; ‡Paired t test/Wilcoxon Signed rank test; Boldface indicates statistical significance. BNI-PIS: barrow neurological index – pain intensity score; CKRS: CyberKnife Radiosurgery IQR: inter-quartile range.
Analysis of Diffusivity Metrics
MRI scan done at the fourth month after CKRS showed that all 28 participants exhibited T1 Gado-enhancement at the treated site DREZ of trigeminal nerve. Hence it was not associated with clinical outcome, nor was it an independent prognostic factor.
Diffusivity Metrics Captured at 4 Months Post-CKRS – FA
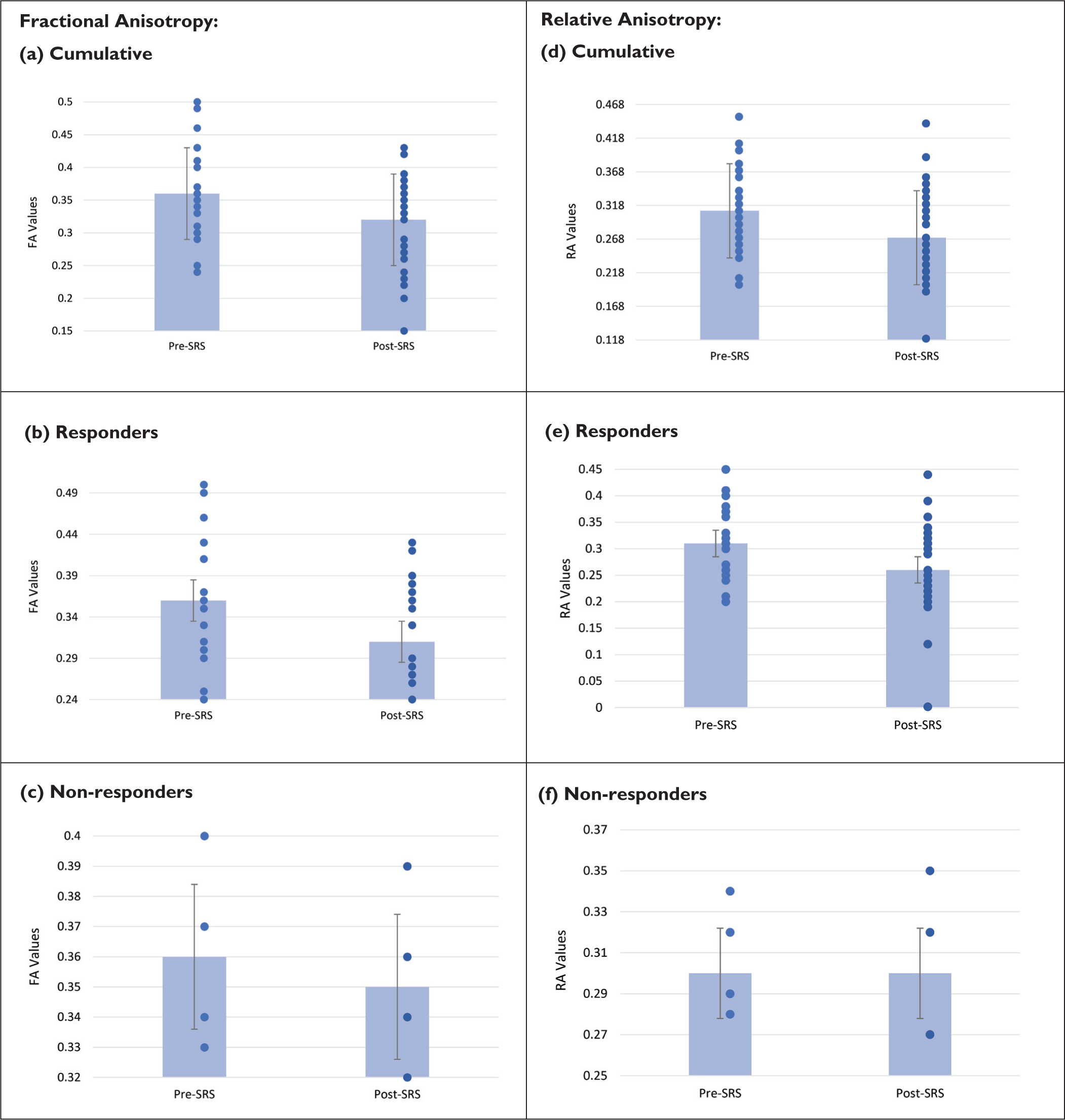
FA values obtained Pre-CKRS and Post-CKRS were analysed against pain responses reported by the study participants which was statistically significant (P < .001) by Paired t test/Wilcoxon Signed rank test [Table 3 and Figure 4A]. A mean drop of 11% in FA values was seen in the entire study cohort. In the responder group, a drop of 13.8% was noted in FA values [Table 3 and Figure 4B]. Moreover, a drop of only 2.7% in FA values was seen in the non-responder group [Table 3 and Figure 4C].
Cumulative Change in FA Values
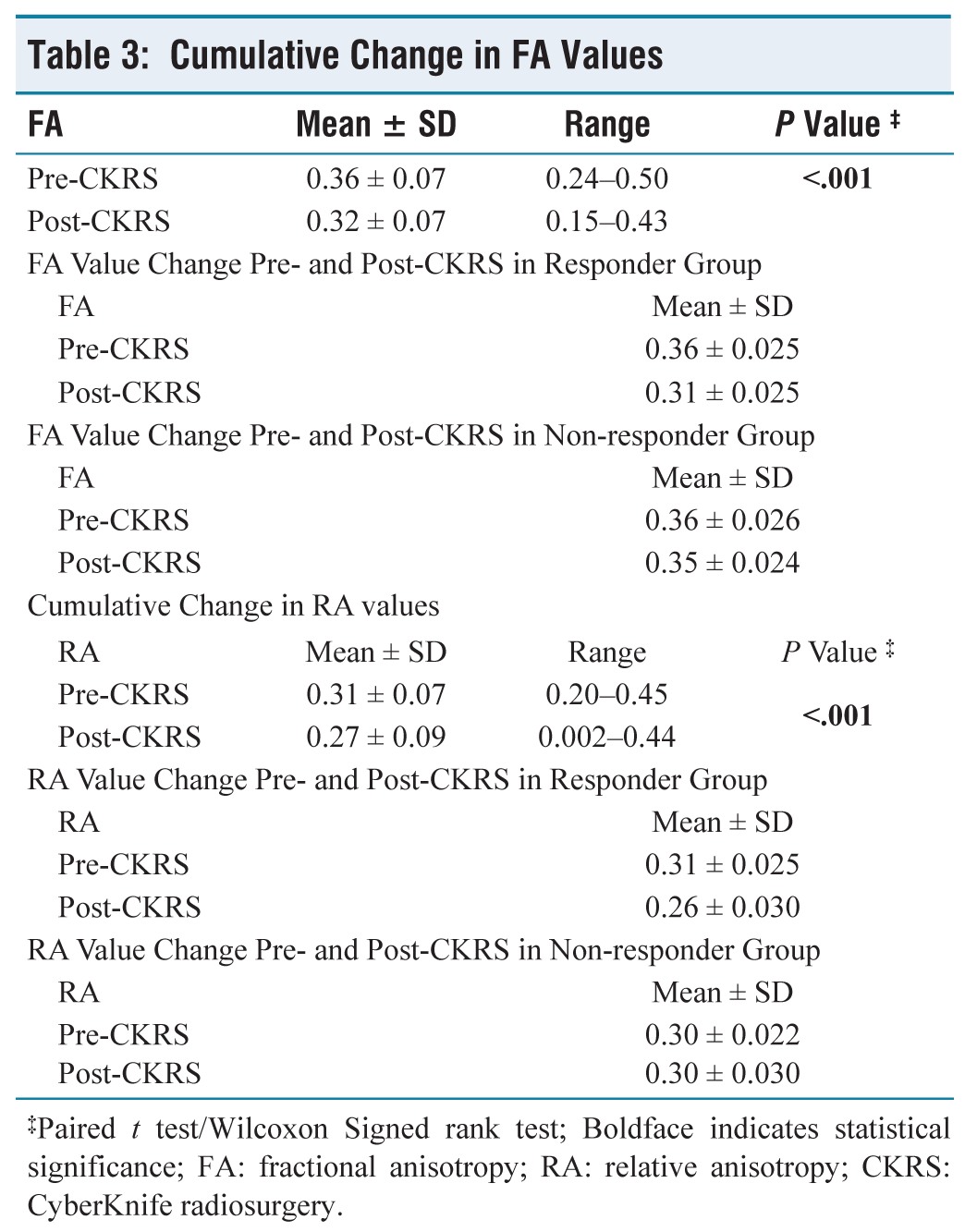
‡Paired t test/Wilcoxon Signed rank test; Boldface indicates statistical significance; FA: fractional anisotropy; RA: relative anisotropy; CKRS: CyberKnife radiosurgery.
Diffusivity Metrics Captured at 4 Months Post-CKRS – Relative Anisotropy (RA)
Relative anisotropy (RA) values obtained pre-CKRS and post-CKRS were analyzed against pain responses reported by the study participants which was statistically significant (P < .001) by paired t test/Wilcoxon signed rank test [Table 3 and Figure 4D]. A mean drop of 12.9% in RA values was seen in the entire study cohort. In the responder group, a drop of 16.1% was noted in the RA values [Table 3 and Figure 4E]. However, there was no drop in RA values noted in the non-responder group [Table 3 and Figure 4F].
Bar graph showing the average DTI-derived values ± standard error of mean (SEM) with overlaid individual patient data
Diffusivity Metrics Captured at 4 Months Post-CKRS – AD, Relative Diffusivity (RD) and MD
No significant changes were observed in the AD, RD and MD values noted pre- and post-CKRS [Table 4].
Cumulative Change in AD Values
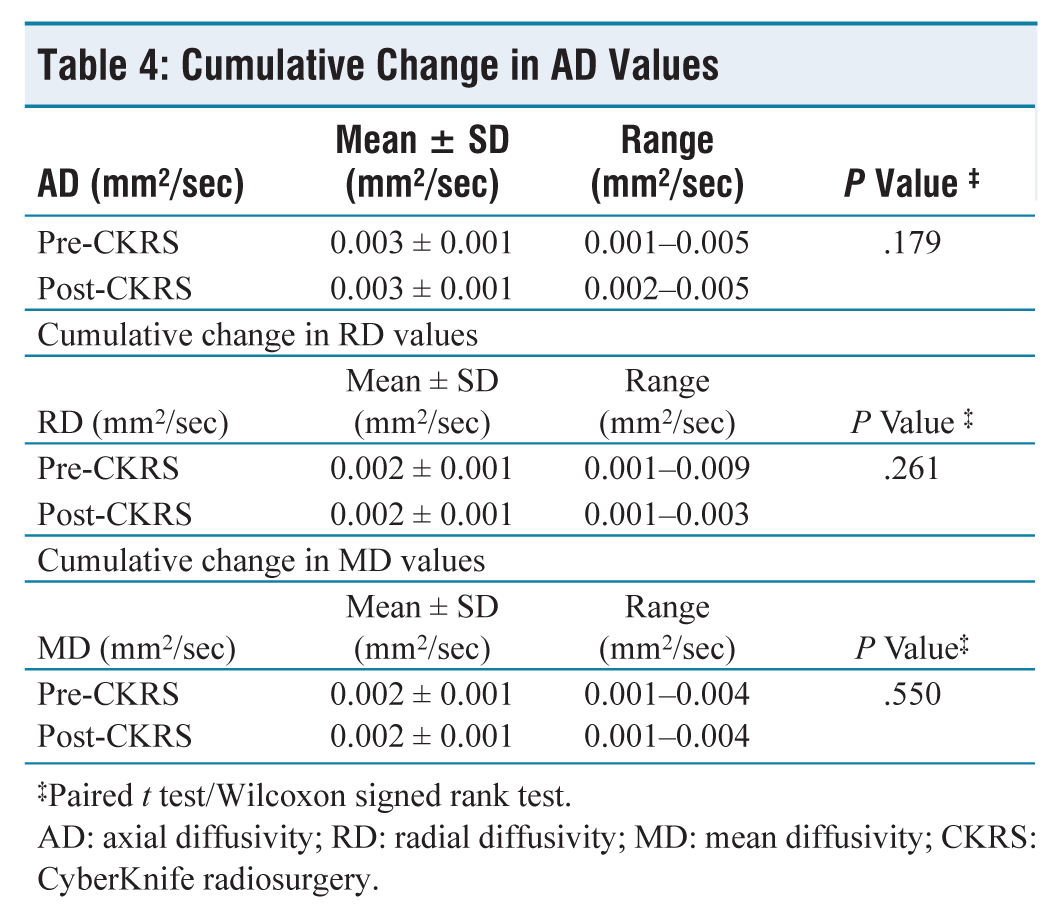
‡Paired t test/Wilcoxon signed rank test.
AD: axial diffusivity; RD: radial diffusivity; MD: mean diffusivity; CKRS: CyberKnife radiosurgery.
Discussion
In the responder group, there was a notable reduction in FA at the radiosurgical target within their affected nerve, when compared to non-responders. Non-responders did not exhibit any distinct FA changes. Our findings imply that, in order for patients to attain enduring pain relief, substantial microstructural alterations at the radiosurgical target should be evident during early follow-up assessments. There might be a potential threshold effect, wherein the quantification of microstructural changes allows us to predict, on an individual basis, whether long-lasting pain relief will be achieved. In this study cohort, responders demonstrated an average 13.8% reduction in FA, while non-responders experienced a 2.7% decrease. Similarly, there was an average drop of 16.1% in RA values for responders; were as there were no changes noted in the non-responders. FA alteration has been demonstrated as an indicator of various medical conditions, including TN. However, while FA provides insights into the broader microstructure of white matter, it does not delve into specific microstructural changes. Therefore, by examining metrics such as RD, AD and MD, we can gain a deeper understanding of alterations related to myelination, axonal integrity and potentially underlying factors such as neuroinflammation and oedema. These factors are integral to comprehending the overall microstructural organisation and changes that occur following radiation treatment. In the study conducted by Tohyama et al.[7] showed that the diffusivity of trigeminal nerve at the 6-month mark post-gamma knife radiosurgery (GKRS) served as a predictive factor for long-term clinical effectiveness. Among long-term responders (comprising 19 individuals), there was a noticeable decrease in FA at the radiosurgical target within their affected nerve. This contrasted significantly with both their unaffected nerve on the opposite side and the FA observed in non-responders. Additionally, long-term responders showed higher values of RD and MD, which are indicative of myelin alterations and inflammation, within their affected nerve compared to their unaffected counterpart. On the other hand, non-responders (consisting of 18 individuals) did not display any distinctive changes in diffusivity following GKRS. In a research study led by Hodaie et al.,[8] five patients diagnosed with TN were enrolled, consisting of four females and one male with an average age of 67 years. These individuals underwent GKRS with a dose of 80Gy administered at 100% isodose line. The study involved conducting Three-Tesla MRI trigeminal nerve tractography scans both before the treatment and at multiple time points, extending up to 14 months after the procedure. For analysis, researchers computed metrics including FA, RD and AD specifically within the region of interest, which was defined as the radiosurgical target area. Control regions encompassed areas outside this target region and the contralateral nerve. Study outcomes demonstrated that trigeminal tractography effectively and accurately pinpointed the radiosurgical target. Following radiosurgery, there was a noteworthy 47% decrease in FA values, particularly within the target area, signifying highly localised changes resulting from the treatment. It is worth noting that RD, indicative of myelin alterations, displayed substantial changes, whereas AD did not, suggesting that radiosurgery predominantly impacts myelin structures. Furthermore, sensitivity of tractography surpassed that of conventional post-treatment MRI scans enhanced with gadolinium, as it detected FA changes even in cases where there was no observable enhancement of the trigeminal nerve. In patients with extended follow-up, recovery of FA and RD values corresponded with the recurrence of pain, highlighting the potential of DTI and tractography as highly sensitive tools for evaluating treatment outcomes in cases of TN. In a study conducted by Haritha et al.,[9] 10 patients suffering from medically refractory TN underwent Linac radiosurgery, employing stereotactic cones and advanced imaging techniques. They received a prescribed dose of 85 Gy at the 100% isodose line. Before treatment and at intervals of 4, 8 and 12 months following treatment, three MRI scans of the trigeminal nerve tract were conducted. The study used multi-tensor tractography to extract metrics derived from DTI, including AD, RD, MD and FA from the cisternal segment of the trigeminal nerve. For comparative purposes, regions outside the treatment target and the contralateral nerve were used as controls. The results of the study demonstrated the precise identification of the radiosurgical target through trigeminal tractography. At the 3-month mark following radiosurgery, there was a notable and statistically significant 55% reduction in FA values within the target area, indicating highly localised changes as a result of the treatment. Additionally, RD, a marker associated with myelin alterations, exhibited significant changes, implying that radiosurgery primarily influences myelin structures. Significantly, tractography displayed heightened sensitivity compared to traditional gadolinium-enhanced post-treatment MRI scans, as it detected FA changes even in cases where there was no observable enhancement of the trigeminal nerve. During long-term patient follow-up, the recovery of FA and RD values demonstrated a correlation with the recurrence of pain, underscoring the potential of DTI and Tractography as valuable tools for evaluating treatment outcomes in individuals with TN. In another investigation conducted by Pikis et al.,[10] a group of 16 patients diagnosed with TN underwent GKRS. MRI was performed before the procedure and then repeated at the 3-month post-procedure milestone. Various metrics derived from DTI were extracted from different segments of the trigeminal nerves. Among the patients who experienced positive treatment outcomes during the last follow-up, it was observed that they had lower average values of FA at the pontine segment (P = .04) and increased average values of RD at the root entry zones (P = .032) of the treated trigeminal nerve, as compared to those who did not respond to the treatment. These observed changes at the 3-month mark appeared to be associated with pain relief, suggesting that DTI could potentially serve as a non-invasive prognostic tool for TN patients undergoing GKRS. However, further research is necessary to validate these findings. In a separate study conducted in India by Shankar et al.,[11] a group of 44 patients who met specific criteria, including a diagnosis of Classic TN (Type 1), and who had not previously undergone surgical procedures for TN, were subjected to SRS treatment. These patients underwent three Tesla MRI scans at the 6-month mark post-treatment and were followed up clinically for a minimum of 12 months. Individuals with secondary TN due to certain conditions were excluded from the study. Diffusivity metrics (including FA, MD, RD and AD) were extracted from both the affected and unaffected trigeminal nerves at the 6-month post-treatment point, and these metrics were then correlated with the long-term clinical outcomes. The findings suggested that at the 6-month post-SRS evaluation, diffusivity within the trigeminal nerve served as a predictive factor for long-term treatment effectiveness. Among the patient cohort, 36 out of 44 individuals who responded positively over the long term exhibited significantly lower FA within their affected nerves at the radiosurgical target, compared to their unaffected nerves and when compared to those who did not respond (8 out of 44 patients). Moreover, RD and MD, which are linked to myelin alterations and inflammation, were notably higher in the affected nerve of long-term responders in comparison to their unaffected nerve. However, non-responders did not demonstrate any discernible diffusivity changes following SRS.
Limitations
A major limitation of this study is the non-homogenous distribution within responder and non-responder groups with which a uniform analysis of DTI metrics could not be done. A longer follow-up period beyond 9 months will help us understand the sustained pain relief rates of TN patients undergoing SRS. Current framework of this study involves utilisation of Three Tesla-MRI combined with DTI, alongside the expertise required for performing necessary DTI analyses with Neuro-Radiologists. Currently, there is not a fully automated pipeline, as the processes of registration and placement of regions of interest (ROIs) demand manual intervention or third-party applications. When considering integration of DTI into clinical practice, several crucial aspects warrant attention. This includes co-registration of imaging data and meticulous assessment of accuracy, Additionally, it is essential to recognise the inherent anatomical variability and variations in size of trigeminal nerve across different patients. These factors carry significant importance in comprehensive evaluation and assessment of the target area. Despite DTI’s promise, challenges remain due to protocol heterogeneity and data acquisition differences. Standardising DTI parameters and conducting larger prospective studies with homogenous responder and non-responder groups with longer follow-ups will improve its reliability and applicability in assessing microstructural changes and predicting non-responders in patients with TN undergoing SRS.
Footnotes
Acknowledgements
RJR acknowledges the help of Dr. Adithyan Rajendran and the neuro-radiology technicians with acquisition and reading of MRI-DTI datasets, which has helped to generate this data. RJR acknowledges the support of our entire clinical and para-clinical staff at the Department of Radiation Oncology including the medical physics team and technicians who helped in the treatment of patients. RJR acknowledges the technical help in treatment planning, execution and analysis of data from the clinical lead of Dr. Shankar Vangipuram and Dr. Shamsudheen Cholayil. RJR acknowledges Dr. Veronica’s general support and assistance in proof reading with language inputs. Last but not the least, RJR acknowledges the motivation and unwavering passion of Dr. Muralidharan and his team of Mrs. Uma and Mrs. Shalini in the publication of this data.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Institutional Ethical Committee Approval Number
Ethical approval for this study was obtained from the Institute Ethics Committee (IEC Application Number: ASH - DNB-029/06-22).
Informed consent
Written informed consent was obtained for anonymized patient information to be published in this article.
Credit author statement
The manuscript has been read and approved by the author, the requirement of authorship has been met, and the author believe it presents honest work.
Data availability
The anonymized patient data that support the findings of this study are available from the corresponding author, Rabbi Jebus Raja upon reasonable request.
Use of artificial intelligence
The author discloses that there was neither the use of AI and AI-assisted technologies in the writing process nor the use of generative AI and AI-assisted tools in figures, images and artwork.
