Abstract
In the healthy brain, there are close correlations between task-related activation of the primary motor cortex (M1), the magnitude of interhemispheric inhibition, and microstructural properties of transcallosal fiber tracts. After subcortical stroke affecting the pyramidal tract (PT), an abnormal pattern of bilateral activity develops in M1. With this prospective longitudinal study, we aimed to determine whether a morphological correlate of poststroke disinhibition could be measured within 20 days and 6 months of PT stroke. Using diffusion tensor imaging with tractography, we delineated transcallosal motor fibers (CMF) in nine PT stroke patients, six patients with subcortical infarct not affecting the PT (NonPT) and six transient ischemic attack patients. We compared changes in CMF fractional anisotropy ratios (rFA) with rFA in a distinct bundle of callosal occipital fibers (COF). At the initial time point, there were no significant differences in rFA between groups and fiber bundles. At follow-up, PT-group rFACMF was significantly lower than PT-group rFACOF and NonPT-group rFACMF. PT-group rFACMF decreased over time and correlated with rFA of the PT (rFAPT) retrograde to the infarct at 6 months. Our data suggest a progressive degenerative transsynaptic effect of PT stroke on CMF, which could be a morphological correlate of transcallosal disinhibition.
Introduction
The corpus callosum (CC) is the major fiber bundle interconnecting the two hemispheres of the human brain. It consists of long commissural tracts that link corresponding cortical regions of opposite hemispheres (Gazzaniga, 2000). Fibers of the CC are topographically organized, mirroring the rostral-caudal distribution of functional regions within the neocortical gray matter (Huang et al, 2005). Regional distributions of callosal fiber density, diameter, and myelination reflect the speed and frequency of transcallosal communication of the interconnected cortical domains (Aboitiz et al, 1992). The neurons that have their cell bodies in the primary motor cortex (M1) extend large diameter highly myelinated axons, which constitute the posterior midbody/isthmus of the CC (Wahl and Ziemann, 2008). These axons bundle together to form a distinct callosal motor fiber (CMF) tract, interconnecting the two functionally homologous motor cortices located along the precentral gyrus of each cerebral hemisphere.
In the healthy adult brain, manual motor tasks result in increased activation of the M1 region contralateral to the moving hand. Activity in M1 inhibits the homologous region of the opposite hemisphere in a process known as interhemispheric inhibition (IHI; Perez and Cohen, 2009). This phenomenon can be explained by the inhibitory model of hemispheric metacontrol, which suggests that with regards to ‘simple’ behaviors such as hand movement, transferring, and integrating information transcallosally would require more time and energy than inhibiting the activity of one of the hemispheres (van der Knaap and van der Ham, 2011). The IHI is mediated by the glutaminergic fibers of the CMF, originating in M1 of each hemisphere and synapsing on local inhibitory interneurons within the homologous contralateral region (Perez and Cohen, 2008). Motor task-related functional increases in blood oxygenation level-dependent signal are significantly correlated with the degree of IHI (Sarfeld et al, 2011). Thus, the more active the motor cortex, the stronger the excitatory signal sent along the CMF and the stronger the contralateral inhibition.
The CMF can be studied noninvasively in the living human brain using diffusion tensor imaging (DTI) with tractography, which takes advantage of the preferential diffusion of water molecules along white-matter fiber tracts to permit the delineation of anatomical connections between cortical regions (Doron and Gazzaniga, 2008). Fractional anisotropy (FA) can be derived from diffusion parameters and provides a useful integrative measure of fiber tract morphology and microstructure as determined by axon diameter, myelination, membrane structure, axonal bundling, and axonal transport (Beaulieu, 2002). The integrity of the CMF is essential for proper functioning of the human motor system, especially with regards to the fine-tuning of motor skills (Wiesendanger and Serrien, 2004). Indeed, better performance on a bimanual coordination task has been associated with higher FA in the body of the CC at the level of the CMF (Johansen-Berg et al, 2007). Furthermore, in healthy subjects, FA of the CMF correlates positively with the degree of IHI (Wahl et al, 2007) and negatively with cortical activity in the nondominant hemisphere during the performance of lateralized tasks (Putnam et al, 2008).
Decreases in CC fiber FA can be seen in normal aging (Abe et al, 2002), but also in several pathologies directly affecting the CC, such as multiple sclerosis (Lenzi et al, 2007), mild cognitive impairment and Alzheimer's disease (Di Paola et al, 2010), as well as chronic cortical stroke (Gupta et al, 2006; Jason et al, 2011). In the case of cerebral pathologies, such as when an ischemic infarct directly affects a tract, relative FA (rFA; the ratio between FA of a tract in the affected hemisphere over the FA of the homologous tract in the unaffected hemisphere) can serve as a measure of stroke-affected fiber integrity (Radlinska et al, 2010). Given the high degree of interindividual variation in FA within clinical samples (especially in the case of cerebrovascular disease) and intraindividual variability across fiber tracts, rFA provides a useful self-normalized measure of morphological fiber integrity in the pathological brain.
In large cortical lesions, there is also a well-documented loss of IHI resulting in the release of contralateral activation (Perez and Cohen, 2009; Takeuchi et al, 2010b; van der Knaap and van der Ham, 2011), caused by the direct destruction of motor cortex neurons and possibly their connecting transcallosal fibers. Similar effects have also recently been described after subcortical stroke (Duque et al, 2005; Shimizu et al, 2002). Notably, when an infarct affects motor fibers of the pyramidal tract (PT) at the level of the internal capsule (thus not affecting callosal fibers), an abnormal pattern of bilateral blood oxygenation level-dependent activation develops in response to simple motor tasks such as finger tapping (Carey et al, 2002; Nair et al, 2007), correlating with the magnitude of motor deficit (Murase et al, 2004).
This longitudinal prospective study thus aims to investigate the indirect effects of subcortical stroke affecting the PT on the integrity of the CMF. Specifically, we address the question of whether a decrease in the rFA of the CMF can be measured within 20 days or 6 months of subcortical stroke along the PT. Furthermore, we aim to determine whether any changes in the morphological integrity of the CMF are associated with poststroke outcome. This question is of special clinical interest because the degree of poststroke bilateral activity has been shown to correlate with the extent of persistent poststroke impairment (Murase et al, 2004) and poor motor recovery (Ward et al, 2003). Thus, we aim to elucidate an important morphological correlate of poststroke functional change.
We will test the primary hypothesis that subcortical ischemic stroke affecting the PT results in a significant decrease in rFA of the CMF. If the decrease in rFA represents progressive secondary degeneration, then we expect it to deteriorate over time. We also expect the changes in CMF morphological integrity to be significantly related to PT damage and upper limb motor function in the chronic poststroke periods.
Materials and methods
Nineteen right-handed patients (14 female, 5 male; mean age 71.4±12.8 years) with first-ever subcortical ischemic infarct either affecting the PT (PT group, n=12) or not (NonPT group, n=7) were recruited from the specialized stroke unit at the Jewish General Hospital in Montreal (QC), Canada. Sample size was estimated based on recently published data on between subject variability and between session reproducibility of tractography measures along the Genu of the CC in healthy controls (Heiervang et al, 2006). Following these data, a minimum sample size of six subjects per group is required to detect reductions in mean tract FA of 2% to 20% between subjects with a power of 0.8 at a P=0.05. Stroke patients were screened for inclusion and exclusion criteria on admittance to the stroke unit. All patients were right handed, having suffered a first-ever subcortical stroke affecting white matter only. Exclusion criteria included prior stroke or history of other neurologic disease, alcohol or drug abuse and contraindications for magnetic resonance imaging (MRI). The PT involvement was determined from initial and follow-up T1- and T2-weighted images by two independent raters (AT and JM), both experienced stroke Neurologists blinded to patient presentation. There was 100% agreement on all patient classification by the raters. All patients were assessed and scanned within 20 days of stroke and again at 6 months after stroke.
The transient ischemic attack (TIA) group consisted of seven patients (two female, five male; mean age 65.1±6.2 years) with TIA, matched to the patient groups with regards to age and cardiovascular risk factors. These subjects were only scanned once at the initial time point, no follow-up scans were acquired because no structural lesions were present on the initial MRI (Table 1).
Demographics and infarct information
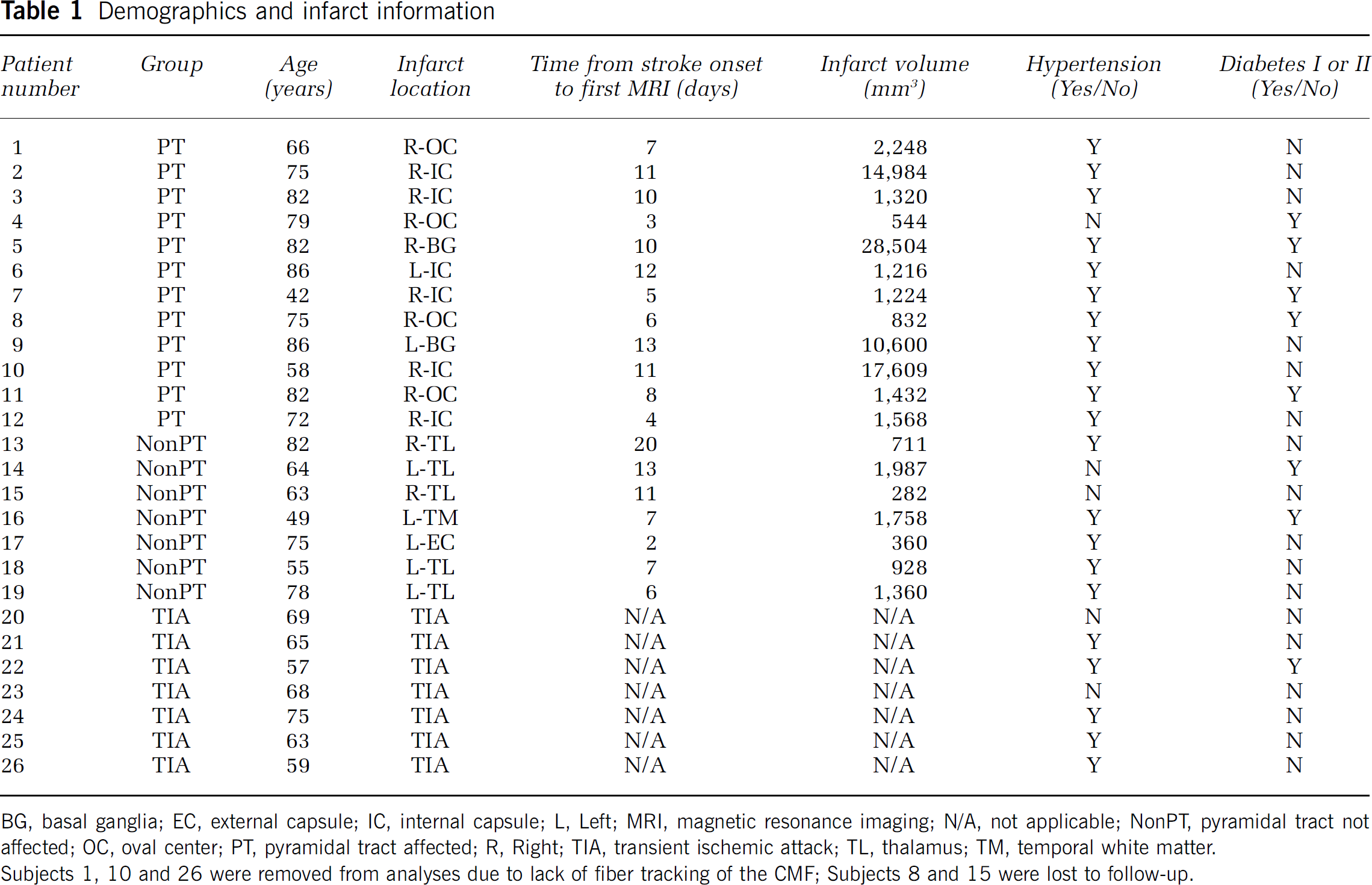
BG, basal ganglia; EC, external capsule; IC, internal capsule; L, Left; MRI, magnetic resonance imaging; N/A, not applicable; NonPT, pyramidal tract not affected; OC, oval center; PT, pyramidal tract affected; R, Right; TIA, transient ischemic attack; TL, thalamus; TM, temporal white matter.
Subjects 1, 10 and 26 were removed from analyses due to lack of fiber tracking of the CMF;
Subjects 8 and 15 were lost to follow-up.
This study was approved by the McGill Faculty of Medicine Institutional Review Board (protocol number: A00-M142-06B). All subjects gave informed consent before participation in accordance with regulations for human subjects’ studies.
Clinical Measures of Stroke Severity
All patients were assessed for clinical stroke severity, in addition to physical and neurologic examination, in the acute and chronic poststroke phases. Stroke-related neurologic disability was evaluated using the NIHSS (National Institute of Health Stroke Scale) and performance in the activities of daily living using the BI (Barthel index). The degree of handicap was assessed by the modified Rankin Scale and upper limb motor function was assessed with the arm subsection of the Rivermead Motor Function Test (RMFT) (Table 2).
Clinical measures across groups and time points
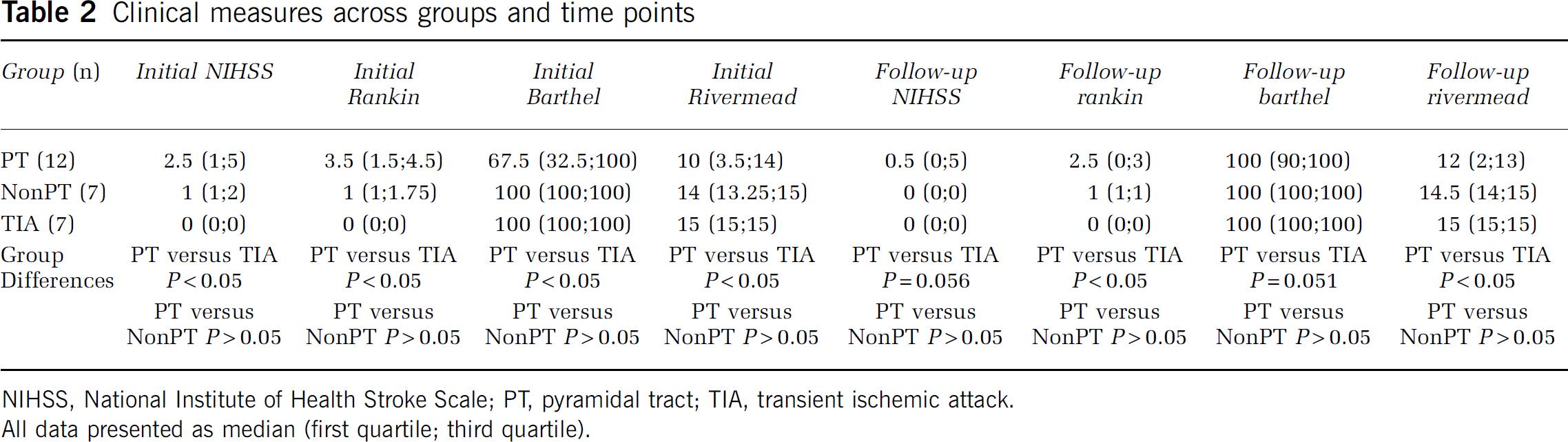
NIHSS, National Institute of Health Stroke Scale; PT, pyramidal tract; TIA, transient ischemic attack.
All data presented as median (first quartile; third quartile).
Magnetic Resonance Imaging
Magnetic Resonance Imaging was performed on a 3-Tesla Siemens Trio Scanner (Siemens Healthcare, Erlangen, Germany), with a 12-channel head coil, at the McConnell Brain Imaging Centre of the Montreal Neurological Institute. A magnetization prepared rapid acquisition gradient echo sequence was used to acquire a 1-mm isotropic high-resolution T1-weighted anatomical reference image (repetition time=2,300 ms, echo time=2.98 ms, TI=900 ms).
Diffusion Tensor Imaging
Diffusion encoding was achieved using a single-shot spin-echo echo planar sequence with twice-refocused balanced diffusion encoding gradients (Reese et al, 2003). Two data sets designed for high angular resolution reconstruction and signal-to-noise ratio were acquired and concatenated, with 50, and later 64, diffusion encoding directions, 2 mm isotropic voxel size, 63 slices, b=1,000 s/mm2, echo time=121 ms, repetition time=11.1 seconds, and Generalized Autocalibrating Partially Parallel Acquisition (GRAPPA) parallel reconstruction. The change in diffusion direction acquisition was an inevitable consequence of midstudy scanner upgrade. Within-subject acquisition was consistent from initial to follow-up time points for all subjects, and differences in diffusion direction acquisitions were evenly distributed between the groups.
Diffusion Data Set Alignment
To carry out within-subject comparisons of diffusion parameters over time, initial and follow-up data sets had to be resampled into the same image space. In our sample, common age-related changes in brain structure such as expansion of ventricles and deepening of sulci, as well as changes in MRI signal caused directly by the lesion, limit the possibility of reliable transformation into Talaraich space. Furthermore, resampling of fiber-tracking output can result in significant partial volume effects, given that the tracts of interest are small and close to the ventricles, where there is a substantial change in signal from one voxel to the next. As such, we created a third, ‘midway’ target T1-weighted image (T1-3), to which both DTI data sets were resampled. The T1-3 was created by aligning the follow-up anatomical image (T1-2) to the initial anatomical image (T1-1) using a 6-parameter linear transformation, then halving the transformation parameters along each axis and applying this new transformation matrix to the original T1-2 image. The raw diffusion data sets from both time points were then aligned to the T1-3 image, and the diffusion directions were adjusted accordingly. Consequently, all clinical diffusion data were altered to the same degree during this initial alignment step. Tensor calculation and fiber tracking at each time point then took place in T1-3 space, and no further transformation or resampling steps were necessary. Figure 1 (steps A to D) illustrates these analysis steps. In TIA patients, where no follow-up MRI was obtained, diffusion data sets were aligned to the initial T1 image.
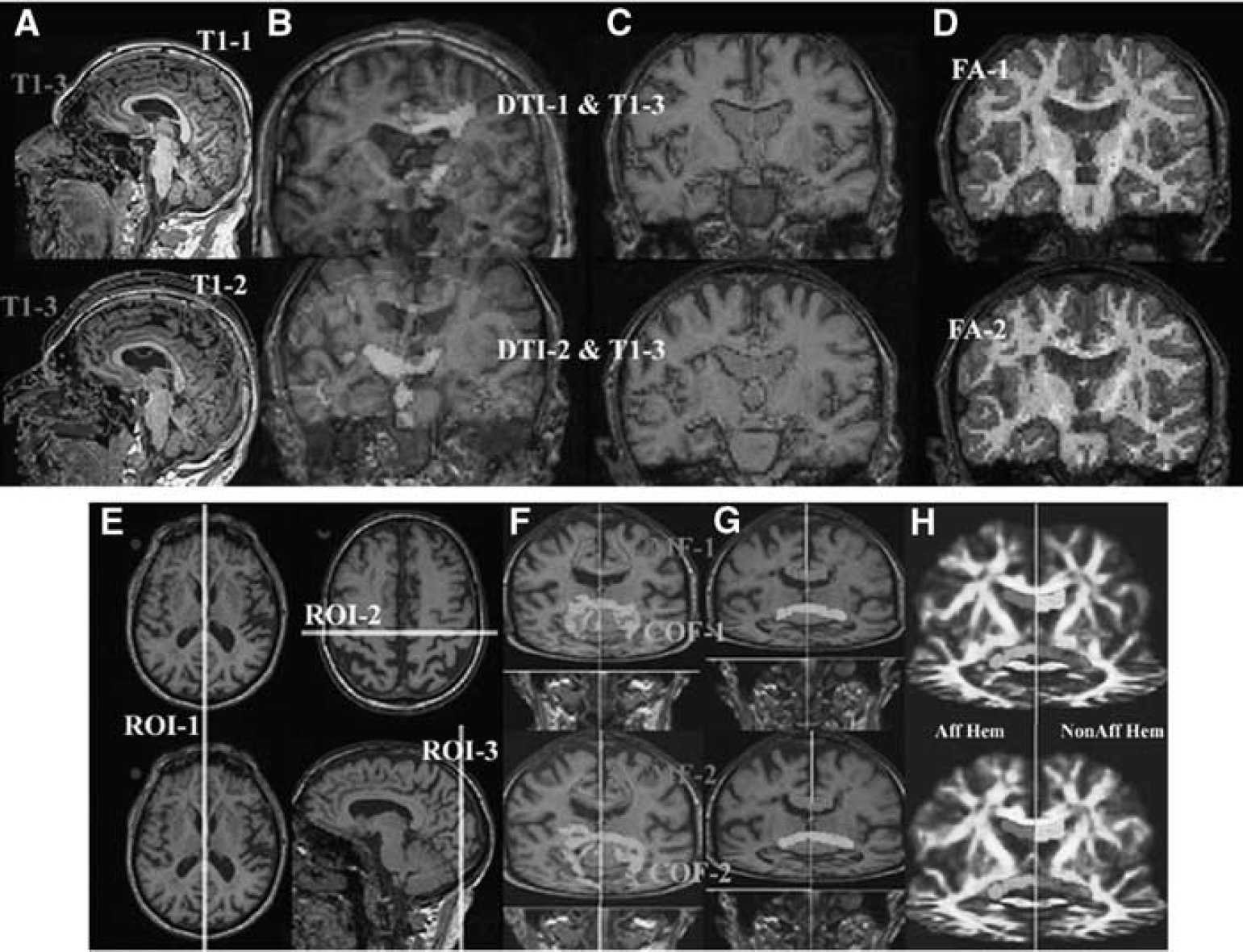
Fiber tracking of the callosal motor fibers (CMF) and callosal occipital fibers (COF). (
Fiber Tracking of Callosal Motor Fibers
A first region of interest (ROI) was placed along the midsagittal plane of the T1-3 image, extending 5 mm along the left-right axis (ROI-1). A second ROI was placed in the T1-3 image centered over the anatomically defined motor cortices (i.e., covering the hand bulbs of both hemispheres), extending 5 mm in the rostral-caudal axis and limited ventrally so as to include only the body of the CC, not the Genu or the splenium (ROI-2) (see Figure 1, step E). Box-shaped ROIs were used to standardize the size of seed regions for fiber tracking, while still taking into consideration anatomical landmarks in all subjects. Fiber tracking was performed using the deterministic FACT algorithm (Mori et al, 1999) and was initiated on a 3 × 3 × 3 grid of start points within each voxel of the ROIs to facilitate branching. Only fibers that passed through both tract-delineating ROIs were retained. The tracking was stopped if the FA was < 0.2 or the curvature from one voxel to the next was >40 degrees. Tracts that branched into the descending fibers of the PT (which were directly affected by the stroke) were excluded from further analysis. Remaining fibers were further truncated at the level of the lateral ventricles, so as to exclude any other fibers of the PT retrograde to the stroke, which were previously shown to have reduced rFA (Radlinska et al, 2010). Fiber tracking was performed in the same way for the initial and follow-up time points. The FA ratio of the CMF (rFACMF) was obtained by dividing the mean FA of CMF in the ipsilesional hemisphere by CMF in the contralesional hemisphere, as separated by the midsagittal plane of the T1-3 anatomical image. These ratios were then related to clinical measures of poststroke recovery. Figure 1 (F and G, in red) illustrates tracing of the CMF.
Fiber Tracking of Callosal Occipital Fibers
A first ROI was placed along the midsagittal plane of the T1-3 image, extending 5 mm along the left-right axis (ROI-1). A second ROI was placed in the T1-3 image over the visual cortex of both hemispheres, extending 5 mm in the rostral-caudal axis and centered around the calcarine fissure in the midsagittal plane at the level of the occipital cortex (ROI-3) (Figure 1, step E). Fiber tracking was performed as described above. Tracts that branched into the fibers of the ventral stream, extending rostrally toward the temporal lobes, were excluded from further analysis. All fibers caudal to the occipital horn of the lateral ventricles were also truncated to minimize the variability inherent in fanning and branching fibers closer to the cortex and to remain consistent with the method used to trace CMF. Fiber tracking was performed in the same way for the initial and follow-up time points. The FA ratio of the COF (rFACOF) was obtained by dividing the mean FA of callosal occipital fibers (COF) in the ipsilesional hemisphere by COF in the contralesional hemisphere, as separated by the midsagittal plane of the T1-3 anatomical image. Figure 1 illustrates the tracing of the COF (steps F and G, in green).
Fiber Tracking of the Pyramidal Tract
The PT delineation was based on a previously published method (Radlinska et al, 2009; Thiel et al, 2010), using the infarct region itself and cerebral peduncles as ROIs for fiber tracking. The FA ratio of the PT (rFAPT) was calculated as a ratio of the affected and nonaffected hemisphere mean FA in the PT and NonPT groups, and as left/right hemisphere FA ratios in the TIA group. In the PT group, tracts were subdivided into anterograde (below stroke) and retrograde (above stroke) portions relative to the lesion site.
Statistical Analyses
At both the initial and follow-up time points, changes in rFACMF and rFACOF were investigated using two-way RM-ANOVA (repeated measures analysis of variance) with factors group (PT, NonPT, and TIA at initial; PT and NonPT at follow-up) and tract (CMF and COF). Pairwise multiple comparisons were performed using the Student–Newman–Keuls Method.
Within the PT group, decreases in rFACMF and rFACOF over time were investigated using one-tailed t-tests. Linear regression analyses were used to model the relationship between rFAPT of the entire PT and RMFT, as well as rFAPT above and below the stroke and rFACMF.
Differences between groups with regards to clinical measures were investigated using one-way ANOVA for each clinical score at both the initial and follow-up time points. Linear regression analyses were used to investigate the relationship between rFACMF and clinical scores. Significance level for all tests was set at P<0.05.
Results
Callosal Motor Fibers and Callosal Occipital Fibers
Callosal motor fibers were successfully traced in all but three patients (two PT group and one TIA group), due to nonspecific damage/severe thinning at the level of the body of the CC in those patients. One PT and one NonPT group patient were lost to follow-up (Table 1). Final analyses were based on nine PT, six NonPT, and six TIA group patients. Callosal occipital fibers were successfully traced in all subjects. The location of CMF and COF in all nine PT-group patients is shown in Figure 2.
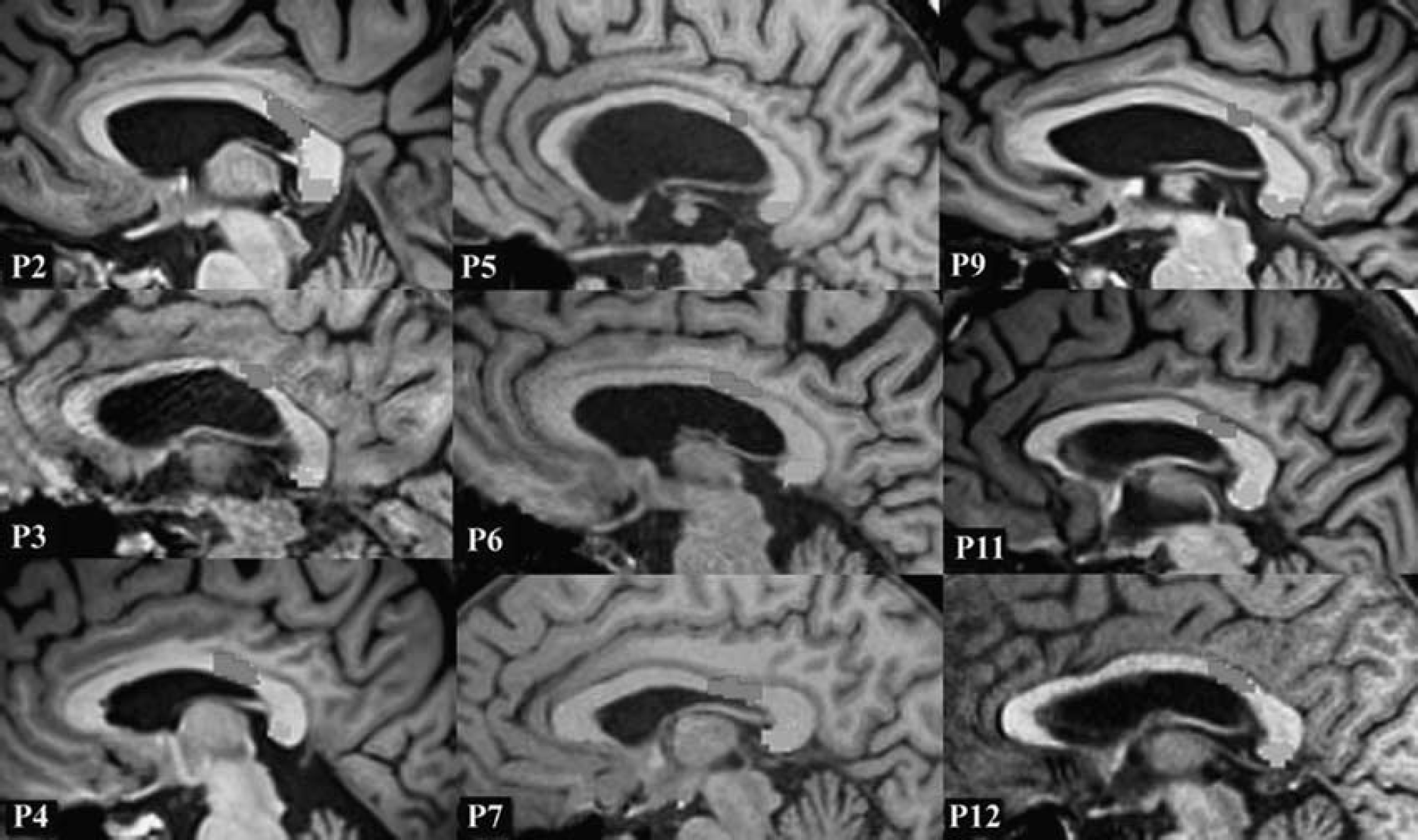
Midsagittal view of the callosal motor fibers (CMF) (red) and callosal occipital fibers (COF) (green) passing through the corpus callosum (CC) in all nine pyramidal tract (PT) group patients. The color reproduction of this figure is available at the Journal of Cerebral Blood Flow and Metabolism journal online.
There were no significant differences between the PT, NonPT, and TIA groups with regards to the rFACMF or rFACOF at the initial time point (two-way RM-ANOVA, P>0.05). An effect of group approached significance (P=0.085) (Figure 3A).
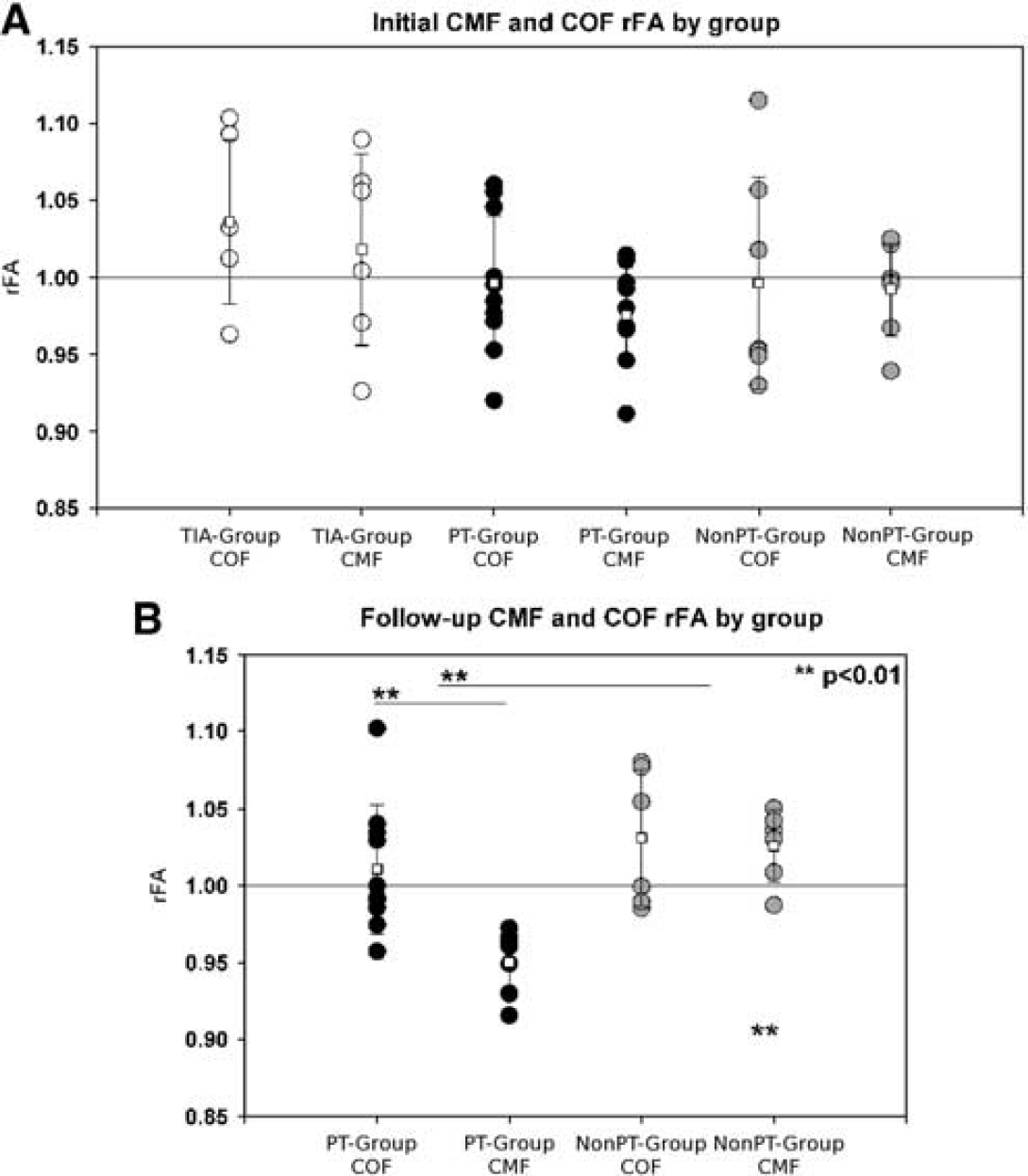
Callosal motor fiber (CMF) fractional anisotropy ratio (rFACMF) and callosal occipital fiber (COF) fractional anisotropy ratio (rFACOF) between groups within 20 days and 6 months after stroke. (
At the follow-up time point, two-way RM-ANOVA with factors group (PT and NonPT) and tract (CMF and COF) revealed significant overall effects of group (P=0.004) and tract (P=0.013), as well as a group × tract interaction (P=0.029). Subsequent pairwise comparisons (Student–Newman–Keuls) revealed decreased rFACMF as compared with rFACOF in the PT group (P=0.001), but not in the NonPT group (P=0.786). Furthermore, differences between groups in mean rFA were significant only within tract CMF (P<0.001), but not within COF (P=0.373) (Figure 3B).
Within the PT group, a paired t-test also confirmed a significant decrease in rFACMF over time (P=0.036, one tailed) in the absence of any change in the rFACOF over time (P>0.05).
No significant changes were found in mean diffusivity values or mean diffusivity ratios (see Supplementary Data).
Pyramidal Tract Fibers
The PT fibers were successfully delineated in both hemispheres of all patients and significant decreases in the rFAPT in PT-group patients as compared with NonPT and TIA groups were found, as reported previously (Radlinska et al, 2010). Initial rFAPT was highly predictive of RMFT score both initially (Rsqr=0.781, P<0.01) and at follow-up (Rsqr 0.757, P<0.01). There were no significant relationships between rFAPT of the entire PT and rFACMF at either time point (P>0.05). Investigation of tract portions above and below the stroke revealed no significant relationships between PT group rFAPT below at either time point (P>0.05) or rFAPT above at the initial time point (Figure 4A). There was, however, a significant relationship between PT group rFAPT above and rFACMF at the follow-up time point (Rsqr=0.533; P=0.04) (Figure 4B).
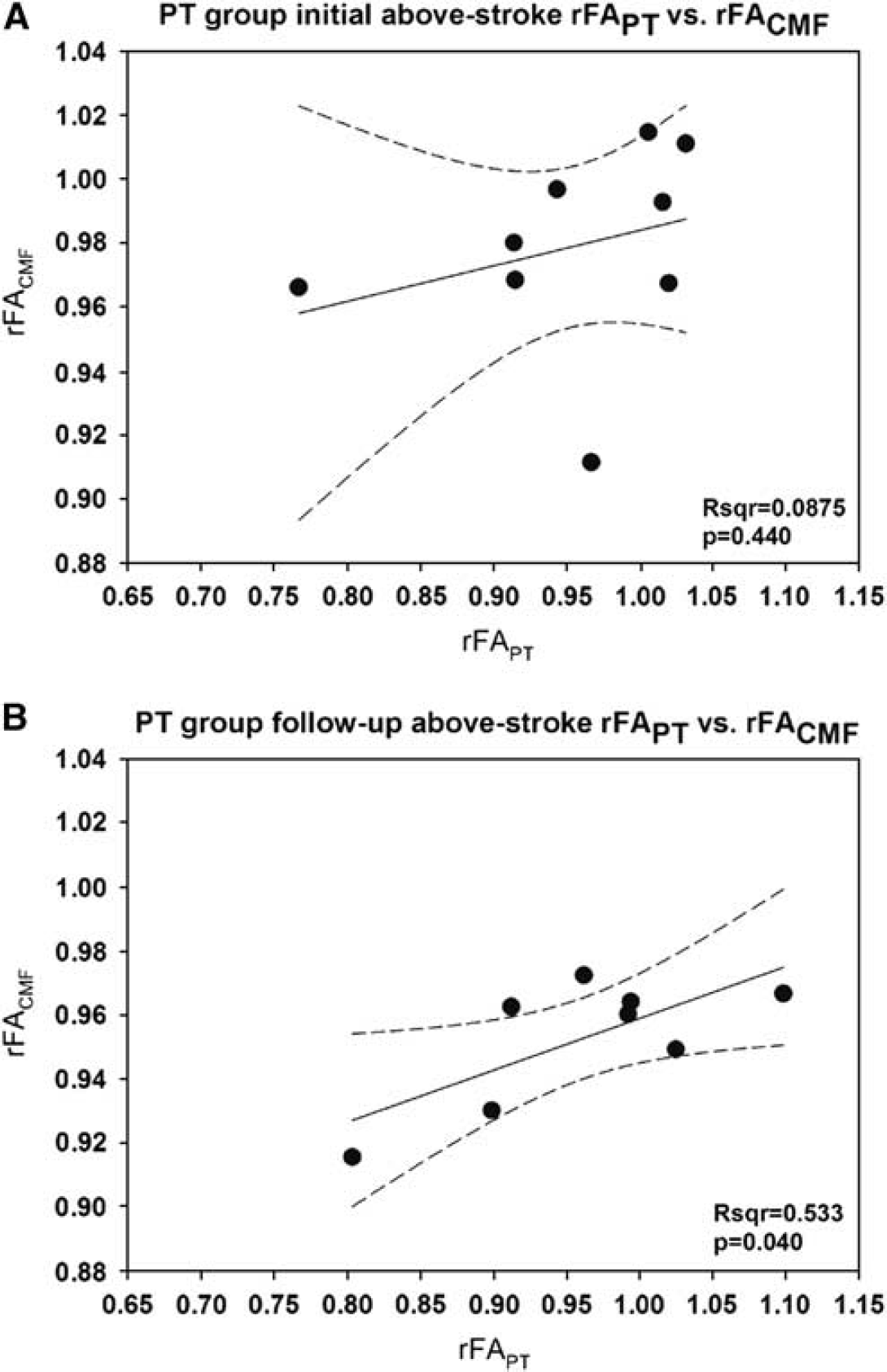
Relationship between pyramidal tract (PT) fractional anisotropy ratio (rFAPT) retrograde to the infarct and callosal motor fiber (CMF) fractional anisotropy ratio (rFACMF) in the PT group. (
Clinical Parameters
The PT group had lower scores than the TIA group on all clinical measures at the initial (P<0.05. for NIHSS, BI, Rankin, and RMFT) and follow-up (P=0.056 for NIHSS; P=0.051 for BI; P<0.05 for Rankin and RMFT) time points. The NonPT group did not differ significantly from the TIA group on any clinical measure at either time point (Table 2). Linear regression revealed no significant relationships between RMFT and rFACMF or rFACOF at the initial (P>0.05) or the follow-up (P>0.05) time points.
Discussion
This study aimed to assess the microstructural integrity of CMF connecting ispilesional and contralesional hemispheres of patients having suffered a subcortical stroke along the PT in the acute (<20 days) and chronic (6 months) poststroke periods. Given that infarcts affecting the PT cause a shift in the relative activity of M1 which is mediated by the CMF, we hypothesized that morphological changes in CMF integrity would be detected by DTI-MRI after subcortical stroke to the PT. Furthermore, the objective of this study was to determine the extent to which indirect effects of PT stroke on the microstructure of cortico-cortical transcallosal motor fibers relate to the direct effects of the stroke on PT integrity, as well as to poststroke clinical symptoms.
To our knowledge, this is the first study to systematically delineate CMF in a group of stroke patients, thus permitting longitudinal investigation of full-tract changes in the integrity of fibers indirectly affected by stroke. The results of our fiber tracking neatly correspond to CMF traced in healthy controls using both deterministic (Hofer and Frahm, 2006) and probabilistic (Johansen-Berg et al, 2007) algorithms, passing through the posterior body/isthmus of the CC. This study also includes two essential controls: the NonPT group and the COF bundle. The inclusion of a group of patients with a subcortical stroke affecting a region other than the PT excludes the possibility that decreases in rFACMF are due to nonspecific changes throughout the stroke-affected hemisphere, or the entire brain. The COF bundle allows us to draw inferences about the specificity of poststroke changes to the fibers of interest within the PT patient group. Indeed, the COF tract provides an excellent comparison given that it is located in a distinct vascular region of the brain, thus minimizing the possibility that these fibers have been affected by a nonembolic microvascular ischemic lesion of the PT. Furthermore, just like the CMF, visual-system fiber bundles in the splenium are large and highly myelinated. Thus, the microstructural origin of differences in the diffusion signal at the level of the CMF as compared with the COF is most probably due to changes in membrane integrity or axonal transport (Beaulieu, 2002) rather than the degree of axonal bundling, which is relatively small in both fiber bundles compared with the fibers of the Genu for example (Aboitiz et al, 1992).
This study adds to a significant body of recent literature supporting the notion that increases in contralesional functional activity after stroke are counterproductive to recovery, rather than an adaptive compensatory mechanism of the brain (Carey et al, 2002; Duque et al, 2005; Mansur et al, 2005). Our data suggest that decreases in rFACMF could be a structural correlate of transcallosal disinhibition via the decreased integrity of excitatory neuronal activity synapsing with inhibitory interneurons in the contralesional hemisphere.
Limitations
The fact that the CMF could not be delineated in three subjects underscores the limitations of the fiber-tracking methodology in this aged, clinical sample. Indeed, even normal aging is characterized by an anterior-to-posterior gradient of microstructural degeneration along the CC (Sullivan et al, 2010) making regions of the isthmus/body of the CC, where CMF cross, particularly vulnerable to thinning. Fibers of the COF, which cross more posteriorly in the splenium of the CC, were not affected and thus delineated in all subjects. The tracing of CMF may have been improved by the application of multifiber reconstruction and probabilistic tractography. In the current study, the single-tensor deterministic approach was chosen because it is generally sufficient to delineate the core of major fiber tracts and we were not interested in the more subtle connections extending to the cortex. Nonetheless, a more complex approach may have improved tracing in the difficult cases and thus maximized data use.
An important limitation of the current study is the small sample size. Indeed, the lack of CMF tracing in three subjects further affected our already limited sample, resulting in small groups for final comparisons. Sample sizes were estimated based on data in healthy controls (Heiervang et al, 2006). However, the effects of cerebrovascular disease in subcortical stroke and TIA patients may result in higher intraindividual and interindividual variability, thus requiring larger sample sizes to make conclusions about the population. We managed to reduce this variability by using FA ratios, and decreases in rFACMF at follow-up in this sample are small, but highly significant.
The FA ratios have been shown to provide a useful normalized measure of lesion-specific damage to a tract when, for example, studying homologous tracts in two hemispheres of the same brain (Radlinska et al, 2010). However, the ratio approach within a single tract may have some disadvantages given that the proposed morphological changes after stroke might potentially propagate along axons and spread beyond the midline to homologous regions of the contralesional hemisphere within the period of study. However, we presume that the changes in FA along the CMF are slowly progressive and occur at a time point between 3 weeks and 6 months after stroke. Indeed, early rodent models showed that the breakdown into ovoids of teased phrenic nerve fibers (a feature of Wallerian degeneration that is reflected by decreased FA) progresses at a speed of 46 to 250 mm/day (Lubinska, 1977). In humans, Thomalla et al (2005) reported decreases in FA at the level of the cerebral peduncles appearing between 3 and 9 months after subcortical stroke affecting the internal capsule; this distance is approximately equal to the distance from the subcortical infarcts of our patients and the fibers of the CMF. Thus, changes in rFA should reflect degeneration secondary to subcortical stroke of the PT affecting only the ipsilesional hemisphere; it should not yet have progressed to the contralesional hemisphere within the 6-month follow-up period. If, however, indirect stroke-related damage has in fact affected fibers of the contralesional hemisphere at the follow-up time point, this would render significant differences more difficult to discern and only bias our results in the conservative direction.
Mechanisms for Change in Callosal Motor Fiber Microstructure
There are two possible mechanisms by which PT stroke could affect the integrity of the CMF. Our previous work has shown that decreases in stroke-affected PT rFA can be seen within just 20 days of subcortical stroke, both retrograde and anterograde to the lesion (Radlinska et al, 2010). This decrease in the integrity of PT fibers may have neuronal-transsynaptic effects on adjacent fiber tracts. Changes in CMF microstructure in the chronic poststroke period may be mediated by such effects on cell bodies in layer V, then transfer to cell bodies in layer III via the inhibitory GABAA nonpyramidal cortical interneurons that connect these regions (Avanzino et al, 2007). The current study revealed a significant relationship between the rFA of the PT retrograde to the lesion and rFACMF at follow-up, thus lending support to this hypothesis and suggesting a progressive process of neuronal-transsynaptic degeneration.
Alternatively, the decreases in rFACMF may be related to functional nonuse atrophy, as mediated by changes in the activity of the cortical regions that are interconnected by these fibers. Indeed, in the healthy brain, the relative activity of M1 is associated with the microstructure of the CMF as measured by DTI (Putnam et al, 2008). Furthermore, increases in FA have been reported after training in both motor and cognitive tasks, such as juggling (Scholz et al, 2009) and working memory (Takeuchi et al, 2010a). Thus, it may be that the same kind of mechanism which can lead to an increase in structural connectivity after training could lead to a decrease in structural connectivity after stroke. This explanation presumes that changes in morphological integrity of the CMF are a consequence of changes in the activity of the motor cortices and suggests that the structural integrity of a given pathway is determined by how often and how well it transmits signals between brain regions. To directly test this hypothesis in future, longitudinal measures of relative functional activity in M1 would need to be collected alongside rFACMF assessment. Given that changes in the relative activity of M1 are closely related to clinical outcome, the data from the current study does not support this hypothesis to the degree that there was no relationship between rFACMF and clinical scores at either time point. However, this relationship may have been better reflected in clinical assessments requiring a higher level of interhemispheric communication, such as complex bimanual tasks. Such tasks should also be included in future studies.
Conclusions
This study shows the occurrence of poststroke changes in the integrity of white-matter transcallosal pathways indirectly affected by a subcortical infarct. Although the precise mechanism of morphological changes in poststroke fiber integrity has yet to be determined, this study contributes to our understanding of the relationship between structure and function in the pathological brain.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
